Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy
Abstract
Simple Summary
Abstract
1. Introduction
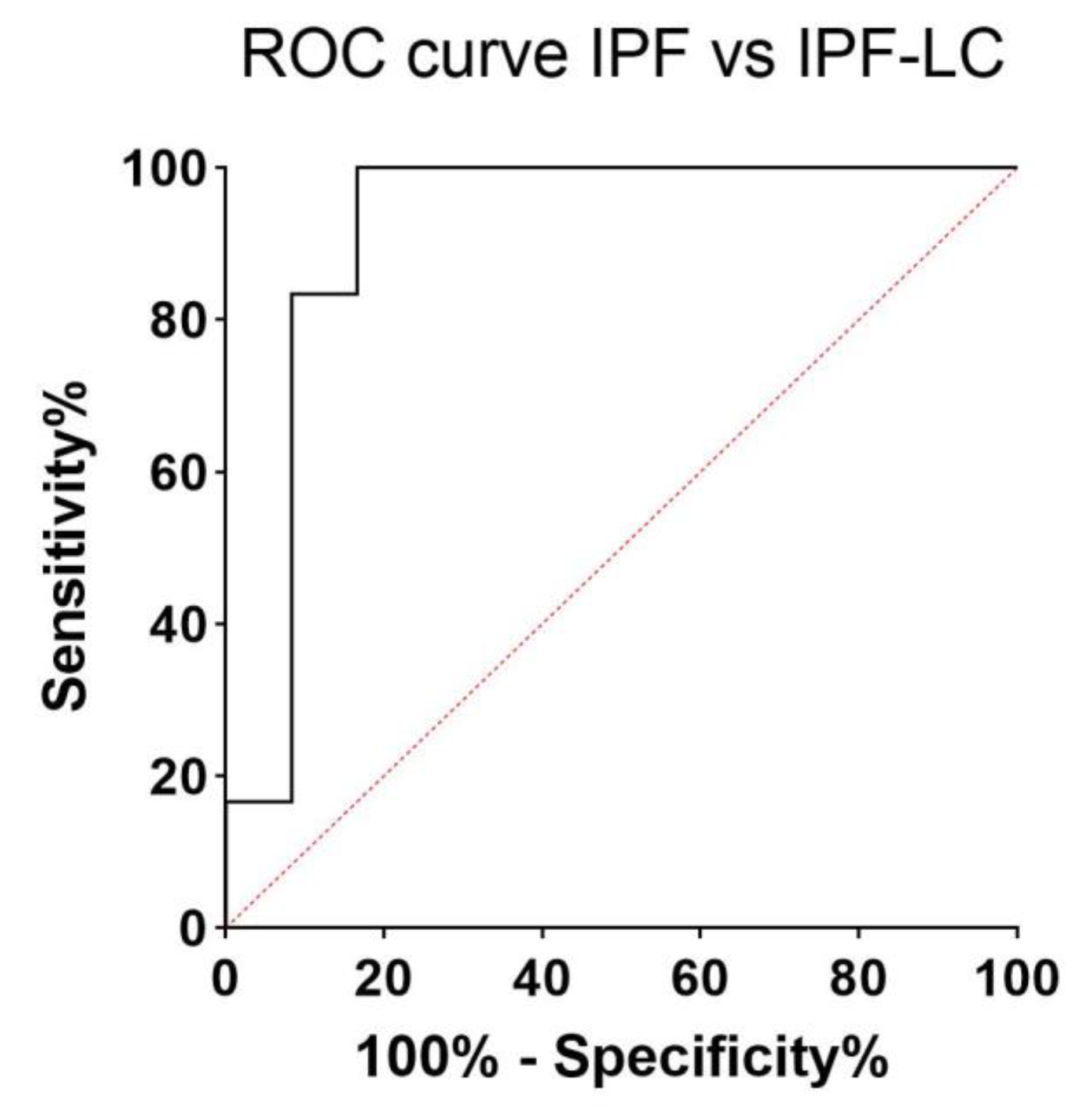
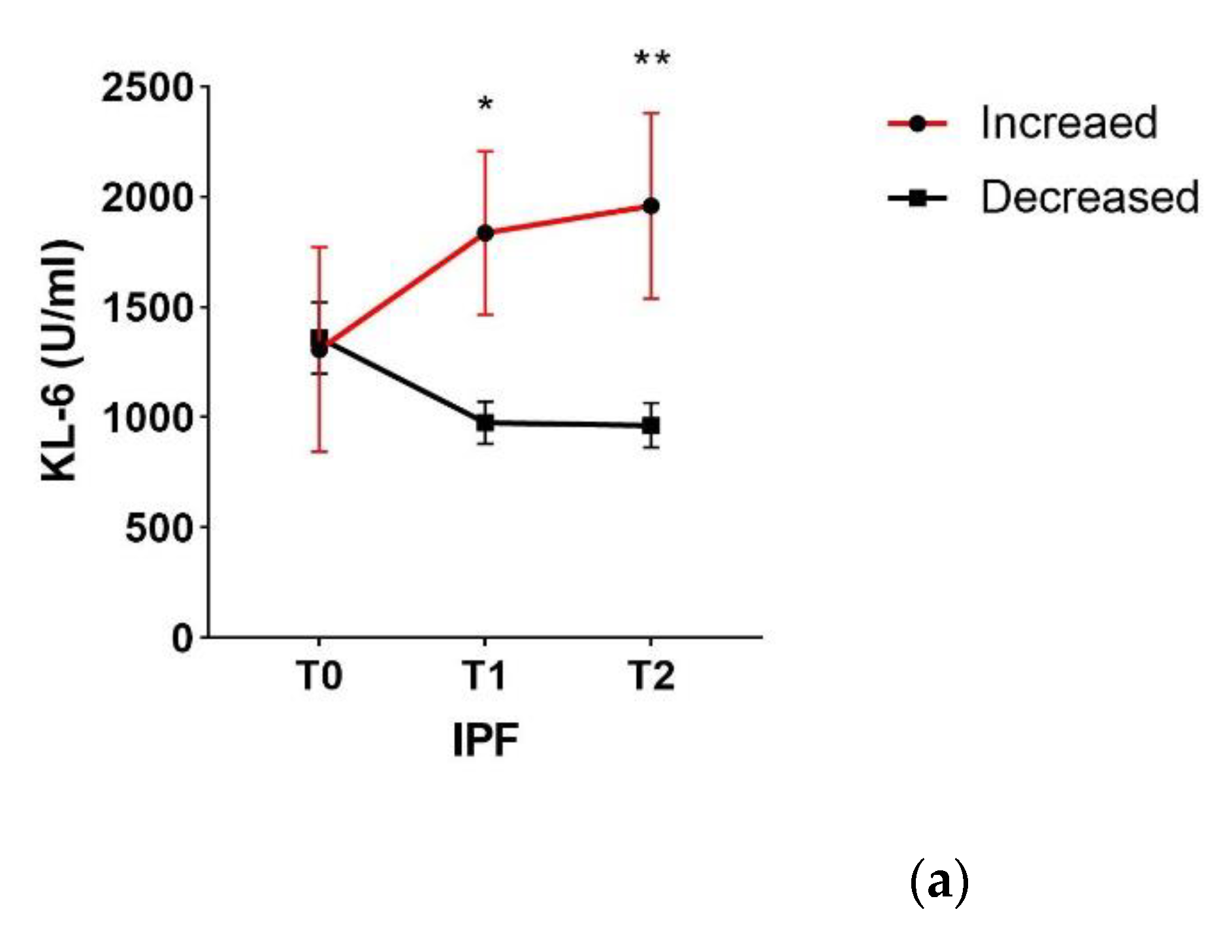
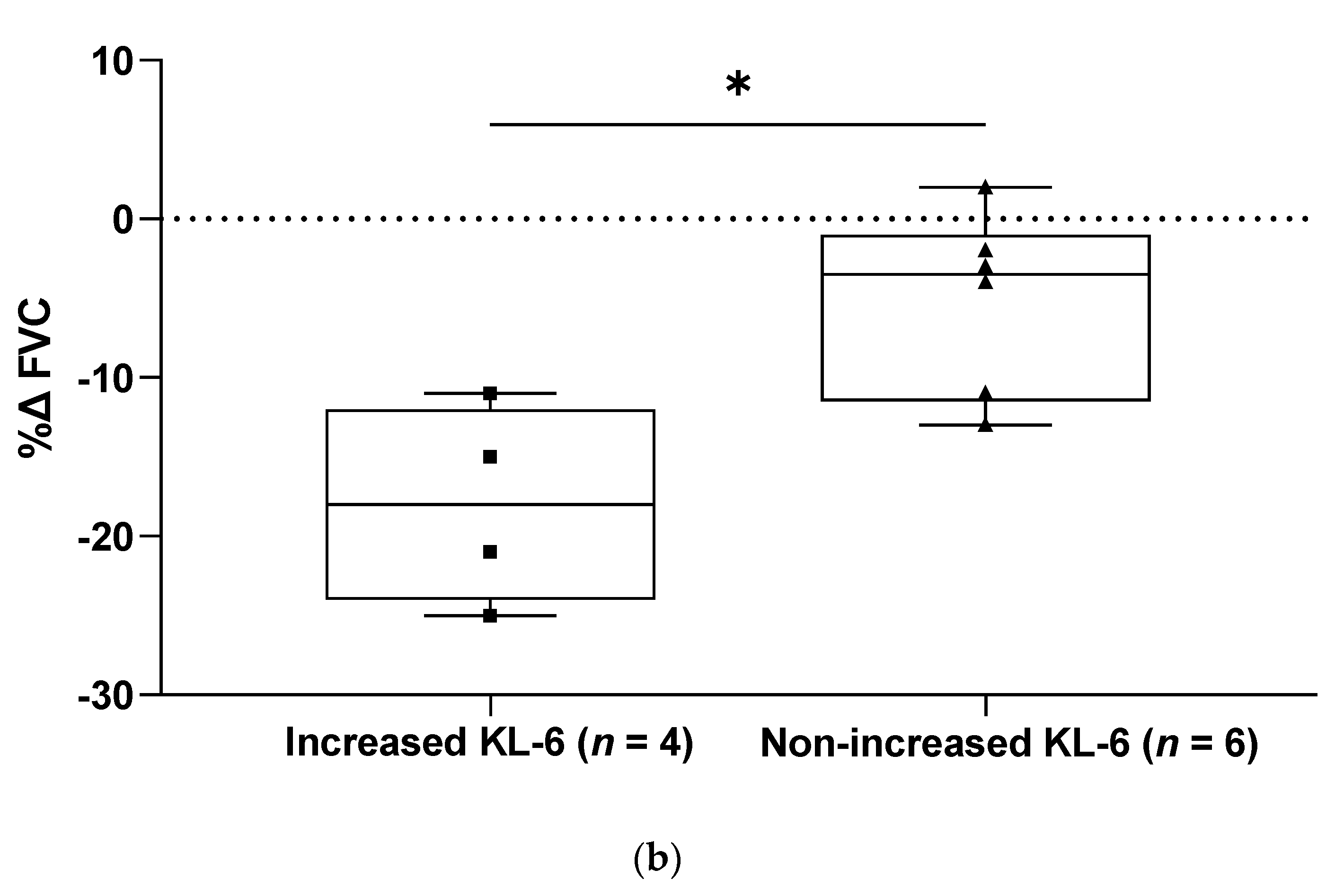
2. Results
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Krebs von den Lungen-6 Assay
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ILD | interstitial lung disease |
| IPF | idiopathic pulmonary fibrosis |
| IPF-LC | IPF associated with lung cancer |
| HRCT | High resolution computed tomography |
| KL-6 | Krebs von den Lungen-6 |
| fHP | fibrotic hypersensitivity pneumonitis |
| PF-AD | pulmonary fibrosis associated with autoimmune diseases |
| ROC | receiver operating characteristic |
| AUC | area under the curve |
References
- Kohno, N.; Kyoizumi, S.; Awaya, Y.; Fukuhara, H.; Yamakido, M.; Akiyama, M. New serum indicator of interstitial pneu-monitis activity. Sialylated carbohydrate antigen KL-6. Chest 1989, 96, 68–73. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Vietri, L.; Lanzarone, N.; Alonzi, V.; Pieroni, M.; Refini, R.M.; Sestini, P.; Bonella, F.; et al. Krebs von den Lungen-6 as a biomarker for disease severity assessment in interstitial lung disease: A comprehensive review. Biomark. Med. 2020, 14, 665–674. [Google Scholar] [CrossRef]
- Kamiya, K.; Watanabe, M.; Kohno, M.; Izumi, Y.; Horinouchi, H.; Kawamura, M.; Shimada, N.; Nomori, H. KL-6 and CEA levels in epithelial lining fluid microsamples predict response to gefitinib in patients with advanced non-small cell lung cancer. Respirology 2011, 16, 976–982. [Google Scholar] [CrossRef] [PubMed]
- Bergantini, L.; Bargagli, E.; Cameli, P.; Cekorja, B.; Lanzarone, N.; Pianigiani, L.; Vietri, L.; Bennett, D.; Sestini, P.; Rottoli, P. Serial KL-6 analysis in patients with idiopathic pulmonary fibrosis treated with nintedanib. Respir. Investig. 2019, 57, 290–291. [Google Scholar] [CrossRef]
- Wakamatsu, K.; Nagata, N.; Kumazoe, H.; Oda, K.; Ishimoto, H.; Yoshimi, M.; Takata, S.; Hamada, M.; Koreeda, Y.; Takakura, K.; et al. Prognostic value of serial serum KL-6 measurements in patients with idiopathic pulmonary fibrosis. Respir. Investig. 2017, 55, 16–23. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Carleo, A.; Cameli, P.; Bergantini, L.; Perrone, A.; Vietri, L.; Lanzarone, N.; Vagaggini, C.; Sestini, P.; Bargagli, E. BAL biomarkers’ panel for differential diagnosis of interstitial lung diseases. Clin. Exp. Med. 2020, 20, 207–216. [Google Scholar] [CrossRef]
- Ohnishi, H.; Yokoyama, A.; Kondo, K.; Hamada, H.; Abe, M.; Nishimura, K.; Hiwada, K.; Kohno, N. Comparative Study of KL-6, Surfactant Protein-A, Surfactant Protein-D, and Monocyte Chemoattractant Protein-1 as Serum Markers for Interstitial Lung Diseases. Am. J. Respir. Crit. Care Med. 2002, 165, 378–381. [Google Scholar] [CrossRef]
- Yokoyama, A.; Kondo, K.; Nakajima, M.; Matsushima, T.; Takahashi, T.; Nishimura, M.; Bando, M.; Sugiyama, Y.; Totani, Y.; Ishizaki, T.; et al. Prognostic value of circulating KL-6 in idiopathic pulmonary fibrosis. Respirology 2006, 11, 164–168. [Google Scholar] [CrossRef]
- Ishikawa, N.; Hattori, N.; Yokoyama, A.; Kohno, N. Utility of KL-6/MUC1 in the clinical management of interstitial lung diseases. Respir. Investig. 2012, 50, 3–13. [Google Scholar] [CrossRef]
- Bargagli, E.; Madioni, C.; Bianchi, N.; Refini, R.M.; Cappelli, R.; Rottoli, P. Serum Analysis of Coagulation Factors in IPF and NSIP. Inflammation 2013, 37, 10–16. [Google Scholar] [CrossRef]
- Vietri, L.; Bennett, D.; Cameli, P.; Bergantini, L.; Cillis, G.; Sestini, P.; Bargagli, E.; Rottoli, P. Serum amyloid A in patients with idiopathic pulmonary fibrosis. Respir. Investig. 2019, 57, 430–434. [Google Scholar] [CrossRef]
- Raghu, G.; Remy-Jardin, M.; Myers, J.L.; Richeldi, L.; Ryerson, C.J.; Lederer, D.J.; Behr, J.; Cottin, V.; Danoff, S.K.; Morell, F.; et al. Diagnosis of Idiopathic Pulmonary Fibrosis. An Official ATS/ERS/JRS/ALAT Clinical Practice Guideline. Am. J. Respir. Crit. Care Med. 2018, 198, e44–e68. [Google Scholar] [CrossRef]
- Landi, C.; Carleo, A.; Vantaggiato, L.; Bergantini, L.; d’Alessandro, M.; Cameli, P.; Bargagli, E. Common molecular pathways targeted by nintedanib in cancer and IPF: A bioinformatic study. Pulm. Pharmacol. Ther. 2020, 64, 101941. [Google Scholar]
- Cameli, P.; Refini, R.M.; Bergantini, L.; D’Alessandro, M.; Alonzi, V.; Magnoni, C.; Rottoli, P.; Sestini, P.; Bargagli, E. Long-Term Follow-Up of Patients With Idiopathic Pulmonary Fibrosis Treated With Pirfenidone or Nintedanib: A Real-Life Comparison Study. Front. Mol. Biosci. 2020, 7, 581828. [Google Scholar] [CrossRef]
- Vietri, L.; Cameli, P.; Perruzza, M.; Cekorja, B.; Bergantini, L.; D’Alessandro, M.; Refini, R.M.; Pieroni, M.; Fossi, A.; Bennett, D.; et al. Pirfenidone in idiopathic pulmonary fibrosis: Real-life experience in the referral centre of Siena. Ther. Adv. Respir. Dis. 2020, 14, 1753466620906326. [Google Scholar] [CrossRef]
- Raghu, G.; Collard, H.R.; Egan, J.J.; Martinez, F.J.; Behr, J.; Brown, K.K.; Colby, T.V.; Cordier, J.-F.; Flaherty, K.R.; Lasky, J.A.; et al. An Official ATS/ERS/JRS/ALAT Statement: Idiopathic Pulmonary Fibrosis: Evidence-based Guidelines for Diagnosis and Management. Am. J. Respir. Crit. Care Med. 2011, 183, 788–824. [Google Scholar] [CrossRef]
- Drakopanagiotakis, F.; Wujak, L.; Wygrecka, M.; Markart, P. Biomarkers in idiopathic pulmonary fibrosis. Matrix Biol. 2018, 68–69, 404–421. [Google Scholar] [CrossRef] [PubMed]
- Bergantini, L.; Cameli, P.; D’Alessandro, M.; Vagaggini, C.; Refini, R.M.; Landi, C.; Pieroni, M.G.; Spalletti, M.; Sestini, P.; Bargagli, E. NK and NKT-like cells in granulomatous and fibrotic lung diseases. Clin. Exp. Med. 2019, 19, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.; Kushima, H.; Kinoshita, Y.; Fujita, M.; Watanabe, K. The serum KL-6 levels in untreated idiopathic pulmonary fibrosis can naturally decline in association with disease progression. Clin. Respir. J. 2018, 12, 2411–2418. [Google Scholar] [CrossRef]
- Satoh, H.; Kurishima, K.; Ishikawa, H.; Ohtsuka, M. Increased levels of KL-6 and subsequent mortality in patients with interstitial lung diseases. J. Intern. Med. 2006, 260, 429–434. [Google Scholar] [CrossRef]
- Travis, W.D.; Brambilla, E.; Nicholson, A.G.; Yatabe, Y.; Austin, J.H.; Beasley, M.B.; Chirieac, L.R.; Dacic, S.; Duhig, E.; Flieder, D.B.; et al. The 2015 World Health Organization Classification of Lung Tumors. J. Thorac. Oncol. 2015, 10, 1243–1260. [Google Scholar] [CrossRef]
- Brambilla, E.; Travis, W.; Colby, T.; Corrin, B.; Shimosato, Y. The new World Health Organization classification of lung tumours. Eur. Respir. J. 2001, 18, 1059–1068. [Google Scholar] [CrossRef]
- Culver, B.H.; Graham, B.L.; Coates, A.L.; Wanger, J.; Berry, C.E.; Clarke, P.K.; Hallstrand, T.S.; Hankinson, J.L.; Kaminsky, D.A.; MacIntyre, N.R.; et al. Recommendations for a Standardized Pulmonary Function Report. An Official American Thoracic Society Technical Statement. Am. J. Respir. Crit. Care Med. 2017, 196, 1463–1472. [Google Scholar] [CrossRef]
- Lanzarone, N.; Gentili, F.; Alonzi, V.; Bergantini, L.; D’Alessandro, M.; Rottoli, P.; Refini, R.M.; Pieroni, M.; Vietri, L.; Bianchi, F.; et al. Bronchoalveolar lavage and serum KL-6 concentrations in chronic hypersensitivity pneumonitis: Correlations with radiological and immunological features. Intern. Emerg. Med. 2020, 15, 1247–1254. [Google Scholar] [CrossRef]
- Bs, M.D.; Cameli, P.; Refini, R.M.; Bs, L.B.; Alonzi, V.; Lanzarone, N.; Bennett, D.; Rana, G.D.; Montagnani, F.; Scolletta, S.; et al. Serum KL-6 concentrations as a novel biomarker of severe COVID-19. J. Med. Virol. 2020, 92, 2216–2220. [Google Scholar] [CrossRef]
- Bergantini, L.; D’Alessandro, M.; Vietri, L.; Rana, G.D.; Cameli, P.; Acerra, S.; Sestini, P.; Bargagli, E. Utility of serological biomarker’ panels for diagnostic accuracy of interstitial lung diseases. Immunol. Res. 2020, 68, 414–421. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Cameli, P.; Bergantini, L.; Franchi, F.; Scolletta, S.; Bargagli, E. Serum concentrations of Krebs von den Lungen-6 in different COVID-19 phenotypes. J. Med. Virol. 2021, 93, 657. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Lanzarone, N.; Mazzei, M.A.; Alonzi, V.; Sestini, P.; Bargagli, E. Serum KL-6 levels in pulmonary Langerhans’ cell histiocytosis. Eur. J. Clin. Investig. 2020, 50, e13242. [Google Scholar] [CrossRef] [PubMed]
- D’Alessandro, M.; Bergantini, L.; Cameli, P.; Curatola, G.; Remediani, L.; Sestini, P.; Bargagli, E.; Unit, S.C. Peripheral biomarkers’ panel for severe COVID-19 patients. J. Med. Virol. 2021, 93, 1230–1232. [Google Scholar] [CrossRef]
- Bergantini, L.; Bargagli, E.; D’Alessandro, M.; Refini, R.; Cameli, P.; Galasso, L.; Scapellato, C.; Montagnani, F.; Scolletta, S.; Franchi, F.; et al. Prognostic bioindicators in severe COVID-19 patients. Cytokine 2021, 141, 155455. [Google Scholar] [CrossRef]
- D’Alessandro, M.; Covid, S.; Bergantini, L.; Cameli, P.; Curatola, G.; Remediani, L.; Bennett, D.; Bianchi, F.; Perillo, F.; Volterrani, L.; et al. Serial KL-6 measurements in COVID-19 patients. Intern. Emerg. Med. 2021, 1–5. [Google Scholar] [CrossRef]
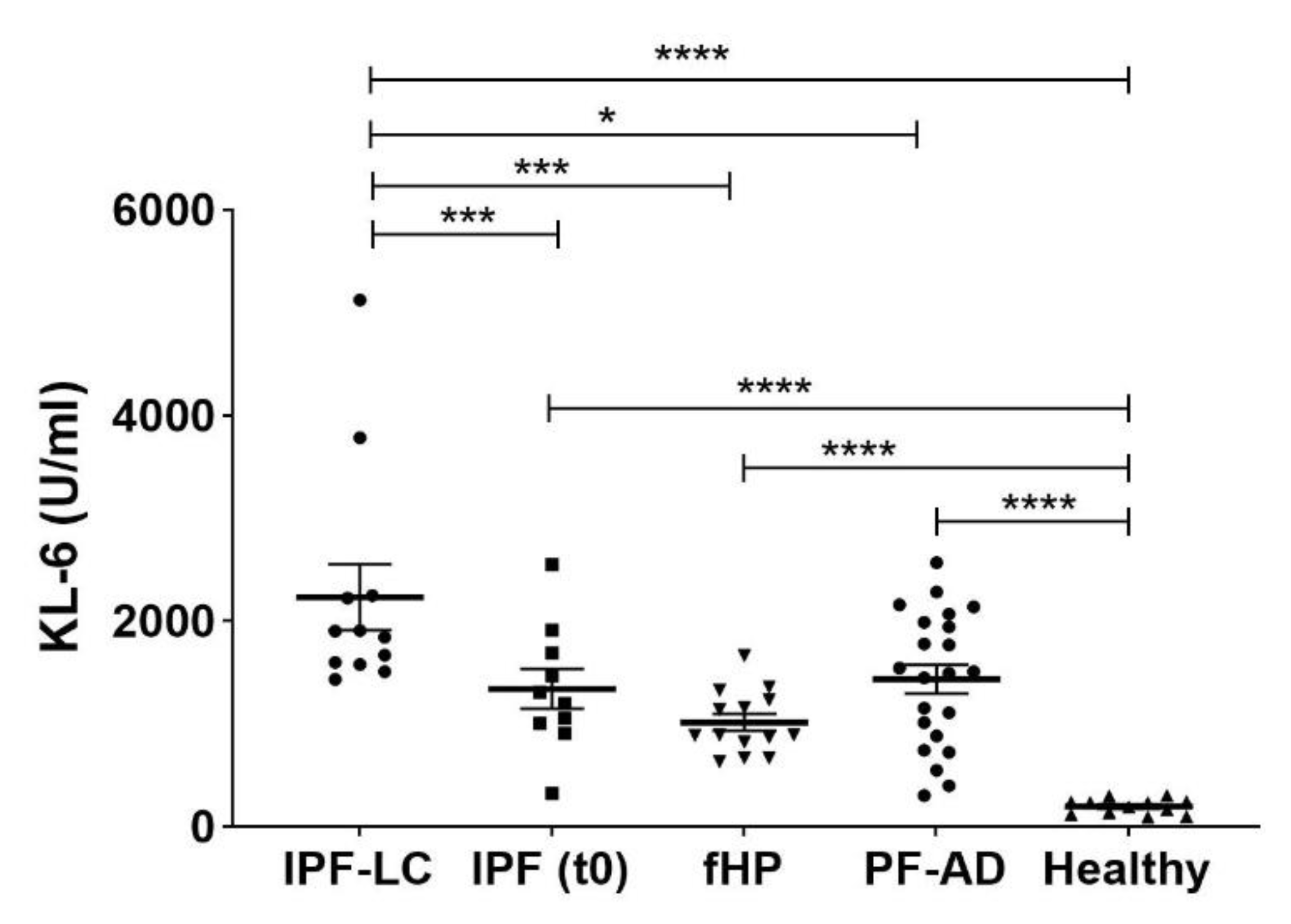
| Parameters | All Patients (n = 142) | IPF (n = 10) | IPF-LC (n = 12) | fHP (n = 14) | PF-AD (n = 22) | HC (n = 12) |
|---|---|---|---|---|---|---|
| Age (median IQR) | 69 (65–76) | 77 (67–80) | 72 (65–76) | 67 (57–72) | 63 (55–68) | 63 (52–65) |
| Gender, M/F | 86/56 | 8/2 | 8/4 | 8/6 | 9/13 | 5/7 |
| Smoking habit (never/former) | 73/69 | 4/6 | 5/7 | 6/8 | 8/14 | 8/4 |
| BMI (kg/m2) | 27 (25–28) | 26 (23–29) | 23 (21–26) | 27 (24–30) | 27 (25–29) | 26 (24–27) |
| Pulmonary function parameters (median, IQR) | ||||||
| FVC % | 83 (75–91) | 86 (72–101) | 63 (50–76) | 70 (58–84) | 89 (79–102) | 101 (88–107) |
| FEV1 % | 83 (73–95) | 87 (73–102) | 63 (52–75) | 66 (60–81) | 85 (78–96) | 98 (89–110) |
| DLCO % | 55 (45–73) | 49 (43–59) | 25 (21–26) | 55 (23–56) | 56 (43–58) | 90 (83–99) |
| KL-6 U/ml | 1252 (977–1742) | 1784 (1523–2242) | 988 (883–1257) | 1500 (846–2008) | 211 (121–241) 1 |
| Serum KL-6 Concentrations | AUC | p Value | Cut-off Value | Sensitivity | Specificity |
|---|---|---|---|---|---|
| IPF-LC vs. HC | 100 | <0.0001 | 1137 | 100 | 92 |
| IPF vs. HC | 100 | <0.0001 | 614 | 100 | 90 |
| fHP vs. HC | 100 | <0.0001 | 649 | 100 | 93 |
| PF-AD vs. HC | 100 | <0.0001 | 743 | 100 | 94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
d’Alessandro, M.; Bergantini, L.; Cameli, P.; Pieroni, M.; Refini, R.M.; Sestini, P.; Bargagli, E. Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy. Cancers 2021, 13, 689. https://doi.org/10.3390/cancers13040689
d’Alessandro M, Bergantini L, Cameli P, Pieroni M, Refini RM, Sestini P, Bargagli E. Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy. Cancers. 2021; 13(4):689. https://doi.org/10.3390/cancers13040689
Chicago/Turabian Styled’Alessandro, Miriana, Laura Bergantini, Paolo Cameli, Maria Pieroni, Rosa Metella Refini, Piersante Sestini, and Elena Bargagli. 2021. "Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy" Cancers 13, no. 4: 689. https://doi.org/10.3390/cancers13040689
APA Styled’Alessandro, M., Bergantini, L., Cameli, P., Pieroni, M., Refini, R. M., Sestini, P., & Bargagli, E. (2021). Serum Concentrations of KL-6 in Patients with IPF and Lung Cancer and Serial Measurements of KL-6 in IPF Patients Treated with Antifibrotic Therapy. Cancers, 13(4), 689. https://doi.org/10.3390/cancers13040689









