Association between Incidental Pelvic Inflammation and Aggressive Prostate Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Variables and Outcome
2.2.1. Definition of Adverse Pathology
2.2.2. Definition of Biochemical Recurrence (BCR)
2.2.3. Grading Pelvic Inflammation
2.3. Statistical Analysis
2.4. Cell Culture
Invasion and Migration Assays
2.5. Quantitative RT PCR
2.6. Microarray Data Analysis
Immunohistochemical Analysis of Prostate Tumor Tissues
2.7. O-Link
3. Results
3.1. Association between Pelvic Inflammation and Adverse Pathology
3.2. Prostate Cancer Patients with Pelvic Inflammation Demonstrate Elevated Levels of Pro-Inflammatory Cytokines in the Blood
3.3. Inflamed Pelvic Tissue Show Elevated Expression of Genes Involved in the STAT Pathway
3.4. Prostate Cancer Patients with Pelvic Inflammation Demonstrate Elevated Levels of Cancer Enabling Transcriptome
3.5. Evidence of Pro-Inflammatory Cytokines in Prostate Tissues of Prostate Cancer Patients with High Pelvic Inflammation
3.6. Inflamed Pelvic Tissue Contributes to Invasive and Migratory Properties of Prostate Cancer Cells and Can Be Suppressed by Fludarabine and Tocilizumab
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Strasner, A.; Karin, M. Immune Infiltration and Prostate Cancer. Front. Oncol. 2015, 5, 128. [Google Scholar] [CrossRef]
- Vitkin, N.; Nersesian, S.; Siemens, D.R.; Koti, M. The Tumor Immune Contexture of Prostate Cancer. Front. Immunol. 2019, 10, 603. [Google Scholar] [CrossRef] [Green Version]
- Nguyen, D.P.; Li, J.; Tewari, A.K. Inflammation and prostate cancer: The role of interleukin 6 (IL-6). BJU Int. 2014, 113, 986–992. [Google Scholar] [CrossRef]
- Rani, A.; Dasgupta, P.; Murphy, J.J. Prostate Cancer: The Role of Inflammation and Chemokines. Am. J. Pathol. 2019, 189, 2119–2137. [Google Scholar] [CrossRef] [Green Version]
- Morselli, S.; Sebastianelli, A.; Campi, R.; Liaci, A.; Gabellini, L.; Tasso, G.; Fantechi, R.; Venturini, S.; Spatafora, P.; Cito, G.; et al. Adverse pathology after radical prostatectomy: The prognostic role of cumulative cancer length >6-mm threshold in prostate cancer-positive biopsies. Prostate Int. 2019, 7, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Shahabi, A.; Satkunasivam, R.; Gill, I.S.; Lieskovsky, G.; Daneshmand, S.; Pinski, J.K.; Stern, M.C. Predictors of time to biochemical recurrence in a radical prostatectomy cohort within the PSA-era. Can Urol. Assoc J. 2016, 10, E17–E22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Audenet, F.; Seringe, E.; Drouin, S.J.; Comperat, E.; Cussenot, O.; Bitker, M.O.; Rouprêt, M. Persistently elevated prostate-specific antigen at six weeks after radical prostatectomy helps in early identification of patients who are likely to recur. World J. Urol. 2012, 30, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, J.F.; Kothari, V.; Drake, J.M.; Zhao, S.; Dylgjeri, E.; Dean, J.L.; Schiewer, M.J.; McNair, C.; Jones, J.K.; Aytes, A.; et al. DNA-PKcs-Mediated Transcriptional Regulation Drives Prostate Cancer Progression and Metastasis. Cancer Cell 2015, 28, 97–113. [Google Scholar] [CrossRef] [Green Version]
- Maranto, C.; Udhane, V.; Hoang, D.T.; Gu, L.; Alexeev, V.; Malas, K.; Cardenas, K.; Brody, J.R.; Rodeck, U.; Bergom, C.; et al. STAT5A/B Blockade Sensitizes Prostate Cancer to Radiation through Inhibition of RAD51 and DNA Repair. Clin. Cancer Res. 2018, 24, 1917–1931. [Google Scholar] [CrossRef] [Green Version]
- Drost, J.; Karthaus, W.R.; Gao, D.; Driehuis, E.; Sawyers, C.L.; Chen, Y.; Clevers, H. Organoid culture systems for prostate epithelial and cancer tissue. Nat. Protoc. 2016, 11, 347–358. [Google Scholar] [CrossRef] [Green Version]
- Karthaus, W.R.; Iaquinta, P.J.; Drost, J.; Gracanin, A.; van Boxtel, R.; Wongvipat, J.; Dowling, C.M.; Gao, D.; Begthel, H.; Sachs, N.; et al. Identification of Multipotent Luminal Progenitor Cells in Human Prostate Organoid Cultures. Cell 2014, 159, 163–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiewer, M.J.; Goodwin, J.F.; Han, S.; Brenner, J.C.; Augello, M.A.; Dean, J.L.; Liu, F.; Planck, J.L.; Ravindranathan, P.; Chinnaiyan, A.M.; et al. Dual Roles of PARP-1 Promote Cancer Growth and Progression. Cancer Discov. 2012, 2, 1134–1149. [Google Scholar] [CrossRef] [Green Version]
- Centenera, M.M.; Raj, G.V.; Knudsen, K.E.; Tilley, W.D.; Butler, L.M. Ex vivo culture of human prostate tissue and drug development. Nat. Rev. Urol. 2013, 10, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Den, R.B.; Santiago-Jimenez, M.; Alter, J.; Schliekelman, M.; Wagner, J.R.; Ii, J.F.R.; Lee, D.I.; Brito, C.G.; Monahan, K.; Gburek, B.; et al. Decipher correlation patterns post prostatectomy: Initial experience from 2 342 prospective patients. Prostate Cancer Prostatic Dis. 2016, 19, 374–379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lockstone, H.E. Exon array data analysis using Affymetrix power tools and R statistical software. Brief. Bioinform. 2011, 12, 634–644. [Google Scholar] [CrossRef] [Green Version]
- Piccolo, S.R.; Sun, Y.; Campbell, J.D.; Lenburg, M.E.; Bild, A.H.; Johnson, W.E. A single-sample microarray normalization method to facilitate personalized-medicine workflows. Genomics 2012, 100, 337–344. [Google Scholar] [CrossRef] [Green Version]
- Chakravarty, D.; Sboner, A.; Nair, S.S.; Giannopoulou, E.G.; Li, R.; Hennig, S.; Mosquera, J.M.; Pauwels, J.; Park, K.; Kossai, M.; et al. The oestrogen receptor alpha-regulated lncRNA NEAT1 is a critical modulator of prostate cancer. Nat. Commun. 2014, 5, 5383. [Google Scholar] [CrossRef]
- Dimple, C.; Nair, S.S.; Rajhans, R.; Pitcheswara, P.R.; Liu, J.; Balasenthil, S.; Le, X.-F.; Burow, M.E.; Auersperg, N.; Tekmal, R.R.; et al. Role of PELP1/MNAR signaling in ovarian tumorigenesis. Cancer Res. 2008, 68, 4902–4909. [Google Scholar] [CrossRef] [Green Version]
- Meisner, S.; Siim, C.; Kjaergaard, J. The role of infections in the pathogenesis of peptic ulcer disease. Acta Chir. Scand. Suppl. 1988, 547, 35–38. [Google Scholar]
- Waingankar, N.; Martini, A.; Griffiths, L.; Shah, P.; Paulucci, D.J.; Kotamarti, S.; Gul, Z.; Elmasri, M.; Yaskiv, O.; Haines, K.; et al. Weighted Gleason Grade Group (WGGG): A new prostate cancer biopsy reporting system with prognostic potential. Urol. Oncol. 2019, 38, 78.e15-78.e21. [Google Scholar] [CrossRef]
- Dean, L.W.; Assel, M.; Sjoberg, D.D.; Vickers, A.J.; Al-Ahmadie, H.A.; Chen, Y.-B.; Gopalan, A.; Sirintrapun, S.J.; Tickoo, S.K.; Eastham, J.A.; et al. Clinical Usefulness of Total Length of Gleason Pattern 4 on Biopsy in Men with Grade Group 2 Prostate Cancer. J. Urol. 2018, 201, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Rayford, W.; Beksac, A.T.; Alger, J.; Alshalalfa, M.; Ahmed, M.; Khan, I.; Falagario, U.G.; Liu, Y.; Davicioni, E.; Spratt, D.E.; et al. Comparative analysis of 1152 African-American and European-American men with prostate cancer identifies distinct genomic and immunological differences. Commun. Biol. 2021, 4, 670. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, N.F.; Gundersen, G.W.; Rahman, A.; Grimes, M.L.; Rikova, K.; Hornbeck, P.; Ma’ayan, C.A. Clustergrammer, a web-based heatmap visualization and analysis tool for high-dimensional biological data. Sci. Data 2017, 4, 170151. [Google Scholar] [CrossRef] [PubMed]
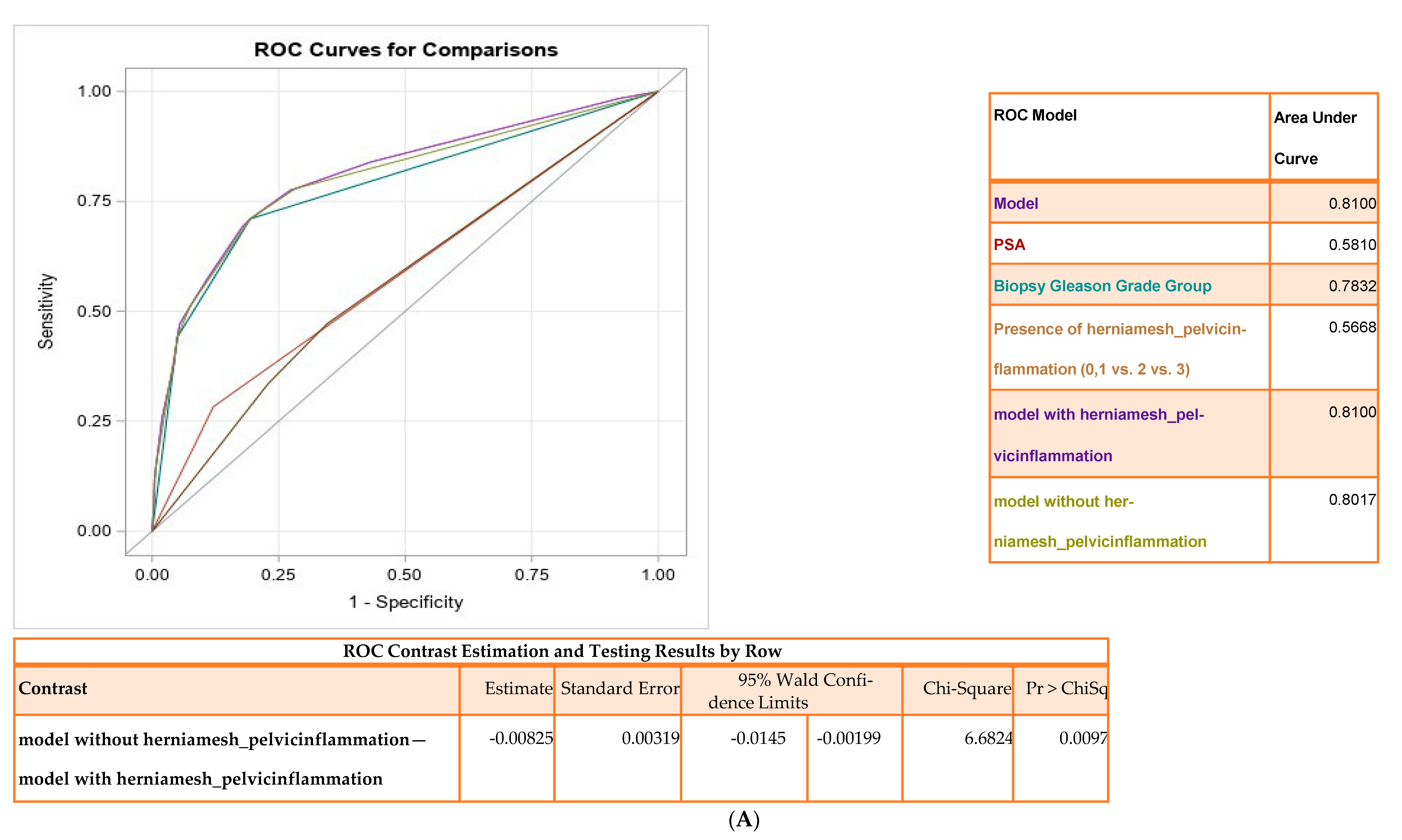
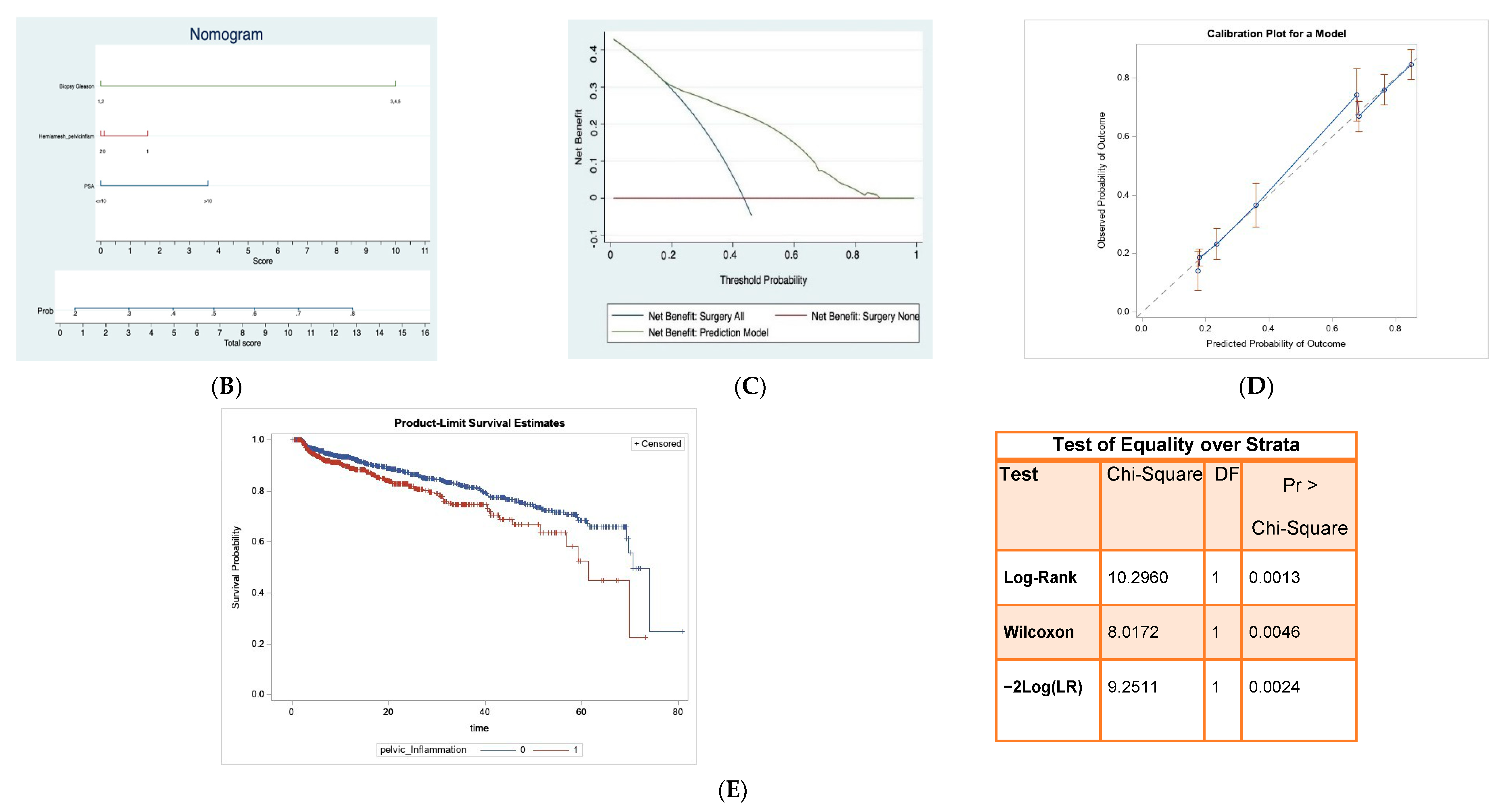
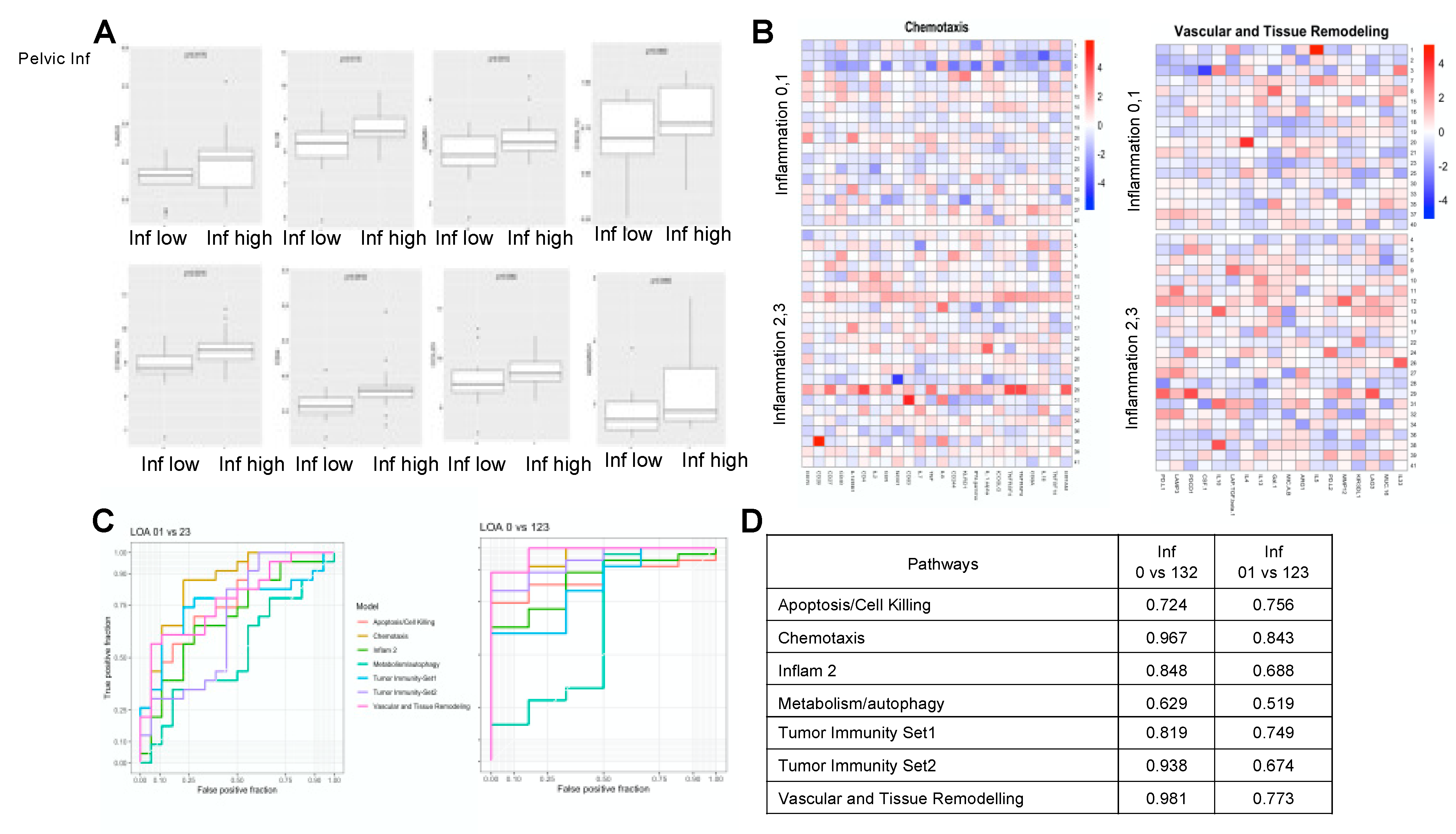

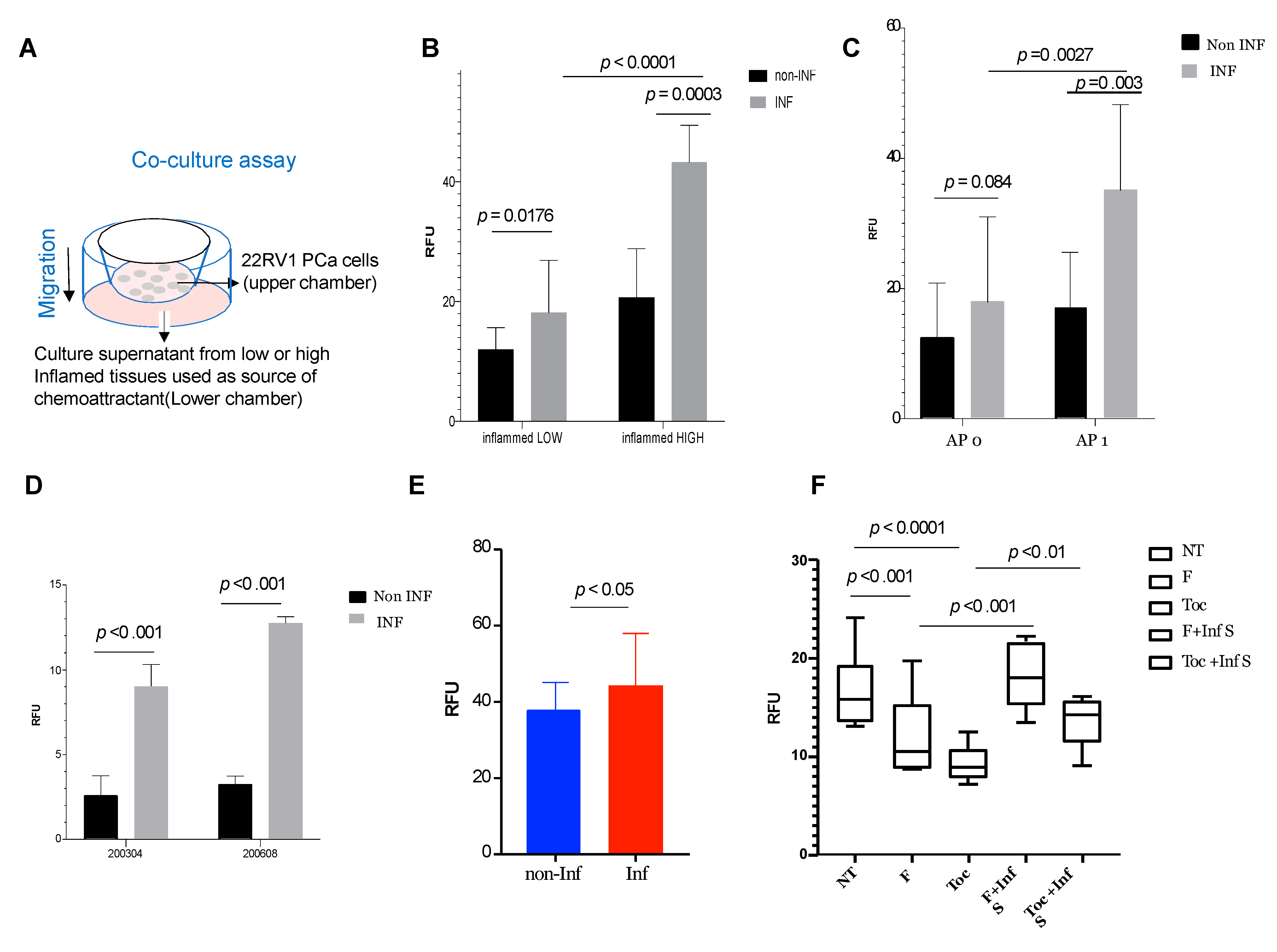
| Covariates | Adverse Pathology Absent (n = 1168) | Adverse Pathology Present (n = 917) | p Value |
|---|---|---|---|
| Age (years) | 61.08 (55.9, 66.8) | 65 (59.4, 69.47) | <0.0001 |
| Surgery_PSA (ng/mL) | 5.4 (4.3, 7.2) | 6.8 (4.9, 10) | <0.0001 |
| MRI Prostate Volume (mL) | 38 (29, 53) | 37(28, 51) | 0.0213 |
| Pelvic Inflammation | <0.0001 | ||
| 0,1 (Low) | 766 (65.58%) | 481 (52.45%) | |
| 2,3 (High) | 402 (34.42%) | 436 (47.55%) | |
| Race | 0.1056 | ||
| African American | 149 (12.76%) | 88 (9.60%) | |
| Caucasian | 857 (73.37%) | 683 (74.48%) | |
| Asian | 61 (5.22%) | 57 (6.22%) | |
| Others | 101 (8.65%) | 89 (9.70%) | |
| MRI PI-RADs | <0.0001 | ||
| 0 | 41 (3.51%) | 14 (1.53%) | |
| 1 | 66 (5.65%) | 39 (4.25%) | |
| 2 | 132 (11.30%) | 50 (5.45%) | |
| 3 | 121 (10.36%) | 42 (4.58%) | |
| 4 | 556 (47.6%) | 330 (35.99%) | |
| 5 | 252 (21.58%) | 442 (48.20%) | |
| MRI ECE | <0.0001 | ||
| absent | 934 (79.97%) | 534 (58.23%) | |
| present | 234 (20.03%) | 383 (41.77%) | |
| Biopsy Gleason group | <0.0001 | ||
| 3 + 3 | 363 (30.91%) | 49 (5.34%) | |
| 3 + 4 | 583 (49.91%) | 215 (23.45%) | |
| 4 + 3 | 166 (14.21%) | 252 (27.48%) | |
| 4 + 4/5 + 3/3 + 5 | 50 (4.28%) | 227 (24.75%) | |
| 4 + 5/5 + 4/5 + 5 | 8 (0.68%) | 174 (18.97%) |
| Univariable Analysis | Multivariable Analysis | |||
|---|---|---|---|---|
| Covariate | Odds Ratio (5% CI) | p Value | Odds Ratio (5% CI) | p Value |
| Age | 1.06 (1.05, 1.07) | <0.0001 | ||
| PSA | ||||
| <10 | Ref | |||
| >10.1 | 2.87 (2.28, 3.62) | <0.0001 | 2.34 (1.79, 3.06) | <0.0001 |
| Race | ||||
| African American | 0.69 (0.47, 1.03) | 0.0108 | ||
| Caucasian | 0.97 (0.71, 1.32) | 0.6654 | ||
| Asian | 1.15 (0.72, 1.85) | 0.1778 | ||
| Others | ||||
| MRI Prostate Volume | 0.99 (0.99, 1.00) | 0.2762 | ||
| Herniamesh pelvicinflam | ||||
| No inflammation w/o hernia mesh | Ref | Ref | ||
| Inflammation without hernia | 1.81 (1.48, 2.22) | 0.0002 | 1.37 (1.08, 1.75) | 0.0077 |
| Inflammation with hernia | 1.45 (1.10, 1.91) | 0.591 | 0.94 (0.68, 1.31) | 0.179 |
| MRI ECE | ||||
| Absent | Ref | |||
| present | 2.98 (2.44, 3.63) | <0.0001 | ||
| MRI PI-RADs | <0.0001 | |||
| 1,2,3 | ||||
| 4,5 | 2.43 (1.95, 3.03) | |||
| Biopsy Gleason Grade Group | <0.0001 | |||
| 1,2 | Ref | Ref | ||
| 3,4,5 | 10.19 (8.27, 12.54) | 9.49 (0.68, 1.31) | <0.0001 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakravarty, D.; Ratnani, P.; Huang, L.; Dovey, Z.; Sobotka, S.; Berryhill, R.; Merisaari, H.; Al Shaarani, M.; Rai, R.; Jambor, I.; et al. Association between Incidental Pelvic Inflammation and Aggressive Prostate Cancer. Cancers 2022, 14, 2734. https://doi.org/10.3390/cancers14112734
Chakravarty D, Ratnani P, Huang L, Dovey Z, Sobotka S, Berryhill R, Merisaari H, Al Shaarani M, Rai R, Jambor I, et al. Association between Incidental Pelvic Inflammation and Aggressive Prostate Cancer. Cancers. 2022; 14(11):2734. https://doi.org/10.3390/cancers14112734
Chicago/Turabian StyleChakravarty, Dimple, Parita Ratnani, Li Huang, Zachary Dovey, Stanislaw Sobotka, Roy Berryhill, Harri Merisaari, Majd Al Shaarani, Richa Rai, Ivan Jambor, and et al. 2022. "Association between Incidental Pelvic Inflammation and Aggressive Prostate Cancer" Cancers 14, no. 11: 2734. https://doi.org/10.3390/cancers14112734
APA StyleChakravarty, D., Ratnani, P., Huang, L., Dovey, Z., Sobotka, S., Berryhill, R., Merisaari, H., Al Shaarani, M., Rai, R., Jambor, I., Yadav, K. K., Mittan, S., Parekh, S., Kodysh, J., Wagaskar, V., Brody, R., Cordon-Cardo, C., Rykunov, D., Reva, B., ... Tewari, A. K. (2022). Association between Incidental Pelvic Inflammation and Aggressive Prostate Cancer. Cancers, 14(11), 2734. https://doi.org/10.3390/cancers14112734








