Association of FOXO3 Expression with Tumor Pathogenesis, Prognosis and Clinicopathological Features in Hepatocellular Carcinoma: A Systematic Review with Meta-Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Objectives
2.2. Literature Search Strategy
2.3. Inclusion and Exclusion Criteria
2.4. Data Extraction and Quality Assessment
2.5. Statistical Analysis
3. Results
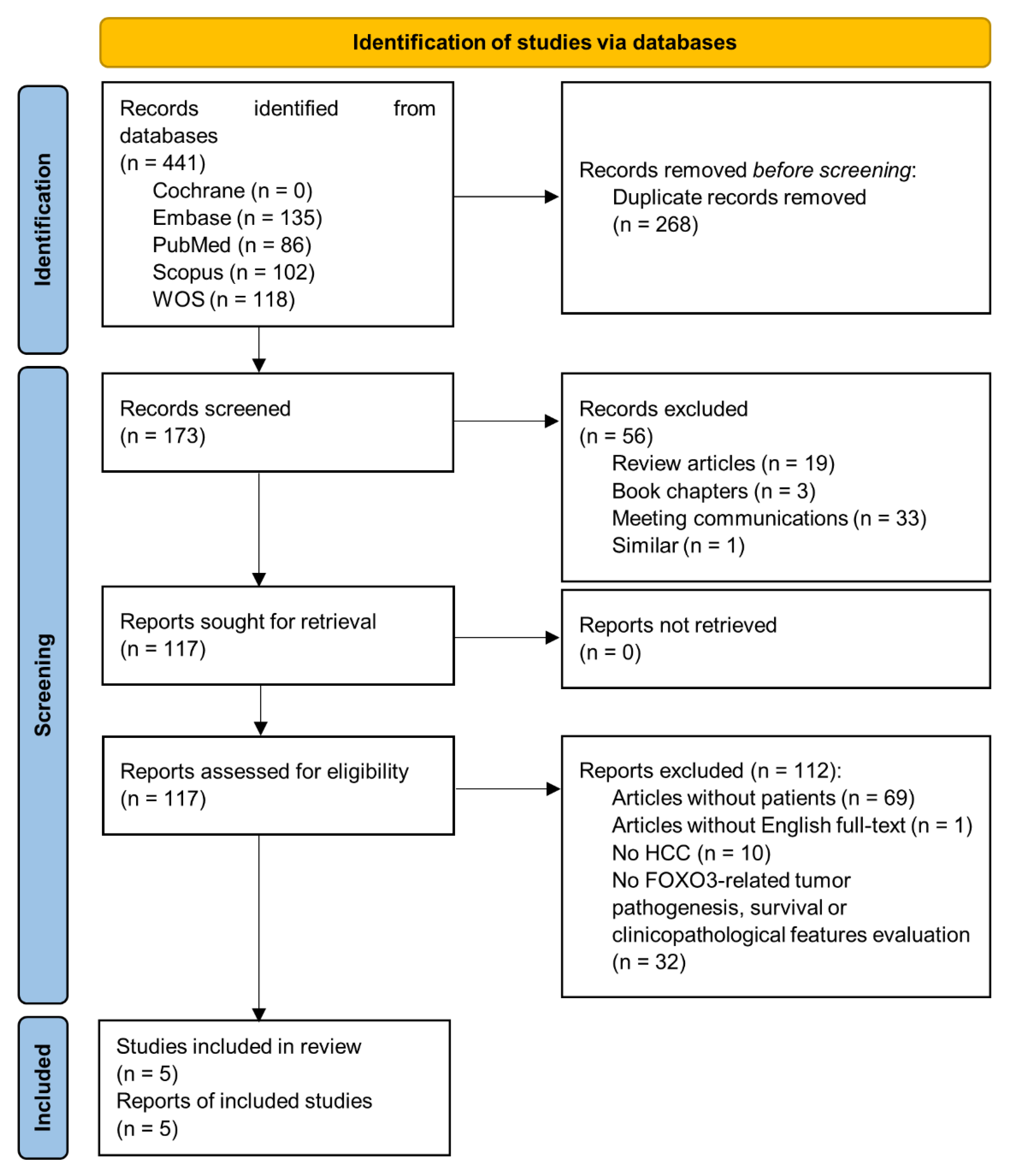
3.1. Study Characteristics
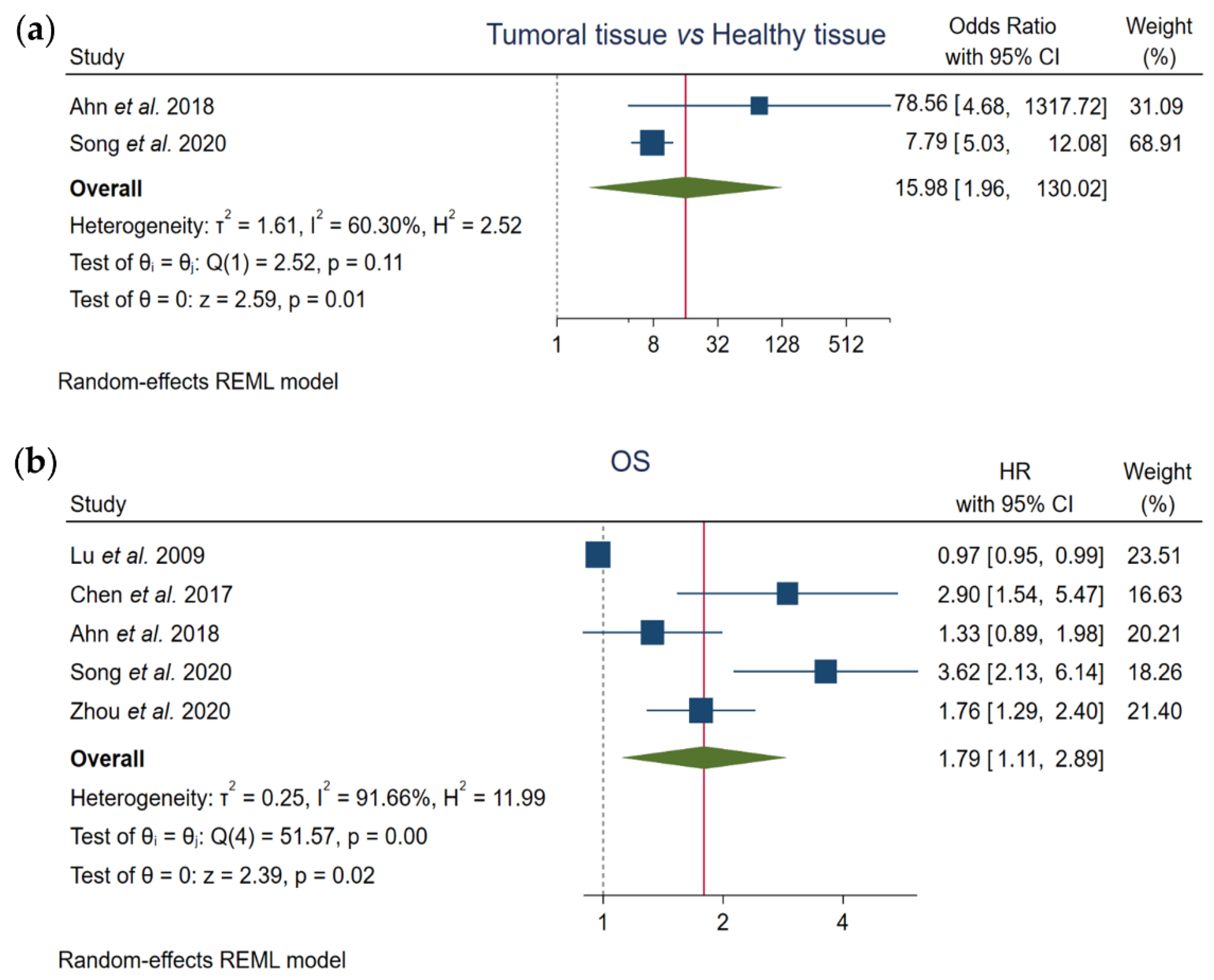
3.2. Association of FOXO3 Expression with HCC Pathogenesis
3.3. Correlation between FOXO3 Expression and OS
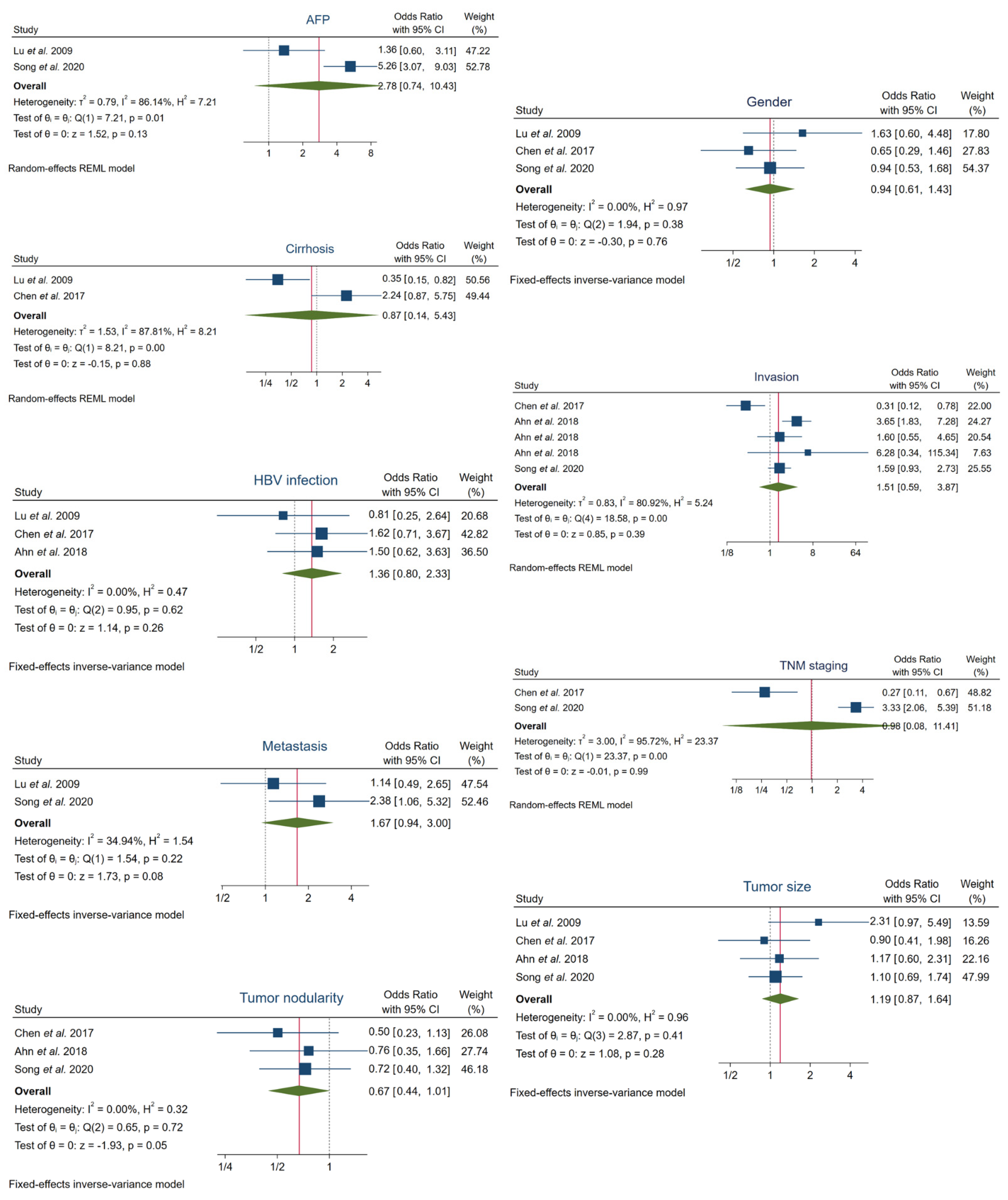
3.4. Correlation of FOXO3 Expression with Clinicopathological Features
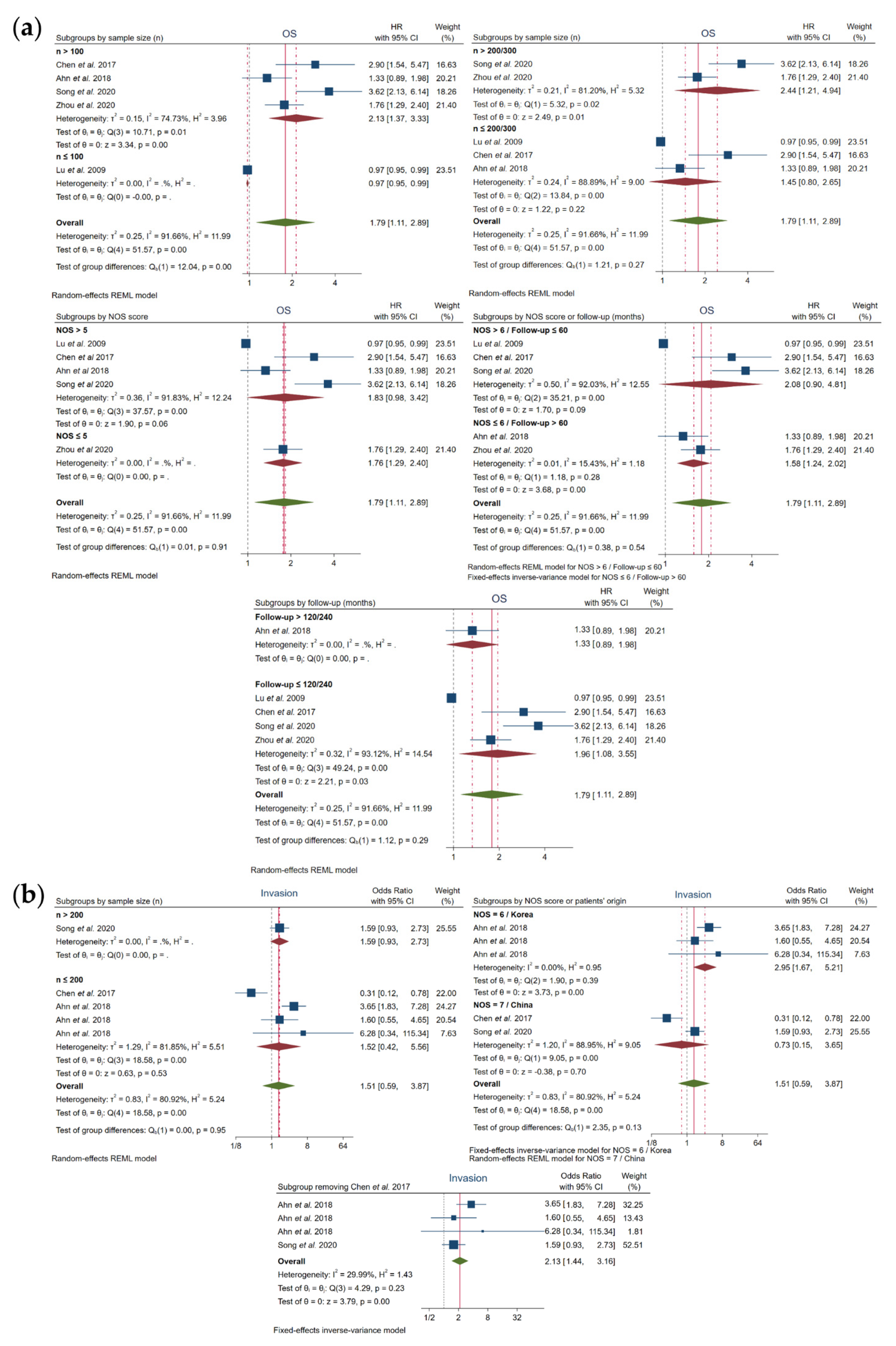
3.5. Subgroup Analysis
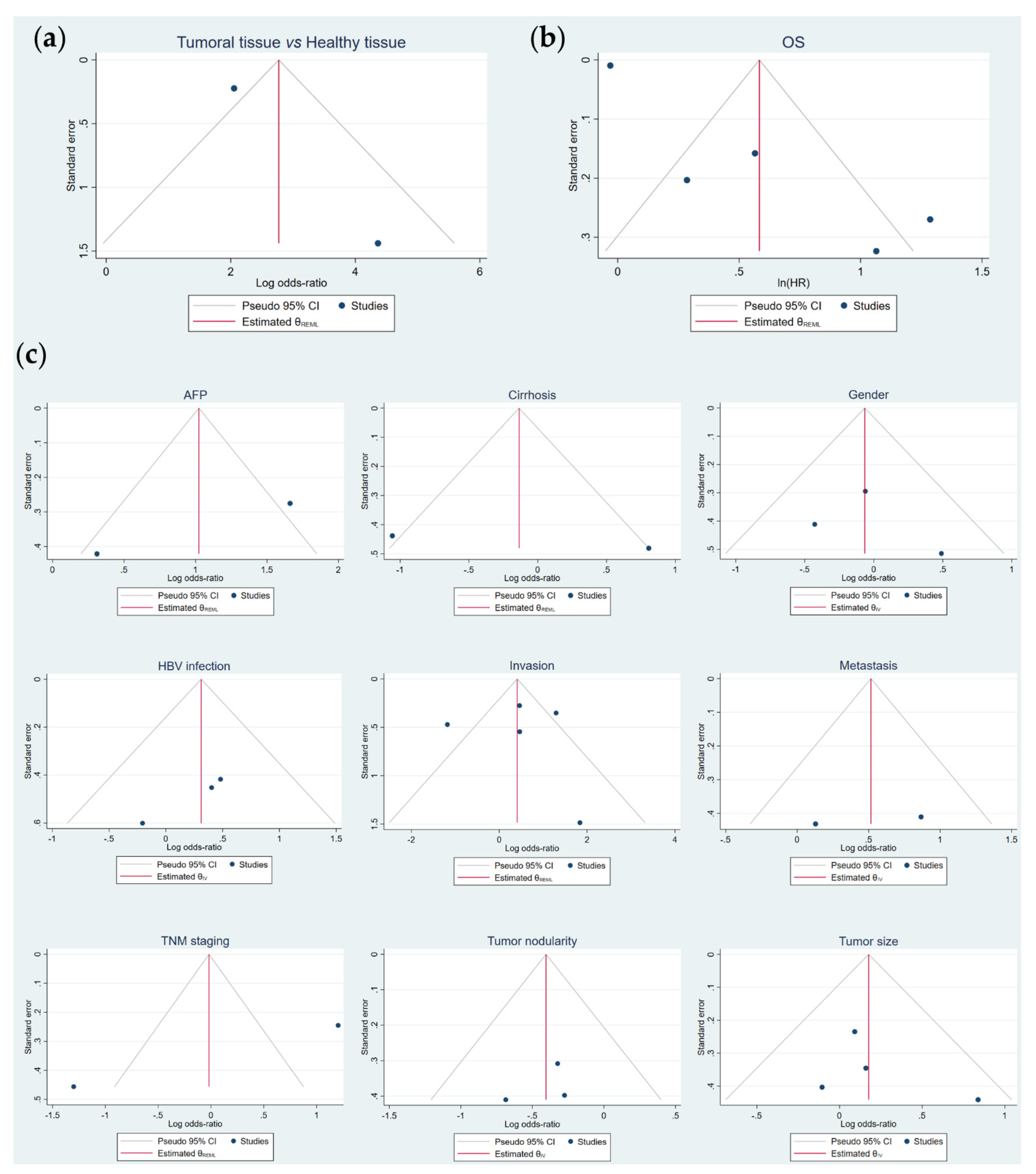
3.6. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Kelley, R.K.; Villanueva, A.; Singal, A.G.; Pikarsky, E.; Roayaie, S.; Lencioni, R.; Koike, K.; Zucman-Rossi, J.; Finn, R.S. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2021, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Forner, A.; Reig, M.; Bruix, J. Hepatocellular carcinoma. Lancet 2018, 391, 1301–1314. [Google Scholar] [CrossRef]
- Kulik, L.; El-Serag, H.B. Epidemiology and management of hepatocellular carcinoma. Gastroenterology 2019, 156, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Florio, A.A.; Znaor, A.; Ruggieri, D.; Laversanne, M.; Alvarez, C.S.; Ferlay, J.; Valery, P.C.; Bray, F.; McGlynn, K.A. International trends in hepatocellular carcinoma incidence, 1978–2012. Int. J. Cancer 2020, 147, 317–330. [Google Scholar] [CrossRef]
- Firkins, J.L.; Tarter, R.; Driessnack, M.; Hansen, L. A closer look at quality of life in the hepatocellular carcinoma literature. Qual. Life Res. 2021, 30, 1525–1535. [Google Scholar] [CrossRef] [PubMed]
- Calissi, G.; Lam, E.W.-F.; Link, W. Therapeutic strategies targeting FOXO transcription factors. Nat. Rev. Drug Discov. 2021, 20, 21–38. [Google Scholar] [CrossRef]
- Coomans De Brachène, A.; Demoulin, J.-B. FOXO transcription factors in cancer development and therapy. Cell. Mol. Life Sci. 2016, 73, 1159–1172. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Mauriz, J.L.; García-Palomo, A.; González-Gallego, J. FoxO proteins: Regulation and molecular targets in liver cancer. Curr. Med. Chem. 2014, 21, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ao, X.; Ding, W.; Ponnusamy, M.; Wu, W.; Hao, X.; Yu, W.; Wang, Y.; Li, P.; Wang, J. Critical role of FOXO3a in carcinogenesis. Mol. Cancer 2018, 17, 104. [Google Scholar] [CrossRef] [PubMed]
- Carbajo-Pescador, S.; Steinmetz, C.; Kashyap, A.; Lorenz, S.; Mauriz, J.L.; Heise, M.; Galle, P.R.; González-Gallego, J.; Strand, S. Melatonin induces transcriptional regulation of Bim by FoxO3a in HepG2 cells. Br. J. Cancer 2013, 108, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Song, S.-S.; Ying, J.-F.; Zhang, Y.-N.; Pan, H.-Y.; He, X.-L.; Hu, Z.-M.; Wang, H.-J.; Dou, X.-B.; Mou, X.-Z. High expression of FOXO3 is associated with poor prognosis in patients with hepatocellular carcinoma. Oncol. Lett. 2020, 19, 3181–3188. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qin, X.; Wu, R.; Wan, L.; Zhang, L.; Liu, R. Circular RNA circFBXO11 modulates hepatocellular carcinoma progress and oxaliplatin resistance through miR-605/FOXO3/ABCB1 axis. J. Cell. Mol. Med. 2020, 24, 5152–5161. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Dong, Z.; Cai, X.; Shen, J.; Xu, Y.; Zhang, M.; Li, H.; Yu, W.; Chen, W. Hypoxia induces sorafenib resistance mediated by autophagy via activating FOXO3a in hepatocellular carcinoma. Cell Death Dis. 2020, 11, 1017. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Li, Z.; Song, L.; Mu, D.; Wang, J.; Tian, L.; Liao, Y. Whole-exome mutational landscape of metastasis in patient-derived hepatocellular carcinoma cells. Genes Dis. 2020, 7, 380–391. [Google Scholar] [CrossRef]
- Ahn, H.; Kim, H.; Abdul, R.; Kim, Y.; Sim, J.; Choi, D.; Paik, S.S.; Shin, S.-J.; Kim, D.-H.; Jang, K. Overexpression of forkhead box O3a and its association with aggressive phenotypes and poor prognosis in human hepatocellular carcinoma. Am. J. Clin. Pathol. 2018, 149, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Pang, L.; Dai, W.; Wu, S.; Ren, T.; Duan, Y.; Zheng, Y.; Bi, S.; Zhang, X.; Kong, J. Role of forkhead box O proteins in hepatocellular carcinoma biology and progression (Review). Front. Oncol. 2021, 11, 667730. [Google Scholar] [CrossRef]
- Lin, Z.; Niu, Y.; Wan, A.; Chen, D.; Liang, H.; Chen, X.; Sun, L.; Zhan, S.; Chen, L.; Cheng, C.; et al. RNA m6A methylation regulates sorafenib resistance in liver cancer through FOXO3-mediated autophagy. EMBO J. 2020, 39, e103181. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, J.; Zhang, L.; Yuan, P.; Lei, L.; Liu, D. Decreased expression of forkhead box O3 in human hepatocellular carcinoma and its prognostic significance. Int. J. Clin. Exp. Med. 2017, 10, 5851–5857. [Google Scholar]
- Lu, M.; Ma, J.; Xue, W.; Cheng, C.; Wang, Y.; Zhao, Y.; Ke, Q.; Liu, H.; Liu, Y.; Li, P.; et al. The expression and prognosis of FOXO3a and Skp2 in human hepatocellular carcinoma. Pathol. Oncol. Res. 2009, 15, 679–687. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 6 May 2021).
- Parmar, M.K.B.; Torri, V.; Stewart, L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat. Med. 1998, 17, 2815–2834. [Google Scholar] [CrossRef]
- Méndez-Blanco, C.; Fernández-Palanca, P.; Fondevila, F.; González-Gallego, J.; Mauriz, J.L. Prognostic and clinicopathological significance of hypoxia-inducible factors 1α and 2α in hepatocellular carcinoma: A systematic review with meta-analysis. Ther. Adv. Med. Oncol. 2021, 13, 1758835920987071. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, V.; Razzaghi, M.; Nikzamir, A.; Ahmadzadeh, A.; Iranshahi, M.; Haghazali, M.; Hamdieh, M. Assessment of liver cancer biomarkers. Gastroenterol. Hepatol. Bed Bench 2020, 13, S29–S39. [Google Scholar] [PubMed]
- Sukowati, C.H.C.; Cabral, L.K.D.; Tiribelli, C.; Pascut, D. Circulating long and circular noncoding RNA as non-invasive diagnostic tools of hepatocellular carcinoma. Biomedicines 2021, 9, 90. [Google Scholar] [CrossRef] [PubMed]
- Pratama, M.Y.; Visintin, A.; Crocè, L.S.; Tiribelli, C.; Pascut, D. Circulatory miRNA as a biomarker for therapy response and disease-free survival in hepatocellular carcinoma. Cancers 2020, 12, 2810. [Google Scholar] [CrossRef] [PubMed]
- Gingold, J.A.; Zhu, D.; Lee, D.-F.; Kaseb, A.; Chen, J. Genomic profiling and metabolic homeostasis in primary liver cancers. Trends Mol. Med. 2018, 24, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Hartmann, D.; Braren, R.; Gupta, A.; Wang, B.; Wang, Y.; Mogler, C.; Cheng, Z.; Wirth, T.; Friess, H.; et al. Oncogenic Akt-FOXO3 loop favors tumor-promoting modes and enhances oxidative damage-associated hepatocellular carcinogenesis. BMC Cancer 2019, 19, 887. [Google Scholar] [CrossRef]
- Yang, L.; Deng, W.; Zhao, B.; Xu, Y.; Wang, X.; Fang, Y.; Xiao, H. FOXO3-induced lncRNA LOC554202 contributes to hepatocellular carcinoma progression via the miR-485-5p/BSG axis. Cancer Gene Ther. 2021. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Du, H.; Zhan, M.; Wang, H.; Chen, P.; Du, D.; Liu, X.; Huang, X.; Ma, P.; Peng, D.; et al. Sepiapterin reductase promotes hepatocellular carcinoma progression via FoxO3a/Bim signaling in a nonenzymatic manner. Cell Death Dis. 2020, 11, 248. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Gomes, A.R.; Monteiro, L.J.; Wong, S.Y.; Wu, L.H.; Ng, T.T.; Karadedou, C.T.; Millour, J.; Ip, Y.-C.; Cheung, Y.N.; et al. Constitutively nuclear FOXO3a localization predicts poor survival and promotes Akt phosphorylation in breast cancer. PLoS ONE 2010, 5, e12293. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Ren, L.; Wu, D.; Yang, X.; Zhou, Z.; Nie, Q.; Jiang, G.; Xue, S.; Weng, W.; Qiu, Y.; et al. Overexpression of FoxO3a is associated with glioblastoma progression and predicts poor patient prognosis. Int. J. Cancer 2017, 140, 2792–2804. [Google Scholar] [CrossRef] [PubMed]
- Rehman, A.; Kim, Y.; Kim, H.; Sim, J.; Ahn, H.; Chung, M.S.; Shin, S.-J.; Jang, K. FOXO3a expression is associated with lymph node metastasis and poor disease-free survival in triple-negative breast cancer. J. Clin. Pathol. 2018, 71, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Dai, Y.; Dai, T.; Xie, T.; Su, X.; Li, J.; Zhou, X.; Meng, K.; Zhao, X. TRIP6 promotes cell proliferation in hepatocellular carcinoma via suppression of FOXO3a. Biochem. Biophys. Res. Commun. 2017, 494, 594–601. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Yu, Y.; Zhang, W.; Yuan, W.; Zhao, N.; Li, Q.; Cui, Y.; Wang, Y.; Li, W.; Sun, Y.; et al. FOXO3a promotes gastric cancer cell migration and invasion through the induction of cathepsin L. Oncotarget 2016, 7, 34773–34784. [Google Scholar] [CrossRef]
- Storz, P.; Döppler, H.; Copland, J.A.; Simpson, K.J.; Toker, A. FOXO3a promotes tumor cell invasion through the induction of matrix metalloproteinases. Mol. Cell. Biol. 2009, 29, 4906–4917. [Google Scholar] [CrossRef]
- Sisci, D.; Maris, P.; Cesario, M.G.; Anselmo, W.; Coroniti, R.; Trombino, G.E.; Romeo, F.; Ferraro, A.; Lanzino, M.; Aquila, S.; et al. The estrogen receptor α is the key regulator of the bifunctional role of FoxO3a transcription factor in breast cancer motility and invasiveness. Cell Cycle 2013, 12, 3405–3420. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Liu, S.; Zhu, M.; Zhang, H.; Wang, J.; Xu, Q.; Lin, K.; Zhou, X.; Tao, M.; Li, C.; et al. PS341 inhibits hepatocellular and colorectal cancer cells through the FOXO3/CTNNB1 signaling pathway. Sci. Rep. 2016, 6, 22090. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Jiang, J.; Gong, L.; Shu, Z.; Xiang, D.; Zhang, X.; Bi, K.; Diao, H. Hepatitis B virus P protein initiates glycolytic bypass in HBV-related hepatocellular carcinoma via a FOXO3/miRNA-30b-5p/MINPP1 axis. J. Exp. Clin. Cancer Res. 2021, 40, 1. [Google Scholar] [CrossRef] [PubMed]
- Shou, Z.; Lin, L.; Liang, J.; Li, J.-L.; Chen, H.-Y. Expression and prognosis of FOXO3a and HIF-1α in nasopharyngeal carcinoma. J. Cancer Res. Clin. Oncol. 2012, 138, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Bullock, M.D.; Bruce, A.; Sreekumar, R.; Curtis, N.; Cheung, T.; Reading, I.; Primrose, J.N.; Ottensmeier, C.; Packham, G.K.; Thomas, G.; et al. FOXO3 expression during colorectal cancer progression: Biomarker potential reflects a tumour suppressor role. Br. J. Cancer 2013, 109, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Yu, J.; Yang, Z.; Zhu, G.; Gao, P.; Wang, H.; Chen, S.; Zhang, J.; Liu, M.; Niu, Y.; et al. Promoter hypomethylation mediated upregulation of MicroRNA-10b-3p targets FOXO3 to promote the progression of esophageal squamous cell carcinoma (ESCC). J. Exp. Clin. Cancer Res. 2018, 37, 301. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Yang, Z.; Liu, X.; Liu, Z.; Miao, X.; Li, D.; Zou, Q.; Yuan, Y. The clinicopathological significance of forkhead box P1 and forkhead box O3a in pancreatic ductal adenocarcinomas. Tumor Biol. 2017, 39, 1010428317699129. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Kang, X.-L.; Zeng, F.-C.; Xu, C.-J.; Zhou, J.-Q.; Luo, D.-N. Correlations of Foxo3 and Foxo4 expressions with clinicopathological features and prognosis of bladder cancer. Pathol. Res. Pract. 2017, 213, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Tenbaum, S.P.; Ordóñez-Morán, P.; Puig, I.; Chicote, I.; Arqués, O.; Landolfi, S.; Fernández, Y.; Herance, J.R.; Gispert, J.D.; Mendizabal, L.; et al. β-Catenin confers resistance to PI3K and AKT inhibitors and subverts FOXO3a to promote metastasis in colon cancer. Nat. Med. 2012, 18, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhao, J.; Huang, C.; Wang, Q.; Pan, K.; Wang, D.; Pan, Q.; Jiang, S.; Lv, L.; Gao, X.; et al. Decreased expression of the FOXO3a gene is associated with poor prognosis in primary gastric adenocarcinoma patients. PLoS ONE 2013, 8, e78158. [Google Scholar] [CrossRef] [PubMed]
| Database | Search Strategy |
|---|---|
| Cochrane Library | (foxo3 OR foxo3a OR fkhrl1 OR Forkhead box protein O3 OR Forkhead box O3 OR forkhead box O-3 OR AF6q21 OR Forkhead in rhabdomyosarcoma-like 1 OR fkhrl1p2 OR foxo2a) AND (hepatocellular carcinoma OR hepatocarcinoma OR HCC): ti, ab, kw |
| Embase | (‘foxo3’ OR ‘foxo3a’ OR ‘fkhrl1′ OR ‘forkhead box protein o3′ OR ‘forkhead box o3’ OR ‘forkhead box o-3’ OR ‘af6q21’ OR ‘forkhead in rhabdomyosarcoma-like 1′ OR ‘fkhrl1p2’ OR ‘foxo2a’) AND (‘hepatocellular carcinoma’ OR ‘hepatocarcinoma’ OR ‘hcc’) |
| PubMed | (“foxo3”[All Fields] OR “foxo3a”[All Fields] OR “fkhrl1”[All Fields] OR “Forkhead box protein O3”[All Fields] OR “Forkhead box O3”[All Fields] OR “forkhead box O-3”[All Fields] OR “AF6q21”[All Fields] OR “Forkhead in rhabdomyosarcoma-like 1”[All Fields] OR “fkhrl1p2”[All Fields] OR “foxo2a”[All Fields]) AND (“hepatocellular carcinoma”[All Fields] OR “hepatocarcinoma”[All Fields] OR “HCC”[All Fields]) |
| Scopus | TITLE-ABS-KEY ((“foxo3” OR “foxo3a” OR “fkhrl1” OR “Forkhead box protein O3” OR “Forkhead box O3” OR “forkhead box O-3” OR “AF6q21” OR “Forkhead in rhabdomyosarcoma-like 1” OR “fkhrl1p2” OR “foxo2a”) AND (“hepatocellular carcinoma” OR “hepatocarcinoma” OR “HCC”)) |
| WOS Core Collection | TS = ((“foxo3” OR “foxo3a” OR “fkhrl1” OR “Forkhead box protein O3” OR “Forkhead box O3” OR “forkhead box O-3” OR “AF6q21” OR “Forkhead in rhabdomyosarcoma-like 1” OR “fkhrl1p2” OR “foxo2a”) AND (“hepatocellular carcinoma” OR “hepatocarcinoma” OR “HCC”)) Indexes = SCI-EXPANDED, SSCI, A&HCI, CPCI-S, CPCI-SSH, BKCI-S, BKCI-SSH, ESCI, CCR-EXPANDED, IC Timespan = All years |
| Study | Lu et al. [20] | Chen et al. [19] | Ahn et al. [16] | Song et al. [12] | Zhou et al. [15] |
|---|---|---|---|---|---|
| Year | 2009 | 2017 | 2018 | 2020 | 2020 |
| Tumor sample size | 91 (74) * | 102 | 187 | 314 | 365 |
| Tumor sample size (M/F) | 71/20 (61/13) * | 62/40 | NR | 255/59 | NR |
| Patients’ origin | China | China | Korea | China | NR |
| Intervention | Surgical resection | Surgical resection | Surgical resection | Surgical resection | NR |
| Pre- or post-surgery treatment | None | None | NR | NR | NR |
| Age range | 32–72 | NR | NR | NR | NR |
| Mean/median age | 51.37 ± 10.50 | NR | NR | NR | NR |
| Study quality | 7/9 | 7/9 | 6/9 | 7/9 | 5/9 |
| FOXO3 levels measurement | IHC | IHC | IHC | IHC | RNA-Seq ** |
| Survival analysis | CS | OS | OS/DFS | OS | OS |
| HR | Reported | Reported | Reported | Estimated | Estimated |
| Healthy liver sample size | NR | NR | 21 | 150 | NR |
| Definition of “high” FOXO3 expression | >0.40 | >4 1 | ≥1 2 | ≥3 3 | NR |
| Number of tumor samples with “high” FOXO3 expression | 45 (37) * | 42 | 121 | 238 | 91 |
| Number of healthy liver samples with “high” FOXO3 expression | NR | NR | 0 | 43 | NR |
| Parameter | Number of Studies (n) | Number of Cases (n) | Samples with High FOXO3 Expression (n) | High FOXO3 Expression (%) | Pooled OR or HR | Test for Heterogeneity | Model Used | ||
|---|---|---|---|---|---|---|---|---|---|
| 95% CI | p-Value | I2 | Q-Test p-Value | ||||||
| HCC pathogenesis | |||||||||
| Tumoral tissue vs. Healthy tissue | 2 | 672 | 402 | 59.82% | 15.98 (1.96–130.02) | 0.01 | 60.30% | 0.11 | REM |
| OS | 5 | 1042 | 529 | 50.77% | 1.79 (1.11–2.89) | 0.02 | 91.66% | 0.00 | REM |
| Clinicopathological features | |||||||||
| AFP | 2 | 346 | 153 | 44.22% | 2.78 (0.74–10.43) | 0.13 | 86.14% | 0.01 | REM |
| Cirrhosis | 2 | 193 | 87 | 45.08% | 0.87 (0.14–5.43) | 0.88 | 87.81% | 0.00 | REM |
| Gender | 3 | 507 | 211 | 41.62% | 0.94 (0.61–1.43) | 0.76 | 0.00% | 0.38 | FEM |
| HBV infection | 3 | 378 | 207 | 54.76% | 1.36 (0.80–2.33) | 0.26 | 0.00% | 0.62 | FEM |
| Invasion | 5 | 890 | 497 | 55.84% | 1.51 (0.59–3.87) | 0.39 | 80.92% | 0.00 | REM |
| Metastasis | 2 | 400 | 168 | 42.00% | 1.67 (0.94–3.00) | 0.08 | 34.94% | 0.22 | FEM |
| TNM staging | 2 | 414 | 166 | 40.10% | 0.98 (0.08–11.41) | 0.99 | 95.72% | 0.00 | REM |
| Tumor nodularity | 3 | 603 | 287 | 47.60% | 0.67 (0.44–1.01) | 0.054 | 0.00% | 0.72 | FEM |
| Tumor size | 4 | 687 | 318 | 46.29% | 1.19 (0.87–1.64) | 0.28 | 0.00% | 0.41 | FEM |
| Subgroups | Number of Studies (n) | Number of Cases (n) | Samples with High FOXO3 Expression (n) | High FOXO3 Expression (%) | Pooled OR or HR | Test for Heterogeneity | Model Used | ||
|---|---|---|---|---|---|---|---|---|---|
| 95% CI | p-Value | I2 | Q-Test p-Value | ||||||
| OS | |||||||||
| Sample size (n) | |||||||||
| n > 100 | 4 | 968 | 492 | 50.83% | 2.13 (1.37–3.33) | 0.00 | 74.73% | 0.01 | REM |
| n ≤ 100 | 1 | 74 | 37 | 50.00% | 0.97 (0.95–0.99) | - | - | - | - |
| n > 200/300 | 2 | 679 | 329 | 48.45% | 2.44 (1.21–4.94) | 0.01 | 81.20% | 0.02 | REM |
| n ≤ 200/300 | 3 | 363 | 200 | 55.10% | 1.45 (0.80–2.65) | 0.22 | 88.89% | 0.00 | REM |
| NOS score (threshold 5) | |||||||||
| NOS > 5 | 4 | 677 | 438 | 64.70% | 1.83 (0.98–3.42) | 0.06 | 91.83% | 0.00 | REM |
| NOS ≤ 5 | 1 | 365 | 91 | 24.93% | 1.76 (1.29–2.40) | - | - | - | - |
| NOS score (threshold 6) | |||||||||
| NOS > 6 | 3 | 490 | 317 | 64.69% | 2.08 (0.90–4.81) | 0.09 | 92.03% | 0.00 | REM |
| NOS ≤ 6 | 2 | 552 | 212 | 38.41% | 1.58 (1.24–2.02) | 0.00 | 15.43% | 0.28 | FEM |
| Follow-up (months) | |||||||||
| >60 | 2 | 552 | 212 | 38.41% | 1.58 (1.24–2.02) | 0.00 | 15.43% | 0.28 | FEM |
| ≤60 | 3 | 490 | 317 | 64.69% | 2.08 (0.90–4.81) | 0.09 | 92.03% | 0.00 | REM |
| >120/240 | 1 | 187 | 121 | 64.71% | 1.33 (0.89–1.98) | - | - | - | - |
| ≤120/240 | 4 | 855 | 408 | 47.72% | 1.96 (1.08–3.55) | 0.03 | 93.12% | 0.00 | REM |
| Invasion | |||||||||
| Sample size (n) | |||||||||
| n > 200 | 1 | 227 | 92 | 40.53% | 1.59 (0.93–2.73) | - | - | - | - |
| n ≤ 200 | 4 | 663 | 405 | 61.09% | 1.52 (0.42–5.56) | 0.53 | 81.85% | 0.00 | REM |
| NOS score | |||||||||
| NOS = 6 | 3 | 561 | 363 | 64.71% | 2.95 (1.67–5.21) | 0.00 | 0.00% | 0.39 | FEM |
| NOS = 7 | 2 | 329 | 134 | 40.73% | 0.73 (0.15–3.65) | 0.70 | 88.95% | 0.00 | REM |
| Patients’ origin | |||||||||
| China | 2 | 329 | 134 | 40.73% | 0.73 (0.15–3.65) | 0.70 | 88.95% | 0.00 | REM |
| Korea | 3 | 561 | 363 | 64.71% | 2.95 (1.67–5.21) | 0.00 | 0.00% | 0.39 | FEM |
| Without Chen et al. [19] | |||||||||
| 4 | 788 | 455 | 57.74% | 2.13 (1.44–3.16) | 0.00 | 29.99% | 0.23 | FEM | |
| Parameter | Number of Studies (n) | Egger’s Test p-Value | Model Used | Trim-and-Fill OR or HR (95% CI) | Imputed Studies(n) |
|---|---|---|---|---|---|
| HCC pathogenesis | |||||
| Tumoral tissue vs. Healthy tissue | 2 | * | REM | - | - |
| OS | 5 | 0.00 | REM | 1.79 (1.11–2.89) | 0 |
| Clinicopathological features | |||||
| AFP | 2 | * | REM | - | - |
| Cirrhosis | 2 | * | REM | - | - |
| Gender | 3 | 0.59 | FEM | - | - |
| HBV infection | 3 | 0.33 | FEM | - | - |
| Invasion | 5 | 0.57 | REM | - | - |
| Metastasis | 2 | 0.22 | FEM | - | - |
| TNM staging | 2 | * | REM | - | - |
| Tumor nodularity | 3 | 0.67 | FEM | - | - |
| Tumor size | 4 | 0.43 | FEM | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fondevila, F.; Fernández-Palanca, P.; Méndez-Blanco, C.; Payo-Serafín, T.; Lozano, E.; Marin, J.J.G.; González-Gallego, J.; Mauriz, J.L. Association of FOXO3 Expression with Tumor Pathogenesis, Prognosis and Clinicopathological Features in Hepatocellular Carcinoma: A Systematic Review with Meta-Analysis. Cancers 2021, 13, 5349. https://doi.org/10.3390/cancers13215349
Fondevila F, Fernández-Palanca P, Méndez-Blanco C, Payo-Serafín T, Lozano E, Marin JJG, González-Gallego J, Mauriz JL. Association of FOXO3 Expression with Tumor Pathogenesis, Prognosis and Clinicopathological Features in Hepatocellular Carcinoma: A Systematic Review with Meta-Analysis. Cancers. 2021; 13(21):5349. https://doi.org/10.3390/cancers13215349
Chicago/Turabian StyleFondevila, Flavia, Paula Fernández-Palanca, Carolina Méndez-Blanco, Tania Payo-Serafín, Elisa Lozano, Jose J. G. Marin, Javier González-Gallego, and José L. Mauriz. 2021. "Association of FOXO3 Expression with Tumor Pathogenesis, Prognosis and Clinicopathological Features in Hepatocellular Carcinoma: A Systematic Review with Meta-Analysis" Cancers 13, no. 21: 5349. https://doi.org/10.3390/cancers13215349
APA StyleFondevila, F., Fernández-Palanca, P., Méndez-Blanco, C., Payo-Serafín, T., Lozano, E., Marin, J. J. G., González-Gallego, J., & Mauriz, J. L. (2021). Association of FOXO3 Expression with Tumor Pathogenesis, Prognosis and Clinicopathological Features in Hepatocellular Carcinoma: A Systematic Review with Meta-Analysis. Cancers, 13(21), 5349. https://doi.org/10.3390/cancers13215349











