Socioeconomic Factors and Survival of Multiple Myeloma Patients
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
3. Discussion
4. Methods
4.1. Data Source
4.2. Study Variables
4.3. Statistical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MM | Multiple Myeloma |
| HSCT | Hematopoietic Stem Cell Transplant |
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Noone, A.; Howlader, N.; Krapcho, M.; Miller, D.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; et al. SEER Cancer Statistics Review 1975–2015. Natl. Cancer Inst. 2015, 2015, 655–710. [Google Scholar]
- Singhal, S.; Mehta, J.; Desikan, R.; Ayers, D.; Roberson, P.; Eddlemon, P.; Munshi, N.; Anaissie, E.; Wilson, C.; Dhodapkar, M.; et al. Antitumor Activity of Thalidomide in Refractory Multiple Myeloma. N. Engl. J. Med. 1999, 341, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Miguel, J.F.S.; Schlag, R.; Khuageva, N.K.; Dimopoulos, M.A.; Shpilberg, O.; Kropff, M.; Spicka, I.; Petrucci, M.T.; Palumbo, A.; Olga S Samoilova, O.S.; et al. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N. Engl. J. Med. 2008, 359, 906–917. [Google Scholar] [CrossRef]
- Rajkumar, V.S.; Jacobus, S.; Callander, N.S.; Fonseca, R.; Vesole, D.H.; Williams, M.E.; Abonour, R.; Siegel, D.S.; Katz, M.; Greipp, P.R. Lenalidomide plus high-dose dexamethasone versus lenalidomide plus low-dose dexamethasone as initial therapy for newly diagnosed multiple myeloma: An open-label randomised controlled trial. Lancet Oncol. 2010, 11, 29–37. [Google Scholar] [CrossRef]
- Harousseau, J.-L.; Attal, M.; Avet-Loiseau, H.; Marit, G.; Caillot, D.; Mohty, M.; Lenain, P.; Hulin, C.; Facon, T.; Casassus, P.; et al. Bortezomib Plus Dexamethasone Is Superior to Vincristine Plus Doxorubicin Plus Dexamethasone As Induction Treatment Prior to Autologous Stem-Cell Transplantation in Newly Diagnosed Multiple Myeloma: Results of the IFM 2005-01 Phase III Trial. J. Clin. Oncol. 2010, 28, 4621–4629. [Google Scholar] [CrossRef]
- Warren, J.L.; Harlan, L.C.; Stevens, J.; Little, R.F.; Abel, G.A. Multiple Myeloma Treatment Transformed: A Population-Based Study of Changes in Initial Management Approaches in the United States. J. Clin. Oncol. 2013, 31, 1984–1989. [Google Scholar] [CrossRef]
- Costa, L.J.; Zhang, M.-J.; Zhong, X.; Dispenzieri, A.; Lonial, S.; Krishnan, A.; Freytes, C.; Vesole, D.; Gale, R.P.; Anderson, K.; et al. Trends in Utilization and Outcomes of Autologous Transplantation as Early Therapy for Multiple Myeloma. Biol. Blood Marrow Transplant. 2013, 19, 1615–1624. [Google Scholar] [CrossRef]
- Malek, E.; Gupta, V.; Creger, R.; Caimi, P.; Vatsayan, A.; Covut, F.; Bashir, Q.; Champlin, R.; Delgado, R.; Rondon, G.; et al. Amifostine reduces gastro-intestinal toxicity after autologous transplantation for multiple myeloma. Leuk. Lymphoma 2018, 59, 1905–1912. [Google Scholar] [CrossRef]
- Fonseca, R.; Abouzaid, S.; Bonafede, M.; Cai, Q.; Parikh, K.; Cosler, L.; Richardson, P. Trends in overall survival and costs of multiple myeloma, 2000–2014. Leukemia 2017, 31, 1915–1921. [Google Scholar] [CrossRef]
- Rosenberg, A.R.; Kroon, L.; Chen, L.; Li, C.I.; Jones, B. Insurance status and risk of cancer mortality among adolescents and young adults. Cancer 2014, 121, 1279–1286. [Google Scholar] [CrossRef]
- Robbins, A.S.; Pavluck, A.L.; Fedewa, S.A.; Chen, A.Y.; Ward, E.M. Insurance Status, Comorbidity Level, and Survival Among Colorectal Cancer Patients Age 18 to 64 Years in the National Cancer Data Base From 2003 to 2005. J. Clin. Oncol. 2009, 27, 3627–3633. [Google Scholar] [CrossRef] [PubMed]
- Parikh, R.R.; Grossbard, M.L.; Green, B.L.; Harrison, L.B.; Yahalom, J. Disparities in survival by insurance status in patients with Hodgkin lymphoma. Cancer 2015, 121, 3515–3524. [Google Scholar] [CrossRef] [PubMed]
- Kwok, J.; Langevin, S.M.; Argiris, A.; Grandis, J.R.; Gooding, W.E.; Taioli, E. The impact of health insurance status on the survival of patients with head and neck cancer. Cancer 2009, 116, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Jemal, A.; Flowers, C.R.; Sineshaw, H.; Nastoupil, L.J.; Ward, E.M. Insurance status is related to diffuse large B-cell lymphoma survival. Cancer 2014, 120, 1220–1227. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.; Brill, I.K.; Brown, E.E. Impact of marital status, insurance status, income, and race/ethnicity on the survival of younger patients diagnosed with multiple myeloma in the United States. Cancer 2016, 122, 3183–3190. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Schrag, N.M.; Halpern, M.; Ward, E.M. The impact of health insurance status on stage at diagnosis of oropharyngeal cancer. Cancer 2007, 110, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.-C.T.; Xu, Y.; Liu, L.; Smieliauskas, F. Rising Prices of Targeted Oral Anticancer Medications and Associated Financial Burden on Medicare Beneficiaries. J. Clin. Oncol. 2017, 35, 2482–2489. [Google Scholar] [CrossRef] [PubMed]
- Huntington, S.F.; Weiss, B.M.; Vogl, D.T.; Cohen, A.D.; Garfall, A.L.; A Mangan, P.; Doshi, J.A.; Stadtmauer, E.A. Financial toxicity in insured patients with multiple myeloma: A cross-sectional pilot study. Lancet Haematol. 2015, 2, e408–e416. [Google Scholar] [CrossRef]
- Ailawadhi, S.; Frank, R.D.; Advani, P.; Swaika, A.; Temkit, M.; Menghani, R.; Sharma, M.; Meghji, Z.; Paulus, S.; Khera, N.; et al. Racial disparity in utilization of therapeutic modalities among multiple myeloma patients: A SEER-medicare analysis. Cancer Med. 2017, 6, 2876–2885. [Google Scholar] [CrossRef]
- Go, R.S.; Bartley, A.C.; Crowson, C.S.; Shah, N.D.; Habermann, E.B.; Holton, S.J.; Holmes, I.D.R. Association Between Treatment Facility Volume and Mortality of Patients With Multiple Myeloma. J. Clin. Oncol. 2017, 35, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Streeter, S.B.; Schwartzberg, L.; Husain, N.; Johnsrud, M. Patient and Plan Characteristics Affecting Abandonment of Oral Oncolytic Prescriptions. J. Oncol. Pract. 2011, 7, 46s–51s. [Google Scholar] [CrossRef] [PubMed]
- Fillmore, N.R.; Yellapragada, S.V.; Ifeorah, C.; Mehta, A.; Cirstea, D.; White, P.S.; Rivero, G.; Zimolzak, A.; Saiju Pyarajan, S.; Nhan Do, N.; et al. With equal access, African American patients have superior survival compared to white patients with multiple myeloma: A VA study. Blood 2019, 133, 2615–2618. [Google Scholar] [CrossRef] [PubMed]
- Abouzaid, S.; Parikh, K.; Zhou, Z.-Y.; Zhou, Z.; Tang, W.; Xie, J.; Patel, M.I. Disparities in treatment patterns and outcomes between Caucasian and African American patients with multiple myeloma (MM). J. Clin. Oncol. 2016, 34, 8022. [Google Scholar] [CrossRef]
- Fiala, M.A.; Wildes, T.M. Racial Disparities in Treatment Utilization for Multiple Myeloma. Cancer 2017, 123, 1590–1596. [Google Scholar] [CrossRef] [PubMed]
- Braunlin, M.; Belani, R.; Buchanan, J.; Wheeling, T.; Kim, C. Trends in the multiple myeloma treatment landscape and survival: A U.S. analysis using 2011–2019 oncology clinic electronic health record data. Leuk. Lymphoma 2020, 134, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jagosky, M.H.; Usmani, S.Z. Extramedullary Disease in Multiple Myeloma. Curr. Hematol. Malig. Rep. 2020, 15, 62–71. [Google Scholar] [CrossRef]
- Waxman, A.J.; Mink, P.J.; Devesa, S.S.; Anderson, W.F.; Weiss, B.M.; Kristinsson, S.Y.; McGlynn, K.A.; Landgren, O. Racial disparities in incidence and outcome in multiple myeloma: A population-based study. Blood 2010, 116, 5501–5506. [Google Scholar] [CrossRef]
- Winn, A.N.; Dusetzina, S.B. More evidence on the limited impact of state oral oncology parity laws. Cancer 2019, 125, 335–336. [Google Scholar] [CrossRef]
- Rajkumar, S.V. Value and Cost of Myeloma Therapy. Am. Soc. Clin. Oncol. Educ. Book 2018, 38, 662–666. [Google Scholar] [CrossRef]
- Pinsky, B.; Huang, H.; Teitelbaum, A.; Esseltine, D.-L.; Henk, H.J. Multiple Myeloma: Patient out-of-Pocket Costs and Health Care Utilization. Blood 2009, 114, 1366. [Google Scholar] [CrossRef]
- National Cancer Data Base—Data Dictionary PUF; The American College of Surgeons: Chicago, IL, USA, 2016.
- United States Department of Agriculture Economic Research Service 2013 Rural-Urban Continuum Codes. Available online: https://www.ers.usda.gov/data-products/rural-urban-continuum-codes/documentation.aspx (accessed on 21 June 2019).
- Charlson, M.; Szatrowski, T.P.; Peterson, J.; Gold, J. Validation of a combined comorbidity index. J. Clin. Epidemiol. 1994, 47, 1245–1251. [Google Scholar] [CrossRef]
- Durie, B.G. The role of anatomic and functional staging in myeloma: Description of Durie/Salmon plus staging system. Eur. J. Cancer 2006, 42, 1539–1543. [Google Scholar] [CrossRef] [PubMed]
- Grambsch, P.; Therneau, T. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 1994, 81, 515–526. [Google Scholar] [CrossRef]
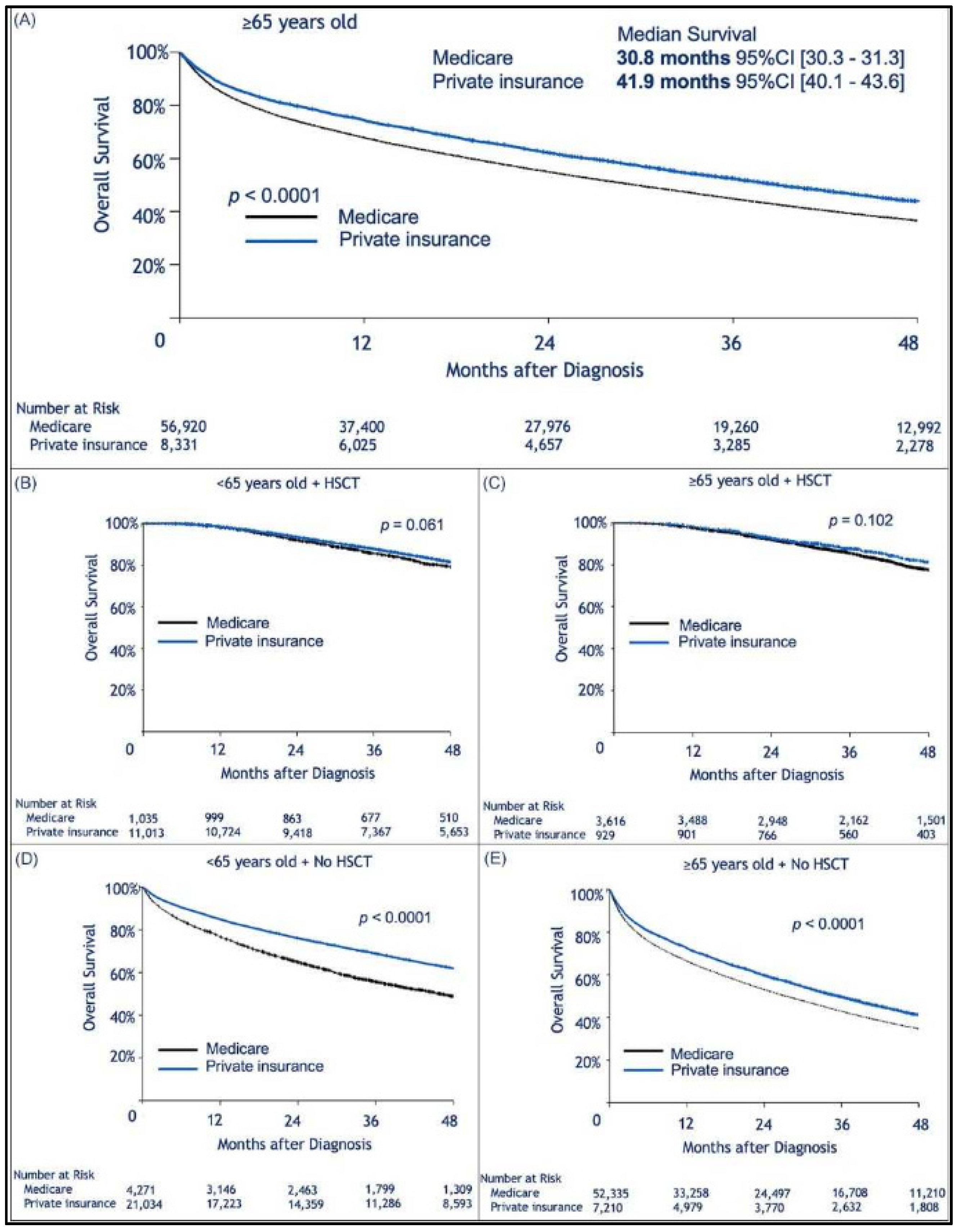
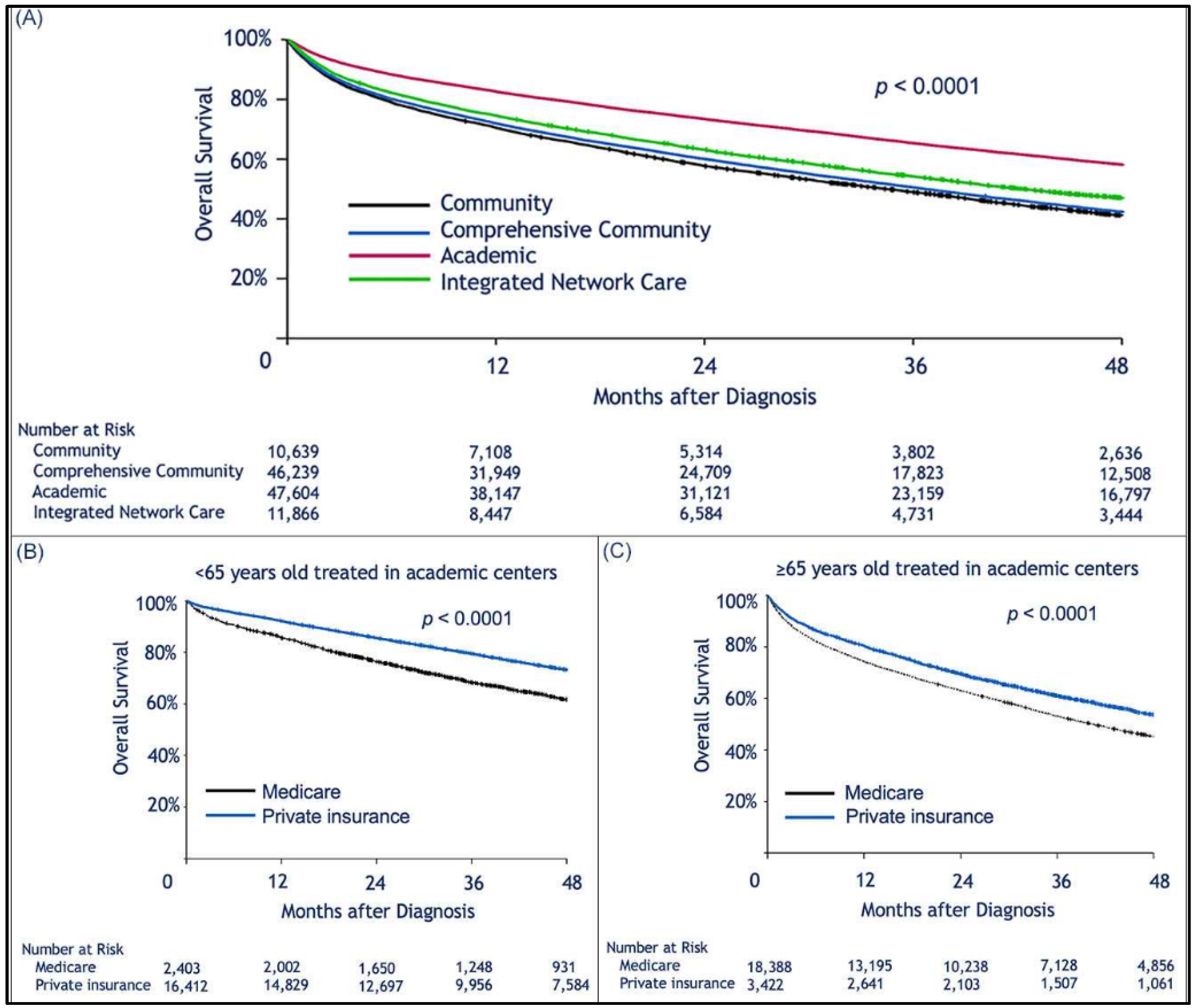

| Characteristic | Total Number of Patients | Medicare N (%), Median [Range] | Private Insurance N (%), Median [Range] | Medicaid N (%), Median [Range] | Not insured N (%), Median [Range] | Other gov. ins. N (%), Median [Range] |
|---|---|---|---|---|---|---|
| Total Number | 115,099 | 62,340 (53) | 41,340 (35) | 6410 (5.4) | 3796 (3.2) | 1213 (1) |
| Age at diagnosis | 67 (19–90) | 74 (23–90) | 57 (21–90) | 58 (19–90) | 57 (21–90) | 61 (27–90) |
| Gender | ||||||
| Male | 62,798 (55) | 33,071 (53) | 23,438 (57) | 3338 (52) | 2155 (57) | 796 (66) |
| Female | 52,301(45) | 29,269 (47) | 17,902 (43) | 3072 (48) | 1641 (43) | 417 (34) |
| Race | ||||||
| White | 87,263 (77) | 49,313 (79) | 31,472 (76) | 3446 (54) | 2214 (58) | 818 (67) |
| Black | 22,976 (20) | 10,982 (18) | 7910 (19) | 2432 (38) | 1347 (35) | 305 (25) |
| Other | 3616 (3) | 1489 (2) | 1416 (3) | 449 (7) | 193 (5) | 69 (6) |
| Median income (zip-code-based) * | ||||||
| <30 k | 16,419 (15) | 9178 (15) | 4497 (11) | 1712 (27) | 858 (23) | 174 (14) |
| 30–34.9 k | 19,878 (18) | 11,258 (18) | 6341 (15) | 1258 (20) | 784 (21) | 237 (20) |
| 35–45.9 k | 30,160 (27) | 16,717 (27) | 10,394 (25) | 1687 (26) | 951 (25) | 411 (34) |
| ≥46 k | 44,367 (40) | 23,000 (37) | 18,419 (45) | 1567 (24) | 1044 (28) | 337 (28) |
| Education level (zip-code-based) ^ | ||||||
| Level 1 | 20,276 (18) | 10,731 (17) | 6021 (15) | 2156 (34) | 1175 (31) | 193 (16) |
| Level 2 | 25,513 (23) | 14,000 (22) | 8546 (21) | 1700 (27) | 997 (26) | 270 (22) |
| Level 3 | 25,223 (23) | 14,011 (22) | 9054 (22) | 1215 (19) | 623 (16) | 320 (26) |
| Level 4 | 39,807 (36) | 21,410 (34) | 16,026 (39) | 1153 (18) | 842 (22) | 376 (31) |
| Area of residence | ||||||
| Metro | 92,236 (83) | 48,638 (78) | 34,110 (83) | 5437 (85) | 3147 (83) | 904 (75) |
| Urban | 16,795 (15) | 10,172 (16) | 5159 (12) | 739 (12) | 485 (13) | 240 (20) |
| Rural | 2357 (2) | 1502 (2) | 689 (2) | 83 (1) | 55 (1) | 28 (2) |
| Facility location | ||||||
| Northeast | 24,688 (22) | 13,135 (21) | 9466 (23) | 1560 (24) | 459 (12) | 68 (6) |
| Midwest | 29,936 (26) | 17,379 (28) | 10,342 (25) | 1331 (21) | 678 (18) | 206 (17) |
| West | 18,113 (16) | 9032 (14) | 7071 (17) | 1252 (20) | 482 (13) | 276 (23) |
| South | 40,834 (36) | 22,722 (36) | 13,479 (33) | 1950 (30) | 2036 (54) | 647 (53) |
| Disease | ||||||
| Primary | 97,693 (65) | 49,969 (80) | 37,167 (90) | 5929 (92) | 3564 (94) | 1064 (88) |
| Secondary | 17,394 (15) | 12,366 (20) | 4167 (10) | 481 (8) | 231 (6) | 149 (12) |
| Durie–Salmon Stage | ||||||
| 1 | 2118 (1.8) | 954 (2) | 1010 (2) | 92 (1) | 36 (1) | 26 (2) |
| 2 | 2970 (2.5) | 1371 (2) | 1326 (3) | 152 (2) | 85 (2) | 36 (3) |
| 3 | 7502 (6.4) | 3297 (5) | 3334 (8) | 518 (8) | 258 (7) | 95 (8) |
| Not available | 105,336 (89.3) | 56,718 (91) | 35,670 (87) | 5648 (89) | 3417 (90) | 1056 (87) |
| Charlson/Deyo comorbidity score | ||||||
| 0 | 87,121 (76) | 44,534 (71) | 33,963 (82) | 4683 (73) | 2998 (79) | 943 (78) |
| 1 | 18,799 (16) | 11,626 (19) | 5347 (13) | 1110 (17) | 538 (14) | 178 (15) |
| 2 | 6405 (6) | 4303 (7) | 1477 (4) | 403 (6) | 158 (4) | 64 (5) |
| 3 or more | 2774 (2) | 1877 (3) | 553 (1) | 214 (3) | 102 (3) | 28 (2) |
| Hematopoietic stem cell transplant | ||||||
| Yes | 18,213 (16) | 4651 (7) | 11,942 (29) | 1075 (17) | 272 (7) | 273 (23) |
| No | 94,354 (84) | 56,606 (91) | 28,244 (68) | 5175 (81) | 3414 (90) | 915 (75) |
| Facility type £ | ||||||
| Community | 10,460 (9) | 6789 (11) | 2713 (7) | 573 (9) | 270 (7) | 115 (9) |
| Comprehensive community | 45,428 (40) | 28,007 (45) | 13,962 (34) | 1687 (26) | 1292 (34) | 480 (40) |
| Academic | 45,990 (41) | 20,791 (33) | 19,834 (48) | 3190 (50) | 1677 (44) | 498 (41) |
| Integrated network cancer program | 11,693 (10) | 6681 (11) | 3849 (9) | 643 (10) | 416 (11) | 104 (9) |
| Facility volume | ||||||
| <10 patient/year | 44,040 (38) | 27,113 (43) | 12,697 (31) | 2271 (35) | 1538 (41) | 421 (35) |
| 10–50 patient/year | 55,648 (48) | 29,354 (47) | 20,390 (49) | 3287 (51) | 1978 (52) | 639 (53) |
| >50 patient/year | 15,411 (14) | 5873 (9) | 8253 (20) | 852 (13) | 280 (7) | 153 (13) |
| Distance traveled | ||||||
| Distance traveled (miles) | 10 (0–4961) | 8 (0–4961) | 12 (0–3367) | 7 (0–2439) | 9 (0–2424) | 14 (0–3329) |
| Traveled > 120 miles | 4711 (4) | 1964 (3) | 2384 (5.7) | 184 (2.8) | 97 (2.4) | 82 (6.7) |
| Factor | Hazard Ratio (95% CI) | p-Value |
|---|---|---|
| Age (per year increase) | 1.044 (1.039, 1.049) | 0.000 |
| Male vs. Female | 1.06 (0.99, 1.14) | 0.098 |
| White vs. Black | 1.07 (0.98, 1.17) | 0.124 |
| Primary Myeloma vs. secondary Myeloma | 0.95 (0.86, 1.04) | 0.242 |
| Charlson–Deyo score (0 vs. 3) | 0.48 (0.39, 0.59) | 0.000 |
| Charlson–Deyo score (1 vs. 3) | 0.7 (0.57, 0.87) | 0.002 |
| Charlson–Deyo score (2 vs. 3) | 0.92 (0.72, 1.16) | 0.472 |
| Other hospital types vs. Academic | 1.49 (1.39, 1.59) | 0.000 |
| Zipcode-based median income: <46 k vs. ≥46 k | 1.16 (1.08, 1.25) | 0.000 |
| Medicare vs. private insurance | 1.09 (0.99, 1.2) | 0.073 |
| Medicaid vs. private insurance | 1.59 (1.36, 1.87) | 0.000 |
| Not insured vs. private insurance | 1.62 (1.32, 1.99) | 0.000 |
| Other gov. insurance vs. private insurance | 0.95 (0.66, 1.39) | 0.802 |
| Factor | Hazard Ratio (95% CI) | p-Value |
|---|---|---|
| Age (per year increase) | 1.06 (1.055, 1.068) | <0.001 |
| Male vs. female | 1.05 (0.96, 1.15) | 0.263 |
| White vs. Black | 1.05 (0.93, 1.18) | 0.436 |
| Primary myeloma vs. secondary myeloma | 0.96 (0.86, 1.06) | 0.387 |
| Charlson–Deyo score (0 vs. 3) | 0.52 (0.41, 0.68) | <0.001 |
| Charlson–Deyo score (1 vs. 3) | 0.77 (0.59, 1) | 0.054 |
| Charlson–Deyo score (2 vs. 3) | 0.9 (0.67, 1.2) | 0.482 |
| Zipcode-based median income: <46 k vs. ≥46 k | 1.09 (1, 1.19) | 0.064 |
| Other hospital types vs. academic | 1.39 (1.28, 1.52) | <0.001 |
| Medicare vs. private | 1.04 (0.9, 1.2) | 0.568 |
| Medicaid vs. private | 1.23 (0.87, 1.75) | 0.246 |
| Not insured vs. private | 1.95 (1.26, 3.03) | 0.003 |
| Other gov. insurance vs. private | 1.1 (0.6, 2.01) | 0.763 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chamoun, K.; Firoozmand, A.; Caimi, P.; Fu, P.; Cao, S.; Otegbeye, F.; Metheny, L.; Patel, S.; Gerson, S.L.; Boughan, K.; et al. Socioeconomic Factors and Survival of Multiple Myeloma Patients. Cancers 2021, 13, 590. https://doi.org/10.3390/cancers13040590
Chamoun K, Firoozmand A, Caimi P, Fu P, Cao S, Otegbeye F, Metheny L, Patel S, Gerson SL, Boughan K, et al. Socioeconomic Factors and Survival of Multiple Myeloma Patients. Cancers. 2021; 13(4):590. https://doi.org/10.3390/cancers13040590
Chicago/Turabian StyleChamoun, Kamal, Amin Firoozmand, Paolo Caimi, Pingfu Fu, Shufen Cao, Folashade Otegbeye, Leland Metheny, Seema Patel, Stanton L. Gerson, Kirsten Boughan, and et al. 2021. "Socioeconomic Factors and Survival of Multiple Myeloma Patients" Cancers 13, no. 4: 590. https://doi.org/10.3390/cancers13040590
APA StyleChamoun, K., Firoozmand, A., Caimi, P., Fu, P., Cao, S., Otegbeye, F., Metheny, L., Patel, S., Gerson, S. L., Boughan, K., De Lima, M., & Malek, E. (2021). Socioeconomic Factors and Survival of Multiple Myeloma Patients. Cancers, 13(4), 590. https://doi.org/10.3390/cancers13040590







