Cognitive Impairment in Older Cancer Patients Treated with First-Line Chemotherapy
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
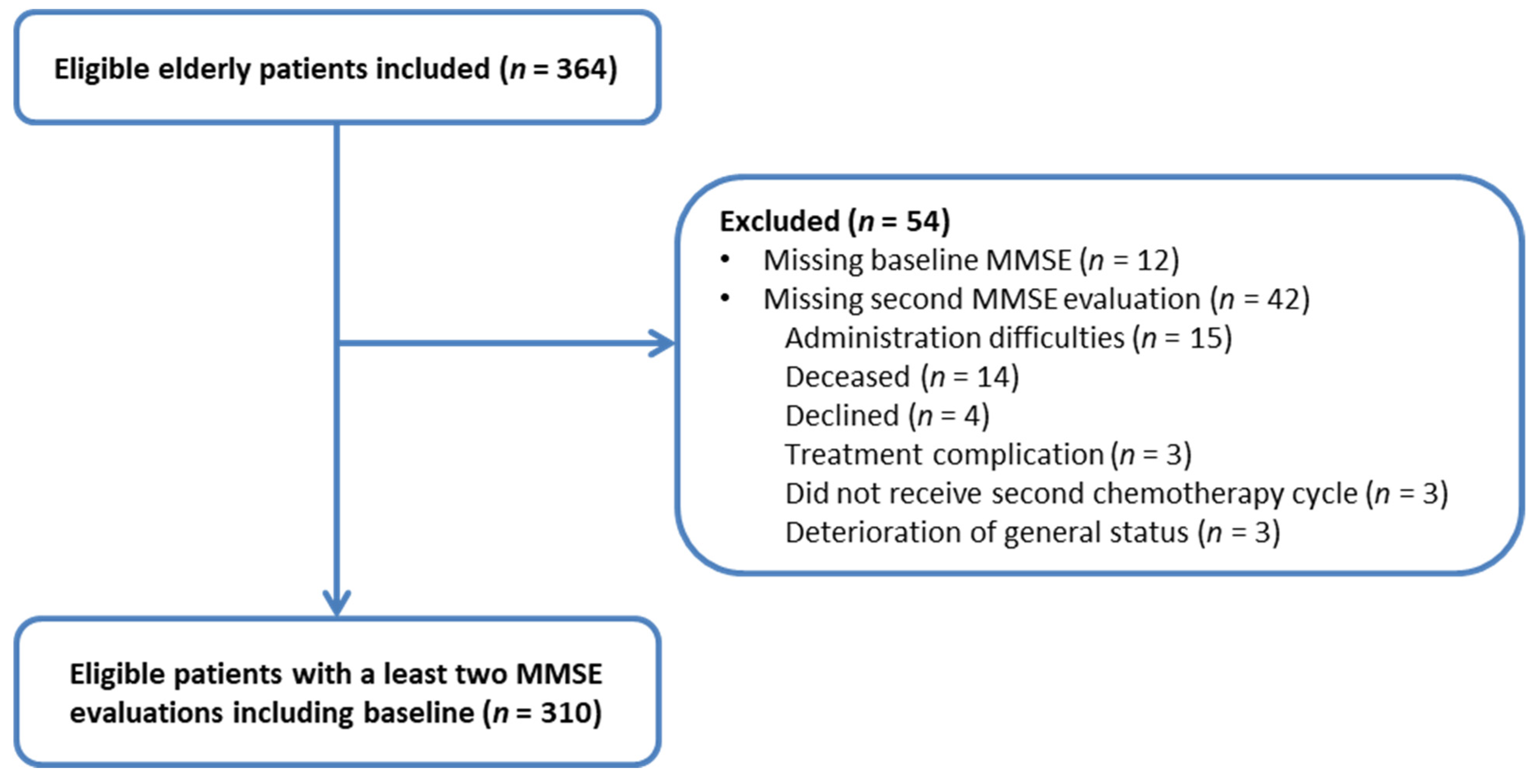
2.1. Participants
2.2. Study Design and Measurements
- -
- MMSE [15] for cognitive function, with adjusted scores for age and sociocultural level (SCL) according to the French standardization and range of the GRECO work group with abnormal score ≤ 10th percentile of normative values (Appendix A) [16];
- -
- Instrumental Activities of Daily Living (IADL) [17], focusing on the ability to perform eight household tasks with abnormal score ≤7;
- -
- Geriatric Depression Scale 15-item version (GDS15) [18] for depressive symptoms with abnormal score ≥6;
- -
- Mini-Nutritional Assessment (MNA) [19] for nutritional status with abnormal score ≤23.5.
2.3. Endpoints
2.4. Statistical Analysis
3. Results
3.1. Baseline MGA
3.2. Baseline QoL Scores
3.3. MMSE Impairment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Percentile | 50–79 Years | ≥80 Years | ||||||
|---|---|---|---|---|---|---|---|---|
| SCL 1 | SCL 2 | SCL 3 | SCL 4 | SCL 1 | SCL 2 | SCL 3 | SCL 4 | |
| 90 | 30 | 30 | 30 | 30 | 29 | 29 | 29 | 29 |
| 75 | 29 | 29 | 30 | 30 | 28 | 28 | 29 | 29 |
| 50 | 28 | 28 | 28 | 29 | 27 | 27 | 27 | 28 |
| 25 | 27 | 27 | 27 | 28 | 26 | 26 | 26 | 27 |
| 10 | 24 | 25 | 26 | 27 | 23 | 24 | 25 | 26 |
| 5 | 22 | 23 | 25 | 26 | 21 | 22 | 24 | 25 |
| 1 | 20 | 21 | 24 | 24 | 19 | 20 | 23 | 23 |
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Pilleron, S.; Sarfati, D.; Janssen-Heijnen, M.; Vignat, J.; Ferlay, J.; Bray, F.; Soerjomataram, I. Global cancer incidence in older adults, 2012 and 2035: A population-based study. Int. J. Cancer 2019, 144, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Joly, F.; Giffard, B.; Rigal, O.; De Ruiter, M.B.; Small, B.J.; Dubois, M.; LeFel, J.; Schagen, S.B.; Ahles, T.A.; Wefel, J.S.; et al. Impact of Cancer and Its Treatments on Cognitive Function: Advances in Research from the Paris International Cognition and Cancer Task Force Symposium and Update Since 2012. J. Pain Symptom Manag. 2015, 50, 830–841. [Google Scholar] [CrossRef] [Green Version]
- Lange, M.; Licaj, I.; Clarisse, B.; Humbert, X.; Grellard, J.-M.; Tron, L.; Joly, F. Cognitive complaints in cancer survivors and expectations for support: Results from a web-based survey. Cancer Med. 2019, 8, 2654–2663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bettio, L.E.B.; Rajendran, L.; Gil-Mohapel, J. The effects of aging in the hippocampus and cognitive decline. Neurosci. Biobehav. Rev. 2017, 79, 66–86. [Google Scholar] [CrossRef] [PubMed]
- Ahles, T.A.; Saykin, A.J.; McDonald, B.C.; Li, Y.; Furstenberg, C.T.; Hanscom, B.S.; Mulrooney, T.J.; Schwartz, G.N.; Kaufman, P.A. Longitudinal assessment of cognitive changes associated with adjuvant treatment for breast cancer: Impact of age and cognitive reserve. J. Clin. Oncol. 2010, 28, 4434–4440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lange, M.; Rigal, O.; Clarisse, B.; Giffard, B.; Sevin, E.; Barillet, M.; Eustache, F.; Joly, F. Cognitive dysfunctions in elderly cancer patients: A new challenge for oncologists. Cancer Treat. Rev. 2014, 40, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Mandelblatt, J.S.; Jacobsen, P.B.; Ahles, T. Cognitive effects of cancer systemic therapy: Implications for the care of older patients and survivors. J. Clin. Oncol. 2014, 32, 2617–2626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handforth, C.; Clegg, A.; Young, C.; Simpkins, S.; Seymour, M.T.; Selby, P.J.; Young, J. The prevalence and outcomes of frailty in older cancer patients: A systematic review. Ann. Oncol. 2015, 26, 1091–1101. [Google Scholar] [CrossRef]
- Mitka, M. Too few older patients in cancer trials: Experts say disparity affects research results and care. JAMA 2003, 290, 27–28. [Google Scholar] [CrossRef] [PubMed]
- Wefel, J.S.; Vardy, J.; Ahles, T.; Schagen, S.B. International Cognition and Cancer Task Force recommendations to harmonise studies of cognitive function in patients with cancer. Lancet Oncol. 2011, 12, 703–708. [Google Scholar] [CrossRef]
- Cieri, F.; Esposito, R.; Cera, N.; Pieramico, V.; Tartaro, A.; di Giannantonio, M. Late-Life Depression: Modifications of Brain Resting State Activity. J. Geriatr. Psychiatry Neurol. 2017, 30, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Soubeyran, P.; Fonck, M.; Blanc-Bisson, C.; Blanc, J.-F.; Ceccaldi, J.; Mertens, C.; Imbert, Y.; Cany, L.; Vogt, L.; Dauba, J.; et al. Predictors of early death risk in older patients treated with first-line chemotherapy for cancer. J. Clin. Oncol. 2012, 30, 1829–1834. [Google Scholar] [CrossRef] [PubMed]
- Chatfield, M.; Matthews, F.E.; Brayne, C. Using the Mini-Mental State Examination for tracking cognition in the older population based on longitudinal data. J. Am. Geriatr. Soc. 2007, 55, 1066–1071. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Kalafat, M.; Hugonot-Diener, L.; Poitrenaud, J. French standardization and range for the GRECO version of the “Mini Mental State” (MMS). Rev. Neuropsychol. 2003, 13, 209–236. [Google Scholar]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontol. 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Herrmann, N.; Mittmann, N.; Silver, I.L.; Shulman, K.I.; Busto, U.A.; Shear, N.H.; Naranjo, C.A. A validation study of The Geriatric Depression Scale short form. Int. J. Geriatr. Psychiatry 1996, 11, 457–460. [Google Scholar] [CrossRef]
- Vellas, B.; Guigoz, Y.; Garry, P.J.; Nourhashemi, F.; Bennahum, D.; Lauque, S.; Albarede, J.-L. The mini nutritional assessment (MNA) and its use in grading the nutritional state of elderly patients. Nutrition 1999, 15, 116–122. [Google Scholar] [CrossRef]
- Aaronson, N.K.; Ahmedzai, S.; Bergman, B.; Bullinger, M.; Cull, A.; Duez, N.J.; Filiberti, A.; Flechtner, H.; Fleishman, S.B.; de Haes, J.C.J.M.; et al. The European Organization for Research and Treatment of Cancer QLQ-C30: A Quality-of-Life Instrument for Use in International Clinical Trials in Oncology. JNCI J. Natl. Cancer Inst. 1993, 85, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Crum, R.M.; Anthony, J.C.; Bassett, S.S.; Folstein, M.F. Population-based norms for the Mini-Mental State Examination by age and educational level. JAMA 1993, 269, 2386–2391. [Google Scholar] [CrossRef] [PubMed]
- Veierød, M.B.; Lydersen, S.; Laake, P. Medical Statistics: In Clinical and Epidemiological Research; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Scott, N.W.; Fayers, P.; Aaronson, N.K.; Bottomley, A.; de Graeff, A.; Groenvold, M.; Gundy, C.; Koller, M.; Petersen, M.A.; Sprangers, M.A.; et al. EORTC QLQ-C30 Reference Values Manual; EORTC Quality of Life Group: Brussels, Belgium, 2008. [Google Scholar]
- Brain, E.G.C.; Mertens, C.; Girre, V.; Rousseau, F.; Blot, E.; Abadie, S.; Uwer, L.; Bourbouloux, E.; Van Praagh-Doreau, I.; Mourey, L.; et al. Impact of liposomal doxorubicin-based adjuvant chemotherapy on autonomy in women over 70 with hormone-receptor-negative breast carcinoma: A French Geriatric Oncology Group (GERICO) phase II multicentre trial. Crit. Rev. Oncol. Hematol. 2011, 80, 160–170. [Google Scholar] [CrossRef]
- Hurria, A.; Rosen, C.; Hudis, C.; Zuckerman, E.; Panageas, K.S.; Lachs, M.S.; Witmer, M.; van Gorp, W.G.; Fornier, M.; D’Andrea, G.; et al. Cognitive function of older patients receiving adjuvant chemotherapy for breast cancer: A pilot prospective longitudinal study. J. Am. Geriatr. Soc. 2006, 54, 925–931. [Google Scholar] [CrossRef]
- Kvale, E.A.; Clay, O.J.; Ross-Meadows, L.A.; McGee, J.S.; Edwards, J.D.; Unverzagt, F.W.; Ritchie, C.S.; Ball, K.K. Cognitive speed of processing and functional declines in older cancer survivors: An analysis of data from the ACTIVE trial. Eur. J. Cancer Care 2010, 19, 110–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minisini, A.M.; De Faccio, S.; Ermacora, P.; Andreetta, C.; Fantinel, R.; Balestrieri, M.; Piga, A.; Puglisi, F. Cognitive functions and elderly cancer patients receiving anticancer treatment: A prospective study. Crit. Rev. Oncol. Hematol. 2008, 67, 71–79. [Google Scholar] [CrossRef]
- Janelsins, M.C.; Kesler, S.R.; Ahles, T.A.; Morrow, G.R. Prevalence, mechanisms, and management of cancer-related cognitive impairment. Int. Rev. Psychiatry Abingdon Engl. 2014, 26, 102–113. [Google Scholar] [CrossRef] [Green Version]
- Ouvrard, C.; Berr, C.; Meillon, C.; Ribet, C.; Goldberg, M.; Zins, M.; Amieva, H. Norms for standard neuropsychological tests from the French CONSTANCES cohort. Eur. J. Neurol. 2019, 26, 786–793. [Google Scholar] [CrossRef] [PubMed]
- DeCarli, C. Mild cognitive impairment: Prevalence, prognosis, aetiology, and treatment. Lancet Neurol. 2003, 2, 15–21. [Google Scholar] [CrossRef]
- Medical Research Council Cognitive Function and Ageing Study; Matthews, F.; Marioni, R.; Brayne, C. Examining the influence of gender, education, social class and birth cohort on MMSE tracking over time: A population-based prospective cohort study. BMC Geriatr. 2012, 12, 45. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.S.; Hebert, L.E.; Scherr, P.A.; Barnes, L.L.; Mendes de Leon, C.F.; Evans, D.A. Educational attainment and cognitive decline in old age. Neurology 2009, 72, 460–465. [Google Scholar] [CrossRef] [Green Version]
- Wärnberg, J.; Gomez-Martinez, S.; Romeo, J.; Díaz, L.-E.; Marcos, A. Nutrition, inflammation, and cognitive function. Ann. N. Y. Acad. Sci. 2009, 1153, 164–175. [Google Scholar] [CrossRef]
- Bicakli, D.H.; Ozveren, A.; Uslu, R.; Dalak, R.M.; Cehreli, R.; Uyar, M.; Karabulut, B.; Akcicek, F. The effect of chemotherapy on nutritional status and weakness in geriatric gastrointestinal system cancer patients. Nutrition 2018, 47, 39–42. [Google Scholar] [CrossRef]
- Bruijnen, C.P.; van Harten-Krouwel, D.G.; Koldenhof, J.J.; Emmelot-Vonk, M.H.; Witteveen, P.O. Predictive value of each geriatric assessment domain for older patients with cancer: A systematic review. J. Geriatr. Oncol. 2019, 10, 859–873. [Google Scholar] [CrossRef] [PubMed]
- Aaldriks, A.A.; Maartense, E.; le Cessie, S.; Giltay, E.J.; Verlaan, H.A.C.M.; van der Geest, L.G.M.; Kloosterman-Boele, W.M.; Peters-Dijkshoorn, M.T.; Blansjaar, B.A.; van Schaick, H.W.; et al. Predictive value of geriatric assessment for patients older than 70 years, treated with chemotherapy. Crit. Rev. Oncol. Hematol. 2011, 79, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Van Blarigan, E.L.; Fuchs, C.S.; Niedzwiecki, D.; Zhang, S.; Saltz, L.B.; Mayer, R.J.; Mowat, R.B.; Whittom, R.; Hantel, A.; Benson, A.; et al. Association of Survival with Adherence to the American Cancer Society Nutrition and Physical Activity Guidelines for Cancer Survivors After Colon Cancer Diagnosis: The CALGB 89803/Alliance Trial. JAMA Oncol. 2018, 4, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Hartman, S.J.; Nelson, S.H.; Myers, E.; Natarajan, L.; Sears, D.D.; Palmer, B.W.; Weiner, L.S.; Parker, B.A.; Patterson, R.E. Randomized controlled trial of increasing physical activity on objectively measured and self-reported cognitive functioning among breast cancer survivors: The memory & motion study. Cancer 2018, 124, 192–202. [Google Scholar] [PubMed]
- Hurria, A.; Togawa, K.; Mohile, S.G.; Owusu, C.; Klepin, H.D.; Gross, C.P.; Lichtman, S.M.; Gajra, A.; Bhatia, S.; Katheria, V.; et al. Predicting chemotherapy toxicity in older adults with cancer: A prospective multicenter study. J. Clin. Oncol. 2011, 29, 3457–3465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Libert, Y.; Dubruille, S.; Borghgraef, C.; Etienne, A.-M.; Merckaert, I.; Paesmans, M.; Reynaert, C.; Roos, M.; Slachmuylder, J.-L.; Vandenbossche, S.; et al. Vulnerabilities in Older Patients when Cancer Treatment is Initiated: Does a Cognitive Impairment Impact the Two-Year Survival? PLoS ONE 2016, 11, e0159734. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hart, R.P.; Wade, J.B.; Martelli, M.F. Cognitive impairment in patients with chronic pain: The significance of stress. Curr. Pain Headache Rep. 2003, 7, 116–126. [Google Scholar] [CrossRef] [PubMed]
- van der Leeuw, G.; Ayers, E.; Leveille, S.G.; Blankenstein, A.H.; van der Horst, H.E.; Verghese, J. The Effect of Pain on Major Cognitive Impairment in Older Adults. J. Pain 2018, 19, 1435–1444. [Google Scholar] [CrossRef] [PubMed]
- Kurita, G.P.; Sjøgren, P.; Ekholm, O.; Kaasa, S.; Loge, J.H.; Poviloniene, I.; Klepstad, P. Prevalence and predictors of cognitive dysfunction in opioid-treated patients with cancer: A multinational study. J. Clin. Oncol. 2011, 29, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Marco, C.A.; Mann, D.; Rasp, J.; Ballester, M.; Perkins, O.; Holbrook, M.B.; Rako, K. Effects of opioid medications on cognitive skills among Emergency Department patients. Am. J. Emerg. Med. 2018, 36, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool for Mild Cognitive Impairment: MOCA: A BRIEF SCREENING TOOL FOR MCI. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Rambeau, A.; Beauplet, B.; Laviec, H.; Licaj, I.; Leconte, A.; Chatel, C.; Le Bon, P.; Denhaerynck, J.; Clarisse, B.; Frenkiel, N.; et al. Prospective comparison of the Montreal Cognitive Assessment (MoCA) and the Mini Mental State Examination (MMSE) in geriatric oncology. J. Geriatr. Oncol. 2019, 10, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Hartung, T.J.; Brähler, E.; Faller, H.; Härter, M.; Hinz, A.; Johansen, C.; Keller, M.; Koch, U.; Schulz, H.; Weis, J.; et al. The risk of being depressed is significantly higher in cancer patients than in the general population: Prevalence and severity of depressive symptoms across major cancer types. Eur. J. Cancer 2017, 72, 46–53. [Google Scholar] [CrossRef]
- Boscher, C.; Joly, F.; Clarisse, B.; Humbert, X.; Grellard, J.-M.; Binarelli, G.; Tron, L.; Licaj, I.; Lange, M. Perceived Cognitive Impairment in Breast Cancer Survivors and Its Relationships with Psychological Factors. Cancers 2020, 12, 3000. [Google Scholar] [CrossRef] [PubMed]
- Apple, A.C.; Schroeder, M.P.; Ryals, A.J.; Wagner, L.I.; Cella, D.; Shih, P.-A.; Reilly, J.; Penedo, F.J.; Voss, J.L.; Wang, L. Hippocampal functional connectivity is related to self-reported cognitive concerns in breast cancer patients undergoing adjuvant therapy. NeuroImage Clin. 2018, 20, 110–118. [Google Scholar] [CrossRef] [PubMed]
| Demographic and Clinical Characteristics | No. | % |
|---|---|---|
| Median age, years (range) | 77.4 (70–93) | |
| ≥80 | 96 | 31.0 |
| Sex | ||
| Male | 183 | 59.0 |
| Female | 127 | 41.0 |
| ECOG performance status | ||
| 0–1 | 234 | 75.5 |
| 2 | 52 | 16.7 |
| 3–4 | 17 | 5.5 |
| Missing data | 7 | 2.3 |
| Living alone | 84 | 27.3 |
| Marital status | ||
| Single | 12 | 3.9 |
| Married | 200 | 64.5 |
| Widower | 79 | 25.5 |
| Divorced | 18 | 5.8 |
| Missing data | 1 | 0.3 |
| Education level | ||
| Pre-primary | 64 | 20.6 |
| Primary school certificate | 151 | 48.7 |
| Secondary studies | 61 | 19.7 |
| Higher studies | 33 | 10.7 |
| Missing data | 1 | 0.3 |
| Tumor site | ||
| NHL | 97 | 31.3 |
| Colon | 83 | 26.8 |
| Stomach | 32 | 10.3 |
| Lung | 31 | 10.0 |
| Pancreas | 18 | 5.8 |
| Prostate | 16 | 5.2 |
| Bladder | 15 | 4.8 |
| Ovary | 14 | 4.5 |
| Primary unknown | 4 | 1.3 |
| Disease extension | ||
| Solid tumors | 213 | 68.7 |
| Localized | 74 | 23.9 |
| Metastatic | 139 | 44.8 |
| NHL | 97 | 31.3 |
| aaIPI score 0–1 | 56 | 18.1 |
| aaIPI score 2–3 | 41 | 13.2 |
| Baseline Geriatric Evaluation | Normal Score | Abnormal Score * | Missing | Overall | ||||
|---|---|---|---|---|---|---|---|---|
| No. | % | No. | % | No. | % | Median | Range | |
| MMSE | 224 | 72.3 | 86 | 27.7 | 0 | 0 | 27.5 | 9 to 30 |
| GDS 15 | 167 | 53.9 | 137 | 44.2 | 6 | 1.9 | 5 | 0 to 13 |
| IADL | 86 | 27.8 | 223 | 71.9 | 1 | 0.3 | 6 | 0 to 8 |
| MNA | 111 | 35.8 | 195 | 62.9 | 4 | 1.3 | 21.5 | 6 to 28 |
| EORTC QLQ-C30 | Study Sample | Reference Values * | |||
|---|---|---|---|---|---|
| No. | Mean | No. | Mean | No. | |
| Global health status/QoL | 304 | 57.2 | 20.2 | 60.6 | 25.1 |
| Physical functioning | 305 | 74.8 | 22.7 | 72.1 | 25.4 |
| Role functioning | 304 | 69.4 | 33 | 70.7 | 34.1 |
| Emotional functioning | 305 | 73.5 | 22.8 | 76.1 | 23.2 |
| Cognitive functioning | 305 | 77.6 | 23.6 | 81 | 22.4 |
| Social functioning | 303 | 81.6 | 26.7 | 78.2 | 28.2 |
| Fatigue | 305 | 38.3 | 28.8 | 35.7 | 29 |
| Nausea and vomiting | 305 | 7.8 | 18 | 9.1 | 19.2 |
| Pain | 305 | 25.6 | 31.7 | 25.9 | 30.5 |
| Dyspnoea | 305 | 24.8 | 30.3 | 23.1 | 29.6 |
| Insomnia | 303 | 30.2 | 3.7 | 26.4 | 31.3 |
| Appetite loss | 304 | 34 | 37.5 | 22.4 | 33.2 |
| Constipation | 302 | 22.7 | 33.1 | 21.7 | 31.2 |
| Diarrhoea | 302 | 13.2 | 26.9 | 8.9 | 20.7 |
| Financial difficulties | 303 | 2.4 | 10.6 | 8.5 | 20.6 |
| Factor | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| OR | 95% CI | p | OR. | 95% CI | p | |
| Age (years) | 0.353 | 0.372 | ||||
| <80 | 1 (reference) | |||||
| ≥80 | 0.74 | 0.38–1.40 | 0.72 | 0.35–1.48 | ||
| Sexe | 0.603 | 0.632 | ||||
| Male | 1 (reference) | |||||
| Female | 1.17 | 0.65–2.10 | 1.18 | 0.60–2.32 | ||
| ECOG performance status | Excluded | |||||
| 0–1 | 1 (reference) | |||||
| 2 | 1.45 | 0.70–3.02 | 0.314 | |||
| 3–4 | 2.02 | 0.67–6.06 | 0.210 | |||
| Living alone | 0.029 | 0.097 | ||||
| No | 1 (reference) | |||||
| Yes | 2.33 | 1.09–4.99 | 2.02 | 0.88–4.65 | ||
| Tumor site | Excluded | |||||
| Hematologic | 1 (reference) | |||||
| Digestive | 0.92 | 0.46–1.81 | 0.805 | |||
| Lung | 1.05 | 0.38–2.95 | 0.921 | |||
| Uro Gynecologic | 1.25 | 0.52–2.99 | 0.611 | |||
| Disease extension | 0.494 | Excluded | ||||
| Localized/IPI 0–1 | 1 (reference) | |||||
| Metastatic/IPI 2–3 | 1.23 | 0.68–2.21 | ||||
| MMSE at baseline * | 0.048 | 0.022 | ||||
| Normal | 1 (reference) | 1 (reference) | ||||
| Abnormal | 2.07 | 1.09–4.31 | 2.58 | 1.14–5.84 | ||
| GDS15 | 0.089 | Excluded | ||||
| <6 | 1 (reference) | |||||
| ≥6 | 1.66 | 0.93–2.98 | ||||
| IADL | 0.099 | Excluded | ||||
| >7 | 1 (reference) | |||||
| ≤7 | 1.82 | 0.89–3.71 | ||||
| MNA | 0.031 | 0.021 | ||||
| >23.5 | 1 (reference) | |||||
| ≤23.5 | 1.91 | 1.21–3.24 | 1.87 | 1.18–3.58 | ||
| EORTC QLQ-C30 | ||||||
| Global health status/QoL | 0.8 | 0.65–0.97 | 0.029 | 0.81 | 0.65–1.00 | 0.054 |
| Physical functioning | 0.77 | 0.62–0.95 | 0.016 | 0.77 | 0.59–0.98 | 0.048 |
| Role functioning | 0.79 | 0.65–0.96 | 0.017 | 0.82 | 0.66–1.03 | 0.087 |
| Emotional functioning | 0.81 | 0.67–0.98 | 0.034 | 0.82 | 0.68–0.93 | 0.02 |
| Cognitive functioning | 0.87 | 0.73–1.05 | 0.162 | Excluded | ||
| Social functioning | 0.77 | 0.64–0.92 | 0.004 | 0.8 | 0.67–0.97 | 0.021 |
| Fatigue | 1.21 | 0.98–1.50 | 0.075 | 1.17 | 0.92–1.47 | 0.198 |
| Nausea and vomiting | 0.8 | 0.39–1.65 | 0.548 | Excluded | ||
| Pain | 1.81 | 1.27–2.59 | 0.001 | 1.77 | 1.21–2.59 | 0.003 |
| Dyspnoea | 0.99 | 0.74–1.32 | 0.933 | Excluded | ||
| Insomnia | 1.28 | 0.92–1.80 | 0.146 | Excluded | ||
| Appetite loss | 1.25 | 0.90–1.75 | 0.183 | Excluded | ||
| Constipation | 1.28 | 0.71–2.29 | 0.412 | Excluded | ||
| Diarrhoea | 0.87 | 0.44–1.73 | 0.695 | Excluded | ||
| Financial difficulties | 0.87 | 0.73–1.05 | 0.164 | Excluded | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dos Santos, M.; Licaj, I.; Bellera, C.; Cany, L.; Binarelli, G.; Soubeyran, P.; Joly, F. Cognitive Impairment in Older Cancer Patients Treated with First-Line Chemotherapy. Cancers 2021, 13, 6171. https://doi.org/10.3390/cancers13246171
Dos Santos M, Licaj I, Bellera C, Cany L, Binarelli G, Soubeyran P, Joly F. Cognitive Impairment in Older Cancer Patients Treated with First-Line Chemotherapy. Cancers. 2021; 13(24):6171. https://doi.org/10.3390/cancers13246171
Chicago/Turabian StyleDos Santos, Mélanie, Idlir Licaj, Carine Bellera, Laurent Cany, Giulia Binarelli, Pierre Soubeyran, and Florence Joly. 2021. "Cognitive Impairment in Older Cancer Patients Treated with First-Line Chemotherapy" Cancers 13, no. 24: 6171. https://doi.org/10.3390/cancers13246171
APA StyleDos Santos, M., Licaj, I., Bellera, C., Cany, L., Binarelli, G., Soubeyran, P., & Joly, F. (2021). Cognitive Impairment in Older Cancer Patients Treated with First-Line Chemotherapy. Cancers, 13(24), 6171. https://doi.org/10.3390/cancers13246171







