CDK4/6 Inhibitor Treatments in Patients with Hormone Receptor Positive, Her2 Negative Advanced Breast Cancer: Potential Molecular Mechanisms, Clinical Implications and Future Perspectives
Abstract
Simple Summary
Abstract
1. Introduction
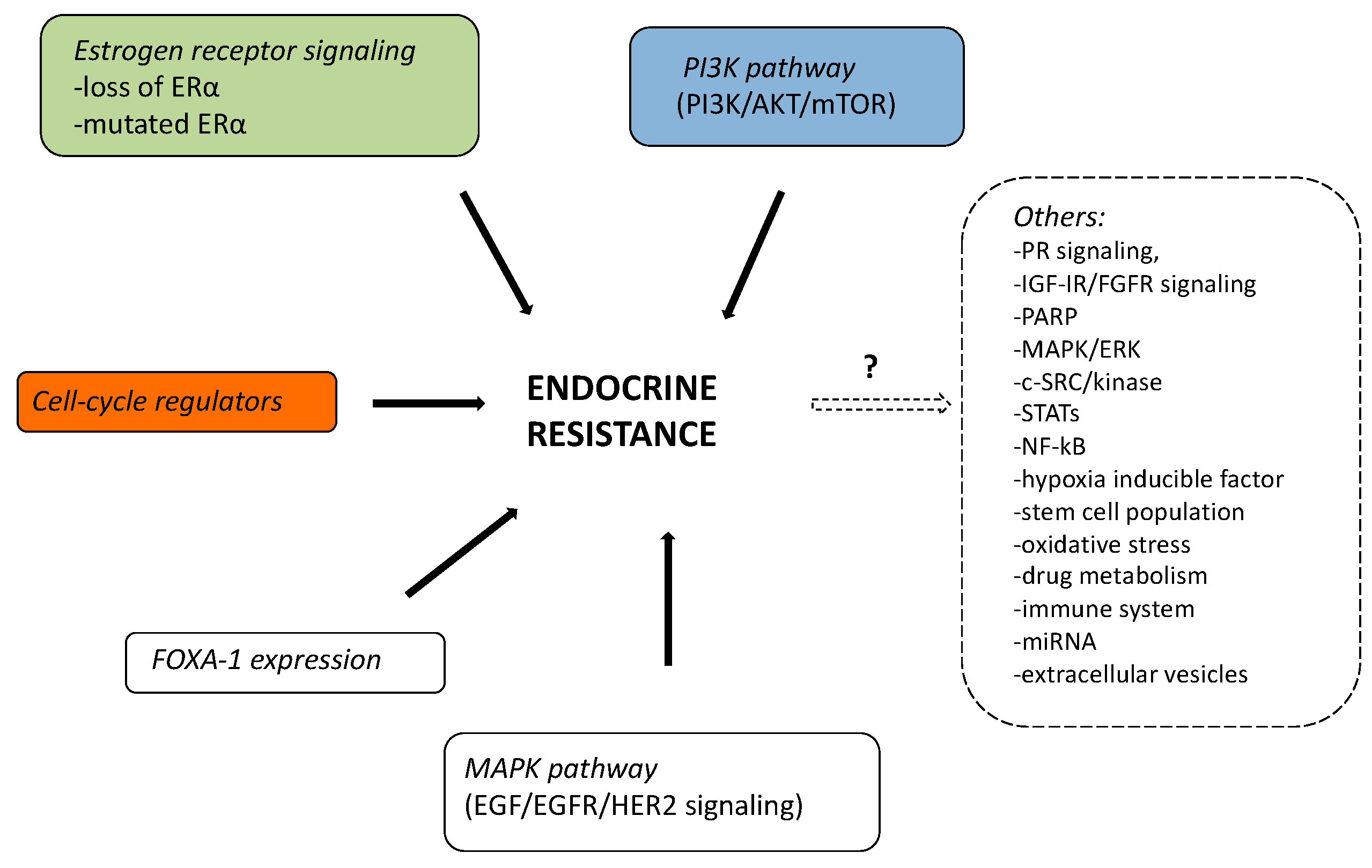
1.1. Mechanism of Endocrine Therapy Resistance
1.1.1. Mutations of ER-α
1.1.2. Loss of ER-α
1.1.3. MAPK Pathway (EGF/EGFR/HER2 Signaling)
1.1.4. PI3K Pathway (PI3K/AKT/mTOR)
1.1.5. FOXA-1 Expression
1.1.6. Cell-Cycle Regulators and Endocrine Resistance
1.2. Clinical Implications
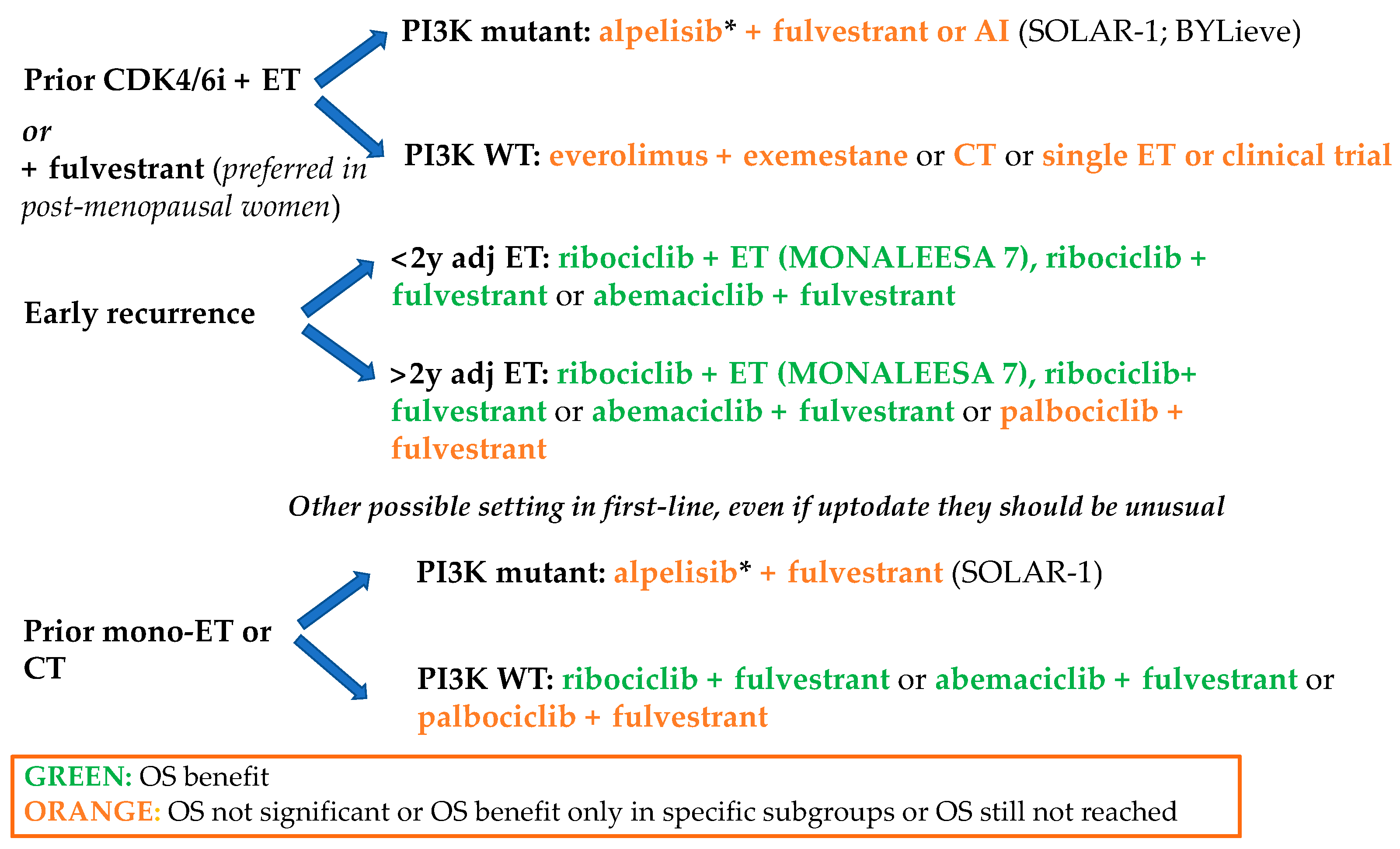
1.2.1. First Line
1.2.2. Second Line and Early Relapse
2. Future Perspectives
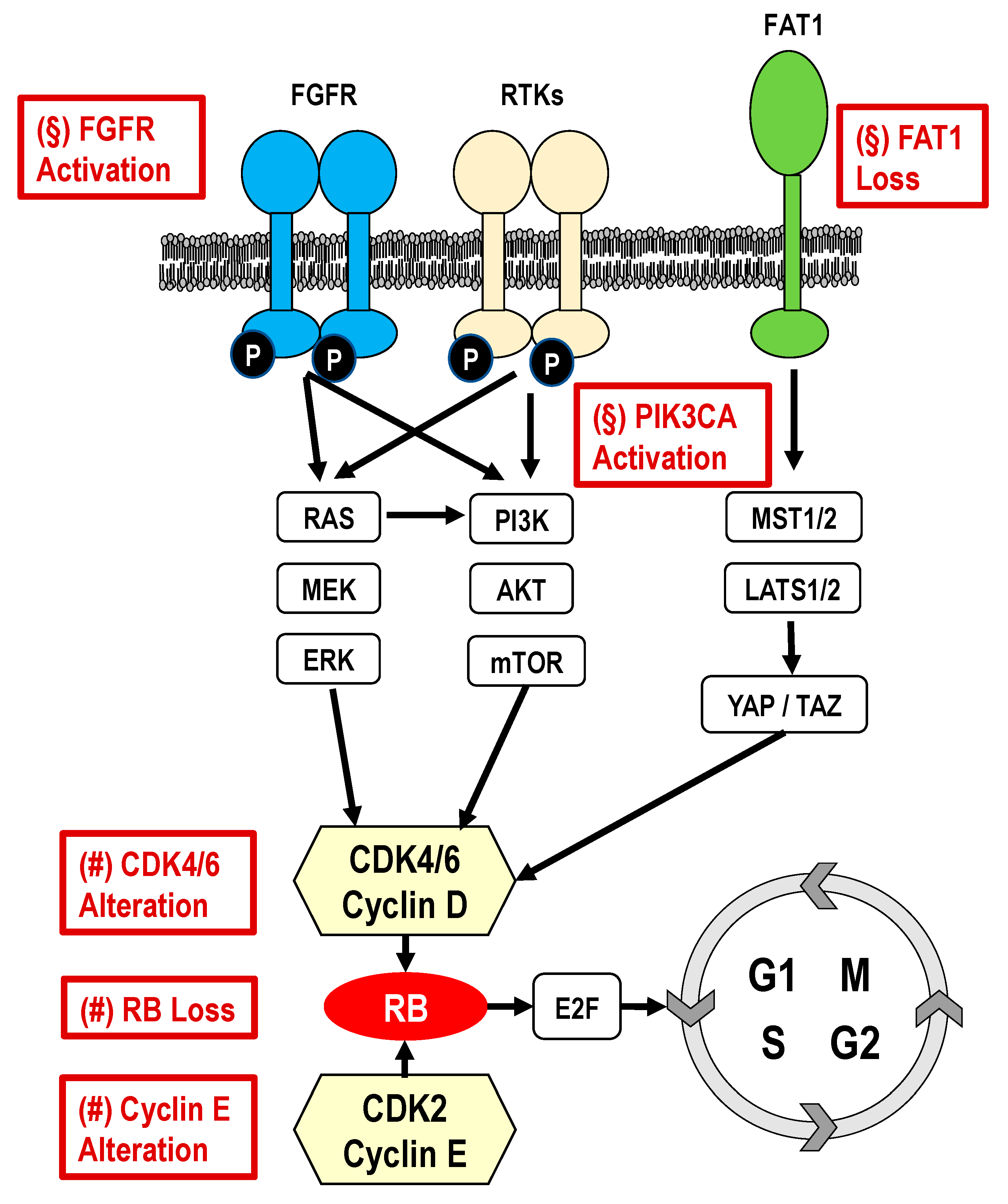
2.1. Cell-cycle-Specific Mechanisms of Resistance to CDK4/6i
- RB loss
- CCND1 amplification/p16 loss
- CCNE1/2 amplification/overexpression
- CDK4/6 amplification/overxpression
2.2. Cell-cycle-Nonspecific Mechanisms of Resistance to CDK4/6 Inhibitors
- PIK3CA pathway activation
- FGFR pathway activation
- MDM2 overexpression
- ESR1 expression and mutation
- PD-1 expression
- Thymidine kinase-1 (TK1)
- FAT1 loss
- Autophagy activation
2.3. Other Potential Mechanism of Resistance
2.4. Strategies after CDK4/6 Inhibitor Progression and New Therapeutic Combinations
3. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Razavi, P.; Chang, M.T.; Xu, G.; Bandlamudi, C.; Ross, D.S.; Vasan, N.; Cai, Y.; Bielski, C.M.; Donoghue, M.T.A.; Jonsson, P.; et al. The Genomic Landscape of Endocrine-Resistant Advanced Breast Cancers. Cancer Cell 2018, 34, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Musgrove, E.A.; Sutherland, R.L. Biological determinants of endocrine resistance in breast cancer. Nat. Rev. Cancer 2009, 9, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.G.; Dickler, M.N. Endocrine resistance in hormone-responsive breast cancer: Mechanisms and therapeutic strategies. Endocr. Relat. Cancer 2016, 23, 337–352. [Google Scholar] [CrossRef] [PubMed]
- Toy, W.; Shen, Y.; Won, H.; Green, B.; Sakr, R.A.; Will, M.; Li, Z.; Gala, K.; Fanning, S.; King, T.A.; et al. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat. Genet. 2013, 45, 1439–1445. [Google Scholar] [CrossRef]
- Hayes, E.L.; Lewis-Wambi, J.S. Mechanisms of endocrine resistance in breast cancer: An overview of the proposed roles of noncoding RNA. Breast Cancer Res. 2015, 17, 40. [Google Scholar] [CrossRef]
- Giacinti, L.; Claudio, P.P.; Lopez, M.; Giordano, A. Epigenetic Information and Estrogen Receptor Alpha Expression in Breast Cancer. Oncologist 2006, 11, 1–8. [Google Scholar] [CrossRef]
- Fan, A.X.C.; Radpour, R.; Haghighi, M.M.; Kohler, C.; Xia, P.; Hahn, S.; Holzgreve, W.; Zhong, X.Y. Mitochondrial DNA content in paired normal and cancerous breast tissue samples from patients with breast cancer. J. Cancer Res. Clin. Oncol. 2009, 135, 983–989. [Google Scholar] [CrossRef]
- Leary, A.F.; Drury, S.; Detre, S.; Pancholi, S.; Lykkesfeldt, A.E.; Martin, L.A.; Dowsett, M.; Johnston, S.R.D. Lapatinib restores hormone sensitivity with differential effects on estrogen receptor signaling in cell models of human epidermal growth factor receptor 2-negative breast cancer with acquired endocrine resistance. Clin. Cancer Res. 2010, 16, 1486–1497. [Google Scholar] [CrossRef]
- Miller, T.W.; Hennessy, B.T.; González-Angulo, A.M.; Fox, E.M.; Mills, G.B.; Chen, H.; Higham, C.; García-Echeverría, C.; Shyr, Y.; Arteaga, C.L. Hyperactivation of phosphatidylinositol-3 kinase promotes escape from hormone dependence in estrogen receptor-positive human breast cancer. J. Clin. Investig. 2010, 120, 2406–2413. [Google Scholar] [CrossRef]
- Bosch, A.; Li, Z.; Bergamaschi, A.; Ellis, H.; Toska, E.; Prat, A.; Tao, J.J.; Spratt, D.E.; Viola-Villegas, N.T.; Castel, P.; et al. PI3K inhibition results in enhanced estrogen receptor function and dependence in hormone receptor-positive breast cancer. Sci. Transl. Med. 2015, 7, 283ra51. [Google Scholar] [CrossRef]
- Fu, X.; Jeselsohn, R.; Pereira, R.; Hollingsworth, E.F.; Creighton, C.J.; Li, F.; Shea, M.; Nardone, A.; De Angelis, C.; Heiser, L.M.; et al. FOXA1 overexpression mediates endocrine resistance by altering the ER transcriptome and IL-8 expression in ER-positive breast cancer. Proc. Natl. Acad. Sci. USA 2016, 113, E6600–E6609. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, B.; Finn, R.S.; Turner, N.C. Treating cancer with selective CDK4/6 inhibitors. Nat. Rev. Clin. Oncol. 2016, 13, 417–430. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.W.; Balko, J.M.; Fox, E.M.; Ghazoui, Z.; Dunbier, A.; Anderson, H.; Dowsett, M.; Jiang, A.; Adam Smith, R.; Maira, S.M.; et al. ERα-dependent E2F transcription can mediate resistance to estrogen deprivation in human breast cancer. Cancer Discov. 2011, 1, 338–351. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Zotano, A.L.; Stricker, T.P.; Formisano, L.; Hutchinson, K.E.; Stover, D.G.; Lee, K.M.; Schwarz, L.J.; Giltnane, J.M.; Estrada, M.V.; Jansen, V.M.; et al. ERþ Breast cancers resistant to prolonged neoadjuvant letrozole exhibit an e2f4 transcriptional program sensitive to cdk4/6 inhibitors. Clin. Cancer Res. 2018, 24, 2517–2529. [Google Scholar] [CrossRef] [PubMed]
- Gelbert, L.M.; Cai, S.; Lin, X.; Sanchez-Martinez, C.; Del Prado, M.; Lallena, M.J.; Torres, R.; Ajamie, R.T.; Wishart, G.N.; Flack, R.S.; et al. Preclinical characterization of the CDK4/6 inhibitor LY2835219: In-vivo cell cycle-dependent/independent anti-tumor activities alone/in combination with gemcitabine. Investig. New Drugs 2014, 32, 825–837. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Lee, N.V.; Hu, W.; Xu, M.; Ferre, R.A.; Lam, H.; Bergqvist, S.; Solowiej, J.; Diehl, W.; He, Y.A.; et al. Spectrum and degree of CDK drug interactions predicts clinical performance. Mol. Cancer Ther. 2016, 15, 2273–2281. [Google Scholar] [CrossRef]
- Hortobagyi, G.N.; Stemmer, S.M.; Burris, H.A.; Yap, Y.S.; Sonke, G.S.; Paluch-Shimon, S.; Campone, M.; Petrakova, K.; Blackwell, K.L.; Winer, E.P.; et al. Updated results from MONALEESA-2, a phase III trial of first-line ribociclib plus letrozole versus placebo plus letrozole in hormone receptor-positive, HER2-negative advanced breast cancer. Ann. Oncol. 2018, 29, 1541–1547. [Google Scholar] [CrossRef]
- Tripathy, D.; Im, S.A.; Colleoni, M.; Franke, F.; Bardia, A.; Harbeck, N.; Hurvitz, S.A.; Chow, L.; Sohn, J.; Lee, K.S.; et al. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): A randomised phase 3 trial. Lancet Oncol. 2018, 19, 904–915. [Google Scholar] [CrossRef]
- Slamon, D.J.; Neven, P.; Chia, S.; Fasching, P.A.; De Laurentiis, M.; Im, S.A.; Petrakova, K.; Val Bianchi, G.; Esteva, F.J.; Martín, M.; et al. Phase III randomized study of ribociclib and fulvestrant in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: MONALEESA-3. J. Clin. Oncol. 2018, 36, 2465–2472. [Google Scholar] [CrossRef]
- Goetz, M.P.; Toi, M.; Campone, M.; Trédan, O.; Bourayou, N.; Sohn, J.; Park, I.H.; Paluch-Shimon, S.; Huober, J.; Chen, S.C.; et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast cancer. J. Clin. Oncol. 2017, 35, 3638–3646. [Google Scholar] [CrossRef]
- Sledge, G.W.; Toi, M.; Neven, P.; Sohn, J.; Inoue, K.; Pivot, X.; Burdaeva, O.; Okera, M.; Masuda, N.; Kaufman, P.A.; et al. MONARCH 2: Abemaciclib in combination with fulvestrant in women with HR+/HER2-advanced breast cancer who had progressed while receiving endocrine therapy. J. Clin. Oncol. 2017, 35, 2875–2884. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Crown, J.P.; Lang, I.; Boer, K.; Bondarenko, I.M.; Kulyk, S.O.; Ettl, J.; Patel, R.; Pinter, T.; Schmidt, M.; et al. The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): A randomised phase 2 study. Lancet Oncol. 2015, 16, 25–35. [Google Scholar] [CrossRef]
- Turner, N.C.; Ro, J.; André, F.; Loi, S.; Verma, S.; Iwata, H.; Harbeck, N.; Loibl, S.; Bartlett, C.H.; Zhang, K.; et al. Palbociclib in hormone-receptor-positive advanced breast cancer. N. Engl. J. Med. 2015, 373, 209–219. [Google Scholar] [CrossRef]
- Turner, N.C.; Slamon, D.J.; Ro, J.; Bondarenko, I.; Im, S.A.; Masuda, N.; Colleoni, M.; DeMichele, A.; Loi, S.; Verma, S.; et al. Overall survival with palbociclib and fulvestrant in advanced breast cancer. N. Engl. J. Med. 2018, 379, 1926–1936. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Turner, N.C.; Bondarenko, I.; Ro, J.; Im, S.A.; Masuda, N.; Colleoni, M.; DeMichele, A.; Loi, S.; Verma, S.; et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): Final analysis of the multicentre, double-blind, phas. Lancet Oncol. 2016, 17, 425–439. [Google Scholar] [CrossRef]
- Giuliano, M.; Schettini, F.; Rognoni, C.; Milani, M.; Jerusalem, G.; Bachelot, T.; De Laurentiis, M.; Thomas, G.; De Placido, P.; Arpino, G.; et al. Endocrine treatment versus chemotherapy in postmenopausal women with hormone receptor-positive, HER2-negative, metastatic breast cancer: A systematic review and network meta-analysis. Lancet Oncol. 2019, 20, 1360–1369. [Google Scholar] [CrossRef]
- Rossi, V.; Berchialla, P.; Giannarelli, D.; Nisticò, C.; Ferretti, G.; Gasparro, S.; Russillo, M.; Catania, G.; Vigna, L.; Mancusi, R.L.; et al. Should All Patients With HR-Positive HER2-Negative Metastatic Breast Cancer Receive CDK 4/6 Inhibitor As First-Line Based Therapy? A Network Meta-Analysis of Data from the PALOMA 2, MONALEESA 2, MONALEESA 7, MONARCH 3, FALCON, SWOG and FACT Trials. Cancers 2019, 11, 1661. [Google Scholar] [CrossRef]
- Im, S.-A.; Lu, Y.-S.; Bardia, A.; Harbeck, N.; Colleoni, M.; Franke, F.; Chow, L.; Sohn, J.; Lee, K.-S.; Campos-Gomez, S.; et al. Overall Survival with Ribociclib plus Endocrine Therapy in Breast Cancer. N. Engl. J. Med. 2019, 381, 307–316. [Google Scholar] [CrossRef]
- DeMichele, A.; Cristofanilli, M.; Brufsky, A.; Liu, X.; Mardekian, J.; McRoy, L.; Layman, R.M.; Rugo, H.S.; Finn, R.S. Overall Survival for First-Line Palbociclib Plus Letrozole vs Letrozole Alone for HR+/HER2–Metastatic Breast Cancer Patients in US Real-World Clinical Practice. Available online: Sabcs.posterview.com (accessed on 20 December 2020).
- Carter, G.C.; Sheffield, K.M.; Gossai, A.; Huang, Y.-J.; Zhu, Y.E.; Bowman, L.; Smith, E.N.; Mathur, R.; Cohen, A.B.; Baxi, S.; et al. Initial real world treatment patterns and outcomes of Abemaciclib for the treatment of HR+, HER2-metastatic breast cancer. Cancer Res. 2020, 80. [Google Scholar] [CrossRef]
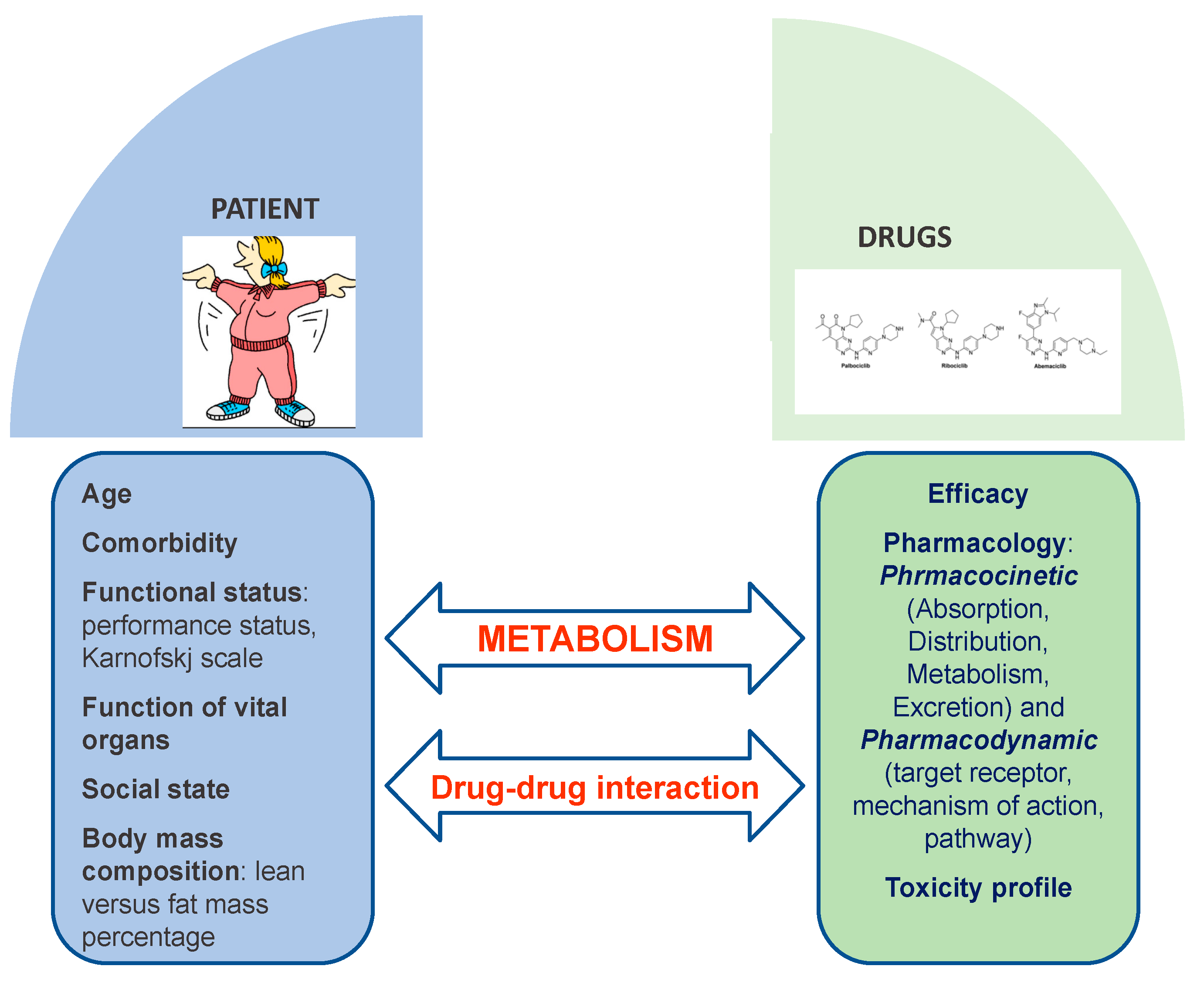
- Fogli, S.; Del Re, M.; Curigliano, G.; Van Schaik, R.H.; Lancellotti, P.; Danesi, R. Complications of Treatment Drug-drug interactions in breast cancer patients treated with CDK4/6 inhibitors. Cancer Treat. Rev. 2019, 74, 21–28. [Google Scholar] [CrossRef]
- Roncato, R.; Angelini, J.; Pani, A.; Cecchin, E.; Sartore-Bianchi, A.; Siena, S.; De Mattia, E.; Scaglione, F.; Toffoli, G. CDK4/6 Inhibitors in Breast Cancer Treatment: Potential Interactions with Drug, Gene, and Pathophysiological Conditions. Int. J. Mol. Sci. 2020, 21, 6350. [Google Scholar] [CrossRef] [PubMed]
- Roberto, M.; Rossi, A.; Panebianco, M.; Pomes, L.M.; Arrivi, G.; Ierinò, D.; Simmaco, M.; Marchetti, P.; Mazzuca, F. Drug–Drug Interactions and Pharmacogenomic Evaluation in Colorectal Cancer Patients: The New Drug-PIN® System Comprehensive Approach. Pharmaceuticals 2021, 14, 67. [Google Scholar] [CrossRef]
- Cardoso, F.; Senkus, E.; Costa, A.; Papadopoulos, E.; Aapro, M.; André, F.; Harbeck, N.; Aguilar Lopez, B.; Barrios, C.H.; Bergh, J.; et al. 4th ESO-ESMO international consensus guidelines for advanced breast cancer (ABC 4). Ann. Oncol. 2018, 29, 1634–1657. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.C.; Liu, Y.; Zhu, Z.; Loi, S.; Colleoni, M.; Loibl, S.; DeMichele, A.; Harbeck, N.; André, F.; Bayar, M.A.M.; et al. Cyclin E1 expression and palbociclib efficacy in previously treated hormone receptor-positive metastatic breast cancer. J. Clin. Oncol. 2019, 37, 1169–1178. [Google Scholar] [CrossRef] [PubMed]
- Hurvitz, S.A.; Im, S.-A.; Lu, Y.-S.; Colleoni, M.; Franke, F.A.; Bardia, A.; Harbeck, N.; Chow, L.; Sohn, J.; Lee, K.S.; et al. Phase III MONALEESA-7 trial of premenopausal patients with HR+/HER2− advanced breast cancer (ABC) treated with endocrine therapy ± ribociclib: Overall survival (OS) results. J. Clin. Oncol. 2019, 37, LBA1008. [Google Scholar] [CrossRef]
- Xi, J.; Oza, A.; Thomas, S.; Ademuyiwa, F.; Weilbaecher, K.; Suresh, R.; Bose, R.; Cherian, M.; Hernandez-Aya, L.; Frith, A.; et al. Retrospective analysis of treatment patterns and effectiveness of palbociclib and subsequent regimens in metastatic breast cancer. JNCCN J. Natl. Compr. Cancer Netw. 2019, 17, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Cook, M.; Al Rabadi, L.; Mitri, Z.I. Everolimus and exemestane for the treatment of metastatic hormone receptor-positive breast cancer patients previously treated with CDK4/6 inhibitor based therapies. J. Clin. Oncol. 2019, 37, 1058. [Google Scholar] [CrossRef]
- Wander, S.A.; Zangardi, M.; Niemierko, A.; Kambadakone, A.; Kim, L.S.; Xi, J.; Pandey, A.K.; Spring, L.; Stein, C.; Juric, D.; et al. A multicenter analysis of abemaciclib after progression on palbociclib in patients (pts) with hormone receptor-positive (HR+)/HER2- metastatic breast cancer (MBC). J. Clin. Oncol. 2019, 37, 1057. [Google Scholar] [CrossRef]
- Pandey, K.; An, H.J.; Kim, S.K.; Lee, S.A.; Kim, S.; Lim, S.M.; Kim, G.M.; Sohn, J.; Moon, Y.W. Molecular mechanisms of resistance to CDK4/6 inhibitors in breast cancer: A review. Int. J. Cancer 2019, 145, 1179–1188. [Google Scholar] [CrossRef]
- Herrera-Abreu, M.T.; Palafox, M.; Asghar, U.; Rivas, M.A.; Cutts, R.J.; Garcia-Murillas, I.; Pearson, A.; Guzman, M.; Rodriguez, O.; Grueso, J.; et al. Early adaptation and acquired resistance to CDK4/6 inhibition in estrogen receptor-positive breast cancer. Cancer Res. 2016, 76, 2301–2313. [Google Scholar] [CrossRef]
- Formisano, L.; Stauffer, K.M.; Young, C.D.; Bhola, N.E.; Guerrero-Zotano, A.L.; Jansen, V.M.; Estrada, M.M.; Hutchinson, K.E.; Giltnane, J.M.; Schwarz, L.J.; et al. Association of FGFR1 with ERα maintains ligand-independent ER transcription and mediates resistance to estrogen deprivation in ER+ breast cancer. Clin. Cancer Res. 2017, 23, 6138–6151. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Fernández, M.; Malumbres, M. Mechanisms of Sensitivity and Resistance to CDK4/6 Inhibition. Cancer Cell 2020, 37, 514–529. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Liu, Y.; Zhu, Z.; Martin, M.; Rugo, H.S.; Diéras, V.; Im, S.-A.; Gelmon, K.A.; Harbeck, N.; Lu, D.R.; et al. Biomarker Analyses of Response to Cyclin-Dependent Kinase 4/6 Inhibition and Endocrine Therapy in Women with Treatment-Naïve Metastatic Breast Cancer. Clin. Cancer Res. 2019, 26, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Taylor-Harding, B.; Aspuria, P.J.; Agadjanian, H.; Cheon, D.J.; Mizuno, T.; Greenberg, D.; Allen, J.R.; Spurka, L.; Funari, V.; Spiteri, E.; et al. Cyclin E1 and RTK/RAS signaling drive CDK inhibitor resistance via activation of E2F and ETS. Oncotarget 2015, 6, 696–714. [Google Scholar] [CrossRef] [PubMed]
- Tolaney, S.M.; Wardley, A.M.; Zambelli, S.; Hilton, J.F.; Troso-Sandoval, T.A.; Ricci, F.; Im, S.A.; Kim, S.B.; Johnston, S.R.; Chan, A.; et al. Abemaciclib plus trastuzumab with or without fulvestrant versus trastuzumab plus standard-of-care chemotherapy in women with hormone receptor-positive, HER2-positive advanced breast cancer (monarcHER): A randomised, open-label, phase 2 trial. Lancet Oncol. 2020, 21, 763–775. [Google Scholar] [CrossRef]
- Bardia, A.; Su, F.; Solovieff, N.; Im, S.-A.; Sohn, J.; Lee, K.S.; Campos-Gomez, S.; Jung, K.H.; Vazquez, R.V.; Lu, Y.-S.; et al. Abstract CT141: Genetic landscape of premenopausal HR+/HER2- advanced breast cancer (ABC) based on comprehensive circulating tumor DNA analysis and association with clinical outcomes in the Phase III MONALEESA-7 trial. Cancer Res. Am. Assoc. Cancer Res. 2019, 79, 141. [Google Scholar]
- Chia, S.; Su, F.; Neven, P.; Im, S.-A.; Petrakova, K.; Bianchi, G.V.; He, W.; Rodriguez-Lorenc, K.; Taran, T.; Babbar, N.; et al. Abstract PD2-08: Gene expression analysis and association with treatment response in postmenopausal patients with hormone receptor-positive, HER2-negative advanced breast cancer in the MONALEESA-3 study. Cancer Res. Am. Assoc. Cancer Res. 2020, 80, 8. [Google Scholar]
- Tolaney, S.; Sahebjam, S.; Le Rhun, E.; Lin, N.; Markel Bear, M.; Yang, Z.; Chen, Y.; Anders, C. Abstract P1-19-01: A phase 2 study of abemaciclib in patients with leptomeningeal metastases secondary to HR+, HER2- breast cancer. Cancer Res. Am. Assoc. Cancer Res. 2019, 79, 19. [Google Scholar]
- O’leary, B.; Cutts, R.J.; Liu, Y.; Hrebien, S.; Huang, X.; Fenwick, K.; André, F.; Loibl, S.; Loi, S.; Garcia-Murillas, I.; et al. The Genetic Landscape and Clonal Evolution of Breast Cancer Resistance to Palbociclib plus Fulvestrant in the PALOMA-3 Trial. Cancer Discov. 2018, 8, 1390–1403. [Google Scholar] [CrossRef]
- Mao, P.; Cohen, O.; Kowalski, K.J.; Kusiel, J.G.; Buendia-Buendia, J.E.; Cuoco, M.S.; Exman, P.; Wander, S.A.; Waks, A.G.; Nayar, U.; et al. Acquired FGFR and FGF Alterations Confer Resistance to Estrogen Receptor (ER) Targeted Therapy in ER + Metastatic Breast Cancer. Clin. Cancer Res. 2020, 26, 5974–5989. [Google Scholar] [CrossRef]
- Formisano, L.; Lu, Y.; Servetto, A.; Hanker, A.B.; Jansen, V.M.; Bauer, J.A.; Sudhan, D.R.; Guerrero-Zotano, A.L.; Croessmann, S.; Guo, Y.; et al. Aberrant FGFR signaling mediates resistance to CDK4/6 inhibitors in ER+ breast cancer. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef]
- O’Leary, B.; Cutts, R.; Huang, X.; Hrebien, S.; Liu, Y.; Garcia-Murillas, I.; Andre, F.; Loi, S.; Loibl, S.; Cristofanilli, M.; et al. Genomic markers of early progression on fulvestrant with or without palbociclib for ER+ advanced breast cancer in the PALOMA-3 trial. J. Clin. Oncol. 2019, 37, 1010. [Google Scholar] [CrossRef]
- Portman, N.; Milioli, H.H.; Alexandrou, S.; Coulson, R.; Yong, A.; Fernandez, K.J.; Chia, K.M.; Halilovic, E.; Segara, D.; Parker, A.; et al. MDM2 inhibition in combination with endocrine therapy and CDK4/6 inhibition for the treatment of ER-positive breast cancer. Breast Cancer Res. 2020, 22, 1–17. [Google Scholar] [CrossRef]
- Vilgelm, A.E.; Saleh, N.; Shattuck-Brandt, R.; Riemenschneider, K.; Slesur, L.; Chen, S.C.; Johnson, C.A.; Yang, J.; Blevins, A.; Yan, C.; et al. MDM2 antagonists overcome intrinsic resistance to CDK4/6 inhibition by inducing p21. Sci. Transl. Med. 2019, 11, eaav7171. [Google Scholar] [CrossRef] [PubMed]
- Bonechi, M.; Galardi, F.; Biagioni, C.; De Luca, F.; Bergqvist, M.; Neumüller, M.; Guarducci, C.; Boccalini, G.; Gabellini, S.; Migliaccio, I.; et al. Plasma thymidine kinase-1 activity predicts outcome in patients with hormone receptor positive and HER2 negative metastatic breast cancer treated with endocrine therapy. Oncotarget 2018, 9, 16389–16399. [Google Scholar] [CrossRef] [PubMed]
- McCartney, A.; Biagioni, C.; Schiavon, G.; Bergqvist, M.; Mattsson, K.; Migliaccio, I.; Benelli, M.; Romagnoli, D.; Bonechi, M.; Boccalini, G.; et al. Prognostic role of serum thymidine kinase 1 activity in patients with hormone receptor–positive metastatic breast cancer: Analysis of the randomised phase III Evaluation of Faslodex versus Exemestane Clinical Trial (EFECT). Eur. J. Cancer 2019, 114, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Razavi, P.; Li, Q.; Toy, W.; Liu, B.; Ping, C.; Hsieh, W.; Sanchez-Vega, F.; Brown, D.N.; Da Cruz Paula, A.F.; et al. Loss of the FAT1 Tumor Suppressor Promotes Resistance to CDK4/6 Inhibitors via the Hippo Pathway. Cancer Cell 2018, 34, 893–905.e8. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, S.; Karakas, C.; Doostan, I.; Chen, X.; Bui, T.; Yi, M.; Raghavendra, A.S.; Zhao, Y.; Bashour, S.I.; Ibrahim, N.K.; et al. CDK4/6 and autophagy inhibitors synergistically induce senescence in Rb positive cytoplasmic cyclin e negative cancers. Nat. Commun. 2017, 8, 15916. [Google Scholar] [CrossRef]
- Buono, G.; Gerratana, L.; Bulfoni, M.; Provinciali, N.; Basile, D.; Giuliano, M.; Corvaja, C.; Arpino, G.; Del Mastro, L.; De Placido, S.; et al. Circulating tumor DNA analysis in breast cancer: Is it ready for prime-time? Cancer Treat. Rev. 2019, 73, 73–83. [Google Scholar] [CrossRef]
- McCartney, A.; Migliaccio, I.; Bonechi, M.; Biagioni, C.; Romagnoli, D.; De Luca, F.; Galardi, F.; Risi, E.; De Santo, I.; Benelli, M.; et al. Mechanisms of Resistance to CDK4/6 Inhibitors: Potential Implications and Biomarkers for Clinical Practice. Front. Oncol. 2019, 9, 666. [Google Scholar] [CrossRef]
- André, F.; Ciruelos, E.; Rubovszky, G.; Campone, M.; Loibl, S.; Rugo, H.S.; Iwata, H.; Conte, P.; Mayer, I.A.; Kaufman, B.; et al. Alpelisib for PIK3CA -Mutated, Hormone Receptor–Positive Advanced Breast Cancer. N. Engl. J. Med. 2019, 380, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Rugo, H.S.; Ruiz Borrego, M.; Chia, S.K.L.; Juric, D.; Turner, N.C.; Drullinsky, P.; Lerebours, F.; Bianchi, G.V.; Nienstedt, C.C.; Ridolfi, A.; et al. Alpelisib (ALP) + endocrine therapy (ET) in patients (pts) with PIK3CA-mutated hormone receptor-positive (HR+), human epidermal growth factor-2-negative (HER2-) advanced breast cancer (ABC): First interim BYLieve study results. J. Clin. Oncol. 2019, 37, 1040. [Google Scholar] [CrossRef]
- Franzoi, M.A.; Eiger, D.; Ameye, L.; Ponde, N.; Caparica, R.; De Angelis, C.; Brandão, M.; Desmedt, C.; Di Cosimo, S.; Kotecki, N.; et al. Clinical Implications of Body Mass Index in Metastatic Breast Cancer Patients Treated With Abemaciclib and Endocrine Therapy. J. Natl. Cancer Inst. 2020, 116. [Google Scholar] [CrossRef]
| Drug | Trial | Setting | Endocrine Sensitivity/Resistance (%) | Efficacy | Adverse Events of Interest | ||||
|---|---|---|---|---|---|---|---|---|---|
| De Novo | Late Relapse | Early Relapse | Second Line | PFS (Months) | OS (Months) | ||||
| Ribociclib | MONALEESA-2 [17] | First line | 34 | 64.7 | — | — | RIBO + LET: 25.3 PBO + LET: 16.0 (HR, 0.56; 95% CI, 0.43–0.72; p < 0.001) | Immature |
|
| MONALEESA-7 [18] | First and second line | 40 | 52.5 | — | 14 (after CT) | RIBO + TAM/NSAI: 23.8 months PBO + TAM/NSAI: 13.0 months (HR, 0.55; 95% CI, 0.44–0.69; p < 0.0001) | HR, 0.712; 95% CI, 0.535–0.948; p = 0.00973 |
| |
| MONALEESA-3 [19] | First and second line | 20 | 29 | 28 | 20 | RIB + FUL: 20.5 (33.6 in first line) PBO + FUL: 12.8 (19.2 in first line) (HR, 0.593; 95% CI, 0.480–0.732; p < 0.001) | HR, 0.724; 95% CI, 0.568– 0.924; p = 0.00455 |
| |
| Abemaciclib | MONARCH-3 [20] | First line | 41.2 | 58.8 | — | — | ABE + NSAI: 28.18 months PBO + NSAI: 14.76 months (HR, 0.540; 95% CI, 0.418–0.698; p = 0.000002) | Immature |
|
| MONARCH-2 [21] | Second line | — | — | 60 | 38 | ABE + FUL: 16.4 PBO + FUL: 9.3 (HR, 0.553; 95% CI, 0.449–0.681; p < 0.001) | HR, 0.757; 95% CI, 0.606–0.945; p = 0.0137 |
| |
| Palbociclib | PALOMA-2 [22] | First line | 37.6 | 40.01 | — | — | PAL + LET: 24.8 PBO + LET: 14.5 (HR, 0.58; 95% CI, 0.46–0.72; p <0.001) | Immature |
|
| PALOMA-3 [23,24,25] | Second line | — | — | 21 | 79 | PAL + FUL: 9.5 PBO + FUL: 4.6 (HR, 0.46; 95% CI, 0.36–0.59; p < 0.0001) | HR, 0.81; 95% CI, 0.64–1.03; p = 0.09 |
| |
| Trial | Population | n/N | Treatment | PFS (Months) | OS (Months) | ORR (%) |
|---|---|---|---|---|---|---|
| MONALEESA-3 |
| 346/726 | Ribociclib plus fulvestrant versus placebo + fulvestrant | 14.6 (HR, 0.57) | 40.2 (HR, 0.73) | 40.9 |
| MONALEESA-7 |
| 94/672 | Ribociclib plus ET plus goserelin versus placebo + ET and goserelin | 16.6 (HR 0.54) | NR (HR 0.67) | 26 |
| MONARCH-2 |
| 669 | Abemaciclib plus fulvestrant versus placebo + fulvestrant | 16.4 (HR, 0.55) | 46.7 (HR 0.75) | 48.1 |
| PALOMA-3 |
| 521 | Palbociclib plus fulvestrant versus placebo + fulvestrant | 11.2 (HR, 0.50) | 34.9 (HR, 0.81; NS, p = 0.09) | 25 |
| Trial | Phase | Study Arms | Previous CDK4/6 |
|---|---|---|---|
| MAINTAIN (NCT02632045) | 2 | Ribociclib + fulvestrant versus placebo + fulvestrant | AI + palbociclib/ribociclib |
| NCT02738866 | 2 | Palbociclib + fulvestrant | Palbociclib + AI |
| PACE (NCT03147287) | 2 | Fulvestrant versus fulvestrant + palbociclib versus ± avelumab | CDK4/6 inhibitor-based regimen |
| NCT02871791 | ½ | Palbociclib + everolimus + exemestane | CDK4/6 inhibitor-based regimen |
| TRINITI-1 (NCT02732119) | 1/2 | Ribociclib + everolimus + exemestane | CDK4/6 inhibitor-based regimen |
| NCT01857193 | 1B | Ribociclib + exemestane versus ± everolimus | Naive or refractory to CDK4/6-inhibitor |
| PALMIRA (NCT03809988) | 2 | Palbociclib + ET | Had clinical benefits with palbociclib + ET in first line |
| Trial | Phase | Study Arms | Previous CDK4/6 |
|---|---|---|---|
| CDK7 inhibitor after CDK4/6 inhibitor progression | |||
| NCT03134638 | 1 | SY-1365 + fulvestrant | CDK inhibitor + AI |
| NCT03363893 | 1 | CT-7001 + fulvestrant | CDK inhibitor |
| CDK2 inhibitor after CDK4/6 inhibitor progression | |||
| NCT03519178 | 1/2A | PF-06873600 versus ± ET | CDK inhibitor + ET |
| Selective estrogen receptor downregulator (SERD) (elacestrant) after CDK4/6 inhibitor progression | |||
| EMERALD (NCT03778931) | 3 | Elacestrant | CDK inhibitor + AI or fulvestrant |
| BCL-2 inhibitor (venetoclax) afterCDK4/6 inhibitor progression | |||
| VERONICA (NCT03519178) | 2 | Venetoclax + fulvestrant | CDK inhibitor-based regimen |
| Fibroblast growth factor receptor (FGFR) inhibitor after CDK4/6 inhibitor progression | |||
| NCT03238196 | 1 | Fulvestrant + palbociclib + erdafitinib | Previous palbociclib allowed |
| Immune checkpoint inhibitor after CDK4/6 inhibitor progression | |||
| PACE (NCT03147287) | 2 | Fulvestrant versus fulvestrant + palbociclib versus ± avelumab | CDK4/6 inhibitor-based regimen |
| NCT0329469 | 1 | Fulvestrant + ribociclib + PDR001 | Not specified |
| MORPHEUS HR+BC (NCT03280563) | 1/2 | Fulvestrant Atezolizumab + entinostat Atezolizumab + fulvestrant Atezolizumab + ipatasertib Atezolizumab + ipatasertib+ fulvestrant Atezolizumab + bevacizumab + ET Atezolizumab + abemaciclib + fulvestrant | CDK4/6 inhibitor in first or second line |
| Trial | Treatment Arms | Phase | Study Population | Primary Endpoint | Status |
|---|---|---|---|---|---|
| Study of AZD2014 and palbociclib in patients with estrogen receptor positive (ER+) metastatic breast cancer (PASTOR) | Vistusertib (mTOR inhibitor) + palbociclib + fulvestrant Placebo + palbociclib + fulvestrant | 1/2 | ER+ locally advanced or MBC in postmenopausal patients pretreated with hormonal therapy | PFS | Completed no results posted |
| Copanlisib, letrozole, and palbociclib in treating patients with hormone receptor positive HER2 negative stage I-IV breast cancer | Copanlisib (PI3K inhibitor) + letrozole Copanlisib + letrozole + palbociclib | 1b/2 | ER+/HER2− postmenopausal any stage breast cancer | Change in Ki-67, DLT | Recruiting |
| Ipatasertib plus Palbociclib and fulvestrant versus placebo plus Palbociclib and fulvestrant in hormone receptor positive and HER2 negative locally advanced unresectable or metastatic breast cancer (IPATunity150) | Ipatasertib (AKT inhibitor) + palbociclib + fulvestrant versus placebo + palbociclib + fulvestrant | 1b/3 randomized | HR+ HER2− ABC progressed during adjuvant ET or the initial 12 months of first-line ET | PFS | Recruiting |
| Ribociclib in combination with everolimus (RAD001) and exemestane in the treatment of postmenopausal women with hormone receptor positive, HER2 negative locally advanced or metastatic breast cancer | Ribociclib, exemestane and everolimus versus ribociclib and exemestane | Nonrandomized, two arms, parallel assignment | Recurrence while on, or within 12 months of end of, adjuvant treatment with letrozole or anastrozole, or progression while on, or within 1 month of end of, letrozole or anastrozole treatment for ABC | DLT and DCR | Completed |
| LEE011 in combination with fulvestrant and alpelisib or buparlisib in the treatment of postmenopausal women with hormone receptor positive, HER2 negative locally recurrent or advanced metastatic breast cancer | LEE011 + fulvestrant + alpelisib or LEE011 + fulvestrant + buparlisib or LEE011 + fulvestrant | 1b/2 nonrandomized | HR+, HER2− locally recurrent or advanced metastatic breast cancer | DLT/PFS | Completed |
| A study to assess the tolerability and clinical activity of gedatolisib in combination with palbociclib/letrozole or palbociclib/fulvestrant in women with metastatic breast cancer | Gedatolisib + palbociclib/letrozole or gedatolisib +palbociclib/fulvestrant | 1b nonrandomized | HR+, HER2− locally recurrent or advanced metastatic breast cancer | DLT ORR | Recruiting |
| Abemaciclib in combination with therapies for patients with metastatic breast cancer | Abemaciclib + letrozole or anastrozole or tamoxifen or exemestane or everolimus or trastuzumab or fulvestrant or pertuzumab | 1b | Metastatic breast cancer | DLT | Active, not recruiting |
| PIPA: combination of PI3 kinase inhibitors and palbociclib with the subsequent addition of fulvestrant in PIK3CA-mutant breast cancers (PIPA) | Palbociclib + taselisib or pictilisib (+ fulvestant) | 1b | Malignant solid tumors; ABC: ER+ progressed on at least one line of previous ET, or PIK3CA mutant breast cancer progressed on at least one line of previous ET or CT, breast cancer refractory to standard treatment | DLT safety | Active, not recruiting |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roberto, M.; Astone, A.; Botticelli, A.; Carbognin, L.; Cassano, A.; D’Auria, G.; Fabbri, A.; Fabi, A.; Gamucci, T.; Krasniqi, E.; et al. CDK4/6 Inhibitor Treatments in Patients with Hormone Receptor Positive, Her2 Negative Advanced Breast Cancer: Potential Molecular Mechanisms, Clinical Implications and Future Perspectives. Cancers 2021, 13, 332. https://doi.org/10.3390/cancers13020332
Roberto M, Astone A, Botticelli A, Carbognin L, Cassano A, D’Auria G, Fabbri A, Fabi A, Gamucci T, Krasniqi E, et al. CDK4/6 Inhibitor Treatments in Patients with Hormone Receptor Positive, Her2 Negative Advanced Breast Cancer: Potential Molecular Mechanisms, Clinical Implications and Future Perspectives. Cancers. 2021; 13(2):332. https://doi.org/10.3390/cancers13020332
Chicago/Turabian StyleRoberto, Michela, Antonio Astone, Andrea Botticelli, Luisa Carbognin, Alessandra Cassano, Giuliana D’Auria, Agnese Fabbri, Alessandra Fabi, Teresa Gamucci, Eriseld Krasniqi, and et al. 2021. "CDK4/6 Inhibitor Treatments in Patients with Hormone Receptor Positive, Her2 Negative Advanced Breast Cancer: Potential Molecular Mechanisms, Clinical Implications and Future Perspectives" Cancers 13, no. 2: 332. https://doi.org/10.3390/cancers13020332
APA StyleRoberto, M., Astone, A., Botticelli, A., Carbognin, L., Cassano, A., D’Auria, G., Fabbri, A., Fabi, A., Gamucci, T., Krasniqi, E., Minelli, M., Orlandi, A., Pantano, F., Paris, I., Pizzuti, L., Portarena, I., Salesi, N., Scagnoli, S., Scavina, P., ... Marchetti, P. (2021). CDK4/6 Inhibitor Treatments in Patients with Hormone Receptor Positive, Her2 Negative Advanced Breast Cancer: Potential Molecular Mechanisms, Clinical Implications and Future Perspectives. Cancers, 13(2), 332. https://doi.org/10.3390/cancers13020332








