Health-Related Quality of Life in Very Long-Term Cancer Survivors 14–24 Years Post-Diagnosis Compared to Population Controls: A Population-Based Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
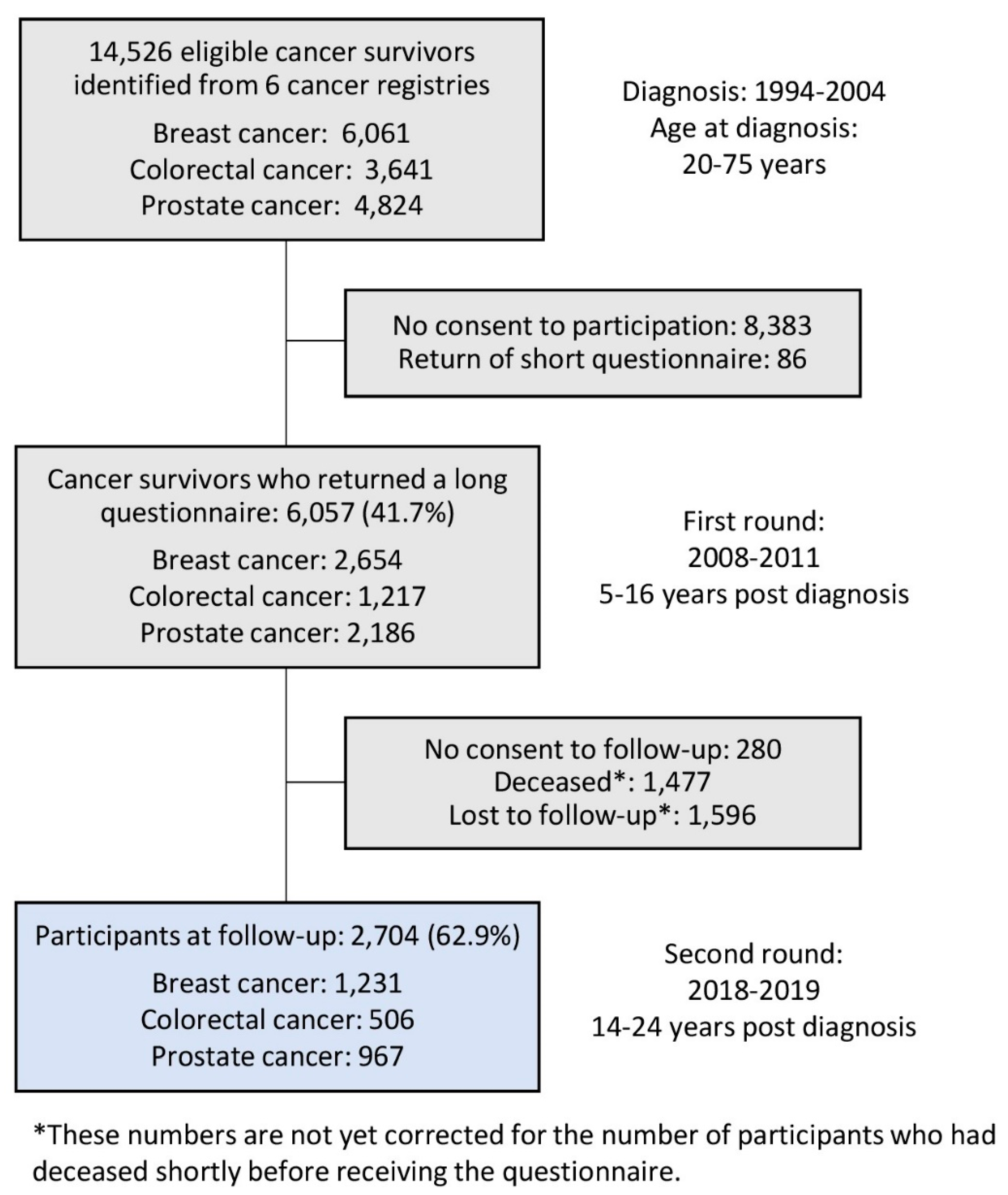
2.1. CAESAR (Cancer Survivors)
2.2. LINDE (Population Controls)
2.3. Measurements
2.4. Sociodemographic and Clinical Data
2.5. Statistical Analyses
3. Results
3.1. Non-Respondent Analysis
3.2. Study Population Characteristics
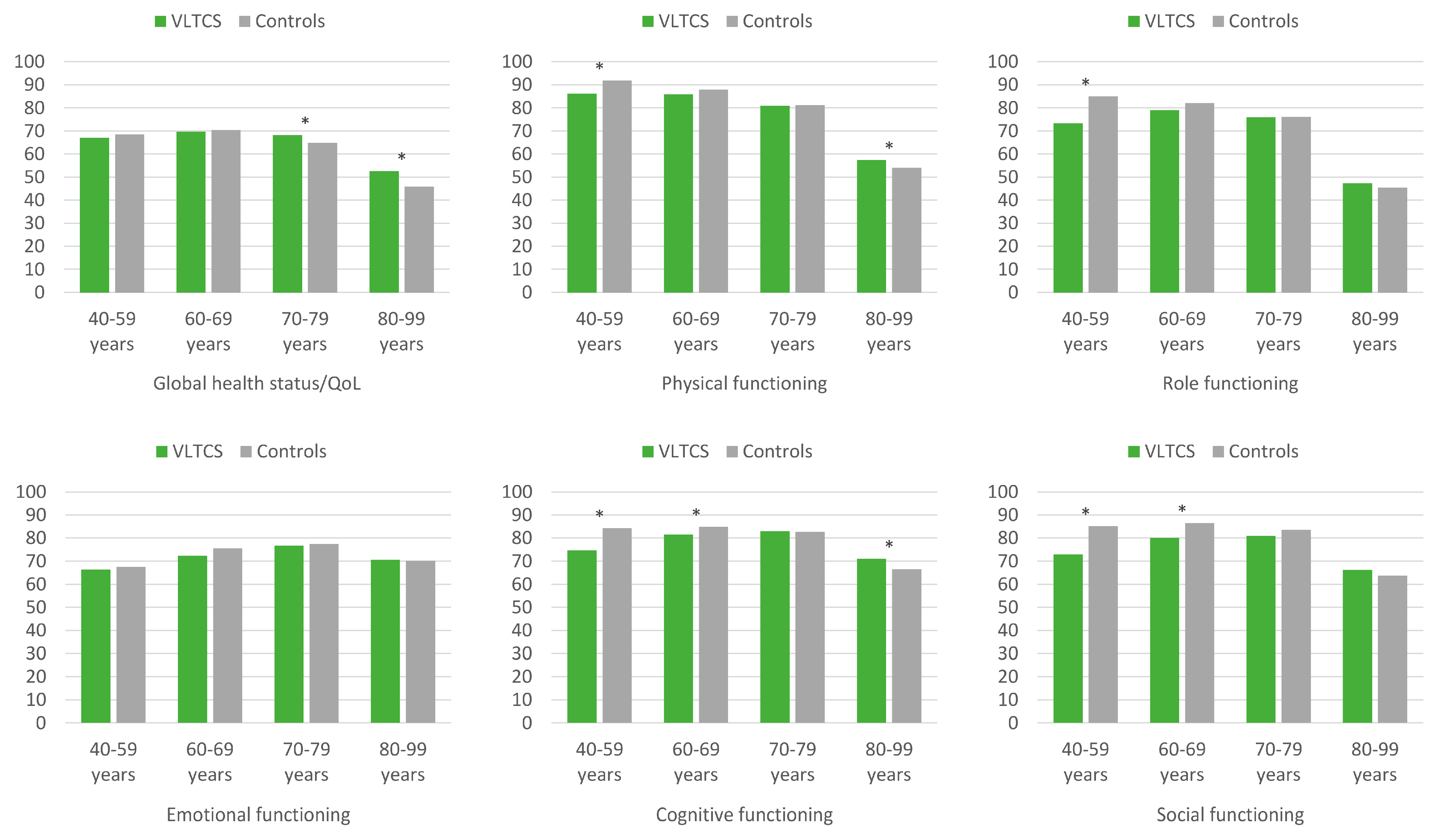
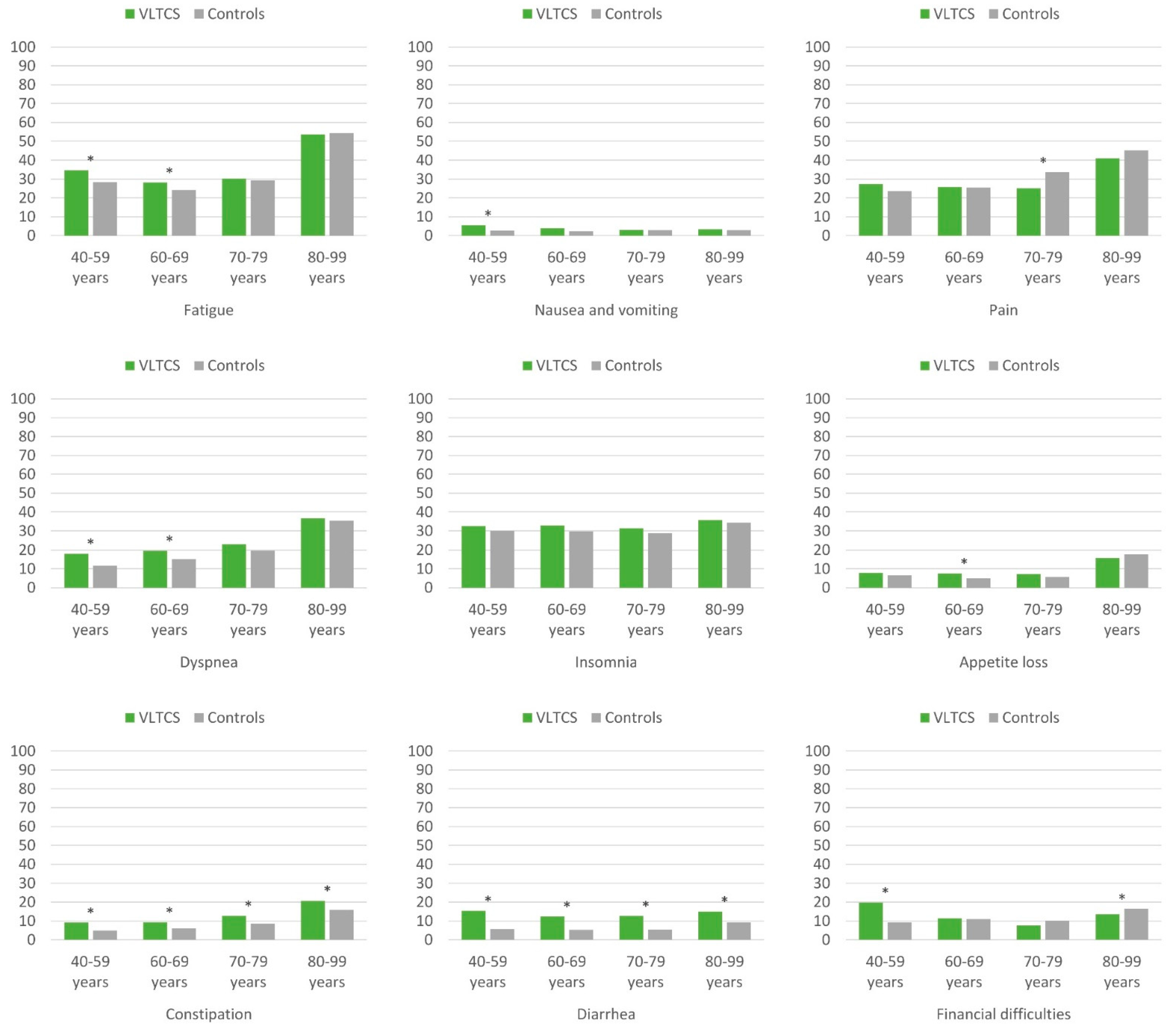
3.3. HRQoL of VLTCS and Population Controls
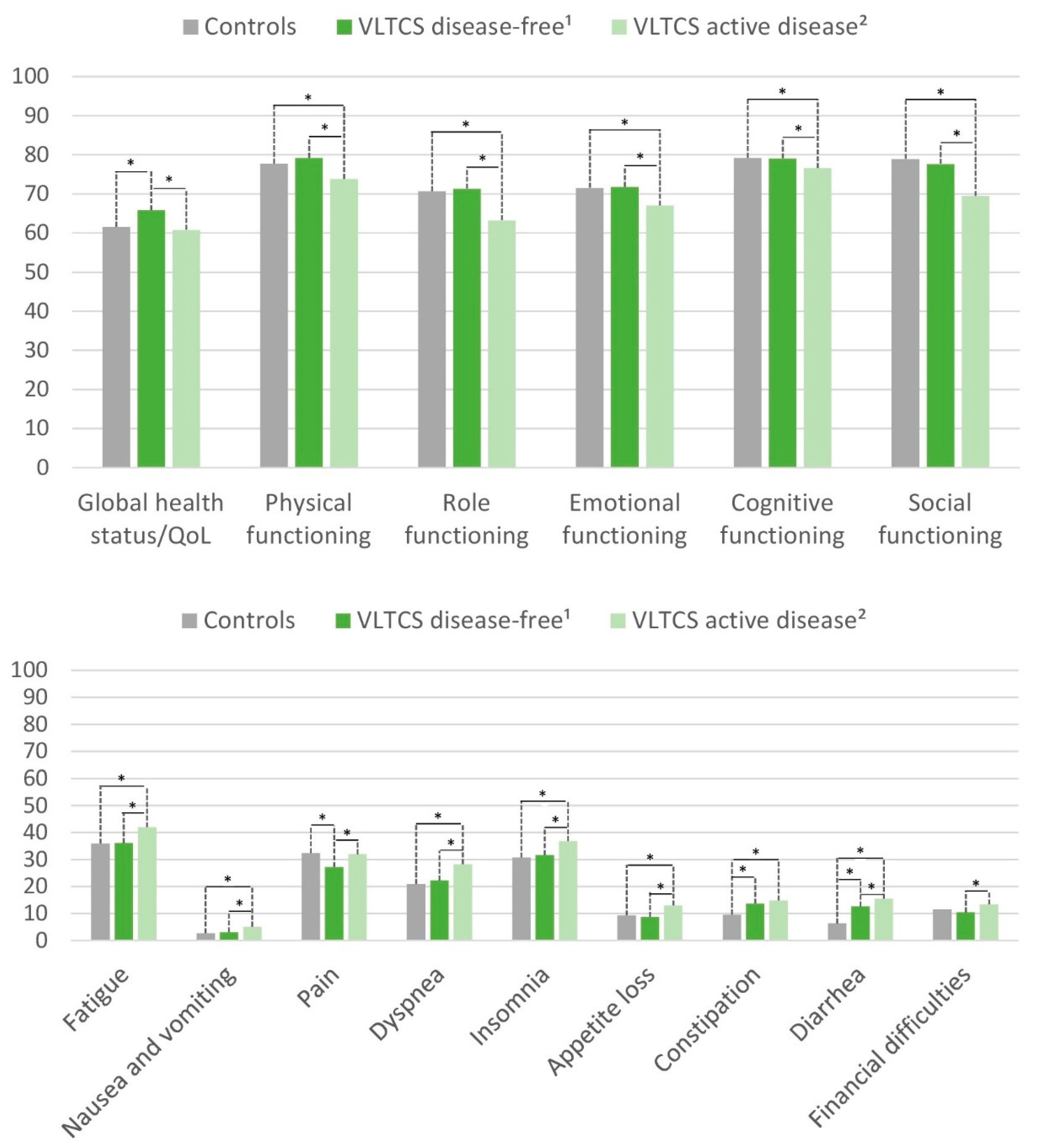
3.4. HRQoL of Disease-Free VLTCS and Those with Active Disease
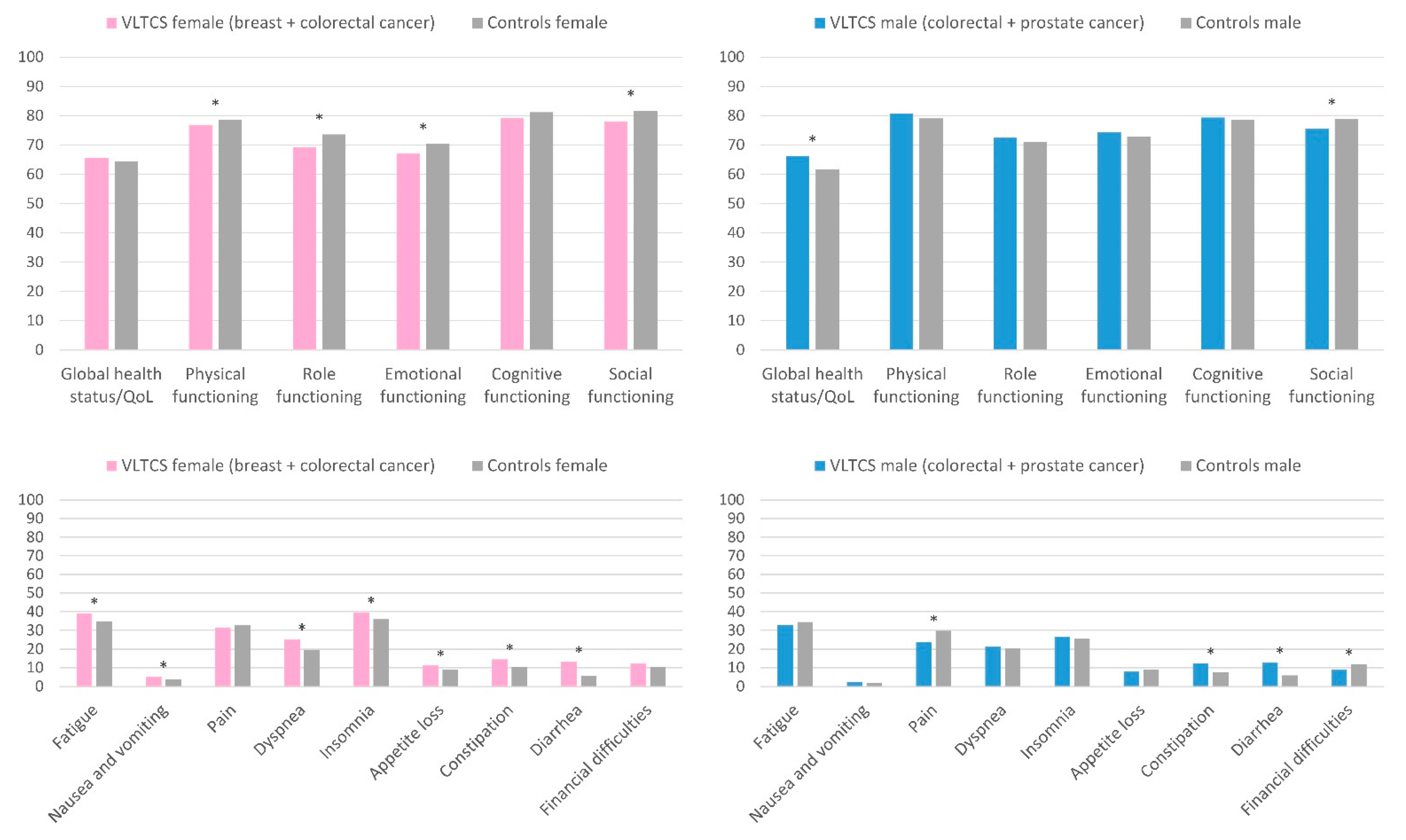
3.5. HRQoL of Female and Male VLTCS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Parry, C.; Kent, E.E.; Mariotto, A.B.; Alfano, C.M.; Rowland, J.H. Cancer Survivors: A Booming Population. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1996–2005. [Google Scholar] [CrossRef] [PubMed]
- Arndt, V. Cancer survivorship in Deutschland–Epidemiologie und Definitionen. Forum 2019, 34, 158–164. [Google Scholar] [CrossRef]
- Firkins, J.; Hansen, L.; Driessnack, M.; Dieckmann, N. Quality of life in “chronic” cancer survivors: A meta-analysis. J. Cancer Surviv. 2020, 14, 504–517. [Google Scholar] [CrossRef] [PubMed]
- Surbone, A.; Tralongo, P. Categorization of Cancer Survivors: Why We Need It. J. Clin. Oncol. 2016, 34, 3372–3374. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.F.; Mant, D.; Carpenter, L.; Forman, D.; Rose, P.W. Long-term health outcomes in a British cohort of breast, colorectal and prostate cancer survivors: A database study. Br. J. Cancer 2011, 105, S29–S37. [Google Scholar] [CrossRef]
- Harrington, C.B.; Hansen, J.A.; Moskowitz, M.; Todd, B.L.; Feuerstein, M. It’s not over when it’s over: Long-term symptoms in cancer survivors-a systematic review. Int. J. Psychiatry Med. 2010, 40, 163–181. [Google Scholar] [CrossRef]
- Wu, H.S.; Harden, J.K. Symptom burden and quality of life in survivorship: A review of the literature. Cancer Nurs. 2015, 38, E29–E54. [Google Scholar] [CrossRef]
- Arndt, V.; Koch-Gallenkamp, L.; Jansen, L.; Bertram, H.; Eberle, A.; Holleczek, B.; Schmid-Höpfner, S.; Waldmann, A.; Zeissig, S.R.; Brenner, H. Quality of life in long-term and very long-term cancer survivors versus population controls in Germany. Acta Oncol. 2017, 56, 190–197. [Google Scholar] [CrossRef]
- Fosså, S.D.; Loge, J.H.; Dahl, A.A. Long-term survivorship after cancer: How far have we come? Ann. Oncol. 2008, 19, v25–v29. [Google Scholar] [CrossRef]
- Mols, F.; Vingerhoets, A.J.; Coebergh, J.W.; Van De Poll-Franse, L.V. Quality of life among long-term breast cancer survivors: A systematic review. Eur. J. Cancer 2005, 41, 2613–2619. [Google Scholar] [CrossRef] [PubMed]
- Ahles, T.A.; Saykin, A.J.; Furstenberg, C.T.; Cole, B.; Mott, L.A.; Skalla, K.; Whedon, M.B.; Bivens, A.; Mitchell, T.; Greenberg, E.R.; et al. Neuropsychologic Impact of Standard-Dose Systemic Chemotherapy in Long-Term Survivors of Breast Cancer and Lymphoma. J. Clin. Oncol. 2002, 20, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Thong, M.S.Y.; Mols, F.; Coebergh, J.-W.W.; Roukema, J.A.; Van De Poll-Franse, L.V. The impact of disease progression on perceived health status and quality of life of long-term cancer survivors. J. Cancer Surviv. 2009, 3, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Kunitake, H.; Russell, M.M.; Zheng, P.; Yothers, G.; Land, S.R.; Petersen, L.; Fehrenbacher, L.; Giguere, J.K.; Wickerham, D.L.; Ko, C.Y.; et al. Quality of life and symptoms in long-term survivors of colorectal cancer: Results from NSABP protocol LTS-01. J. Cancer Surviv. 2017, 11, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Götze, H.; Taubenheim, S.; Dietz, A.; Lordick, F.; Mehnert, A. Comorbid conditions and health-related quality of life in long-term cancer survivors—Associations with demographic and medical characteristics. J. Cancer Surviv. 2018, 12, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, M.A.; Muzzatti, B.; Flaiban, C.; Gipponi, K.; Carnaghi, C.; Tralongo, P.; Caruso, M.; Cavina, R.; Tirelli, U. Long-term quality of life profile in oncology: A comparison between cancer survivors and the general population. Support. Care Cancer 2017, 26, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Hart, T.L.; Charles, S.T.; Gunaratne, M.; Baxter, N.N.; Cotterchio, M.; Cohen, Z.; Gallinger, S. Symptom Severity and Quality of Life Among Long-term Colorectal Cancer Survivors Compared With Matched Control Subjects: A Population-Based Study. Dis. Colon Rectum 2018, 61, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, V.; Oerlemans, S.; van de Poll-Franse, L.V.; Vingerhoets, A.J.; Mols, F. Suffering in long-term cancer survivors: An evaluation of the PRISM-R2 in a population-based cohort. Qual. Life Res. 2011, 20, 1645–1654. [Google Scholar] [CrossRef]
- Hedman, C.; Djärv, T.; Strang, P.; Lundgren, C.I. Determinants of long-term quality of life in patients with differentiated thyroid carcinoma—A population-based cohort study in Sweden. Acta Oncol. 2015, 55, 365–369. [Google Scholar] [CrossRef]
- Joly, F.; Espié, M.; Marty, M.; Héron, J.-F.; Henry-Amar, M. Long-term quality of life in premenopausal women with node-negative localized breast cancer treated with or without adjuvant chemotherapy. Br. J. Cancer 2000, 83, 577–582. [Google Scholar] [CrossRef]
- Kendall, A.R.; Mahue-Giangreco, M.; Carpenter, C.; Ganz, P.A.; Bernstein, L. Influence of exercise activity on quality of life in long-term breast cancer survivors. Qual. Life Res. 2005, 14, 361–371. [Google Scholar] [CrossRef]
- Mazariego, C.G.; Juraskova, I.; Campbell, R.; Smith, D.P. Long-term unmet supportive care needs of prostate cancer survivors: 15-year follow-up from the NSW Prostate Cancer Care and Outcomes Study. Support. Care Cancer 2020, 28, 5511–5520. [Google Scholar] [CrossRef]
- Paskett, E.D.; Herndon, J.E.; Day, J.M.; Stark, N.N.; Winer, E.P.; Grubbs, S.S.; Pavy, M.D.; Shapiro, C.L.; List, M.A.; Hensley, M.L.; et al. Applying a conceptual model for examining health-related quality of life in long-term breast cancer survivors: CALGB study 79804. Psycho-Oncology 2008, 17, 1108–1120. [Google Scholar] [CrossRef]
- Hammerer, P.G.; Wirth, M.P.; On Behalf of the ENA E005/DE-N-LEU-019 Study Group. Health-Related Quality of Life in 536 Long-Term Prostate Cancer Survivors after Treatment with Leuprorelin Acetate: A Combined Retrospective and Prospective Analysis. Urol. Int. 2017, 100, 72–78. [Google Scholar] [CrossRef]
- Mosher, C.E.; Sloane, R.; Morey, M.C.; Snyder, D.C.; Cohen, H.J.; Ms, P.E.M.; Demark-Wahnefried, W. Associations between lifestyle factors and quality of life among older long-term breast, prostate, and colorectal cancer survivors. Cancer 2009, 115, 4001–4009. [Google Scholar] [CrossRef]
- Van Dipten, C.; Hartman, T.C.O.; Biermans, M.C.J.; Assendelft, W.J.J. Substitution scenario in follow-up of chronic cancer patients in primary care: Prevalence, disease duration and estimated extra consultation time. Fam. Pract. 2015, 33, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Fayers, P.M.; Aaronson, N.K.; Bjordal, K.; Groenvold, M.; Curran, D.; Bottomley, A.; On behalf of the EORTC Quality of Life Group. The EORTC QLQ-C30 Scoring Manual, 3rd ed.; European Organisation for Research and Treatment of Cancer: Brussels, Belgium, 2001. [Google Scholar]
- Mols, F.; van de Poll-Franse, L.V.; Vingerhoets, A.J.J.M.; Hendrikx, A.; Aaronson, N.K.; Houterman, S.; Coebergh, J.W.W.; Essink-Bot, M.L. Long-term quality of life among Dutch prostate cancer survivors: Results of a population-based study. Cancer 2006, 107, 2186–2196. [Google Scholar] [CrossRef] [PubMed]
- Adam, S.; Doege, D.; Koch-Gallenkamp, L.; Thong, M.S.Y.; Bertram, H.; Eberle, A.; Holleczek, B.; Pritzkuleit, R.; Waldeyer-Sauerland, M.; Waldmann, A.; et al. Age-specific health-related quality of life in disease-free long-term prostate cancer survivors versus male population controls—Results from a population-based study. Support. Care Cancer 2019, 28, 2875–2885. [Google Scholar] [CrossRef]
- Andersen, B.L.; Shapiro, C.L.; Farrar, W.B.; Crespin, T.; Wells-DiGregorio, S. Psychological responses to cancer recurrence. Cancer 2005, 104, 1540–1547. [Google Scholar] [CrossRef] [PubMed]
- Wiltink, L.M.; White, K.; King, M.T.; Rutherford, C. Systematic review of clinical practice guidelines for colorectal and anal cancer: The extent of recommendations for managing long-term symptoms and functional impairments. Support. Care Cancer 2020, 28, 2523–2532. [Google Scholar] [CrossRef] [PubMed]
- Arndt, V.; Koch-Gallenkamp, L.; Bertram, H.; Eberle, A.; Holleczek, B.; Pritzkuleit, R.; Waldeyer-Sauerland, M.; Waldmann, A.; Zeissig, S.R.; Doege, D.; et al. Return to work after cancer. A multi-regional population-based study from Germany. Acta Oncol. 2019, 58, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Pearce, A.; Tomalin, B.; Kaambwa, B.; Horevoorts, N.; Duijts, S.; Mols, F.; Van De Poll-Franse, L.; Koczwara, B. Financial toxicity is more than costs of care: The relationship between employment and financial toxicity in long-term cancer survivors. J. Cancer Surviv. 2019, 13, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, R.; Hinz, A. Reference data for the quality of life questionnaire EORTC QLQ-C30 in the general German population. Eur. J. Cancer 2001, 37, 1345–1351. [Google Scholar] [CrossRef]
- Leach, C.R.; Weaver, K.E.; Aziz, N.M.; Alfano, C.M.; Bellizzi, K.M.; Kent, E.E.; Forsythe, L.P.; Rowland, J.H. The complex health profile of long-term cancer survivors: Prevalence and predictors of comorbid conditions. J. Cancer Surviv. 2014, 9, 239–251. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Cancer Survivors | Population Controls | pcrude (χ2) | padj (CMH) | |||
|---|---|---|---|---|---|---|---|
| n | % | n | % | %adj a | |||
| Total | 2704 | 100.0 | 1765 | 100.0 | |||
| Age (at survey) | |||||||
| 40–59 years | 169 | 6.3 | 726 | 41.1 | 6.3 | <0.0001 | - |
| 60–69 years | 468 | 17.3 | 399 | 22.6 | 17.3 | ||
| 70–79 years | 1114 | 41.2 | 345 | 19.5 | 41.2 | ||
| 80–99 years | 953 | 35.2 | 295 | 16.7 | 35.2 | ||
| Mean age (SD) | 75.2 | (8.7) | 63.4 | (13.8) | |||
| Sex | |||||||
| Female | 1438 | 53.2 | 942 | 53.4 | 53.2 | 0.90 | - |
| Male | 1266 | 46.8 | 823 | 46.6 | 46.8 | ||
| Education | |||||||
| ≤9 years | 1267 | 46.9 | 671 | 38.0 | 49.5 | <0.0001 | 0.08 |
| 10 years | 707 | 26.1 | 484 | 27.4 | 22.0 | ||
| ≥12 years | 730 | 27.0 | 610 | 34.6 | 28.5 | ||
| Employment (at survey) | |||||||
| Full-time | 90 | 3.3 | 455 | 25.8 | 5.2 | <0.0001 | <0.0001 |
| Part-time | 139 | 5.1 | 255 | 14.4 | 5.6 | ||
| Unemployed | 6 | 0.2 | 53 | 3.0 | 1.3 | ||
| Housewife | 286 | 10.6 | 180 | 10.2 | 13.3 | ||
| (Early) Retirement | 1978 | 73.1 | 752 | 42.6 | 70.9 | ||
| Other | 47 | 1.7 | 50 | 2.8 | 2.3 | ||
| Multiple answers | 158 | 5.8 | 20 | 1.1 | 1.4 | ||
| Having a partner (at survey) | 2046 | 75.7 | 1335 | 75.7 | 69.8 | 0.96 | 0.0002 |
| Having children | 2317 | 85.7 | 1503 | 85.2 | 89.8 | 0.61 | 0.003 |
| Current marital status | |||||||
| Unmarried | 118 | 4.4 | 166 | 9.4 | 3.9 | <0.0001 | <0.0001 |
| Married | 1914 | 70.8 | 1180 | 66.8 | 63.8 | ||
| Divorced | 183 | 6.8 | 167 | 9.5 | 6.6 | ||
| Widowed | 489 | 18.1 | 252 | 14.3 | 25.7 | ||
| History of comorbidities (self-report) | |||||||
| Coronary heart disease | 254 | 9.4 | 157 | 8.9 | 13.9 | 0.63 | <0.0001 |
| Osteoporosis | 365 | 13.5 | 167 | 9.5 | 15.7 | <0.0001 | 0.33 |
| Diabetes | 378 | 14.0 | 222 | 13.0 | 17.7 | 0.19 | 0.01 |
| Chronic back pain | 914 | 33.8 | 567 | 32.1 | 38.6 | 0.41 | 0.02 |
| Depression (ever) | 304 | 11.3 | 285 | 16.1 | 14.1 | <0.0001 | 0.04 |
| Characteristics | Disease-Free Survivors | Survivors with Active Disease | |||
|---|---|---|---|---|---|
| n | % | n | % | p (χ2) | |
| Total | 1972 | 72.9 | 732 | 27.1 | |
| Age at (current) survey | |||||
| 40–59 years | 118 | 6.0 | 51 | 7.0 | 0.10 |
| 60–69 years | 362 | 18.4 | 106 | 14.5 | |
| 70–79 years | 807 | 40.9 | 307 | 41.9 | |
| 80–99 years | 685 | 34.7 | 268 | 36.6 | |
| Mean age (SD) | 75.0 | (8.6) | 75.7 | (8.7) | |
| Tumor | |||||
| Breast cancer | 942 | 47.8 | 289 | 39.4 | 0.0003 |
| Colorectal cancer | 363 | 18.4 | 143 | 19.5 | |
| Prostate cancer | 666 | 33.8 | 301 | 41.1 | |
| Sex | |||||
| Female | 1098 | 55.7 | 340 | 46.5 | <0.0001 |
| Male | 874 | 44.3 | 392 | 53.5 | |
| Stage at diagnosis | |||||
| I | 611 | 31.0 | 182 | 24.9 | <0.0001 |
| II | 973 | 49.3 | 317 | 43.2 | |
| III | 389 | 19.7 | 159 | 21.7 | |
| IV | 75 | 10.2 | |||
| Recurrence/metastasis after diagnosis | 455 | 62.2 | |||
| Second primary tumor | 317 | 43.3 | |||
| Primary therapy | |||||
| Surgery | 1538 | 78.0 | 561 | 76.7 | 0.47 |
| Radiotherapy | 1100 | 55.8 | 447 | 61.1 | 0.01 |
| Chemotherapy | 806 | 40.9 | 273 | 37.3 | 0.09 |
| Hormone (endocrine) therapy | 595 | 32.2 | 263 | 38.5 | 0.003 |
| Immuno-/Antibody therapy | 109 | 6.3 | 41 | 6.5 | 0.87 |
| EORTC QLQ-C30 Scales/Scores | Cancer Survivors | Population Controls | ||||
|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Diff. | p (glm) | |
| Global health status/QoL | 64.4 | 1.2 | 61.5 | 1.2 | 2.9 | 0.0002 |
| Physical functioning | 77.7 | 1.1 | 77.6 | 1.1 | 0.0 | 0.97 |
| Role functioning | 69.0 | 1.5 | 70.6 | 1.6 | −1.5 | 0.13 |
| Emotional functioning | 70.4 | 1.3 | 71.5 | 1.3 | −1.1 | 0.20 |
| Cognitive functioning | 78.4 | 1.2 | 79.2 | 1.2 | −0.8 | 0.30 |
| Social functioning | 75.3 | 1.5 | 78.8 | 1.5 | −3.5 | 0.0003 |
| Fatigue | 37.8 | 1.4 | 36.0 | 1.4 | 1.8 | 0.045 |
| Nausea and vomiting | 3.6 | 0.6 | 2.7 | 0.6 | 0.9 | 0.02 |
| Pain | 28.6 | 1.6 | 32.5 | 1.6 | −3.9 | 0.0003 |
| Dyspnea | 24.0 | 1.5 | 21.0 | 1.6 | 3.0 | 0.003 |
| Insomnia | 33.2 | 1.7 | 30.8 | 1.8 | 2.4 | 0.04 |
| Appetite loss | 10.0 | 1.1 | 9.4 | 1.1 | 0.6 | 0.40 |
| Constipation | 14.0 | 1.3 | 9.6 | 1.3 | 4.4 | <0.0001 |
| Diarrhea | 13.5 | 1.1 | 6.4 | 1.1 | 7.1 | <0.0001 |
| Financial difficulties | 11.3 | 1.2 | 11.5 | 1.2 | −0.3 | 0.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doege, D.; Thong, M.S.Y.; Weißer, L.; Koch-Gallenkamp, L.; Jansen, L.; Bertram, H.; Eberle, A.; Holleczek, B.; Nennecke, A.; Pritzkuleit, R.; et al. Health-Related Quality of Life in Very Long-Term Cancer Survivors 14–24 Years Post-Diagnosis Compared to Population Controls: A Population-Based Study. Cancers 2021, 13, 2754. https://doi.org/10.3390/cancers13112754
Doege D, Thong MSY, Weißer L, Koch-Gallenkamp L, Jansen L, Bertram H, Eberle A, Holleczek B, Nennecke A, Pritzkuleit R, et al. Health-Related Quality of Life in Very Long-Term Cancer Survivors 14–24 Years Post-Diagnosis Compared to Population Controls: A Population-Based Study. Cancers. 2021; 13(11):2754. https://doi.org/10.3390/cancers13112754
Chicago/Turabian StyleDoege, Daniela, Melissa S. Y. Thong, Linda Weißer, Lena Koch-Gallenkamp, Lina Jansen, Heike Bertram, Andrea Eberle, Bernd Holleczek, Alice Nennecke, Ron Pritzkuleit, and et al. 2021. "Health-Related Quality of Life in Very Long-Term Cancer Survivors 14–24 Years Post-Diagnosis Compared to Population Controls: A Population-Based Study" Cancers 13, no. 11: 2754. https://doi.org/10.3390/cancers13112754
APA StyleDoege, D., Thong, M. S. Y., Weißer, L., Koch-Gallenkamp, L., Jansen, L., Bertram, H., Eberle, A., Holleczek, B., Nennecke, A., Pritzkuleit, R., Waldmann, A., Zeissig, S. R., Brenner, H., & Arndt, V. (2021). Health-Related Quality of Life in Very Long-Term Cancer Survivors 14–24 Years Post-Diagnosis Compared to Population Controls: A Population-Based Study. Cancers, 13(11), 2754. https://doi.org/10.3390/cancers13112754






