The Clinical Feature and Treatment Outcome of Ocular Melanoma: A 34-Year Experience in a Tertiary Referral Center
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Enrollment
2.2. Data Collection
2.3. Statistical Analysis
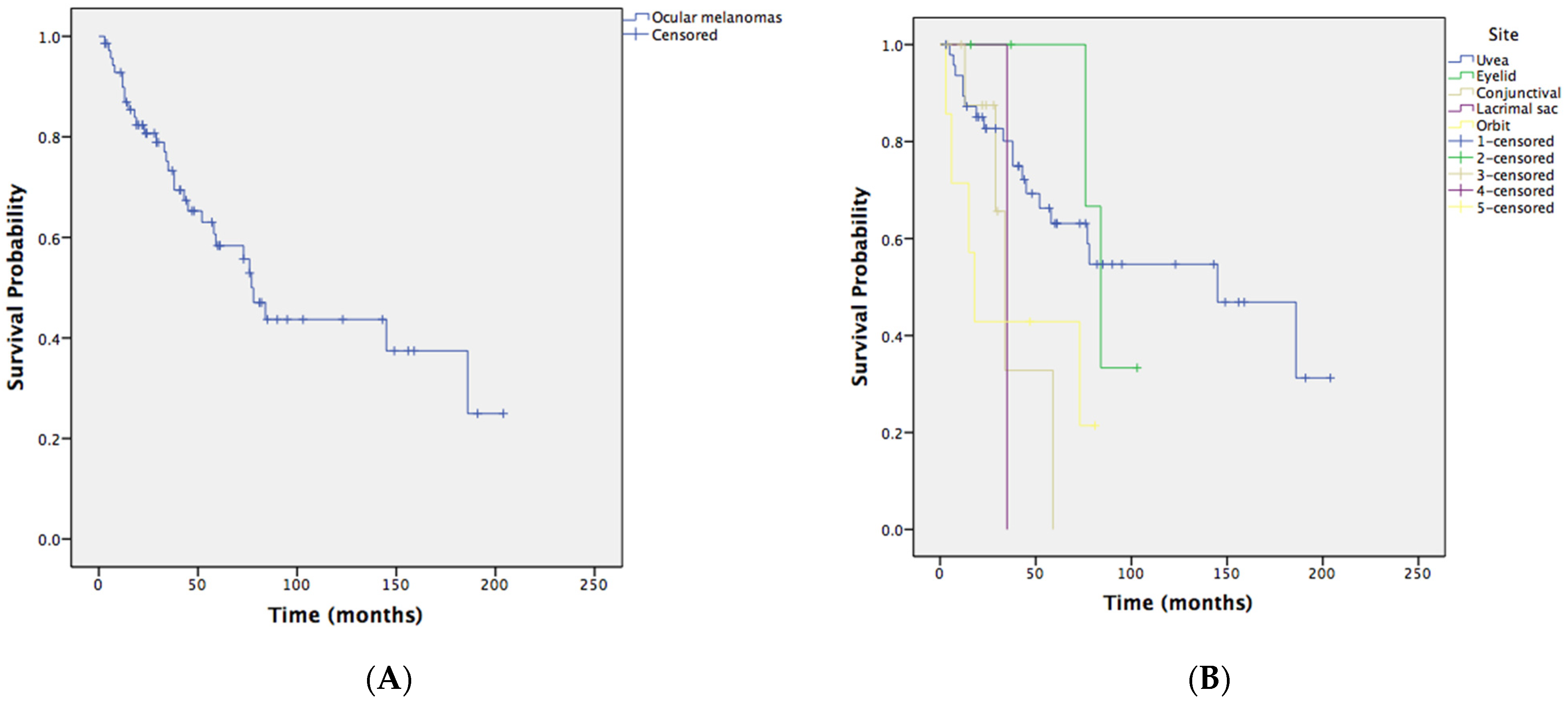
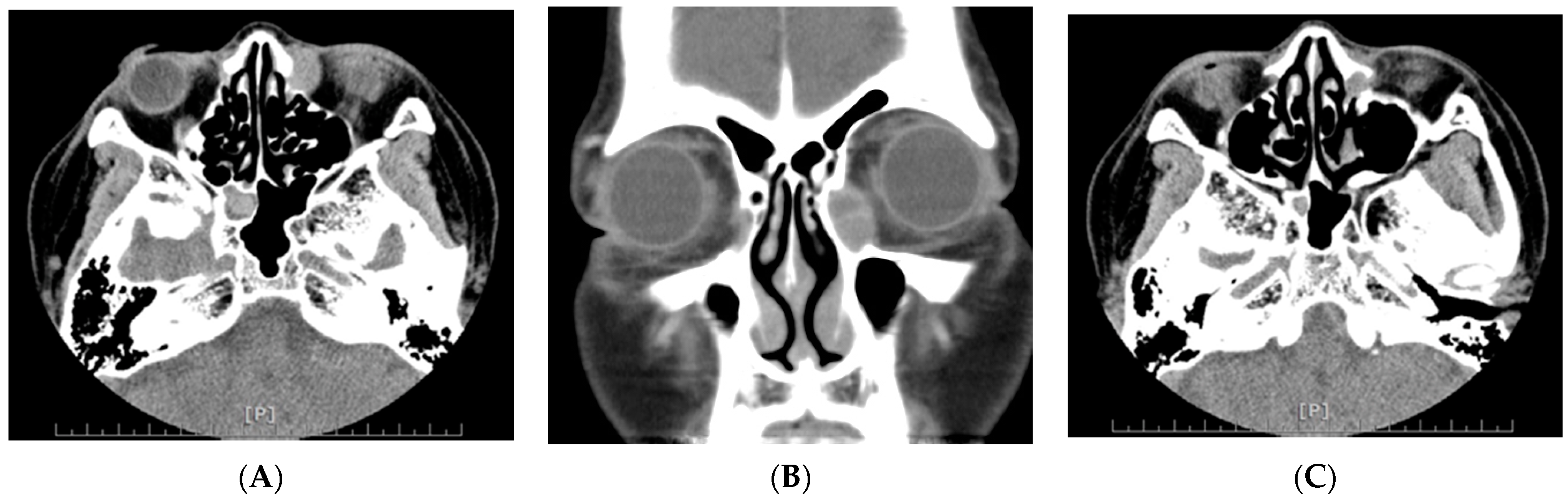
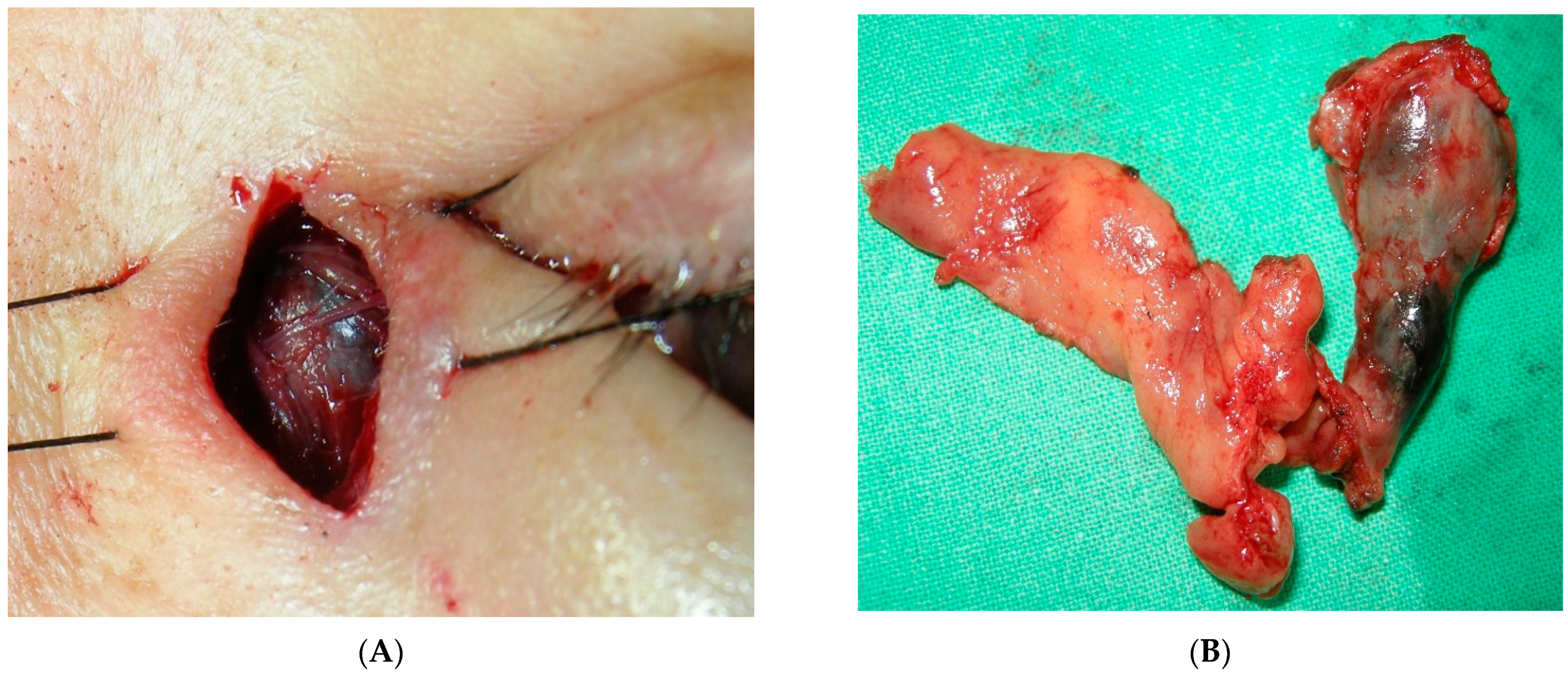
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chang, A.E.; Karnell, L.H.; Menck, H.R. The national cancer data base report on cutaneous and noncutaneous melanoma: A summary of 84,836 cases from the past decade. The American College of Surgeons Commission on Cancer and the American Cancer Society. Cancer 1998, 83, 1664–1678. [Google Scholar] [CrossRef] [Green Version]
- Kaliki, S.; Shields, C.L. Uveal melanoma: Relatively rare but deadly cancer. Eye 2017, 31, 241–257. [Google Scholar] [CrossRef] [Green Version]
- McLaughlin, C.C.; Wu, X.C.; Jemal, A.; Martin, H.J.; Roche, L.M.; Chen, V.W. Incidence of noncutaneous melanomas in the U.S. Cancer 2005, 103, 1000–1007. [Google Scholar] [CrossRef]
- Isager, P.; Østerlind, A.; Engholm, G.; Heegaard, S.; Lindegaard, J.; Overgaard, J.; Storm, H.H. Uveal and conjunctival malignant melanoma in Denmark, 1943–1997: Incidence and validation study. Ophthalmic Epidemiol. 2005, 12, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Shields, C.L.; Furuta, M.; Thangappan, A.; Nagori, S.; Mashayekhi, A.; Lally, D.R.; Kelly, C.C.; Rudich, D.S. Metastasis of uveal melanoma millimeter-by-millimeter in 8033 consecutive eyes. Arch. Ophthalmol. 2009, 127, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.L.Y.; Hong, J.; Goh, W.L.; Chang, E.W.Y.; Yang, V.S.; Poon, E.; Somasundaram, N.; Farid, M.; Chan, A.S.Y.; Chan, J.Y. Clinical features and survival outcomes of ocular melanoma in a multi-ethnic Asian cohort. Sci. Rep. 2020, 10, 16367. [Google Scholar] [CrossRef] [PubMed]
- Vajdic, C.M.; Kricker, A.; Giblin, M.; McKenzie, J.; Aitken, J.; Giles, G.G.; Armstrong, B.K. Incidence of ocular melanoma in Australia from 1990 to 1998. Int. J. Cancer 2003, 105, 117–122. [Google Scholar] [CrossRef]
- Jovanovic, P.; Mihajlovic, M.; Djordjevic-Jocic, J.; Vlajkovic, S.; Cekic, S.; Stefanovic, V. Ocular melanoma: An overview of the current status. Int. J. Clin. Exp. Pathol. 2013, 6, 1230–1244. [Google Scholar] [PubMed]
- Damato, B.E.; Coupland, S.E. Differences in uveal melanomas between men and women from the British Isles. Eye 2012, 26, 292–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yue, H.; Qian, J.; Yuan, Y.; Zhang, R.; Bi, Y.; Meng, F.; Xuan, Y. Clinicopathological Characteristics and Prognosis for Survival after Enucleation of Uveal Melanoma in Chinese Patients: Long-term Follow-up. Curr. Eye Res. 2017, 42, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Kaštelan, S.; Gverović Antunica, A.; Beketić Orešković, L.; Salopek Rabatić, J.; Kasun, B.; Bakija, I. Conjunctival Melanoma—Epidemiological Trends and Features. Pathol. Oncol. Res. 2018, 24, 787–796. [Google Scholar] [CrossRef]
- Zhou, C.; Wang, Y.; Jia, R.; Fan, X. Conjunctival Melanoma in Chinese Patients: Local Recurrence, Metastasis, Mortality, and Comparisons with Caucasian Patients. Investig. Ophthalmol. Vis. Sci. 2017, 58, 5452–5459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chan, F.M.; O’Donnell, B.A.; Whitehead, K.; Ryman, W.; Sullivan, T.J. Treatment and outcomes of malignant melanoma of the eyelid: A review of 29 cases in Australia. Ophthalmology 2007, 114, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, M.O.; McGeehan, B.; Lee, V.; Maguire, M.G.; Briceño, C.A. Primary orbital melanoma: A report of a case and comprehensive review of the literature. Orbit 2021, 40, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Xu, Y.; Lu, Y.; Chen, M.; Cao, J.; Wang, Y.; Lin, X.; Ye, J. Epidemiology and Survival Outcomes for Eyelid Primary Malignant Melanoma: An Analysis of 1397 Cases in the SEER Database. J. Ophthalmol. 2020, 2020, 4858636. [Google Scholar] [CrossRef]
- Chen, P.Y.; Liao, Y.L.; Chu, Y.C.; Tsai, Y.J. Conjunctival melanoma: A 20-year survey in a comprehensive medical center. J. Formos. Med. Assoc. 2021, 120, 250–255. [Google Scholar] [CrossRef]
- Cerman, E.; Çekiç, O. Clinical use of photodynamic therapy in ocular tumors. Surv. Ophthalmol. 2015, 60, 557–574. [Google Scholar] [CrossRef]
- Shields, C.L.; Shields, J.A.; Perez, N.; Singh, A.D.; Cater, J. Primary transpupillary thermotherapy for small choroidal melanoma in 256 consecutive cases: Outcomes and limitations. Ophthalmology 2002, 109, 225–234. [Google Scholar] [CrossRef]
- Mashayekhi, A.; Shields, C.L.; Rishi, P.; Atalay, H.T.; Pellegrini, M.; McLaughlin, J.P.; Patrick, K.A.; Morton, S.J.; Remmer, M.H.; Parendo, A.; et al. Primary transpupillary thermotherapy for choroidal melanoma in 391 cases: Importance of risk factors in tumor control. Ophthalmology 2015, 122, 600–609. [Google Scholar] [CrossRef]
- Jain, P.; Finger, P.T.; Fili, M.; Damato, B.; Coupland, S.E.; Heimann, H.; Kenawy, N.; Brouwer, N.J.; Marinkovic, M.; Van Duinen, S.G.; et al. Conjunctival melanoma treatment outcomes in 288 patients: A multicentre international data-sharing study. Br. J. Ophthalmol. 2020, 105, 1358–1364. [Google Scholar] [CrossRef]
- Yin, V.T.; Warneke, C.L.; Merritt, H.A.; Esmaeli, B. Number of excisions required to obtain clear surgical margins and prognostic value of AJCC T category for patients with eyelid melanoma. Br. J. Ophthalmol. 2014, 98, 1681–1685. [Google Scholar] [CrossRef]
- Shields, C.L.; Shields, J.A.; Gündüz, K.; Cater, J.; Mercado, G.V.; Gross, N.; Lally, B. Conjunctival melanoma: Risk factors for recurrence, exenteration, metastasis, and death in 150 consecutive patients. Arch. Ophthalmol. 2000, 118, 1497–1507. [Google Scholar] [CrossRef]
- Mancera, N.; Smalley, K.S.M.; Margo, C.E. Melanoma of the eyelid and periocular skin: Histopathologic classification and molecular pathology. Surv. Ophthalmol. 2019, 64, 272–288. [Google Scholar] [CrossRef]
- Manchegowda, P.; Singh, A.D.; Shields, C.; Kaliki, S.; Shah, P.; Gopal, L.; Rishi, P. Uveal Melanoma in Asians: A Review. Ocul. Oncol. Pathol. 2021, 7, 159–167. [Google Scholar] [CrossRef]
- Wong, J.R.; Nanji, A.A.; Galor, A.; Karp, C.L. Management of conjunctival malignant melanoma: A review and update. Expert Rev. Ophthalmol. 2014, 9, 185–204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ralli, M.; Botticelli, A.; Visconti, I.C.; Angeletti, D.; Fiore, M.; Marchetti, P.; Lambiase, A.; De Vincentiis, M.; Greco, A. Immunotherapy in the Treatment of Metastatic Melanoma: Current Knowledge and Future Directions. J. Immunol. Res. 2020, 2020, 9235638. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Vidal, C.; Fernandez-Diaz, D.; Fernandez-Marta, B.; Lago-Baameiro, N.; Pardo, M.; Silva, P.; Paniagua, L.; Blanco-Teijeiro, M.J.; Piñeiro, A.; Bande, M. Treatment of Metastatic Uveal Melanoma: Systematic Review. Cancers 2020, 12, 2557. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Piperno-Neumann, S.; Kapiteijn, E.; Chapman, P.B.; Frank, S.; Joshua, A.M.; Piulats, J.M.; Wolter, P.; Cocquyt, V.; Chmielowski, B.; et al. Selumetinib in Combination with Dacarbazine in Patients with Metastatic Uveal Melanoma: A Phase III, Multicenter, Randomized Trial (SUMIT). J. Clin. Oncol. 2018, 36, 1232–1239. [Google Scholar] [CrossRef] [Green Version]
- Shah, S.; Luke, J.J.; Jacene, H.A.; Chen, T.; Giobbie-Hurder, A.; Ibrahim, N.; Buchbinder, E.L.; McDermott, D.F.; Flaherty, K.T.; Sullivan, R.J.; et al. Results from phase II trial of HSP90 inhibitor, STA-9090 (ganetespib), in metastatic uveal melanoma. Melanoma Res. 2018, 28, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Subramaniam, S.S.; Anand, R.; Mellor, T.K.; Brennan, P.A. Primary Lacrimal Sac Melanoma with Metastatic Cervical Disease: A Review of the Literature and Case Report. J. Oral Maxillofac. Surg. 2017, 75, 1438–1441. [Google Scholar] [CrossRef]
- Flanagan, J.C.; Stokes, D.P. Lacrimal sac tumors. Ophthalmology 1978, 85, 1282–1287. [Google Scholar] [CrossRef]
- Heindl, L.M.; Junemann, A.G.; Kruse, F.E.; Holbach, L.M. Tumors of the lacrimal drainage system. Orbit 2010, 29, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.W.; Yin, J.H.; Xiang, S.T.; He, Q.; Zhou, H.; Su, W. CT and MRI findings in relapsing primary malignant melanoma of the lacrimal sac: A case report and brief literature review. BMC Ophthalmol. 2020, 20, 191. [Google Scholar] [CrossRef] [PubMed]
- Diener-West, M.; Reynolds, S.M.; Agugliaro, D.J.; Caldwell, R.; Cumming, K.; Earle, J.D.; Hawkins, B.S.; Hayman, J.A.; Jaiyesimi, I.; Jampol, L.M.; et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch. Ophthalmol. 2005, 123, 1639–1643. [Google Scholar] [PubMed]
| Tumor Origin | n (%) | Gender, n (M/F) | Median Age, Years (IQR) | Laterality, n (R/L/B) |
|---|---|---|---|---|
| Uvea | 55 (62.5%) | 26/29 | 47.0 (42–60) | 30/25/0 |
| Eyelid | 6 (6.8%) | 4/2 | 78.5 (56–82) | 3/3/0 |
| Conjunctiva | 18 (20.5%) | 11/7 | 66.0 (46–75) | 11/7/0 |
| Lacrimal sac | 1 (1.1%) | 0/1 | 57.0 (NA) | 0/1/0 |
| Orbit | 8 (9.1%) | 6/2 | 79.5 (77–85) | 4/3/1 |
| Total | 88 (100%) | 47/41 | 54.5 (44–72) | 48/39/1 |
| Tumor Origin | Treatment | Local Recurrence | Metastasis | Mortality | Follow-Up | |||
|---|---|---|---|---|---|---|---|---|
| Wide Excision | Exenteration/Enucleation | Chemotherapy/Radiation/CCRT | Gamma Knife/TTT | n (%) | n (%) | n (%) | Months Median (IQR) | |
| Uvea | 0 | 2/44 | 13/3/0 | 4/4 | 2 (4%) | 24 (44%) | 22 (40%) | 46.5 (22–84) |
| Eyelid | 3 | 1/0 | 1/0/0 | 0/0 | 2 (33%) | 2 (33%) | 2 (33%) | 76.0 (27–94) |
| Conjunctiva | 7 | 10/0 | 1/0/1 | 0/0 | 1 (6%) | 7 (39%) | 7 (39%) | 26.0 (13–31) |
| Lacrimal sac | 1 | 0/0 | 0/0/1 | 0/0 | 0 (0%) | 1 (100%) | 1 (100%) | 35.0 (NA) |
| Orbit | 5 | 2/0 | 0/4/2 | 0/0 | 3 (38%) | 4 (50%) | 4 (50%) | 18 (6–73) |
| Total | 16 | 15/44 | 15/7/4 | 4/4 | 8 (9%) | 38 (43%) | 36 (41%) | 41.0 (19–77) |
| Tumor Origin | Metastasis, n (%) | Metastasis at Dx, n | Dx to Metastasis, Months * | Metastasis to Mortality, Months | Dx to Mortality, Months |
|---|---|---|---|---|---|
| Uvea | 24 (44%) | 5 | 30.5 | 7.0 | 38.0 |
| Eyelid | 2 (33%) | 0 | 74.0 | 6.5 | 80.0 |
| Conjunctiva | 7 (39%) | 2 | 16.1 | 9.0 | 31.5 |
| Lacrimal sac | 1 (100%) | 0 | 17.0 | 18.0 | 35.0 |
| Orbit | 4 (50%) | 0 | 6.0 | 4.0 | 10.5 |
| Total | 38 (43%) | 7 | 27.2 | 7.0 | 34.0 |
| Tumor Origin | 1-Year Survival Rate | 3-Year Survival Rate | 5-Year Survival Rate | 10-Year Survival Rate |
|---|---|---|---|---|
| Uvea | 89.4% | 80.1% | 63.1% | 54.7% |
| Eyelid | 100% | 100% | 100% | 33.3% |
| Conjunctiva | 100% | 32.8% | 0% | 0% |
| Lacrimal sac | 100% | 0% | 0% | 0% |
| Orbit | 71.4% | 42.9% | 42.9% | 21.4% |
| Total | 89.8% | 73.3% | 58.4% | 43.7% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Y.-Y.; Hou, T.-Y.; Yu, W.-K.; Tsai, C.-C.; Kao, S.-C.; Hsu, W.-M.; Liu, J.-L. The Clinical Feature and Treatment Outcome of Ocular Melanoma: A 34-Year Experience in a Tertiary Referral Center. Cancers 2021, 13, 5926. https://doi.org/10.3390/cancers13235926
Huang Y-Y, Hou T-Y, Yu W-K, Tsai C-C, Kao S-C, Hsu W-M, Liu J-L. The Clinical Feature and Treatment Outcome of Ocular Melanoma: A 34-Year Experience in a Tertiary Referral Center. Cancers. 2021; 13(23):5926. https://doi.org/10.3390/cancers13235926
Chicago/Turabian StyleHuang, Yu-Yun, Tzu-Yu Hou, Wei-Kuang Yu, Chieh-Chih Tsai, Shu-Ching Kao, Wen-Ming Hsu, and Jui-Ling (Catherine) Liu. 2021. "The Clinical Feature and Treatment Outcome of Ocular Melanoma: A 34-Year Experience in a Tertiary Referral Center" Cancers 13, no. 23: 5926. https://doi.org/10.3390/cancers13235926
APA StyleHuang, Y.-Y., Hou, T.-Y., Yu, W.-K., Tsai, C.-C., Kao, S.-C., Hsu, W.-M., & Liu, J.-L. (2021). The Clinical Feature and Treatment Outcome of Ocular Melanoma: A 34-Year Experience in a Tertiary Referral Center. Cancers, 13(23), 5926. https://doi.org/10.3390/cancers13235926







