Recent Advances in the Genetic of MALT Lymphomas
Abstract
Simple Summary
Abstract
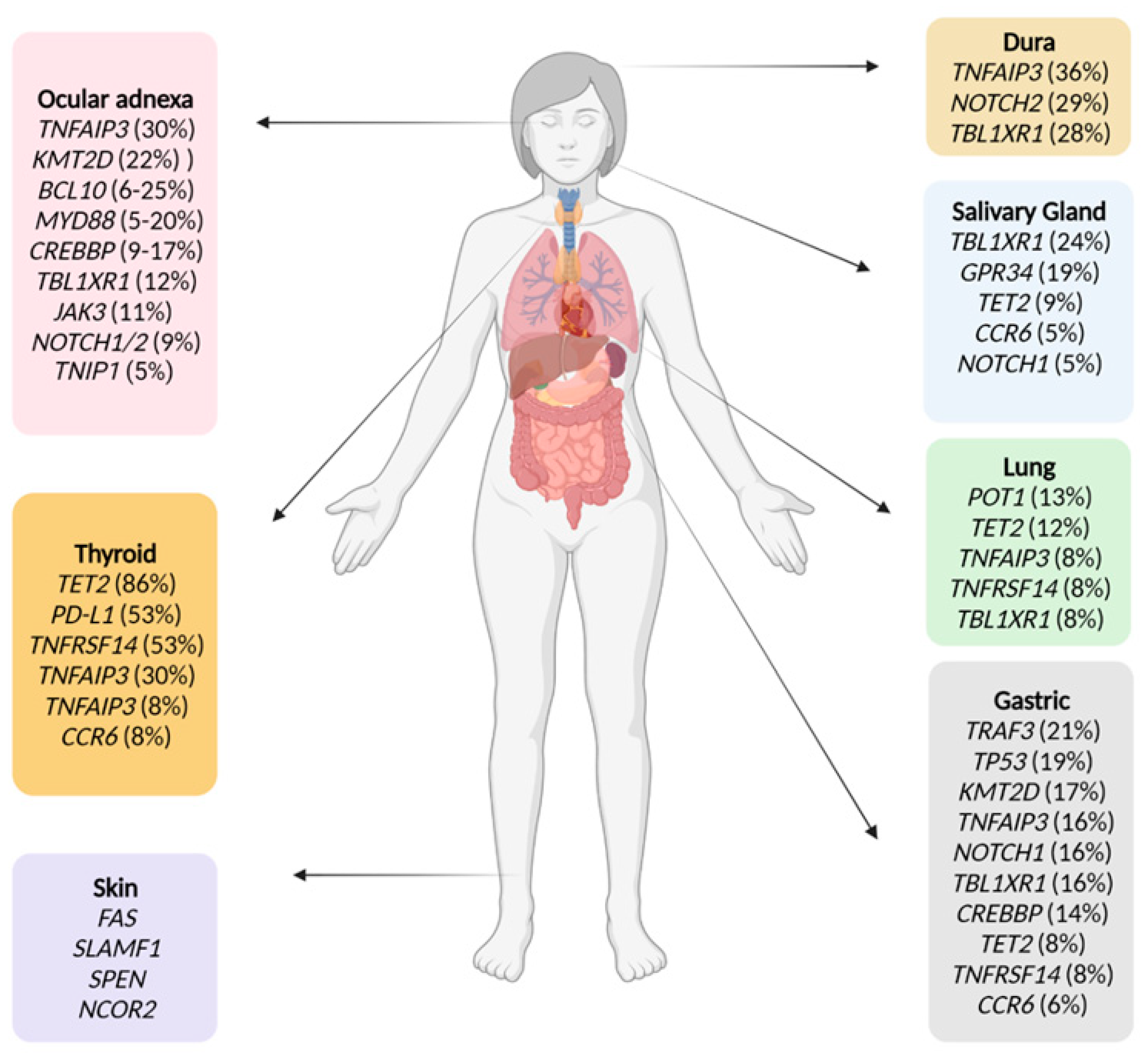
1. Introduction
2. IGHV Usage
3. Cytogenetics
4. Mutations
5. MicroRNAs
6. Transcriptomics
7. Epigenetics and Methylation
8. Applicability in the Real-World
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Isaacson, P.; Wright, D.H. Malignant lymphoma of mucosa-associated lymphoid tissue. A distinctive type of B-cell lymphoma. Cancer 1983, 52, 1410–1416. [Google Scholar] [CrossRef]
- Swerdlow, S.H.; World Health Organization, International Agency for Research on Cancer. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; World Health Organization: Geneva, Switzerland; IARC: Lyon, France, 2017. [Google Scholar]
- Teras, L.R.; DeSantis, C.E.; Cerhan, J.R.; Morton, L.M.; Jemal, A.; Flowers, C.R. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J. Clin. 2016, 66, 443–459. [Google Scholar] [CrossRef]
- Cerhan, J.R.; Habermann, T.M. Epidemiology of Marginal Zone Lymphoma. Ann. Lymphoma 2021, 5, 1. [Google Scholar] [CrossRef] [PubMed]
- Luminari, S.; Cesaretti, M.; Marcheselli, L.; Rashid, I.; Madrigali, S.; Maiorana, A.; Federico, M. Decreasing incidence of gastric MALT lymphomas in the era of anti-Helicobacter pylori interventions: Results from a population-based study on extranodal marginal zone lymphomas. Ann. Oncol. 2010, 21, 855–859. [Google Scholar] [CrossRef] [PubMed]
- Zucca, E.; Bertoni, F. The spectrum of MALT lymphoma at different sites: Biological and therapeutic relevance. Blood 2016, 127, 2082–2092. [Google Scholar] [CrossRef]
- Isaacson, P.G.; Du, M.-Q. MALT lymphoma: From morphology to molecules. Nat. Rev. Cancer 2004, 4, 644–653. [Google Scholar] [CrossRef]
- Ferreri, A.J.M.; Govi, S.; Ponzoni, M. Marginal zone lymphomas and infectious agents. Semin. Cancer Biol. 2013, 23, 431–440. [Google Scholar] [CrossRef]
- Zucca, E.; Conconi, A.; Pedrinis, E.; Cortelazzo, S.; Motta, T.; Gospodarowicz, M.K.; Patterson, B.J.; Ferreri, J.M.; Ponzoni, M.; Devizzi, L.; et al. Nongastric marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue. Arbor Cienc. Pensam. Cult. 2003, 101, 2489–2495. [Google Scholar] [CrossRef]
- Isaacson, P.G. Gastrointestinal lymphoma. Hum. Pathol. 1994, 25, 1020–1029. [Google Scholar] [CrossRef]
- Morgner, A.; Lehn, N.; Andersen, L.P.; Thiede, C.; Bennedsen, M.; Trebesius, K.; Neubauer, B.; Neubauer, A.; Stolte, M.; Bayerdörffer, E. Helicobacter heilmannii-associated primary gastric low-grade MALT lymphoma: Complete remission after curing the infection. Gastroenterology 2000, 118, 821–828. [Google Scholar] [CrossRef]
- Ponzoni, M.; Ferreri, A.J.M.; Guidoboni, M.; Lettini, A.A.; Cangi, M.G.; Pasini, E.; Sacchi, L.; Pecciarini, L.; Grassi, S.; Dal Cin, E.; et al. Chlamydia Infection and Lymphomas: Association Beyond Ocular Adnexal Lymphomas Highlighted by Multiple Detection Methods. Clin. Cancer Res. 2008, 14, 5794–5800. [Google Scholar] [CrossRef]
- Ferreri, A.J.M.; Guidoboni, M.; Ponzoni, M.; De Conciliis, C.; Dell’Oro, S.; Fleischhauer, K.; Caggiari, L.; Lettini, A.A.; Dal Cin, E.; Ieri, R.; et al. Evidence for an association between Chlamydia psittaci and ocular adnexal lymphomas. J. Natl. Cancer Inst. 2004, 96, 586–594. [Google Scholar] [CrossRef]
- Goodlad, J.R.; Davidson, M.M.; Hollowood, K.; Ling, C.; MacKenzie, C.; Christie, I.; Batstone, P.J.; Ho-Yen, D.O. Primary cutaneous B-cell lymphoma and Borrelia burgdorferi infection in patients from the highlands of Scotland. Am. J. Surg. Pathol. 2000, 24, 1279–1285. [Google Scholar] [CrossRef]
- Cerroni, L.; Zöchling, N.; Pütz, B.; Kerl, H. Infection by Borrelia burgdorferi and cutaneous B-cell lymphoma. J. Cutan. Pathol. 1997, 24, 457–461. [Google Scholar] [CrossRef]
- Lecuit, M.; Abachin, E.; Martin, A.; Poyart, C.; Pochart, P.; Suarez, F.; Bengoufa, D.; Feuillard, J.; Lavergne, A.; Gordon, J.I.; et al. Immunoproliferative Small Intestinal Disease Associated with Campylobacter jejuni. N. Engl. J. Med. 2004, 350, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Adam, P.; Czapiewski, P.; Colak, S.; Kosmidis, P.; Tousseyn, T.; Sagaert, X.; Boudova, L.; Okoń, K.; Morresi-Hauf, A.; Agostinelli, C.; et al. Prevalence of Achromobacter xylosoxidans in pulmonary mucosa-associated lymphoid tissue lymphoma in different regions of Europe. Br. J. Haematol. 2014, 164, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Wöhrer, S.; Troch, M.; Streubel, B.; Zwerina, J.; Skrabs, C.; Formanek, M.; Hauff, W.; Hoffmann, M.; Müllauer, L.; Chott, A.; et al. MALT lymphoma in patients with autoimmune diseases: A comparative analysis of characteristics and clinical course. Leukemia 2007, 21, 1812–1818. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Galanina, N.; Bejar, R.; Choi, M.; Goodman, A.; Wieduwilt, M.; Mulroney, C.; Kim, L.; Yeerna, H.; Tamayo, P.; Vergilio, J.A.; et al. Comprehensive genomic profiling reveals diverse but actionable molecular portfolios across hematologic malignancies: Implications for next generation clinical trials. Cancers 2019, 11, 11. [Google Scholar] [CrossRef] [PubMed]
- Hussell, T.; Isaacson, P.G.; Spencer, J. Proliferation and differentiation of tumour cells from B-cell lymphoma of mucosa-associated lymphoid tissue in vitro. J. Pathol. 1993, 169, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Noy, A.; de Vos, S.; Thieblemont, C.; Martin, P.; Flowers, C.R.; Morschhauser, F.; Collins, G.P.; Ma, S.; Coleman, M.; Peles, S.; et al. Targeting Bruton tyrosine kinase with ibrutinib in relapsed/refractory marginal zone lymphoma. Blood 2017, 129, 2224–2232. [Google Scholar] [CrossRef]
- Aarts, W.M.; Bende, R.J.; Steenbergen, E.J.; Kluin, P.M.; Ooms, E.C.M.; Pals, S.T.; Van Noesel, C.J.M. Variable heavy chain gene analysis of follicular lymphomas: Correlation between heavy chain isotype expression and somatic mutation load. Blood 2000, 95, 2922–2929. [Google Scholar] [CrossRef]
- Hallas, C.; Greiner, A.; Peters, K.; Müller-Hermelink, H.K. Immunoglobulin V(H) genes of high-grade mucosa-associated lymphoid tissue lymphomas show a high load of somatic mutations and evidence of antigen-dependent affinity maturation. Lab. Investig. 1998, 78, 277–287. [Google Scholar] [PubMed]
- Qin, Y.; Greiner, A.; Trunk, M.J.F.; Schmausser, B.; Ott, M.M.; Müller-Hermelink, H.K. Somatic Hypermutation in Low-Grade Mucosa-Associated Lymphoid Tissue-Type B-Cell Lymphoma. Blood 1995, 86, 3528–3534. [Google Scholar] [CrossRef] [PubMed]
- Bende, R.J.; Aarts, W.M.; Riedl, R.G.; De Jong, D.; Pals, S.T.; Van Noesel, C.J.M. Among B cell non-Hodgkin’s lymphomas, MALT lymphomas express a unique antibody repertoire with frequent rheumatoid factor reactivity. J. Exp. Med. 2005, 201, 1229–1241. [Google Scholar] [CrossRef]
- Du, M.; Diss, T.C.; Xu, C.; Peng, H.; Isaacson, P.G.; Pan, L. Ongoing mutation in MALT lymphoma immunoglobulin gene suggests that antigen stimulation plays a role in the clonal expansion. Leukemia 1996, 10, 1190–1197. [Google Scholar]
- Bertoni, F.; Cazzaniga, G.; Bosshard, G.; Roggero, E.; Barbazza, R.; De Boni, M.; Capella, C.; Pedrinis, E.; Cavalli, F.; Biondi, A.; et al. Immunoglobulin heavy chain diversity genes rearrangement pattern indicates that MALT-type gastric lymphoma B cells have undergone an antigen selection process. Br. J. Haematol. 1997, 97, 830–836. [Google Scholar] [CrossRef]
- Tierens, A.; Delabie, J.; Pittaluga, S.; Driessen, A.; De Wolf-Peeters, C.; DeWolf-Peeters, C. Mutation analysis of the rearranged immunoglobulin heavy chain genes of marginal zone cell lymphomas indicates an origin from different marginal zone B lymphocyte subsets. Blood 1998, 91, 2381–2386. [Google Scholar] [CrossRef] [PubMed]
- Xochelli, A.; Bikos, V.; Polychronidou, E.; Galigalidou, C.; Agathangelidis, A.; Charlotte, F.; Moschonas, P.; Davis, Z.; Colombo, M.; Roumelioti, M.; et al. Disease-biased and shared characteristics of the immunoglobulin gene repertoires in marginal zone B cell lymphoproliferations. J. Pathol. 2019, 247, 416–421. [Google Scholar] [CrossRef]
- Craig, V.J.; Arnold, I.; Gerke, C.; Huynh, M.Q.; Wündisch, T.; Neubauer, A.; Renner, C.; Falkow, S.; Müller, A. Gastric MALT lymphoma B cells express polyreactive, somatically mutated immunoglobulins. Blood 2010, 115, 581–591. [Google Scholar] [CrossRef]
- Du, M.-Q. Mucosa-associated lymphoid tissue lymphoma of various sites: Common molecular mechanisms but different players. Ann. Lymphoma 2020, 4, 8. [Google Scholar] [CrossRef]
- Bende, R.J.; Janssen, J.; Beentjes, A.; Wormhoudt, T.A.M.; Wagner, K.; Haacke, E.A.; Kroese, F.G.M.; Guikema, J.E.; van Noesel, C.J.M. Salivary gland MALT lymphomas of Sjögren’s syndrome patients in majority express rheumatoid factors affinity-selected for IgG. Arthritis Rheumatol. 2020, in press. [Google Scholar] [CrossRef]
- Hoogeboom, R.; Bende, R.J.; Van Noesel, C.J.M. MALT lymphoma-derived rheumatoid factors are nonpolyreactive high-affinity antibodies. Blood 2010, 116, 1818–1819. [Google Scholar] [CrossRef][Green Version]
- Coupland, S.E.; Foss, H.D.; Anagnostopoulos, I.; Hummel, M.; Stein, H. Immunoglobulin V(H) gene expression among extranodal marginal zone B-cell lymphomas of the ocular adnexa. Investig. Ophthalmol. Vis. Sci. 1999, 40, 555–562. [Google Scholar]
- Mannami, T.; Yoshino, T.; Oshima, K.; Takase, S.; Kondo, E.; Ohara, N.; Nakagawa, H.; Ohtsuki, H.; Harada, M.; Akagi, T. Clinical, histopathological, and immunogenetic analysis of ocular adnexal lymphoproliferative disorders: Characterization of MALT lymphoma and reactive lymphoid hyperplasia. Mod. Pathol. 2001, 14, 641–649. [Google Scholar] [CrossRef]
- Bahler, D.W.; Szankasi, P.; Kulkarni, S.; Tubbs, R.R.; Cook, J.R.; Swerdlow, S.H. Use of similar immunoglobulin VH gene segments by MALT lymphomas of the ocular adnexa. Mod. Pathol. 2009, 22, 833–838. [Google Scholar] [CrossRef][Green Version]
- Moody, S.; Escudero-Ibarz, L.; Wang, M.; Clipson, A.; Ruiz, E.O.; Dunn-Walters, D.; Xue, X.; Zeng, N.; Robson, A.; Chuang, S.-S.; et al. Significant association between TNFAIP3 inactivation and biased immunoglobulin heavy chain variable region 4-34 usage in mucosa-associated lymphoid tissue lymphoma. J. Pathol. 2017, 243, 3–8. [Google Scholar] [CrossRef]
- van Maldegem, F.; Wormhoudt, T.A.M.; Mulder, M.M.S.; Oud, M.E.C.M.; Schilder-Tol, E.; Musler, A.R.; Aten, J.; Saeed, P.; Kersten, M.J.; Pals, S.T.; et al. Chlamydia psittaci-negative ocular adnexal marginal zone B-cell lymphomas have biased VH4-34 immunoglobulin gene expression and proliferate in a distinct inflammatory environment. Leukemia 2012, 26, 1647–1653. [Google Scholar] [CrossRef] [PubMed]
- Dagklis, A.; Ponzoni, M.; Govi, S.; Cangi, M.G.; Pasini, E.; Charlotte, F.; Vino, A.; Doglioni, C.; Davi, F.; Lossos, I.S.; et al. Immunoglobulin gene repertoire in ocular adnexal lymphomas: Hints on the nature of the antigenic stimulation. Leukemia 2012, 26, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Richardson, C.; Chida, A.S.; Adlowitz, D.; Silver, L.; Fox, E.; Jenks, S.A.; Palmer, E.; Wang, Y.; Heimburg-Molinaro, J.; Li, Q.-Z.; et al. Molecular Basis of 9G4 B Cell Autoreactivity in Human Systemic Lupus Erythematosus. J. Immunol. 2013, 191, 4926–4939. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Ikpatt, O.F.; Dubovy, S.R.; Lossos, C.; Natkunam, Y.; Chapman-Fredricks, J.R.; Fan, Y.-S.; Lossos, I.S. Molecular and genomic aberrations in Chlamydophila psittaci negative ocular adnexal marginal zone lymphomas. Am. J. Hematol. 2013, 88, 730–735. [Google Scholar] [CrossRef]
- Thiede, C.; Alpen, B.; Morgner, A.; Schmidt, M.; Ritter, M.; Ehninger, G.; Stolte, M.; Bayerdörffer, E.; Neubauer, A. Ongoing somatic mutations and clonal expansions after cure of Helicobacter pylori infection in gastric mucosa-assiociated lymphoid tissue B-cell lymphoma. J. Clin. Oncol. 1998, 16, 3822–3831. [Google Scholar] [CrossRef] [PubMed]
- Michaeli, M.; Tabibian-Keissar, H.; Schiby, G.; Shahaf, G.; Pickman, Y.; Hazanov, L.; Rosenblatt, K.; Dunn-Walters, D.K.; Barshack, I.; Mehr, R. Immunoglobulin gene repertoire diversification and selection in the stomach—From gastritis to gastric lymphomas. Front. Immunol. 2014, 5, 264. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Nakamura, N.; Kuze, T.; Ono, N.; Abe, M. Multiple lymphomatous polyposis of the gastrointestinal tract is a heterogenous group that includes mantle cell lymphoma and follicular lymphoma: Analysis of somatic mutation of immunoglobulin heavy chain gene variable region. Hum. Pathol. 1999, 30, 581–587. [Google Scholar] [CrossRef]
- Matsubara, J.; Ono, M.; Negishi, A.; Ueno, H.; Okusaka, T.; Furuse, J.; Furuta, K.; Sugiyama, E.; Saito, Y.; Kaniwa, N.; et al. Identification of a predictive biomarker for hematologic toxicities of gemcitabine. J. Clin. Oncol. 2009, 27, 2261–2268. [Google Scholar] [CrossRef] [PubMed]
- Sakuma, H.; Nakamura, T.; Uemura, N.; Chiba, T.; Sugiyama, T.; Asaka, M.; Akamatsu, T.; Ueda, R.; Eimoto, T.; Goto, H.; et al. Immunoglobulin VH gene analysis in gastric MALT lymphomas. Mod. Pathol. 2007, 20, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Thieblemont, C.; Bertoni, F.; Copie-Bergman, C.; Ferreri, A.J.M.; Ponzoni, M. Chronic inflammation and extra-nodal marginal-zone lymphomas of MALT-type. Semin. Cancer Biol. 2014, 24, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Bhatt, S.; Lu, X.; Guo, F.; Veelken, H.; Hsu, D.K.; Liu, F.T.; Alvarez Cubela, S.; Kunkalla, K.; Vega, F.; et al. Chlamydophila psittaci-negative ocular adnexal marginal zone lymphomas express self polyreactive B-cell receptors. Leukemia 2015, 29, 1587–1599. [Google Scholar] [CrossRef]
- Du, M.Q. MALT lymphoma: Recent advances in aetiology and molecular genetics. J. Clin. Exp. Hematop. 2007, 47, 31–42. [Google Scholar] [CrossRef]
- Streubel, B.; Simonitsch-Klupp, I.; Müllauer, L.; Lamprecht, A.; Huber, D.; Siebert, R.; Stolte, M.; Trautinger, F.; Lukas, J.; Püspök, A.; et al. Variable frequencies of MALT lymphoma-associated genetic aberrations in MALT lymphomas of different sites. Leukemia 2004, 18, 1722–1726. [Google Scholar] [CrossRef]
- Farinha, P.; Gascoyne, R.D. Molecular Pathogenesis of Mucosa-Associated Lymphoid Tissue Lymphoma. J. Clin. Oncol. 2005, 23, 6370–6378. [Google Scholar] [CrossRef]
- Ruefli-Brasse, A.A.; French, D.M.; Dixit, V.M. Regulation of NF-κB-Dependent Lymphocyte Activation and Development by Paracaspase. Science 2003, 302, 1581–1584. [Google Scholar] [CrossRef] [PubMed]
- Lucas, P.C.; Yonezumi, M.; Inohara, N.; McAllister-Lucas, L.M.; Abazeed, M.E.; Chen, F.F.; Yamaoka, S.; Seto, M.; Núñez, G. Bcl10 and MALT1, Independent Targets of Chromosomal Translocation in MALT Lymphoma, Cooperate in a Novel NF-κB Signaling Pathway. J. Biol. Chem. 2001, 276, 19012–19019. [Google Scholar] [CrossRef] [PubMed]
- Akagi, T.; Motegi, M.; Tamura, A.; Suzuki, R.; Hosokawa, Y.; Suzuki, H.; Ota, H.; Nakamura, S.; Morishima, Y.; Taniwaki, M.; et al. A novel gene, MALT1 at 18q21, is involved in t(11;18) (q21;q21) found in low-grade B-cell lymphoma of mucosa-associated lymphoid tissue. Oncogene 1999, 18, 5785–5794. [Google Scholar] [CrossRef]
- Morgan, J.; Yin, Y.; Borowsky, A.; Kuo, F.; Nourmand, N.; Koontz, J.; Reynolds, C.; Soreng, L.; Griffin, C.; Graeme-Cook, F.; et al. Breakpoints of the t(11;18)(q21;q21) in mucosa-associated lymphoid tissue (MALT) lymphoma lie within or near the previously undescribed gene MALT1 in chromosome 18. Cancer Res. 1999, 59, 6205–6213. [Google Scholar] [PubMed]
- Baens, M.; Maes, B.; Steyls, A.; Geboes, K.; Marynen, P.; De Wolf-Peeters, C. The product of the t(11;18), an API2-MLT fusion, marks nearly half of gastric MALT type lymphomas without large cell proliferation. Am. J. Pathol. 2000, 156, 1433–1439. [Google Scholar] [CrossRef][Green Version]
- Sanchez-Izquierdo, D.; Buchonnet, G.; Siebert, R.; Gascoyne, R.D.; Climent, J.; Karran, L.; Marin, M.; Blesa, D.; Horsman, D.; Rosenwald, A.; et al. MALT1 is deregulated by both chromosomal translocation and amplification in B-cell non-Hodgkin lymphoma. Blood 2003, 101, 4539–4546. [Google Scholar] [CrossRef] [PubMed]
- Ruland, J.; Duncan, G.S.; Wakeham, A.; Mak, T.W. Differential requirement for Malt1 in T and B cell antigen receptor signaling. Immunity 2003, 19, 749–758. [Google Scholar] [CrossRef]
- Remstein, E.D.; James, C.D.; Kurtine, P.J. Incidence and subtype specificity of API2-MALT1 fusion translocations in extranodal, nodal, and splenic marginal zone lymphomas. Am. J. Pathol. 2000, 156, 1183–1188. [Google Scholar] [CrossRef]
- Lucas, P.C.; Kuffa, P.; Gu, S.; Kohrt, D.; Kim, D.S.L.; Siu, K.; Jin, X.; Swenson, J.; McAllister-Lucas, L.M. A dual role for the API2 moiety in API2-MALT1-dependent NF-kappaB activation: Heterotypic oligomerization and TRAF2 recruitment. Oncogene 2007, 26, 5643–5654. [Google Scholar] [CrossRef]
- Zhou, H.; Du, M.-Q.; Dixit, V.M. Constitutive NF-??B activation by the t(11;18)(q21;q21) product in MALT lymphoma is linked to deregulated ubiquitin ligase activity. Cancer Cell 2005, 7, 425–431. [Google Scholar] [CrossRef]
- Baens, M.; Fevery, S.; Sagaert, X.; Noels, H.; Hagens, S.; Broeckx, V.; Billiau, A.D.; De Wolf-Peeters, C.; Marynen, P. Selective expansion of marginal zone B cells in Emicro-API2-MALT1 mice is linked to enhanced IkappaB kinase gamma polyubiquitination. Cancer Res. 2006, 66, 5270–5277. [Google Scholar] [CrossRef]
- Dierlamm, J.; Baens, M.; Wlodarska, I.; Stefanova-Ouzounova, M.; Hernandez, J.M.; Hossfeld, D.K.; De Wolf-Peeters, C.; Hagemeijer, A.; Van den Berghe, H.; Marynen, P. The Apoptosis Inhibitor Gene API2 and a Novel 18q Gene, MLT, Are Recurrently Rearranged in the t(11;18)(q21;q21) Associated With Mucosa-Associated Lymphoid Tissue Lymphomas. Blood 1999, 93, 3601–3609. [Google Scholar] [CrossRef]
- Rosebeck, S.; Madden, L.; Jin, X.; Gu, S.; Apel, I.J.; Appert, A.; Hamoudi, R.A.; Noels, H.; Sagaert, X.; Loo, P.V.; et al. Cleavage of NIK by the API2-MALT1 fusion oncoprotein leads to noncanonical NF-kappaB activation. Science 2011, 331, 468–472. [Google Scholar] [CrossRef]
- Nie, Z.; Du, M.-Q.; McAllister-Lucas, L.M.; Lucas, P.C.; Bailey, N.G.; Hogaboam, C.M.; Lim, M.S.; Elenitoba-Johnson, K.S.J. Conversion of the LIMA1 tumour suppressor into an oncogenic LMO-like protein by API2–MALT1 in MALT lymphoma. Nat. Commun. 2015, 6, 5908. [Google Scholar] [CrossRef]
- Streubel, B.; Seitz, G.; Stolte, M.; Birner, P.; Chott, A.; Raderer, M. MALT lymphoma associated genetic aberrations occur at different frequencies in primary and secondary intestinal MALT lymphomas. Gut 2006, 55, 1581–1585. [Google Scholar] [CrossRef][Green Version]
- Remstein, E.D.; Kurtin, P.J.; Einerson, R.R.; Paternoster, S.F.; Dewald, G.W. Primary pulmonary MALT lymphomas show frequent and heterogeneous cytogenetic abnormalities, including aneuploidy and translocations involving API2 and MALT1 and IGH and MALT1. Leukemia 2004, 18, 156–160. [Google Scholar] [CrossRef]
- Gallardo, F.; Bellosillo, B.; Espinet, B.; Pujol, R.M.; Estrach, T.; Servitje, O.; Romagosa, V.; Barranco, C.; Boluda, S.; García, M.; et al. Aberrant nuclear BCL10 expression and lack of t(11;18)(q21;q21) in primary cutaneous marginal zone B-cell lymphoma. Hum. Pathol. 2006, 37, 867–873. [Google Scholar] [CrossRef]
- Schreuder, M.I.; Hoefnagel, J.J.; Jansen, P.M.; van Krieken, J.H.J.M.; Willemze, R.; Hebeda, K.M. FISH analysis of MALT lymphoma-specific translocations and aneuploidy in primary cutaneous marginal zone lymphoma. J. Pathol. 2005, 205, 302–310. [Google Scholar] [CrossRef]
- Dierlamm, J.; Baens, M.; Stefanova-Ouzounova, M.; Hinz, K.; Wlodarska, I.; Maes, B.; Steyls, A.; Driessen, A.; Verhoef, G.; Gaulard, P.; et al. Detection of t(11;18)(q21;q21) by interphase fluorescence in situ hybridization using API2 and MLTspecific probes. Blood 2000, 96, 2215–2218. [Google Scholar] [CrossRef]
- Murga Penas, E.M.; Hinz, K.; Röser, K.; Copie-Bergman, C.; Wlodarska, I.; Marynen, P.; Hagemeijer, A.; Gaulard, P.; Löning, T.; Hossfeld, D.K.; et al. Translocations t(11;18)(q21;q21) and t(14;18)(q32;q21) are the main chromosomal abnormalities involving MLT/MALT1 in MALT lymphomas. Leukemia 2003, 17, 2225–2229. [Google Scholar] [CrossRef]
- Takino, H.; Li, C.; Hu, S.; Kuo, T.-T.; Geissinger, E.; Muller-Hermelink, H.K.; Kim, B.; Swerdlow, S.H.; Inagaki, K. Primary cutaneous marginal zone B-cell lymphoma: A molecular and clinicopathological study of cases from Asia, Germany, and the United States. Mod. Pathol. 2008, 21, 1517–1526. [Google Scholar] [CrossRef]
- Dierlamm, J.; Murga Penas, E.; Daibata, M.; Tagushi, H.; Hinz, K.; Baens, M.; Cools, J.; Schilling, G.; Michaux, L.; Marynen, P.; et al. The novel t(11;12;18)(q21;q13;q21) represents a variant translocation of the t(11;18)(q21;q21) associated with MALT-type lymphoma. Leukemia 2002, 16, 1863–1864. [Google Scholar] [CrossRef]
- Kubonishi, I.; Sugito, S.; Kobayashi, M.; Asahi, Y.; Tsuchiya, T.; Yamashiro, T.; Miyoshi, I. A unique chromosome translocation, t(11;12;18)(q21;q13;q21) [correction of t(11;12;18)(q13;q13;q12)], in primary lung lymphoma. Cancer Genet. Cytogenet. 1995, 82, 54–56. [Google Scholar] [CrossRef]
- Murga Penas, E.M.; Callet-Bauchu, E.; Ye, H.; Hinz, K.; Albert, N.; Copie-Bergman, C.; Gazzo, S.; Berger, F.; Salles, G.; Bokemeyer, C.; et al. The translocations t(6;18;11)(q24;q21;q21) and t(11;14;18)(q21;q32;q21) lead to a fusion of the API2 and MALT1 genes and occur in MALT lymphomas. Haematologica 2007, 92, 405–409. [Google Scholar] [CrossRef]
- Alpen, B.; Neubauer, A.; Dierlamm, J.; Marynen, P.; Thiede, C.; Bayerdörffer, E.; Stolte, M. Translocation t(11;18) absent in early gastric marginal zone B-cell lymphoma of MALT type responding to eradication of Helicobacter pylori infection. Blood 2000, 95, 4014–4015. [Google Scholar] [CrossRef]
- Liu, H.; Ye, H.; Ruskone-Fourmestraux, A.; De Jong, D.; Pileri, S.; Thiede, C.; Lavergne, A.; Boot, H.; Caletti, G.; Wündisch, T.; et al. T(11;18) is a marker for all stage gastric MALT lymphomas that will not respond to H. pylori eradication. Gastroenterology 2002, 122, 1286–1294. [Google Scholar] [CrossRef]
- Montalban, C.; Santón, A.; Redondo, C.; García-Cosio, M.; Boixeda, D.; Vazquez-Sequeiros, E.; Norman, F.; de Argila, C.M.; Alvarez, I.; Abraira, V.; et al. Long-term persistence of molecular disease after histological remission in low-grade gastric MALT lymphoma treated with H. pylori eradication. Lack of association with translocation t(11;18): A 10-year updated follow-up of a prospective study. Ann. Oncol. 2005, 16, 1539–1544. [Google Scholar] [CrossRef]
- Nakamura, S.; Matsumoto, T.; Nakamura, S.; Jo, Y.; Fujisawa, K.; Suekane, H.; Yao, T.; Tsuneyoshi, M.; Iida, M. Chromosomal translocation t(11;18)(q21;q21) in gastrointestinal mucosa associated lymphoid tissue lymphoma. J. Clin. Pathol. 2003, 56, 36–42. [Google Scholar] [CrossRef]
- Iwano, M.; Okazaki, K.; Uchida, K.; Nakase, H.; Ohana, M.; Matsushima, Y.; Inagaki, H.; Chiba, T. Characteristics of gastric B-cell lymphoma of mucosa-associated lymphoid tissue type involving multiple organs. J. Gastroenterol. 2004, 39, 739–746. [Google Scholar] [CrossRef]
- Yeh, K.H.; Kuo, S.H.; Chen, L.T.; Mao, T.L.; Doong, S.L.; Wu, M.S.; Hsu, H.C.; Tzeng, Y.S.; Chen, C.L.; Lin, J.T.; et al. Nuclear expression of BCL10 or nuclear factor kappa B helps predict Helicobacter pylori-independent status of low-grade gastric mucosa-associated lymphoid tissue lymphomas with or without t(11;18)(q21;q21 ). Blood 2005, 106, 1037–1041. [Google Scholar] [CrossRef]
- Nakamura, S.; Ye, H.; Bacon, C.M.; Goatly, A.; Liu, H.; Banham, A.H.; Ventura, R.; Matsumoto, T.; Iida, M.; Ohji, Y.; et al. Clinical impact of genetic aberrations in gastric MALT lymphoma: A comprehensive analysis using interphase fluorescence in situ hybridisation. Gut 2007, 56, 1358–1363. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Liu, H.; Attygalle, A.; Wotherspoon, A.C.; Nicholson, A.G.; Charlotte, F.; Leblond, V.; Speight, P.; Goodlad, J.; Lavergne-Slove, A.; et al. Variable frequencies of t(11;18)(q21;q21) in MALT lymphomas of different sites: Significant association with CagA strains of H. pylori in gastric MALT lymphoma. Blood 2003, 102, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Kuo, S.H.; Chen, L.T.; Yeh, K.H.; Wu, M.S.; Hsu, H.C.; Yeh, P.Y.; Mao, T.L.; Chen, C.L.; Doong, S.L.; Lin, J.T.; et al. Nuclear expression of BCL10 or nuclear factor kappa B predicts Helicobacter pylori-independent status of early-stage, high-grade gastric mucosa-associated lymphoid tissue lymphomas. J. Clin. Oncol. 2004, 22, 3491–3497. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Gong, L.; Liu, H.; Ruskone-Fourmestraux, A.; De Jong, D.; Pileri, S.; Thiede, C.; Lavergne, A.; Boot, H.; Caletti, G.; et al. Strong BCL10 nuclear expression identifies gastric MALT lymphomas that do not respond to H. pylori eradication. Gut 2006, 55, 137–139. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, K.; Maeshima, A.M.; Nomoto, J.; Suzuki, T.; Yuda, S.; Yamauchi, N.; Taniguchi, H.; Makita, S.; Fukuhara, S.; Munakata, W.; et al. Mucosa-associated lymphoid tissue lymphoma with t(11;18)(q21;q21) translocation: Long-term follow-up results. Ann. Hematol. 2019, 98, 1675–1687. [Google Scholar] [CrossRef] [PubMed]
- Dong, G.; Liu, C.; Ye, H.; Gong, L.; Zheng, J.; Li, M.; Huang, X.; Huang, X.; Huang, Y.; Shi, Y.; et al. BCL10 nuclear expression and t(11;18)(q21;q21) indicate nonresponsiveness to Helicobacter pylori eradication of Chinese primary gastric MALT lymphoma. Int. J. Hematol. 2009, 88, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Salar, A.; Bellosillo, B.B.; Serrano, S.; Besses, C. Persistent residual disease in t(11;18)(q21;q21) positive gastric mucosa-associated lymphoid tissue lymphoma treated with chemotherapy or rituximab. J. Clin. Oncol. 2005, 23, 7361–7362. [Google Scholar] [CrossRef]
- Lévy, M.; Copie-Bergman, C.; Gameiro, C.; Chaumette, M.T.; Delfau-Larue, M.H.; Haioun, C.; Charachon, A.; Hemery, F.; Gaulard, P.; Leroy, K.; et al. Prognostic value of translocation t(11;18) in tumoral response of low-grade gastric lymphoma of mucosa-associated lymphoid tissue type to oral chemotherapy. J. Clin. Oncol. 2005, 23, 5061–5066. [Google Scholar] [CrossRef]
- Martinelli, G.; Laszlo, D.; Ferreri, A.J.M.M.; Pruneri, G.; Ponzoni, M.; Conconi, A.; Crosta, C.; Pedrinis, E.; Bertoni, F.; Calabrese, L.; et al. Clinical activity of rituximab in gastric marginal zone non-Hodgkin’s lymphoma resistant to or not eligible for anti-Helicobacter pylori therapy. J. Clin. Oncol. 2005, 23, 1979–1983. [Google Scholar] [CrossRef]
- Lévy, M.; Copie-Bergman, C.; Molinier-Frenkel, V.; Riou, A.; Haioun, C.; Gaulard, P.; Delfau-Larue, M.-H.H.; Sobhani, I.; Leroy, K.; Delchier, J.-C.C. Treatment of t(11;18)-positive gastric mucosa-associated lymphoid tissue lymphoma with rituximab and chlorambucil: Clinical, histological, and molecular follow-up. Leuk. Lymphoma 2010, 51, 284–290. [Google Scholar] [CrossRef]
- Salar, A.; Domingo-Domenech, E.; Panizo, C.; Nicolás, C.; Bargay, J.; Muntañola, A.; Canales, M.; Bello, J.L.; Sancho, J.M.; Tomás, J.F.; et al. First-line response-adapted treatment with the combination of bendamustine and rituximab in patients with mucosa-associated lymphoid tissue lymphoma (MALT2008-01): A multicentre, single-arm, phase 2 trial. Lancet Haematol. 2014, 1, e104–e111. [Google Scholar] [CrossRef]
- Ruskone-Fourmestraux, A.; Fischbach, W.; Aleman, B.M.P.; Boot, H.; Du, M.Q.; Megraud, F.; Montalban, C.; Raderer, M.; Savio, A.; Wotherspoon, A.; et al. EGILS consensus report. Gastric extranodal marginal zone B-cell lymphoma of MALT. Gut 2011, 60, 747–758. [Google Scholar] [CrossRef]
- Zucca, E.; Arcaini, L.; Buske, C.; Johnson, P.W.; Ponzoni, M.; Raderer, M.; Ricardi, U.; Salar, A.; Stamatopoulos, K.; Thieblemont, C.; et al. Marginal zone lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 17–29. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Nakata, M.; Maekawa, M.; Takahashi, M.; Fujii, H.; Matsuno, Y.; Mitsuhiro, F.; Hiroyuki, O.; Daizo, S.; Takeaki, T.; et al. Detection of t(11; 18) in MALT-type lymphoma with dual-color fluorescence in situ hybridization and reverse transcriptase-polymerase chain reaction analysis. Diagn. Mol. Pathol. 2001, 10, 207–213. [Google Scholar] [CrossRef]
- Fecteau, J.F.; Néron, S. CD40 stimulation of human peripheral B lymphocytes: Distinct response from naive and memory cells. J. Immunol. 2003, 171, 4621–4629. [Google Scholar] [CrossRef]
- Chen, J.; Chen, Z.J. Signaling to NF-kappaB: Regulation by ubiquitination. Cold Spring Harb. Perspect. Biol. 2010, 2, a003350. [Google Scholar]
- Du, M.-Q. MALT lymphoma: Genetic abnormalities, immunological stimulation and molecular mechanism. Best Pract. Res. Clin. Haematol. 2017, 30, 13–23. [Google Scholar] [CrossRef]
- Takada, S.; Yoshino, T.; Taniwaki, M.; Nakamura, N.; Nakamine, H.; Oshima, K.; Sadahira, Y.; Inagaki, H.; Oshima, K.; Tadaatsu, A. Involvement of the chromosomal translocation t(11;18) in some mucosa-associated lymphoid tissue lymphomas and diffuse large B-cell lymphomas of the ocular adnexa: Evidence from multiplex reverse transcriptase-polymerase chain reaction and fluorescence in situ hybridization on using formalin-fixed, paraffin-embedded specimens. Mod. Pathol. 2003, 16, 445–452. [Google Scholar] [PubMed]
- Streubel, B.; Lamprecht, A.; Dierlamm, J.; Cerroni, L.; Stolte, M.; Ott, G.; Raderer, M.; Chott, A. T(14;18)(q32;q21) involving IGH and MALT1 is a frequent chromosomal aberration in MALT lymphoma. Blood 2003, 101, 2335–2339. [Google Scholar] [CrossRef]
- Thome, M. CARMA1, BCL-10 and MALT1 in lymphocyte development and activation. Nat. Rev. Immunol. 2004, 4, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Gong, L.; Liu, H.; Hamoudi, R.A.; Shirali, S.; Ho, L.; Chott, A.; Streubel, B.; Siebert, R.; Gesk, S.; et al. MALT lymphoma with t(14;18)(q32;q21)/IGH-MALT1 is characterized by strong cytoplasmic MALT1 and BCL10 expression. J. Pathol. 2005, 205, 293–301. [Google Scholar] [CrossRef]
- Achuthan, R.; Bell, S.M.; Carr, I.M.; Leek, J.P.; Roberts, P.; Horgan, K.; Markham, A.F.; Selby, P.J.; MacLennan, K.A. BCL10 in malignant lymphomas—An evaluation using fluorescence in situ hybridization. J. Pathol. 2002, 196, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Coornaert, B.; Baens, M.; Heyninck, K.; Bekaert, T.; Haegman, M.; Staal, J.; Sun, L.; Chen, Z.J.; Marynen, P.; Beyaert, R. T cell antigen receptor stimulation induces MALT1 paracaspase—Mediated cleavage of the NF-κB inhibitor A20. Nat. Immunol. 2008, 9, 263–271. [Google Scholar] [CrossRef]
- Düwel, M.; Welteke, V.; Oeckinghaus, A.; Baens, M.; Kloo, B.; Ferch, U.; Darnay, B.G.; Ruland, J.; Marynen, P.; Krappmann, D. A20 negatively regulates T cell receptor signaling to NF-kappaB by cleaving Malt1 ubiquitin chains. J. Immunol. 2009, 182, 7718–7728. [Google Scholar] [CrossRef] [PubMed]
- Kirchhofer, D.; Vucic, D. Protease activity of MALT1: A mystery unravelled. Biochem. J. 2012, 444, e3–e5. [Google Scholar] [CrossRef]
- Hailfinger, S.; Nogai, H.; Pelzer, C.; Jaworski, M.; Cabalzar, K.; Charton, J.E.; Guzzardi, M.; Décaillet, C.; Grau, M.; Dörken, B.; et al. Malt1-dependent RelB cleavage promotes canonical NF-kappaB activation in lymphocytes and lymphoma cell lines. Proc. Natl. Acad. Sci. USA 2011, 108, 14596–14601. [Google Scholar] [CrossRef] [PubMed]
- Tusche, M.W.; Ward, L.A.; Vu, F.; McCarthy, D.; Quintela-Fandino, M.; Ruland, J.; Gommerman, J.L.; Mak, T.W. Differential requirement of MALT1 for BAFF-induced outcomes in B cell subsets. J. Exp. Med. 2009, 206, 2671. [Google Scholar] [CrossRef]
- Zhang, S.; Wei, M.; Liang, Q.; Johnson, D.; Dow, N.; Nelson, A.; Aguilera, N.; Auerbach, A.; Wang, G. The t(14;18)(q32;q21)/IGH-MALT1 translocation in gastrointestinal extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma). Histopathology 2014, 64, 791–798. [Google Scholar] [CrossRef]
- Tanimoto, K.; Sekiguchi, N.; Yokota, Y.; Kaneko, A.; Watanabe, T.; Maeshima, A.M.; Matsuno, Y.; Harada, M.; Tobinai, K.; Kobayashi, Y. Fluorescence in situhybridization (FISH) analysis of primary ocular adnexal MALT lymphoma. BMC Cancer 2006, 6, 249. [Google Scholar] [CrossRef]
- Wongchaowart, N.T.; Kim, B.; Hsi, E.D.; Swerdlow, S.H.; Tubbs, R.R.; Cook, J.R. t(14;18)(q32;q21) involving IGH and MALT1 is uncommon in cutaneous MALT lymphomas and primary cutaneous diffuse large B-cell lymphomas. J. Cutan. Pathol. 2006, 33, 286–292. [Google Scholar] [CrossRef]
- Garcia, M.; Konoplev, S.; Morosan, C.; Abruzzo, L.V.; Bueso-Ramos, C.E.; Medeiros, L.J. MALT lymphoma involving the kidney: A report of 10 cases and review of the literature. Am. J. Clin. Pathol. 2007, 128, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Bhagavathi, S.; Greiner, T.C.; Kazmi, S.A.; Fu, K.; Sanger, W.G.; Chan, W.C. Extranodal marginal zone lymphoma of the dura mater with IgH/MALT1 translocation and review of literature. J. Hematop. 2008, 1, 131–137. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kuper-Hommel, M.J.J.; Schreuder, M.I.; Gemmink, A.H.; van Krieken, J.H.J.M. T(14;18)(q32;q21) involving MALT1 and IGH genes occurs in extranodal diffuse large B-cell lymphomas of the breast and testis. Mod. Pathol. 2013, 26, 421–427. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cook, J.R.; Sherer, M.; Craig, F.E.; Shekhter-Levin, S.; Swerdlow, S.H. T(14;18)(q32;q21) involving MALT1 and IGH genes in an extranodal diffuse large B-cell lymphoma. Hum. Pathol. 2003, 34, 1212–1215. [Google Scholar] [CrossRef] [PubMed]
- Maes, B.; Demunter, A.; Peeters, B.; De Wolf-Peeters, C. BCL10 mutation does not represent an important pathogenic mechanism in gastric MALT-type lymphoma, and the presence of the API2-MLT fusion is associated with aberrant nuclear BCL10 expression. Blood 2002, 99, 1398–1404. [Google Scholar] [CrossRef]
- Ye, H.; Dogan, A.; Karran, L.; Willis, T.G.; Chen, L.; Wlodarska, I.; Dyer, M.J.; Isaacson, P.G.; Du, M.Q. BCL10 expression in normal and neoplastic lymphoid tissue. Nuclear localization in MALT lymphoma. Am. J. Pathol. 2000, 157, 1147–1154. [Google Scholar] [CrossRef]
- Streubel, B.; Vinatzer, U.; Lamprecht, A.; Raderer, M.; Chott, A. T(3;14)(p14.1;q32) involving IGH and FOXP1 is a novel recurrent chromosomal aberration in MALT lymphoma. Leukemia 2005, 19, 652–658. [Google Scholar] [CrossRef]
- Wlodarska, I.; Veyt, E.; De Paepe, P.; Vandenberghe, P.; Nooijen, P.; Theate, I.; Michaux, L.; Sagaert, X.; Marynen, P.; Hagemeijer, A.; et al. FOXP1, a gene highly expressed in a subset of diffuse large B-cell lymphoma, is recurrently targeted by genomic aberrations. Leukemia 2005, 19, 1299–1305. [Google Scholar] [CrossRef]
- Fenton, J.A.L.; Schuuring, E.; Barrans, S.L.; Banham, A.H.; Rollinson, S.J.; Morgan, G.J.; Jack, A.S.; van Krieken, J.H.J.M.; Kluin, P.M. t(3;14)(p14;q32) Results in aberrant expression of FOXP1 in a case of diffuse large B-cell lymphoma. Genes Chromosom. Cancer 2006, 45, 164–168. [Google Scholar] [CrossRef]
- Vinatzer, U.; Gollinger, M.; Müllauer, L.; Raderer, M.; Chott, A.; Streubel, B. Mucosa-associated lymphoid tissue lymphoma: Novel translocations including rearrangements of ODZ2, JMJD2C, and CNN3. Clin. Cancer Res. 2008, 14, 6426–6431. [Google Scholar] [CrossRef]
- Ansell, S.M.; Akasaka, T.; McPhail, E.; Manske, M.; Braggio, E.; Price-Troska, T.; Ziesmer, S.; Secreto, F.; Fonseca, R.; Gupta, M.; et al. t(X;14)(p11;q32) in MALT lymphoma involving GPR34 reveals a role for GPR34 in tumor cell growth. Blood 2012, 120, 3949–3957. [Google Scholar] [CrossRef] [PubMed]
- Baens, M.; Ferreiro, J.F.; Tousseyn, T.; Urbankova, H.; Michaux, L.; de Leval, L.; Dierickx, D.; Wolter, P.; Sagaert, X.; Vandenberghe, P.; et al. t(X;14)(p11.4;q32.33) is recurrent in marginal zone lymphoma and up-regulates GPR34. Haematologica 2012, 97, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.; Liu, H.; Ye, H.; Martín-Subero, J.I.; Siebert, R.; Huang, W. Pulmonary mucosa-associated lymphoid tissue lymphoma with strong nuclear B-cell CLL/lymphoma 10 (BCL10) expression and novel translocation t(1;2)(p22;p12)/immunoglobulin κ chain-BCL10. J. Clin. Pathol. 2007, 60, 727. [Google Scholar] [CrossRef] [PubMed]
- Van Keimpema, M.; Grüneberg, L.J.; Mokry, M.; Van Boxtel, R.; Van Zelm, M.C.; Coffer, P.; Pals, S.T.; Spaargaren, M. The forkhead transcription factor FOXP1 represses human plasma cell differentiation. Blood 2015, 126, 2098–2109. [Google Scholar] [CrossRef] [PubMed]
- van Keimpema, M.; Grüneberg, L.J.; Mokry, M.; van Boxtel, R.; Koster, J.; Coffer, P.J.; Pals, S.T.; Spaargaren, M. FOXP1 directly represses transcription of proapoptotic genes and cooperates with NF-κB to promote survival of human B cells. Blood 2014, 124, 3431. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, Y.; Shiozawa, E.; Watanabe, N.; Homma, M.; Noh, J.Y.; Ito, K.; Takimoto, M.; Yamochi-Onizuka, T. t(3;14)(p14.1;q32)/FOXP1-IGH translocation in thyroid extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma). Leuk. Res. 2020, 95, 106399. [Google Scholar] [CrossRef]
- Wu, F.; Watanabe, N.; Tzioni, M.-M.; Akarca, A.; Zhang, C.; Li, Y.; Chen, Z.; Cucco, F.; Carmell, N.; Noh, J.Y.; et al. Thyroid MALT lymphoma: Self-harm to gain potential T-cell help. Leukemia 2021, 35, 3497–3508. [Google Scholar] [CrossRef]
- Wotherspoon, A.C.; Pan, L.; Diss, T.C.; Isaacson, P.G. Cytogenetic study of B-cell lymphoma of mucosa-associated lymphoid tissue. Cancer Genet. Cytogenet. 1992, 58, 35–38. [Google Scholar] [CrossRef]
- Whang-Peng, J.; Knutsen, T.; Jaffe, E.; Raffeld, M.; Zhao, W.P.; Duffey, P.; Longo, D.L. Cytogenetic study of two cases with lymphoma of mucosa-associated lymphoid tissue. Cancer Genet. Cytogenet. 1994, 77, 74–80. [Google Scholar] [CrossRef]
- Dierlamm, J.; Michaux, L.; Wlodarska, I.; Pittaluga, S.; Zeller, W.; Stul, M.; Criel, A.; Thomas, J.; Boogaerts, M.; Delaere, P.; et al. Trisomy 3 in marginal zone B-cell lymphoma: A study based on cytogenetic analysis and fluorescence in situ hybridization. Br. J. Haematol. 1996, 93, 242–249. [Google Scholar] [CrossRef]
- Taji, S.; Nomura, K.; Matsumoto, Y.; Sakabe, H.; Yoshida, N.; Mitsufiji, S.; Nishida, K.; Horiike, S.; Nakamura, S.; Morita, M.; et al. Trisomy 3 may predict a poor response of gastric MALT lymphoma to Helicobacter pylori eradication therapy. World J. Gastroenterol. 2005, 11, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Ott, G.; Katzenberger, T.; Greiner, A.; Kalla, J.; Rosenwald, A.; Heinrich, U.; Ott, M.M.; Müller-Hermelink, H.K. The t(11;18)(q21;q21) Chromosome Translocation Is a Frequent and Specific Aberration in Low-Grade but not High-Grade Malignant Non-Hodgkin’s Lymphomas of the Mucosa-associated Lymphoid Tissue (MALT-) Type. AACR 1997, 57, 3944–3948. [Google Scholar]
- Clark, H.M.; Jones, D.B.; Wright, D.H. Cytogenetic and molecular studies of t(14;18) and t(14;19) in nodal and extranodal B-cell lymphoma. J. Pathol. 1992, 166, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Wotherspoon, A.C.; Finn, T.M.; Isaacson, P.G. Trisomy 3 in Low-Grade B-Cell Lymphomas of Mucosa-Associated Lymphoid Tissue. Blood 1995, 85, 2000–2004. [Google Scholar] [CrossRef] [PubMed]
- Ott, G.; Kalla, J.; Steinhoff, A.; Rosenwald, A.; Katzenberger, T.; Roblick, U.; Ott, M.M.; Müller-Hermelink, H.K. Trisomy 3 Is Not a Common Feature in Malignant Lymphomas of Mucosa-Associated Lymphoid Tissue Type. Am. J. Pathol. 1998, 153, 689. [Google Scholar] [CrossRef]
- Blanco, R.; Lyda, M.; Davis, B.; Kraus, M.; Fenoglio-Preiser, C. Trisomy 3 in gastric lymphomas of extranodal marginal zone B-cell (mucosa-associated lymphoid tissue) origin demonstrated by FISH in intact paraffin tissue sections. Hum. Pathol. 1999, 30, 706–711. [Google Scholar] [CrossRef]
- Zhang, Y.; Cheung, A.N.; Chan, A.C.; Shen, D.H.; Xu, W.S.; Chung, L.P.; Ho, F.C. Detection of trisomy 3 in primary gastric B-cell lymphoma by using chromosome in situ hybridization on paraffin sections. Am. J. Clin. Pathol. 1998, 110, 347–353. [Google Scholar] [CrossRef][Green Version]
- Brynes, R.K.; Almaguer, P.D.; Leathery, K.E.; McCourty, A.; Arber, D.A.; Medeiros, L.J.; Nathwani, B.N. Numerical cytogenetic abnormalities of chromosomes 3, 7, and 12 in marginal zone B-cell lymphomas. Mod. Pathol. 1996, 9, 995–1000. [Google Scholar] [PubMed]
- Krugmann, J.; Tzankov, A.; Dirnhofer, S.; Fend, F.; Wolf, D.; Siebert, R.; Probst, P.; Erdel, M. Complete or partial trisomy 3 in gastro-intestinal MALT lymphomas co-occurs with aberrations at 18q21 and correlates with advanced disease stage: A study on 25 cases. World J. Gastroenterol. 2005, 11, 7384. [Google Scholar] [CrossRef] [PubMed]
- Dierlamm, J.; Rosenberg, C.; Stul, M.; Pittaluga, S.; Wlodarska, I.; Michaux, L.; Dehaen, M.; Verhoef, G.; Thomas, J.; De Kelver, W.; et al. Characteristic pattern of chromosomal gains and losses in marginal zone B cell lymphoma detected by comparative genomic hybridization. Leukemia 1997, 11, 747–758. [Google Scholar] [CrossRef][Green Version]
- Rinaldi, A.; Mian, M.; Chigrinova, E.; Arcaini, L.; Bhagat, G.; Novak, U.; Rancoita, P.M.V.; de Campos, C.P.; Forconi, F.; Gascoyne, R.D.; et al. Genome-wide DNA profiling of marginal zone lymphomas identifies subtype-specific lesions with an impact on the clinical outcome. Blood 2011, 117, 1595–1604. [Google Scholar] [CrossRef]
- Dierlamm, J.; Wlodarska, I.; Michaux, L.; Stefanova, M.; Hinz, K.; Van Den Berghe, H.; Hagemeijer, A.; Hossfeld, D.K. Genetic abnormalities in marginal zone b-cell lymphoma. Hematol. Oncol. 2000, 18, 1–13. [Google Scholar] [CrossRef]
- Deutsch, A.J.A.; Aigelsreiter, A.; Steinbauer, E.; Fruhwirth, M.; Kerl, H.; Beham-Schmid, C.; Schaider, H.; Neumeister, P. Distinct signatures of B-cell homeostatic and activation-dependent chemokine receptors in the development and progression of extragastric MALT lymphomas. J. Pathol. 2008, 215, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Cascione, L.; Rinaldi, A.; Bruscaggin, A.; Tarantelli, C.; Arribas, A.J.; Kwee, I.; Pecciarini, L.; Mensah, A.A.; Spina, V.; Chung, E.Y.L.; et al. Novel insights into the genetics and epigenetics of MALT lymphoma unveiled by next generation sequencing analyses. Haematologica 2019, 104, E558–E561. [Google Scholar] [CrossRef]
- Remstein, E.D.; Dogan, A.; Einerson, R.R.; Paternoster, S.F.; Fink, S.R.; Law, M.; Gordon, W.; Kurtin, P.J. The incidence and anatomic site specificity of chromosomal translocations in primary extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT lymphoma) in North America. Am. J. Surg. Pathol. 2006, 30, 1546–1553. [Google Scholar] [CrossRef] [PubMed]
- Joao, C.; Farinha, P.; Da Silva, M.G.; Martins, C.; Crespo, M.; Cabecadas, J. Cytogenetic abnormalities in MALT lymphomas and their precursor lesions from different organs. A fluorescence in situ hybridization (FISH) study. Histopathology 2007, 50, 217–224. [Google Scholar] [CrossRef]
- Dierlamm, J.; Pittaluga, S.; Wlodarska, I.; Stul, M.; Thomas, J.; Boogaerts, M.; Michaux, L.; Driessen, A.; Mecucci, C.; Cassiman, J. Marginal Zone B-Cell Lymphomas of Different Sites Share Similar Cytogenetic and Morphologic Features. Blood 1996, 87, 299–307. [Google Scholar] [CrossRef]
- Du, M.; Peng, H.; Singh, N.; Isaacson, P.G.; Pan, L. The Accumulation of p53 Abnormalities Is Associated With Progression of Mucosa-Associated Lymphoid Tissue Lymphoma. Blood 1995, 86, 4587–4593. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Chen, G.; Du, M.; Singh, N.; Isaacson, P.G.; Pan, L. Replication error phenotype and p53 gene mutation in lymphomas of mucosa-associated lymphoid tissue. Am. J. Pathol. 1996, 148, 643. [Google Scholar]
- Rødahl, E.; Lybæk, H.; Arnes, J.; Ness, G.O. Chromosomal imbalances in some benign orbital tumours. Acta Ophthalmol. Scand. 2005, 83, 385–391. [Google Scholar] [CrossRef]
- Matteucci, C.; Galieni, P.; Leoncini, L.; Lazzi, S.; Lauria, F.; Polito, E.; Martelli, M.; Mecucci, C. Typical genomic imbalances in primary MALT lymphoma of the orbit. J. Pathol. 2003, 200, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Chanudet, E.; Ye, H.; Ferry, J.; Bacon, C.; Adam, P.; Müller-Hermelink, H.; Radford, J.; Pileri, S.; Ichimura, K.; Collins, V.; et al. A20 deletion is associated with copy number gain at the TNFA/B/C locus and occurs preferentially in translocation-negative MALT lymphoma of the ocular adnexa and salivary glands. J. Pathol. 2009, 217, 420–430. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.S.; Honma, K.; Karnan, S.; Tagawa, H.; Kim, Y.D.; Oh, Y.L.; Seto, M.; Ko, Y.H. Genome-wide array-based comparative genomic hybridization of ocular marginal zone B cell lymphoma: Comparison with pulmonary and nodal marginal zone B cell lymphoma. Genes Chromosomes Cancer 2007, 46, 776–783. [Google Scholar] [CrossRef]
- Honma, K.; Tsuzuki, S.; Nakagawa, M.; Karnan, S.; Aizawa, Y.; Kim, W.S.; Kim, Y.-D.; Ko, Y.-H.; Seto, M. TNFAIP3 is the target gene of chromosome band 6q23.3-q24.1 loss in ocular adnexal marginal zone B cell lymphoma. Genes Chromosomes Cancer 2008, 47, 1–7. [Google Scholar] [CrossRef]
- Honma, K.; Tsuzuki, S.; Nakagawa, M.; Tagawa, H.; Nakamura, S.; Morishima, Y.; Seto, M. TNFAIP3/A20 functions as a novel tumor suppressor gene in several subtypes of non-Hodgkin lymphomas. Blood 2009, 114, 2467–2475. [Google Scholar] [CrossRef]
- Chanudet, E.; Huang, Y.; Ichimura, K.; Dong, G.; Hamoudi, R.A.; Radford, J.; Wotherspoon, A.C.; Isaacson, P.G.; Ferry, J.; Du, M.-Q. A20 is targeted by promoter methylation, deletion and inactivating mutation in MALT lymphoma. Leukemia 2010, 24, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Catrysse, L.; Vereecke, L.; Beyaert, R.; van Loo, G. A20 in inflammation and autoimmunity. Trends Immunol. 2014, 35, 22–31. [Google Scholar] [CrossRef]
- Moody, S.; Thompson, J.S.; Chuang, S.S.; Liu, H.; Raderer, M.; Vassiliou, G.; Wlodarska, I.; Wu, F.; Cogliatti, S.; Robson, A.; et al. Novel GPR34 and CCR6 mutation and distinct genetic profiles in MALT lymphomas of different sites. Haematologica 2018, 103, 1329–1336. [Google Scholar] [CrossRef]
- Johansson, P.; Klein-Hitpass, L.; Grabellus, F.; Arnold, G.; Klapper, W.; Pförtner, R.; Dührsen, U.; Eckstein, A.; Dürig, J.; Küppers, R. Recurrent mutations in NF-κB pathway components, KMT2D, and NOTCH1/2 in ocular adnexal MALT-type marginal zone lymphomas. Oncotarget 2016, 7, 62627–62639. [Google Scholar] [CrossRef]
- Jung, H.; Yoo, H.Y.; Lee, S.H.; Shin, S.; Kim, S.C.; Lee, S.; Joung, J.-G.; Nam, J.-Y.; Ryu, D.; Yun, J.W.; et al. The mutational landscape of ocular marginal zone lymphoma identifies frequent alterations in TNFAIP3 followed by mutations in TBL1XR1 and CREBBP. Oncotarget 2017, 8, 17038–17049. [Google Scholar] [CrossRef]
- Cani, A.K.; Soliman, M.; Hovelson, D.H.; Liu, C.-J.; McDaniel, A.S.; Haller, M.J.; Bratley, J.; Rahrig, S.; Li, Q.; Briceño, C.A.; et al. Comprehensive Genomic Profiling of Orbital and Ocular Adnexal Lymphomas Identifies Frequent Alterations in MYD88 and Chromatin Modifiers: New Routes to Targeted Therapies. Mod. Pathol. 2016, 29, 685. [Google Scholar] [CrossRef]
- Behdad, A.; Zhou, X.Y.; Gao, J.; Raparia, K.; Dittman, D.; Green, S.J.; Qi, C.; Betz, B.; Bryar, P.; Chen, Q.; et al. High Frequency of MYD88 L265P Mutation in Primary Ocular Adnexal Marginal Zone Lymphoma and Its Clinicopathologic Correlation: A Study From a Single Institution. Arch. Pathol. Lab. Med. 2019, 143, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.M.; Rinaldi, A.; Cavalli, A.; Mensah, A.A.; Ponzoni, M.; Gascoyne, R.D.; Bhagat, G.; Zucca, E.; Bertoni, F. MYD88 somatic mutations in MALT lymphomas. Br. J. Haematol. 2012, 158, 662–664. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Wang, M.; Moody, S.; Xue, X.; Huang, Y.; Bi, Y.; Du, M.Q. Distinct involvement of NF-κB regulators by somatic mutation in ocular adnexal malt lymphoma. Br. J. Haematol. 2013, 160, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Ngo, V.N.; Young, R.M.; Schmitz, R.; Jhavar, S.; Xiao, W.; Lim, K.H.; Kohlhammer, H.; Xu, W.; Yang, Y.; Zhao, H.; et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011, 470, 115–121. [Google Scholar] [CrossRef]
- Bi, Y.; Zeng, N.; Chanudet, E.; Huang, Y.; Hamoudi, R.A.; Liu, H.; Dong, G.; Watkins, A.J.; Ley, S.C.; Zou, L.; et al. A20 inactivation in ocular adnexal MALT lymphoma. Haematologica 2012, 97, 926–930. [Google Scholar] [CrossRef]
- Vela, V.; Juskevicius, D.; Gerlach, M.M.; Meyer, P.; Graber, A.; Cathomas, G.; Dirnhofer, S.; Tzankov, A. High throughput sequencing reveals high specificity of TNFAIP3 mutations in ocular adnexal marginal zone B-cell lymphomas. Hematol. Oncol. 2020, 38, 284–292. [Google Scholar] [CrossRef] [PubMed]
- Ganapathi, K.A.; Jobanputra, V.; Iwamoto, F.; Jain, P.; Chen, J.; Cascione, L.; Nahum, O.; Levy, B.; Xie, Y.; Khattar, P.; et al. The genetic landscape of dural marginal zone lymphomas. Oncotarget 2016, 7, 43052. [Google Scholar] [CrossRef]
- Agathangelidis, A.; Xochelli, A.; Stamatopoulos, K. A gene is known by the company it keeps: Enrichment of TNFAIP3 gene aberrations in MALT lymphomas expressing IGHV4-34 antigen receptors. J. Pathol. 2017, 243, 403–406. [Google Scholar] [CrossRef]
- Hayden, M.S.; Ghosh, S. Regulation of NF-κB by TNF family cytokines. Semin. Immunol. 2014, 26, 253–266. [Google Scholar] [CrossRef]
- Vereecke, L.; Beyaert, R.; van Loo, G. The ubiquitin-editing enzyme A20 (TNFAIP3) is a central regulator of immunopathology. Trends Immunol. 2009, 30, 383–391. [Google Scholar] [CrossRef]
- Boone, D.L.; Turer, E.E.; Lee, E.G.; Ahmad, R.C.; Wheeler, M.T.; Tsui, C.; Hurley, P.; Chien, M.; Chai, S.; Hitotsumatsu, O.; et al. The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat. Immunol. 2004, 5, 1052–1060. [Google Scholar] [CrossRef]
- Thome, M.; Tschopp, J. TCR-induced NF-kappaB activation: A crucial role for Carma1, Bcl10 and MALT1. Trends Immunol. 2003, 24, 419–424. [Google Scholar] [CrossRef]
- Shi, J.H.; Sun, S.C. Tumor Necrosis Factor Receptor-Associated Factor Regulation of Nuclear Factor κB and Mitogen-Activated Protein Kinase Pathways. Front. Immunol. 2018, 9, 1849. [Google Scholar] [CrossRef] [PubMed]
- Hyeon, J.; Lee, B.; Shin, S.H.; Yoo, H.Y.; Kim, S.J.; Kim, W.S.; Park, W.-Y.; Ko, Y.-H. Targeted deep sequencing of gastric marginal zone lymphoma identified alterations of TRAF3 and TNFAIP3 that were mutually exclusive for MALT1 rearrangement. Mod. Pathol. 2018, 31, 1418–1428. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.; Cariappa, A.; Moran, S.T. Marginal Zone B Cells. Annu. Rev. Immunol. 2005, 23, 161–196. [Google Scholar] [CrossRef]
- Pillai, S.; Cariappa, A. The follicular versus marginal zone B lymphocyte cell fate decision. Nat. Rev. Immunol. 2009, 9, 767–777. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Chiba, S.; Ichikawa, M.; Kunisato, A.; Asai, T.; Shimizu, K.; Yamaguchi, T.; Yamamoto, G.; Seo, S.; Kumano, K.; et al. Notch2 Is Preferentially Expressed in Mature B Cells and Indispensable for Marginal Zone B Lineage Development. Immunity 2003, 18, 675–685. [Google Scholar] [CrossRef]
- Hampel, F.; Ehrenberg, S.; Hojer, C.; Draeseke, A.; Marschall-Schröter, G.; Kühn, R.; Mack, B.; Gires, O.; Vahl, C.J.; Schmidt-Supprian, M.; et al. CD19-independent instruction of murine marginal zone B-cell development by constitutive Notch2 signaling. Blood 2011, 118, 6321–6331. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, Y.; Weng, Y.; Lai, Q.; Luo, T.; Zhao, J.; Ren, G.; Li, W.; Pan, H.; Ke, Y.; et al. The Truncate Mutation of Notch2 Enhances Cell Proliferation through Activating the NF-κB Signal Pathway in the Diffuse Large B-Cell Lymphomas. PLoS ONE 2014, 9, e108747. [Google Scholar] [CrossRef][Green Version]
- Puente, X.S.; Pinyol, M.; Quesada, V.; Conde, L.; Ordonez, G.R.; Villamor, N.; Escaramis, G.; Jares, P.; Bea, S.; Gonzalez-Diaz, M.; et al. Whole-genome sequencing identifies recurrent mutations in chronic lymphocytic leukaemia. Nature 2011, 475, 101–105. [Google Scholar] [CrossRef]
- Arcaini, L.; Rossi, D.; Lucioni, M.; Nicola, M.; Bruscaggin, A.; Fiaccadori, V.; Riboni, R.; Ramponi, A.; Ferretti, V.V.; Cresta, S.; et al. The NOTCH pathway is recurrently mutated in diffuse large B-cell lymphoma associated with hepatitis C virus infection. Haematologica 2015, 100, 246–252. [Google Scholar] [CrossRef]
- Rossi, D.; Trifonov, V.; Fangazio, M.; Bruscaggin, A.; Rasi, S.; Spina, V.; Monti, S.; Vaisitti, T.; Arruga, F.; Famà, R.; et al. The coding genome of splenic marginal zone lymphoma: Activation of NOTCH2 and other pathways regulating marginal zone development. J. Exp. Med. 2012, 209, 1537–1551. [Google Scholar] [CrossRef]
- Kiel, M.J.; Velusamy, T.; Betz, B.L.; Zhao, L.; Weigelin, H.G.; Chiang, M.Y.; Huebner-Chan, D.R.; Bailey, N.G.; Yang, D.T.; Bhagat, G.; et al. Whole-genome sequencing identifies recurrent somatic NOTCH2 mutations in splenic marginal zone lymphoma. J. Exp. Med. 2012, 209, 1553–1565. [Google Scholar] [CrossRef]
- Karube, K.; Martínez, D.; Royo, C.; Navarro, A.; Pinyol, M.; Cazorla, M.; Castillo, P.; Valera, A.; Carrió, A.; Costa, D.; et al. Recurrent mutations of NOTCH genes in follicular lymphoma identify a distinctive subset of tumours. J. Pathol. 2014, 234, 423–430. [Google Scholar] [CrossRef]
- Kuksin, C.A.; Minter, L.M. The Link between Autoimmunity and Lymphoma: Does NOTCH Signaling Play a Contributing Role? Front. Oncol. 2015, 5, 51. [Google Scholar] [CrossRef] [PubMed]
- Johansson, P.; Klein-Hitpass, L.; Budeus, B.; Kuhn, M.; Lauber, C.; Seifert, M.; Roeder, I.; Pförtner, R.; Stuschke, M.; Dührsen, U.; et al. Identifying Genetic Lesions in Ocular Adnexal Extranodal Marginal Zone Lymphomas of the MALT Subtype by Whole Genome, Whole Exome and Targeted Sequencing. Cancers 2020, 12, 986. [Google Scholar] [CrossRef] [PubMed]
- Kiesewetter, B.; Copie-Bergman, C.; Levy, M.; Wu, F.; Dupuis, J.; Barau, C.; Arcaini, L.; Paulli, M.; Lucioni, M.; Bonometti, A.; et al. Genetic Characterization and Clinical Features of Helicobacter pylori Negative Gastric Mucosa-Associated Lymphoid Tissue Lymphoma. Cancers 2021, 13, 2993. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wei, R.L.; Pi, Y.L.; Guo, Q. Significance of Bcl10 gene mutations in the clinical diagnosis of MALT-type ocular adnexal lymphoma in the Chinese population. Genet. Mol. Res. 2013, 12, 1194–1204. [Google Scholar] [CrossRef] [PubMed]
- Shingleton, J.R.; Dave, S.S. TET2 Deficiency Sets the Stage for B-cell Lymphoma. Cancer Discov. 2018, 8, 1515–1517. [Google Scholar] [CrossRef] [PubMed]
- Du, M.Q. MALT lymphoma: A paradigm of NF-κB dysregulation. Semin. Cancer Biol. 2016, 39, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.; Liu, T.; Zhang, H.; Zhou, X.; Jin, H.; Sun, M.; Yun, Z.; Luo, H.; Ni, Z.; Zhao, R.; et al. Whole-Exome Sequencing Reveals New Potential Mutations Genes for Primary Mucosa-Associated Lymphoid Tissue Lymphoma Arising From the Kidney. Front. Oncol. 2021, 10, 609839. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Mercado, M.; Manterola, L.; Lawrie, C.H. MicroRNAs in Lymphoma: Regulatory Role and Biomarker Potential. Curr. Genomics 2015, 16, 349. [Google Scholar] [CrossRef] [PubMed]
- Di Lisio, L.; Martinez, N.; Montes-Moreno, S.; Piris-Villaespesa, M.; Sanchez-Beato, M.; Piris, M.A. The role of miRNAs in the pathogenesis and diagnosis of B-cell lymphomas. Blood 2012, 120, 1782–1790. [Google Scholar] [CrossRef] [PubMed]
- Lawrie, C.H. MicroRNAs and lymphomagenesis: A functional review. Br. J. Haematol. 2013, 160, 571–581. [Google Scholar] [CrossRef]
- Craig, V.J.; Cogliatti, S.B.; Rehrauer, H.; Wündisch, T.; Müller, A. Epigenetic Silencing of MicroRNA-203 Dysregulates ABL1 Expression and Drives Helicobacter-Associated Gastric Lymphomagenesis. Cancer Res. 2011, 71, 3616–3624. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, A.; Quan, L.; Qu, Y.; Gu, A. Three novel microRNAs based on microRNA signatures for gastric mucosa-associated lymphoid tissue lymphoma. Neoplasma 2018, 65, 339–348. [Google Scholar] [CrossRef]
- Saito, Y.; Suzuki, H.; Tsugawa, H.; Imaeda, H.; Matsuzaki, J.; Hirata, K.; Hosoe, N.; Nakamura, M.; Mukai, M.; Saito, H.; et al. Overexpression of miR-142-5p and miR-155 in Gastric Mucosa-Associated Lymphoid Tissue (MALT) Lymphoma Resistant to Helicobacter pylori Eradication. PLoS ONE 2012, 7, e47396. [Google Scholar] [CrossRef]
- Krützfeldt, J.; Rajewsky, N.; Braich, R.; Rajeev, K.G.; Tuschl, T.; Manoharan, M.; Stoffel, M. Silencing of microRNAs in vivo with “antagomirs”. Nature 2005, 438, 685–689. [Google Scholar] [CrossRef]
- Babar, I.A.; Cheng, C.J.; Booth, C.J.; Liang, X.; Weidhaas, J.B.; Saltzman, W.M.; Slack, F.J. Nanoparticle-based therapy in an in vivo microRNA-155 (miR-155)-dependent mouse model of lymphoma. Proc. Natl. Acad. Sci. USA 2012, 109, E1695–E1704. [Google Scholar] [CrossRef]
- Fernández, C.; Bellosillo, B.; Ferraro, M.; Seoane, A.; Sánchez-González, B.; Pairet, S.; Pons, A.; Barranco, L.; Vela, M.C.; Gimeno, E.; et al. MicroRNAs 142-3p, miR-155 and miR-203 are deregulated in gastric MALT lymphomas compared to chronic gastritis. Cancer Genom. Proteom. 2017, 14, 75–82. [Google Scholar] [CrossRef][Green Version]
- Cai, J.; Liu, X.; Cheng, J.; Li, Y.; Huang, X.; Li, Y.; Ma, X.; Yu, H.; Liu, H.; Wei, R. MicroRNA-200 is commonly repressed in conjunctival MALT lymphoma, and targets cyclin E2. Graefes Arch. Clin. Exp. Ophthalmol. 2012, 250, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Park, S.M.; Gaur, A.B.; Lengyel, E.; Peter, M.E. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008, 22, 894–907. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Chng, W.J.; Remstein, E.D.; Fonseca, R.; Bergsagel, P.L.; Vrana, J.A.; Kurtin, P.J.; Dogan, A. Gene expression profiling of pulmonary mucosa-associated lymphoid tissue lymphoma identifies new biologic insights with potential diagnostic and therapeutic applications. Blood 2009, 113, 635–645. [Google Scholar] [CrossRef]
- Hamoudi, R.A.; Appert, A.; Ye, H.; Ruskone-Fourmestraux, A.; Streubel, B.; Chott, A.; Raderer, M.; Gong, L.; Wlodarska, I.; De Wolf-Peeters, C. Differential expression of NF-kappaB target genes in MALT lymphoma with and without chromosome translocation: Insights into molecular mechanism. Leukemia 2010, 24, 1487–1497. [Google Scholar] [CrossRef]
- Zou, Q.; Zhang, H.; Meng, F.; He, L.; Zhang, J.; Xiao, D. Proteomic and transcriptomic studies of BGC823 cells stimulated with Helicobacter pylori isolates from gastric MALT lymphoma. PLoS ONE 2020, 15, e0238379. [Google Scholar] [CrossRef]
- Zhang, J.; Wei, J.; Wang, Z.; Feng, Y.; Wei, Z.; Hou, X.; Xu, J.; He, Y.; Yang, D. Transcriptome hallmarks in Helicobacter pylori infection influence gastric cancer and MALT lymphoma. Epigenomics 2020, 12, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.B.; Jeffery, P.; Gahete, M.D.; Whiteside, E.; Walpole, C.; Maugham, M.; Jovanovic, L.; Gunter, J.; Williams, E.; Nelson, C.; et al. The long non-coding RNA GHSROS reprograms prostate cancer cell lines toward a more aggressive phenotype. PeerJ 2021, 9, e10280. [Google Scholar] [CrossRef]
- Lue, J.K.; Amengual, J.E.; O’Connor, O.A. Epigenetics and Lymphoma: Can We Use Epigenetics to Prime or Reset Chemoresistant Lymphoma Programs? Curr. Oncol. Rep. 2015, 17, 40. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Green, M.R. Harnessing lymphoma epigenetics to improve therapies. Blood 2020, 136, 2386–2391. [Google Scholar] [CrossRef] [PubMed]
- Mondello, P.; Tadros, S.; Teater, M.; Fontan, L.; Chang, A.Y.; Jain, N.; Yang, H.; Singh, S.; Ying, H.-Y.; Chu, C.-S. Selective inhibition of HDAC3 targets synthetic vulnerabilities and activates immune surveillance in lymphoma. AACR 2020, 10, 440–459. [Google Scholar] [CrossRef] [PubMed]
- Fung, M.K.L.; Au, W.Y.; Liang, R.; Srivastava, G.; Kwong, Y.L. Aberrant promoter methylation in gastric lymphomas. Haematologica 2003, 88, 231–232. [Google Scholar] [CrossRef]
- Rossi, D.; Capello, D.; Gloghini, A.; Franceschetti, S.; Paulli, M.; Bhatia, K.; Saglio, G.; Vitolo, U.; Pileri, S.A.; Esteller, M.; et al. Aberrant promoter methylation of multiple genes throughout the clinico-pathologic spectrum of B-cell neoplasia. Haematologica 2004, 89, 154–164. [Google Scholar]
- Takino, H.; Okabe, M.; Li, C.; Ohshima, K.; Yoshino, T.; Nakamura, S.; Ueda, R.; Eimoto, T.; Inagaki, H. p16/INK4a gene methylation is a frequent finding in pulmonary MALT lymphomas at diagnosis. Mod. Pathol. 2005, 18, 1187–1192. [Google Scholar] [CrossRef][Green Version]
- Dugge, R.; Wagener, R.; Möller, P.; Barth, T.F.E. Genome-wide DNA methylation analysis along the progression of gastric marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue (MALT) type. Br. J. Haematol. 2021, 193, 369–374. [Google Scholar] [CrossRef]
- Deaton, A.M.; Bird, A. CpG islands and the regulation of transcription. Genes Dev. 2011, 25, 1010–1022. [Google Scholar] [CrossRef]
- Davis, A.R.; Stone, S.L.; Oran, A.R.; Sussman, R.T.; Bhattacharyya, S.; Morrissette, J.J.; Bagg, A. Targeted massively parallel sequencing of mature lymphoid neoplasms: Assessment of empirical application and diagnostic utility in routine clinical practice. Mod. Pathol. 2021, 34, 904–921. [Google Scholar] [CrossRef]
- Pillonel, V.; Juskevicius, D.; Bihl, M.; Stenner, F.; Halter, J.P.; Dirnhofer, S.; Tzankov, A. Routine next generation sequencing of lymphoid malignancies: Clinical utility and challenges from a 3-Year practical experience. Leuk. Lymphoma 2020, 61, 2568–2583. [Google Scholar] [CrossRef]
- Morschhauser, F.; Tilly, H.; Chaidos, A.; McKay, P.; Phillips, T.; Assouline, S.; Batlevi, C.L.; Campbell, P.; Ribrag, V.; Damaj, G.L.; et al. Tazemetostat for patients with relapsed or refractory follicular lymphoma: An open-label, single-arm, multicentre, phase 2 trial. Lancet. Oncol. 2020, 21, 1433–1442. [Google Scholar] [CrossRef]
| Location | Antigen Exposure Association | IGHV Usage | Abnormality | Involved Genes | Copy Number Variations | Other Imbalances |
|---|---|---|---|---|---|---|
| GASTRIC | Helicobacter pylori Helicobacter heilmannii Campylobacter jejuni (small intestine) | IGHV4-34 IGHV3-7 IGHV1-69 IGHV1-2 IGHV3-23 | t(11;18)(q21;q21) 20–25% (intestinal 33%) t(1;14)(p22;q32) 4% | BIRC3-MALT1 IGHV-BCL10 | Trisomy 3 Trisomy 18 | TNFAIP3 deletion |
| OCULARADNEXA | Chlamydia psittaci | IGHV4-34: 18% IGHV3-23: 12–17% IGHV3-30: 10–14% IGHV3-7: 9% | t(11;18)(q21;q21) 10% t(14;18)(q32;q21) 7% t(3;14)(p14.1;q32) | BIRC3-MALT1 IGHV-MALT1 IGHV-FOXP1 | Trisomy 18 6q gain 30% 3q gain 18q gain | TNFAIP3 deletion 19% |
| THRYOID | Hashimoto thyroiditis | IGHV3-30 | t(3;14)(p14.1;q32) 7–56% t(14;18)(q32;q21) | IGHV-FOXP1 IGHV-MALT1 | Trisomy 3 | TNFAIP3 deletion 11% PD-L1 deletion 53% |
| SALIVAL GLAND | Lymphoepithelial sialadenitis Sjögren syndrome | IGHV1-69/J4: 55% IGHV3-7/J3 15% IGHV4-59/J2(J5) IGHV3-30/JH4 | t(X;14)(p11.4;q32) | IGHV-GPR34 | TNFAIP3 deletion 8% | |
| SKIN | Borrelia burgdorferi | IGHV1-69 IGHV4-59 IGHV3-30 | t(14;18)(q32;q21) 10% t(3;14)(p14.1;q32), | IGHV-MALT1 | ||
| LUNG | Achromobacter xylosoxidans | IGHV3 IGHV4-34 | t(11;18)(q21;q21) 40% t(11;12;18)(q21;q13;q21) t(11;14;18)(q21;q32;q21) t(1;14)(p22;q32) 9% t(14;18)(q32;q21) 6–9% | BIRC3-MALT1 IGHV-BCL10 IGHV-MALT1 | 3q gain 18q gain |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Sevilla, J.J.; Salar, A. Recent Advances in the Genetic of MALT Lymphomas. Cancers 2022, 14, 176. https://doi.org/10.3390/cancers14010176
Rodríguez-Sevilla JJ, Salar A. Recent Advances in the Genetic of MALT Lymphomas. Cancers. 2022; 14(1):176. https://doi.org/10.3390/cancers14010176
Chicago/Turabian StyleRodríguez-Sevilla, Juan José, and Antonio Salar. 2022. "Recent Advances in the Genetic of MALT Lymphomas" Cancers 14, no. 1: 176. https://doi.org/10.3390/cancers14010176
APA StyleRodríguez-Sevilla, J. J., & Salar, A. (2022). Recent Advances in the Genetic of MALT Lymphomas. Cancers, 14(1), 176. https://doi.org/10.3390/cancers14010176






