2-[18F]FDG PET/CT as a Predictor of Microvascular Invasion and High Histological Grade in Patients with Hepatocellular Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. PET/CT Acquisition
2.3. Image Evaluation
2.4. Pathological Study of the Resected Lesions
2.5. Post-Surgical Follow-up
2.6. Statistical Analysis
3. Results
3.1. Recurrence and Survival Data
3.2. Clinical and Histological Factors Predicting Tumor Recurrence after the Surgery
3.3. Correlation between Metabolic Parameters on PET/CT and Histological Factors of a Poor Prognosis
3.3.1. Presence of MVI
3.3.2. Poor Cellular Differentiation
3.4. Presence of MVI and/or Poor Cellular Differentiation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Heimbach, J.K.; Kulik, L.M.; Finn, R.S.; Sirlin, C.B.; Abecassis, M.M.; Roberts, L.R.; Zhu, A.X.; Murad, M.H.; Marrero, J.A. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology 2018, 67, 358–380. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Schwartz, M.; Mazzaferro, V. Resection and Liver Transplantation for Hepatocellular Carcinoma. Semin. Liver Dis. 2005, 25, 181–200. [Google Scholar] [CrossRef] [PubMed]
- Poon, R.T.-P.; Fan, S.-T.; Ng, I.O.-L.; Lo, C.-M.; Liu, C.-L.; Wong, J. Long-Term Survival and Pattern of Recurrence After Resection of Small Hepatocellular Carcinoma in Patients With Preserved Liver Function Implications for a Strategy of Salvage Transplantation. Ann. Surg. 2002, 235, 373–382. [Google Scholar] [CrossRef]
- Colecchia, A.; Schiumerini, R.; Cucchetti, A.; Cescon, M.; Taddia, M.; Marasco, G.; Festi, D. Prognostic factors for hepatocellular carcinoma recurrence. World J. Gastroenterol. 2014, 20, 5935–5950. [Google Scholar] [CrossRef]
- Forner, A.; Vilana, R.; Ayuso, C.; Bianchi, L.; Solé, M.; Ayuso, J.R.; Boix, L.; Sala, M.; Varela, M.; Llovet, J.M.; et al. Diagnosis of hepatic nodules 20 mm or smaller in cirrhosis: Prospective validation of the noninvasive diagnostic criteria for hepatocellular carcinoma. Hepatology 2008, 47, 97–104. [Google Scholar] [CrossRef]
- Ünal, E.; İdilman, İ.S.; Akata, D.; Özmen, M.N.; Karçaaltıncaba, M. Microvascular invasion in hepatocellular carcinoma. Diagn. Interv. Radiol. 2016, 22, 125–132. [Google Scholar] [CrossRef]
- Huang, J.; Tian, W.; Zhang, L.; Huang, Q.; Lin, S.; Ding, Y.; Liang, W.; Zheng, S. Preoperative prediction power of imaging methods for microvascular invasion in hepatocellular carcinoma: A systemic review and meta-analysis. Front. Oncol. 2020, 10, 887. [Google Scholar] [CrossRef]
- Castilla-Lièvre, M.A.; Franco, D.; Gervais, P.; Kuhnast, B.; Agostini, H.; Marthey, L.; Désarnaud, S.; Helal, B.O. Diagnostic value of combining 11C-choline and 18F-FDG PET/CT in hepatocellular carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 852–859. [Google Scholar] [CrossRef]
- Cho, K.J.; Choi, N.K.; Shin, M.H.; Chong, A.R. Clinical usefulness of FDG-PET in patients with hepatocellular carcinoma undergoing surgical resection. Ann. Hepato-Biliary-Pancreatic Surg. 2017, 21, 194–198. [Google Scholar] [CrossRef][Green Version]
- Hyun, S.H.; Eo, J.S.; Song, B.-I.; Lee, J.W.; Na, S.J.; Hong, I.K.; Oh, J.K.; Chung, Y.A.; Kim, T.S.; Yun, M. Preoperative prediction of microvascular invasion of hepatocellular carcinoma using 18F-FDG PET/CT: A multicenter retrospective cohort study. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Lim, C.; Salloum, C.; Chalaye, J.; Lahat, E.; Costentin, C.E.; Osseis, M.; Itti, E.; Feray, C.; Azoulay, D. 18F-FDG PET/CT predicts microvascular invasion and early recurrence after liver resection for hepatocellular carcinoma: A prospective observational study. HPB 2019, 21, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Galle, P.R.; Forner, A.; Llovet, J.M.; Mazzaferro, V.; Piscaglia, F.; Raoul, J.L.; Schirmacher, P.; Vilgrain, V. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef] [PubMed]
- Reynés-Llompart, G.; Sabaté-Llobera, A.; Llinares-Tello, E.; Martí-Climent, J.M.; Gámez-Cenzano, C. Image quality evaluation in a modern PET system: Impact of new reconstructions methods and a radiomics approach. Sci. Rep. 2019, 9, 10640. [Google Scholar] [CrossRef]
- Mulé, S.; Chalaye, J.; Legou, F.; Tenenhaus, A.; Calderaro, J.; Galletto Pregliasco, A.; Laurent, A.; Kharrat, R.; Amaddeo, G.; Regnault, H. Hepatobiliary MR contrast agent uptake as a predictive biomarker of aggressive features on pathology and reduced recurrence-free survival in resectable hepatocellular carcinoma: Comparison with dual-tracer 18F-FDG and 18F-FCH PET/CT. Eur. Radiol. 2020, 30, 5348–5357. [Google Scholar] [CrossRef]
- Ahn, S.Y.; Lee, J.M.; Joo, I.; Lee, E.S.; Lee, S.J.; Cheon, G.J.; Choi, B. Prediction of microvascular invasion of hepatocellular carcinoma using gadoxetic acid-enhanced MR and 18F-FDG PET/CT. Abdom. Imaging 2015, 40, 843–851. [Google Scholar] [CrossRef]
- Zhang, Y.; Shu, Z.; Ye, Q.; Chen, J.; Zhong, J.; Jiang, H.; Wu, C.; Yu, T.; Pang, P.; Ma, T.; et al. Preoperative Prediction of Microvascular Invasion in Hepatocellular Carcinoma via Multi-Parametric MRI Radiomics. Front. Oncol. 2021, 11, 633596. [Google Scholar] [CrossRef]
- Gallagher, B.M.; Fowler, J.S.; Gutterson, N.I.; MacGregor, R.R.; Wolf, A.P. Metabolic Trapping as a Principle of Radiopharmaceutical Design: Some Factors Responsible for the Biodistribution of [18F] 2-Deoxy-2-Fluoro-D-Glucose. J. Nucl. Med. 1978, 19, 1154–1161. [Google Scholar]
- Torizuka, T.; Tamaki, N.; Inokuma, T.; Magata, Y.; Sasayama, S. In Vivo Assessment of Glucose Metabolism in Hepatocellular Carcinoma with FDG-PET. J. Nucl. Med. 1995, 36, 1811–1817. [Google Scholar]
- Kroh, A.; Uschner, D.; Lodewick, T.; Eickhoff, R.M.; Schöning, W.; Ulmer, F.T.; Neumann, U.P.; Binnebösel, M. Impact of body composition on survival and morbidity after liver resection in hepatocellular carcinoma patients. Hepatobiliary Pancreat. Dis. Int. 2019, 18, 28–37. [Google Scholar] [CrossRef]
- Sugawara, Y.; Zasadny, K.; Neyhoff, A.; Wahl, R. Nuclear Medicine Reevaluation of the Standardized Uptake Value for FDG: Variations with Body Weight and Methods for Correction 1. Radiology 1999, 213, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Zhang, G.; Guo, J.; Hao, S.; Wang, Z.; Fan, G. Prognostic value of pretreatment PET/CT lean body mass-corrected parameters in patients with hepatocellular carcinoma. Nucl. Med. Commun. 2018, 39, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Azmi, N.H.M.; Suppiah, S.; Liong, C.W.; Noor, N.M.; Said, S.M.; Hanafi, M.H.; Kaewput, C.; Saad, F.F.A.; Vinjamuri, S. Reliability of standardized uptake value normalized to lean body mass using the liver as a reference organ, in contrast-enhanced 18F-FDG PET/CT imaging. Radiat. Phys. Chem. 2018, 147, 35–39. [Google Scholar] [CrossRef]
- Bailly, M.; Venel, Y.; Orain, I.; Salamé, E.; Ribeiro, M.J. 18F-FDG PET in liver transplantation setting of hepatocellular carcinoma predicting histology? Clin. Nucl. Med. 2016, 41, e126–e129. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Kim, H.S.; Lee, S.; Lee, J.W. Emerging role of 18F-fluorodeoxyglucose positron emission tomography for guiding management of hepatocellular carcinoma. World J. Gastroenterol. 2019, 25, 1289–1306. [Google Scholar] [CrossRef]
- Krak, N.C.; Boellaard, R.; Hoekstra, O.S.; Twisk, J.W.R.; Hoekstra, C.J.; Lammertsma, A.A. Effects of ROI definition and reconstruction method on quantitative outcome and applicability in a response monitoring trial. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Ferrer-Fàbrega, J.; Forner, A.; Liccioni, A.; Miquel, R.; Molina, V.; Navasa, M.; Fondevila, C.; García-Valdecasas, J.C.; Bruix, J.; Fuster, J. Prospective validation of ab initio liver transplantation in hepatocellular carcinoma upon detection of risk factors for recurrence after resection. Hepatology 2016, 63, 839–849. [Google Scholar] [CrossRef]
- Allard, M.A.; Sebagh, M.; Ruiz, A.; Guettier, C.; Paule, B.; Vibert, E.; Cunha, A.S.; Cherqui, D.; Samuel, D.; Bismuth, H.; et al. Does pathological response after transarterial chemoembolization for hepatocellular carcinoma in cirrhotic patients with cirrhosis predict outcome after liver resection or transplantation? J. Hepatol. 2015, 63, 83–92. [Google Scholar] [CrossRef]
- Lei, J.Y.; Zhong, J.J.; Yan, L.N.; Zhu, J.Q.; Wang, W.T.; Zeng, Y.; Li, B.; Wen, T.F.; Yang, J.Y.; Liver Surgery Group. Response to transarterial chemoembolization as a selection criterion for resection of hepatocellular carcinomas. Br. J. Surg. 2016, 103, 881–890. [Google Scholar] [CrossRef][Green Version]
- Kobayashi, T.; Aikata, H.; Honda, F.; Nakano, N.; Nakamura, Y.; Hatooka, M.; Morio, K.; Morio, R.; Fukuhara, T.; Masaki, K.; et al. Preoperative Fluorine 18 Fluorodeoxyglucose Positron Emission Tomography/Computed Tomography for Prediction of Microvascular Invasion in Small Hepatocellular Carcinoma. J. Comput. Assist. Tomogr. 2016, 40, 524–530. [Google Scholar] [CrossRef]
- Chou, C.T.; Chen, R.C.; Lin, W.C.; Ko, C.J.; Chen, C.B.; Chen, Y.L. Prediction of microvascular invasion of hepatocellular carcinoma: Preoperative CT and histopathologic correlation. Am. J. Roentgenol. 2014, 202, W253–W259. [Google Scholar] [CrossRef] [PubMed]
- Jeng, L.B.; Changlai, S.P.; Shen, Y.Y.; Lin, C.C.; Tsai, C.H.; Kao, C.H. Limited value of 18F-2-deoxyglucose positron emission tomography to detect hepatocellular carcinoma in hepatitis B virus carriers. Hepatogastroenterology 2003, 50, 2154–2156. [Google Scholar] [PubMed]
- Asman, Y.; Evenson, A.R.; Even-Sapir, E.; Shibolet, O. [18F]fludeoxyglucose positron emission tomography and computed tomography as a prognostic tool before liver transplantation, resection, and loco-ablative therapies for hepatocellular carcinoma. Liver Transplant. 2015, 21, 572–580. [Google Scholar] [CrossRef]
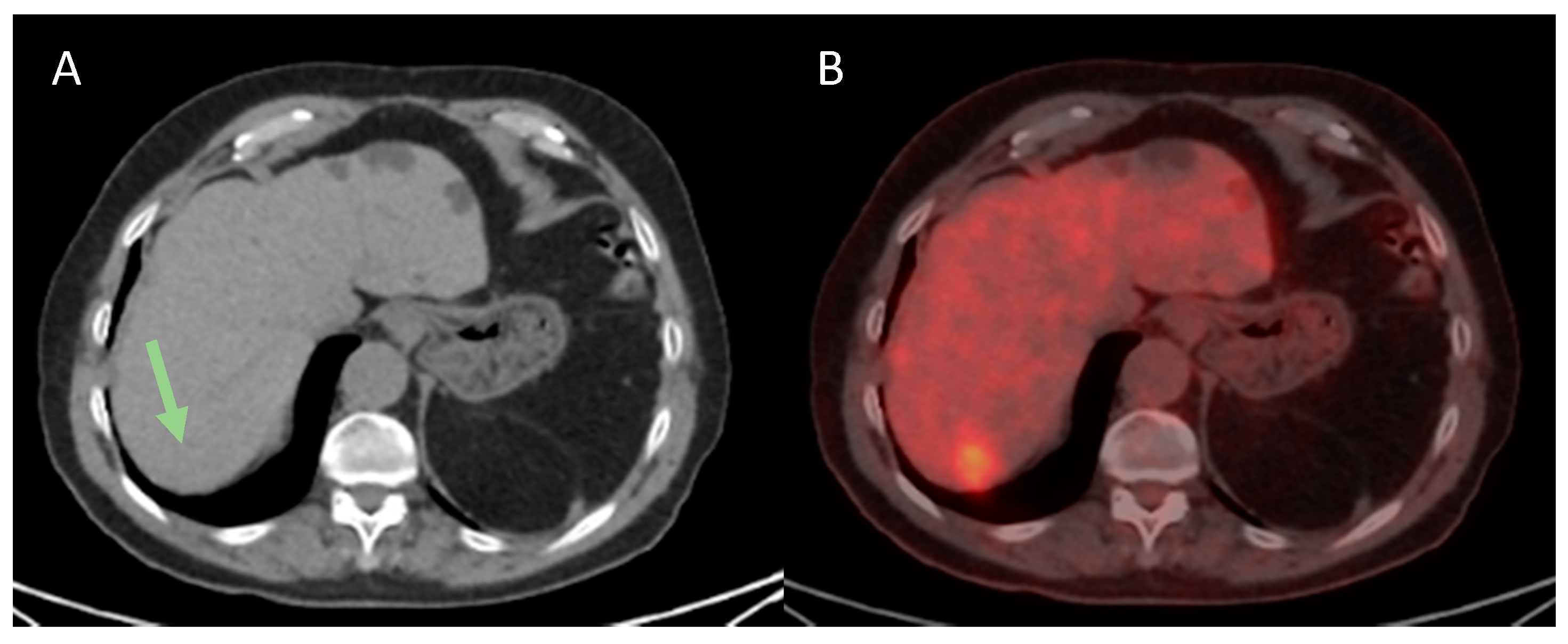
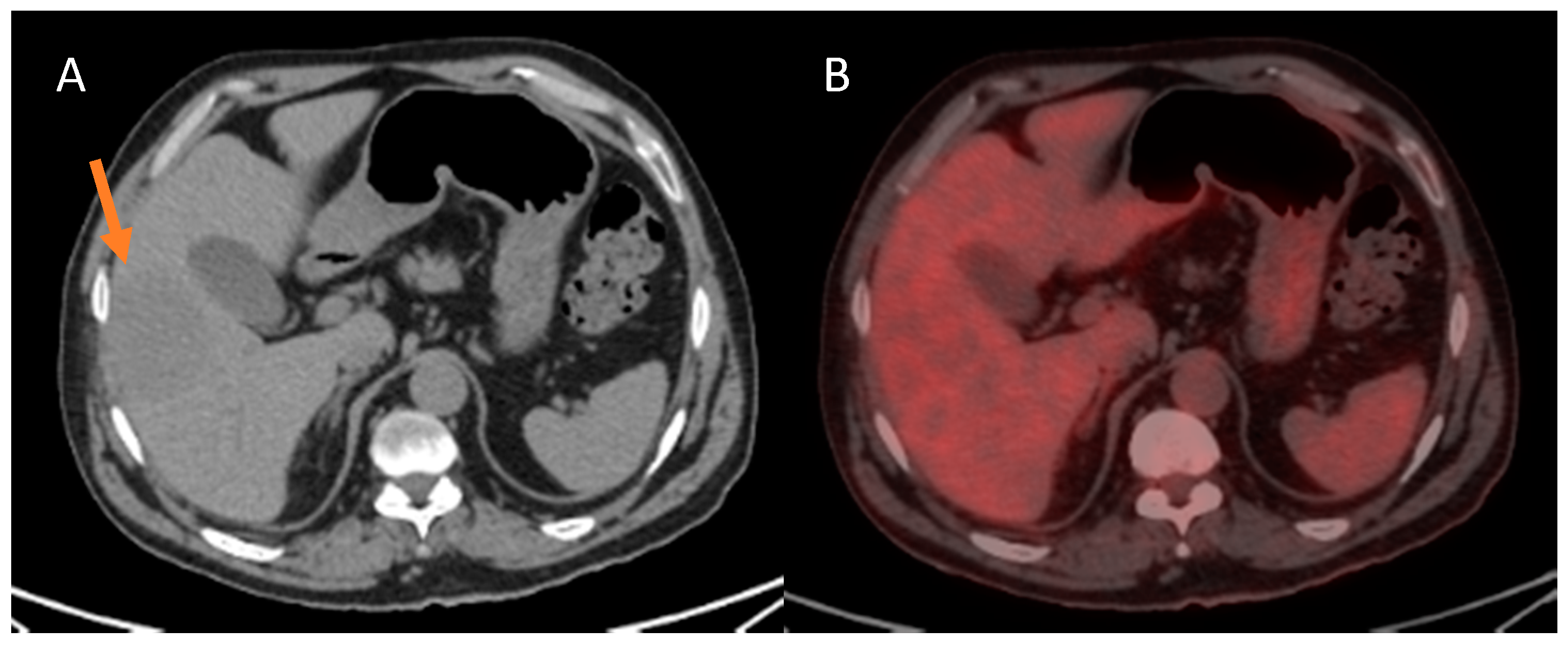
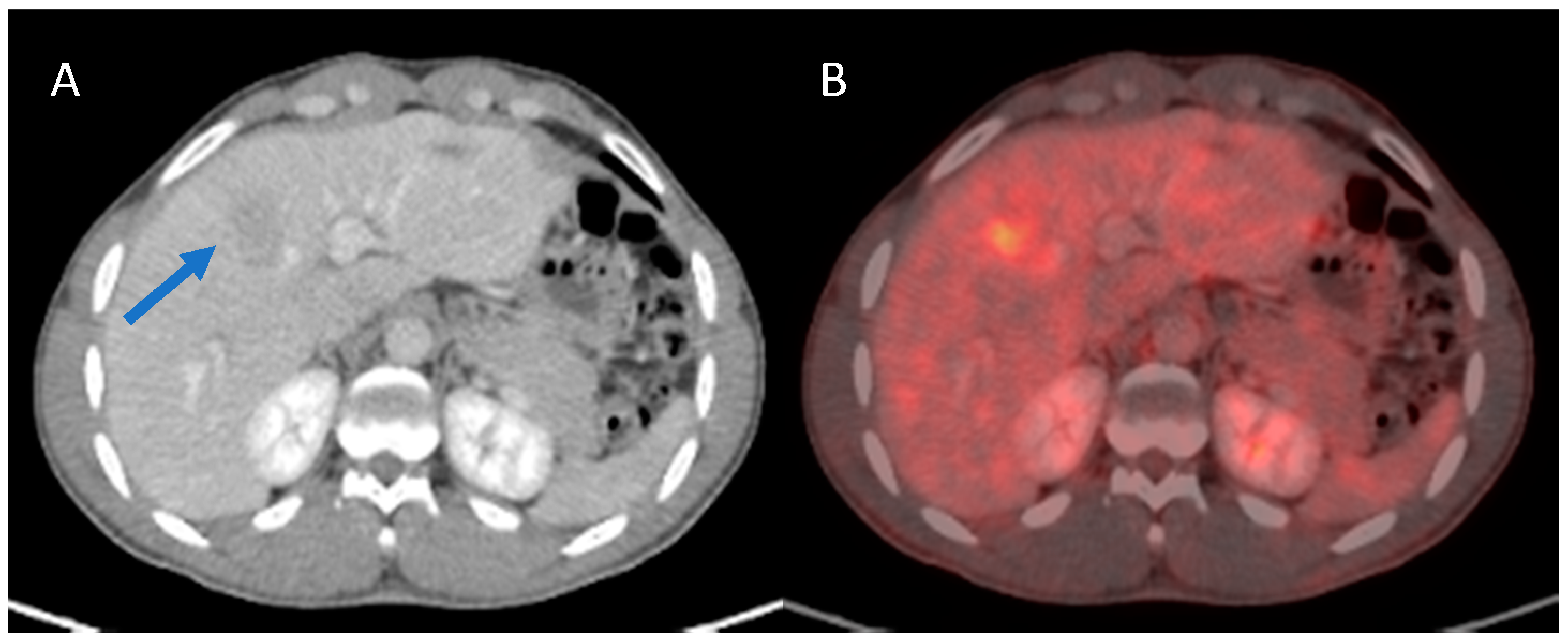
| Variables | n = 41 |
|---|---|
| Sex (M/F) | 39/2 |
| Age (years), mean (SD) | 61 (11.4) |
| BMI (kg/m2), mean (SD) | 27.1 (4.3) |
| HVPG ≥10 mmHg (measured in 10 patients), n | 1 |
| MELD score, mean (SD) | 7.2 (1.5) |
| Liver disease, n | |
| HBV related | 7 |
| HCV related | 8 |
| Alcohol related | 9 |
| NASH | 1 |
| None or unknown | 16 |
| HCV Ab+, n (%) | 12 (29.3) |
| HCV RNA+, n | 2 |
| HBV S Ag+, n (%) | 7 (17.1) |
| HBV DNA+, n | 0 |
| AFP (ng/mL), mean (SD) | 1573.2 (926.1) |
| Tumor characteristics on cross-sectional imaging | |
| Size of the largest nodule (mm), mean (SD) | 42 (22) |
| Liver and kidney function test results | |
| Total bilirubin (µmol/L), mean (SD) | 10.0 (4.3) |
| Albumin (g/L), mean (SD) | 45.9 (3.5) |
| Platelet (×105/mm3), mean (SD) | 209 (68) |
| Time of prothrombin ratio, mean (SD) | 1.11 (0.43) |
| Creatinine (µmol/L), mean (SD) | 81.6 (20.0) |
| ASA classification (I/II/III/IV) | 1/19/21/0 |
| MELD, mean (SD) | 7.2 (1.5) |
| Child–Pugh | 5 (0) |
| Intra-operative and post-operative variables | |
| Open/laparoscopic | 21/20 |
| Major resection, n (%) | 3 (7.3) |
| Anatomical resection, n (%) | 17 (41.5) |
| Pringle maneuver yes/no | 37/4 |
| Perioperative transfusion yes/no | 4/37 |
| Post-operative complications, Clavien classification (No/I/II/IIIa) | 28/6/4/3 |
| Mortality, n | 0 |
| Hospitalization time (days), mean (SD) | 6.9 (6.8) |
| Pathological variables | |
| Fibrosis grade (0/F1 + F2/F3 + F4) | 13/6/22 |
| Histological type (HCC/hepatocholangiocarcinoma) | 39/2 |
| Number of tumors, mean (SD) | 1.12 (0.33) |
| Multiple tumors, n (%) | 5 (12.2) |
| Tumor size (mm), mean (SD) | 43 (24.8) |
| Grade of differentiation (G1 + G2/G3 + G4) | 29/12 |
| Satellite nodules, n (%) | 4 (9.8) |
| Microvascular invasion, n (%) | 17 (41.5) |
| Margin invasion, n (%) | 6 (14.6) |
| Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|
| Preoperative Variables | p-Value | HR 95% CI | p-Value |
| Sex (M/F) | 0.860 | ||
| Age ≥ 70 years | 0.076 | ||
| BMI ≥ 30 kg/m2 | 0.582 | ||
| MELD score ≥ 9 | 0.418 | ||
| ASA classification (I/II/III/IV) | 0.177 | ||
| Liver stiffness (>14 kPa) | 0.715 | ||
| HBV S Ag+ | 0.009 | 1.18 (0.22–6.31) | 0.844 |
| HCV Ab+ | 0.826 | ||
| AFP ≥ 200 ng/mL | 0.563 | ||
| Intra-operative and post-operative variables | |||
| Open/laparoscopic | 0.684 | ||
| Major resection | 0.380 | ||
| Pringle maneuver | 0.065 | ||
| Anatomical resection | 0.187 | ||
| Perioperative transfusion | 0.372 | ||
| Post-operative complications | 0.235 | ||
| Histopathological variables | |||
| Fibrosis grade (0 + F1 + F2/F3 + F4) | 0.641 | ||
| Multiple nodules | 0.119 | ||
| Satellite nodules | 0.280 | ||
| Microvascular invasion (MVI) | 0.014 | 3.93 (0.39–39.21) | 0.243 |
| Differentiation grade (G1 + G2/G3 + G4) | 0.001 | 8.51 (0.85–84.75) | 0.068 |
| Histology (HCC/hepatocholangiocarcinoma) | 0.385 | ||
| R1 resection | 0.591 | ||
| Variables | SULpeak60 ≥ 2.26 (n = 21) | SULpeak60 < 2.26 (n = 20) | p-Value |
|---|---|---|---|
| Sex (M/F) | 21/0 | 18/2 | 0.232 |
| Age (years), mean (SD) | 60.4 (12.8) | 62.6 (10.4) | 0.547 |
| BMI (kg/m2), mean (SD) | 26.6 (4.0) | 27.6 (4.6) | 0.450 |
| HCV Ab+, n (%) HCV RNA+, n (%) | 2 (9.5) 0 | 10 (50) 2 (10) | 0.006 1.000 |
| HBV S Ag+, n (%) | 7 (33.3) | 0 (0) | 0.009 |
| AFP (ng/mL), mean (SD) | 2091.0 (13109.7) | 24 (66.8) | 0.359 |
| AFP ≥ 200 ng/mL, n (%) | 3 (14.3) | 1 (5.6) | 0.609 |
| Tumor characteristics on cross-sectional imaging | |||
| Size of the nodule (mm), mean (SD) | 5.1 (2.6) | 3.2 (1.2) | 0.006 |
| Functional preoperative status | |||
| ASA classification (I/II/III/IV) | 1/6/14/0 | 0/13/7/0 | 0.053 |
| MELD | 7.19 (1.8) | 7.2 (1.2) | 0.985 |
| Intra-operative and post-operative variables | |||
| Open/laparoscopic | 13/8 | 8/12 | 0.217 |
| Major resection, n (%) | 2 (9.5) | 1 (5) | 1.000 |
| Anatomical resection, n (%) | 10 (47.6) | 8 (35) | 0.530 |
| Pringle maneuver yes/no | 18 (85.7) | 19 (95.0) | 1.000 |
| Perioperative transfusion yes/no | 3/18 | 1/19 | 0.606 |
| Post-operative complications. Clavien classification (No/I/II/IIIa) | 14/4/1/2 | 14/2/3/1 | 0.577 |
| Mortality, n | 0 | 0 | |
| Hospitalization time (days), mean (SD) | 6.95 (5.5) | 7.1 (8.0) | 0.982 |
| Pathological variables | |||
| Fibrosis grade (0 + F1 + F2/F3 + F4) | 11/10 | 8/12 | 0.536 |
| Histological type (HCC/hepatocholangiocarcinoma) | 21/0 | 18/2 | 0.232 |
| Number of tumors | 1.1 (0.4) | 1.05 (0.2) | 0.174 |
| Multiple tumors, n (%) | 4 (19) | 1 (5) | 0.343 |
| Tumor size (mm), mean (SD) | 5.2 (2.8) | 3.4 (1.6) | 0.013 |
| Tumor size ≥ 30 mm, n (%) | 18 (85.7) | 10 (50) | 0.020 |
| Grade of differentiation (G1 + G2/G3 + G4) | 12/9 | 17/3 | 0.085 |
| Satellite nodules, n (%) | 3 (14.3) | 1 (5) | 0.606 |
| Microvascular invasion, n (%) | 13 (61.9) | 4 (20) | 0.010 |
| R1 resection, n (%) | 5 (23.8) | 1 (5.3) | 0.186 |
| Variables | TLRpeak60 ≥ 1.20 (n = 20) | TLRpeak60 < 1.20 (n = 21) | p-Value |
|---|---|---|---|
| Sex (M/F) | 20/0 | 19/2 | 0.488 |
| Age (years), mean (SD) | 59.0 (13.5) | 63.8 (8.8) | 0.183 |
| BMI (kg/m2), mean (SD) | 26.4 (4.3) | 27.8 (4.3) | 0.327 |
| HCV Ab+, n (%) HCV RNA+, n (%) | 3 (15) 0 (0) | 9 (42.9) 2 (22.2) | 0.085 1.000 |
| HBV S Ag+, n (%) | 5 (25) | 2 (9.5) | 0.238 |
| AFP (ng/mL), mean (SD) | 3046.4 (13,432.9) | 22.6 (64.4) | 0.327 |
| AFP ≥ 200 ng/mL, n (%) | 3 (15) | 1 (5.3) | 0.605 |
| Tumor characteristics on cross-sectional imaging | |||
| Size of the largest nodule (mm), mean (SD) | 5.2 (2.7) | 3.3 (1.2) | 0.008 |
| Functional preoperative status | |||
| ASA classification (I/II/III/IV) | 1/7/12/0 | 0/12/9/0 | 0.210 |
| MELD | 7.6 (0.9) | 6.8 (1.9) | 0.104 |
| Intra-operative and post-operative variables | |||
| Open/laparoscopic | 11/9 | 10/11 | 0.758 |
| Major resection, n (%) | 2 (10) | 1 (4.8) | 0.606 |
| Anatomical resection, n (%) | 10 (50) | 7 (33.3) | 0.350 |
| Pringle maneuver yes/no | 17/3 | 20/1 | 0.343 |
| Perioperative transfusion yes/no | 3/17 | 1/20 | 0.343 |
| Post-operative complications. Clavien classification (No/I/II/IIIa) | 14/3/1/2 | 14/3/3/1 | 0.727 |
| Mortality, n | 0 | 0 | |
| Hospitalization time (days), mean (SD) | 6.9 (5.6) | 7.0 (7.8) | 0.945 |
| Pathological variables | |||
| Fibrosis grade (0 + F1 + F2/F3 + F4) | 11/9 | 8/13 | 0.354 |
| Histological type (HCC/hepatocholangiocarcinoma) | 20/0 | 19/2 | 0.488 |
| Number of tumors | 1.2 (0.4) | 1.05 (0.2) | 0.151 |
| Multiple tumors, n (%) | 4 (20) | 1 (4.8) | 0.184 |
| Tumor size (mm), mean (SD) | 4.9 (2.6) | 3.9 (2.3) | 0.265 |
| Tumor size > 30 mm, n (%) | 18 (90) | 10 (47.6) | 0.006 |
| Grade of differentiation (G1 + G2/G3 + G4) | 10/10 | 19/2 | 0.006 |
| Satellite nodules yes/no | 4/16 | 0/21 | 0.048 |
| Microvascular invasion, n (%) | 12 (60) | 5 (23.8) | 0.028 |
| R1 resection, n (%) | 5 (25) | 1 (5) | 0.182 |
| Variables | Univariate Analysis | ||
|---|---|---|---|
| HR | 95% CI | p-Value | |
| Sex (M/F) | 0.972 | ||
| Age ≥ 70 years | 0.255 | ||
| BMI ≥ 30 kg/m2 | 0.929 | ||
| Diabetes mellitus | 0.282 | ||
| HCV Ab+, n (%) HCV RNA+, n (%) | 0.559 0.999 | ||
| HBV S Ag+, n (%) | 0.253 | ||
| MELD ≥ 9 | 0.168 | ||
| AFP (ng/mL) | 0.535 | ||
| AFP ≥ 200 ng/mL | 0.337 | ||
| Liver stiffness > 14 kPa | 0.140 | ||
| Preoperative tumor size | 0.154 | ||
| Preoperative tumor size ≥30 mm | 0.469 | ||
| TLRpeak60 p50 | 7.50 | 1.87–29.98 | 0.004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabaté-Llobera, A.; Mestres-Martí, J.; Reynés-Llompart, G.; Lladó, L.; Mils, K.; Serrano, T.; Cortés-Romera, M.; Bertran, E.; Fabregat, I.; Ramos, E. 2-[18F]FDG PET/CT as a Predictor of Microvascular Invasion and High Histological Grade in Patients with Hepatocellular Carcinoma. Cancers 2021, 13, 2554. https://doi.org/10.3390/cancers13112554
Sabaté-Llobera A, Mestres-Martí J, Reynés-Llompart G, Lladó L, Mils K, Serrano T, Cortés-Romera M, Bertran E, Fabregat I, Ramos E. 2-[18F]FDG PET/CT as a Predictor of Microvascular Invasion and High Histological Grade in Patients with Hepatocellular Carcinoma. Cancers. 2021; 13(11):2554. https://doi.org/10.3390/cancers13112554
Chicago/Turabian StyleSabaté-Llobera, Aida, Judit Mestres-Martí, Gabriel Reynés-Llompart, Laura Lladó, Kristel Mils, Teresa Serrano, Montserrat Cortés-Romera, Esther Bertran, Isabel Fabregat, and Emilio Ramos. 2021. "2-[18F]FDG PET/CT as a Predictor of Microvascular Invasion and High Histological Grade in Patients with Hepatocellular Carcinoma" Cancers 13, no. 11: 2554. https://doi.org/10.3390/cancers13112554
APA StyleSabaté-Llobera, A., Mestres-Martí, J., Reynés-Llompart, G., Lladó, L., Mils, K., Serrano, T., Cortés-Romera, M., Bertran, E., Fabregat, I., & Ramos, E. (2021). 2-[18F]FDG PET/CT as a Predictor of Microvascular Invasion and High Histological Grade in Patients with Hepatocellular Carcinoma. Cancers, 13(11), 2554. https://doi.org/10.3390/cancers13112554







