Diagnostic and Prognostic Value of miR-16, miR-146a, miR-192 and miR-221 in Exosomes of Hepatocellular Carcinoma and Liver Cirrhosis Patients
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Populations
2.2. Verification of Hemolysis in Plasma Samples
2.3. Exosome Extraction
2.4. Western Blot
2.5. Extraction of miRNAs from Exosomes
2.6. Conversion of Exosomal miRNAs into cDNA
2.7. Preamplification of cDNA
2.8. PCR-Based TaqMan miRNA Arrays
2.9. Single TaqMan PCR Analyses
2.10. Data Normalization and Statistical Analyses
3. Results
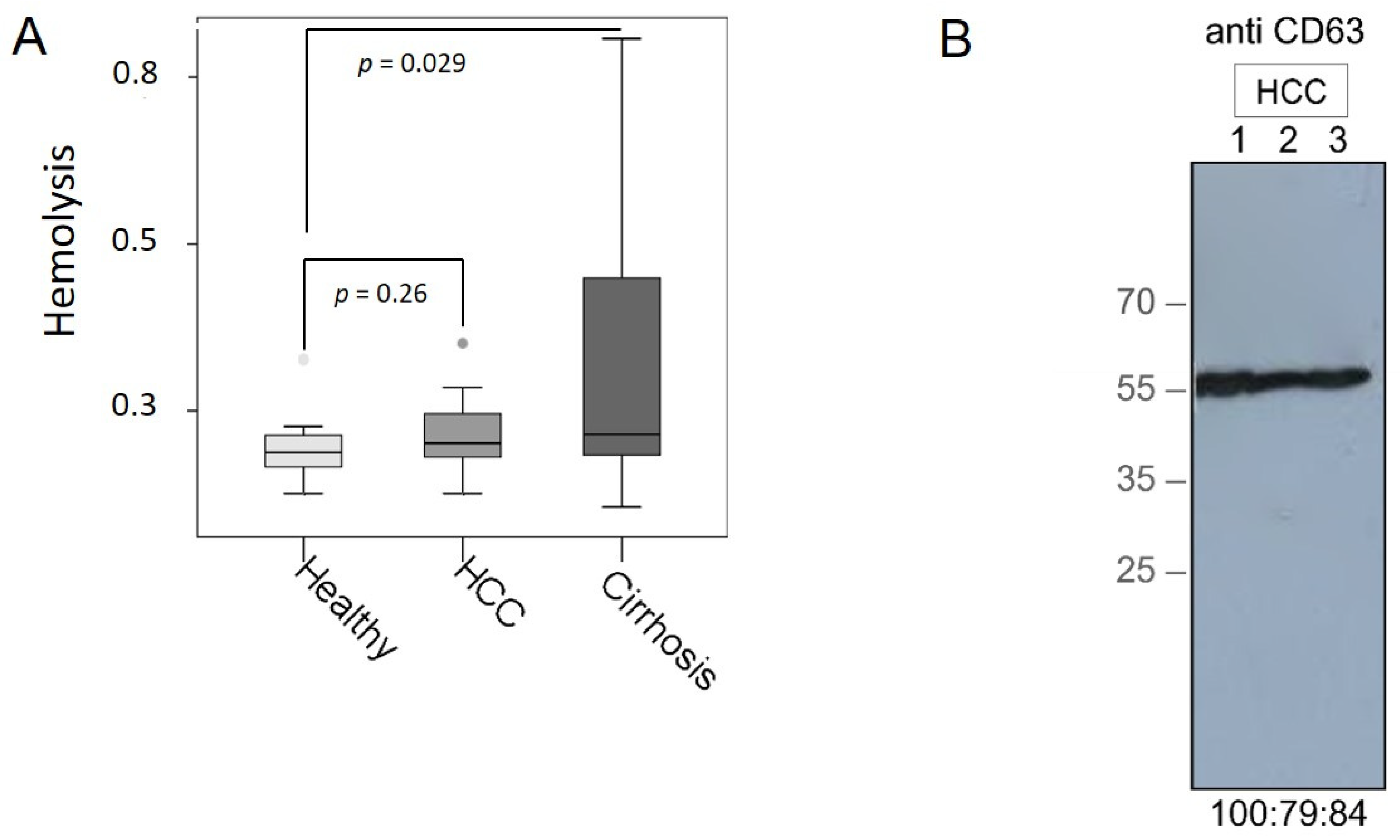
3.1. Verification of Plasma Samples and Exosomes
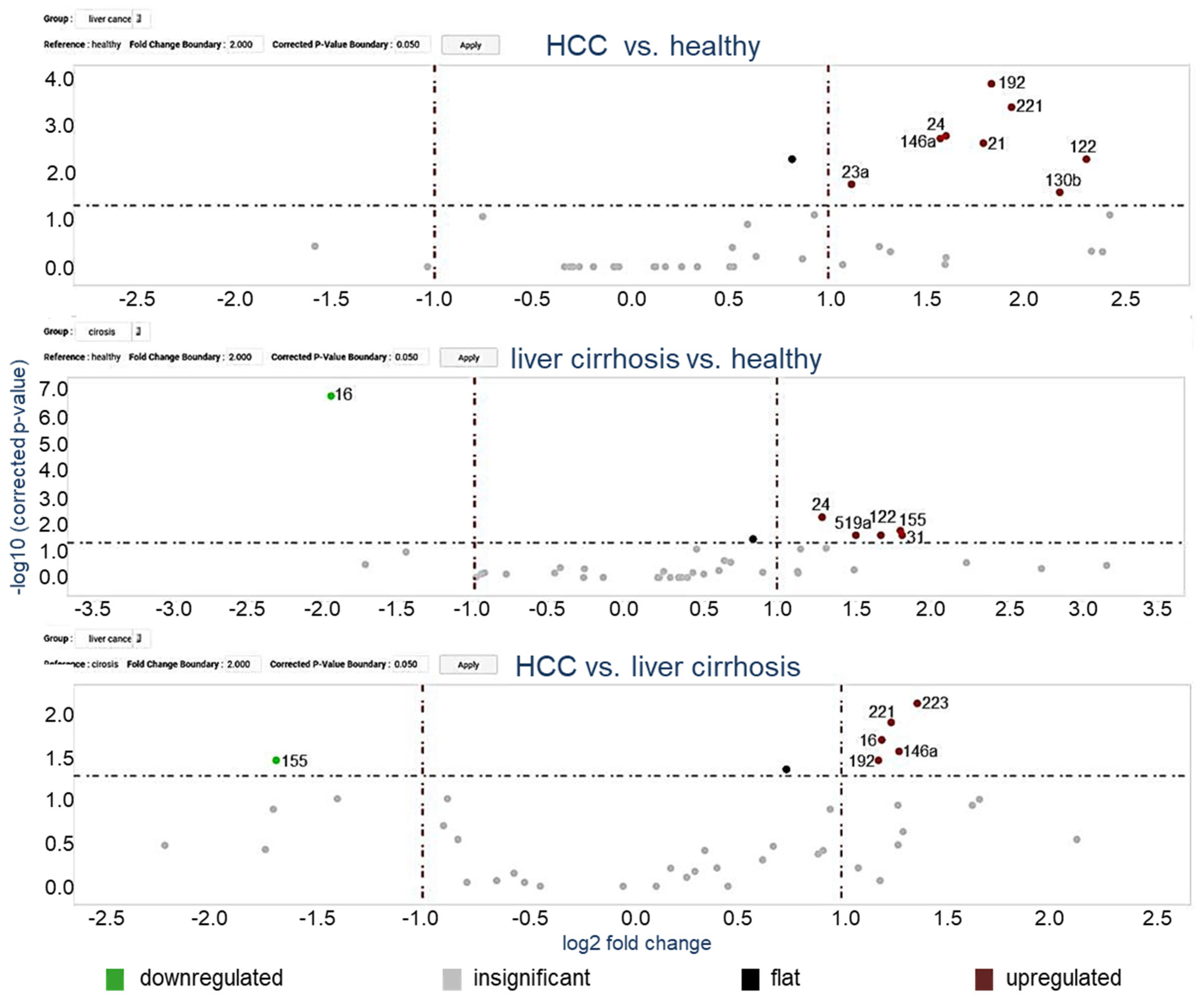
3.2. MiRNA Profiling in Exosomes Using Microarray Cards
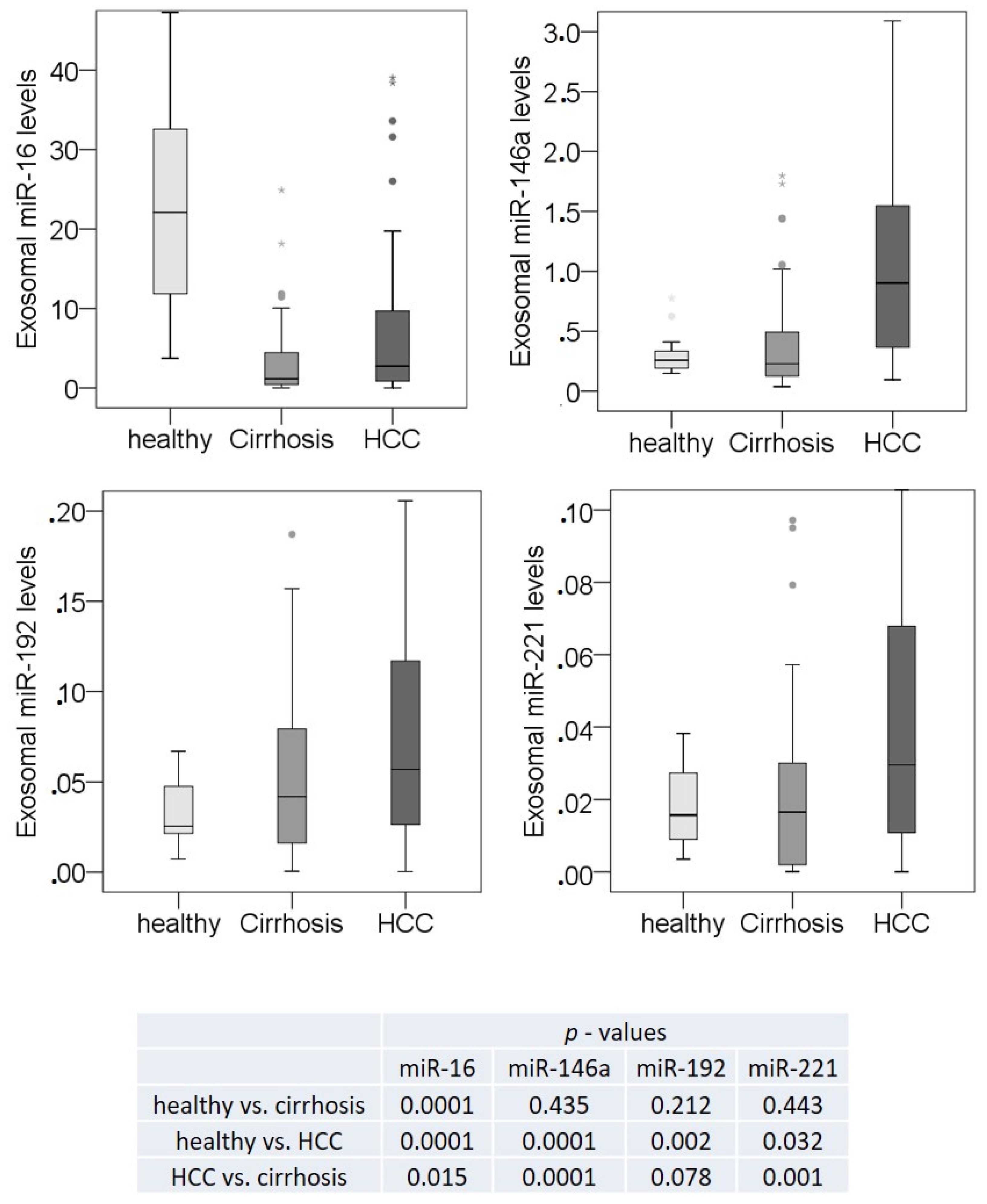
3.3. Exosomal miR-16, miR-146a, miR-192, and miR-221 Profiling Using Single TaqMan Real-Time PCR Assays
3.4. Correlations of Exosomal miR-16, miR-146a, miR-192, and miR-221 with the Clinicopathological Parameters
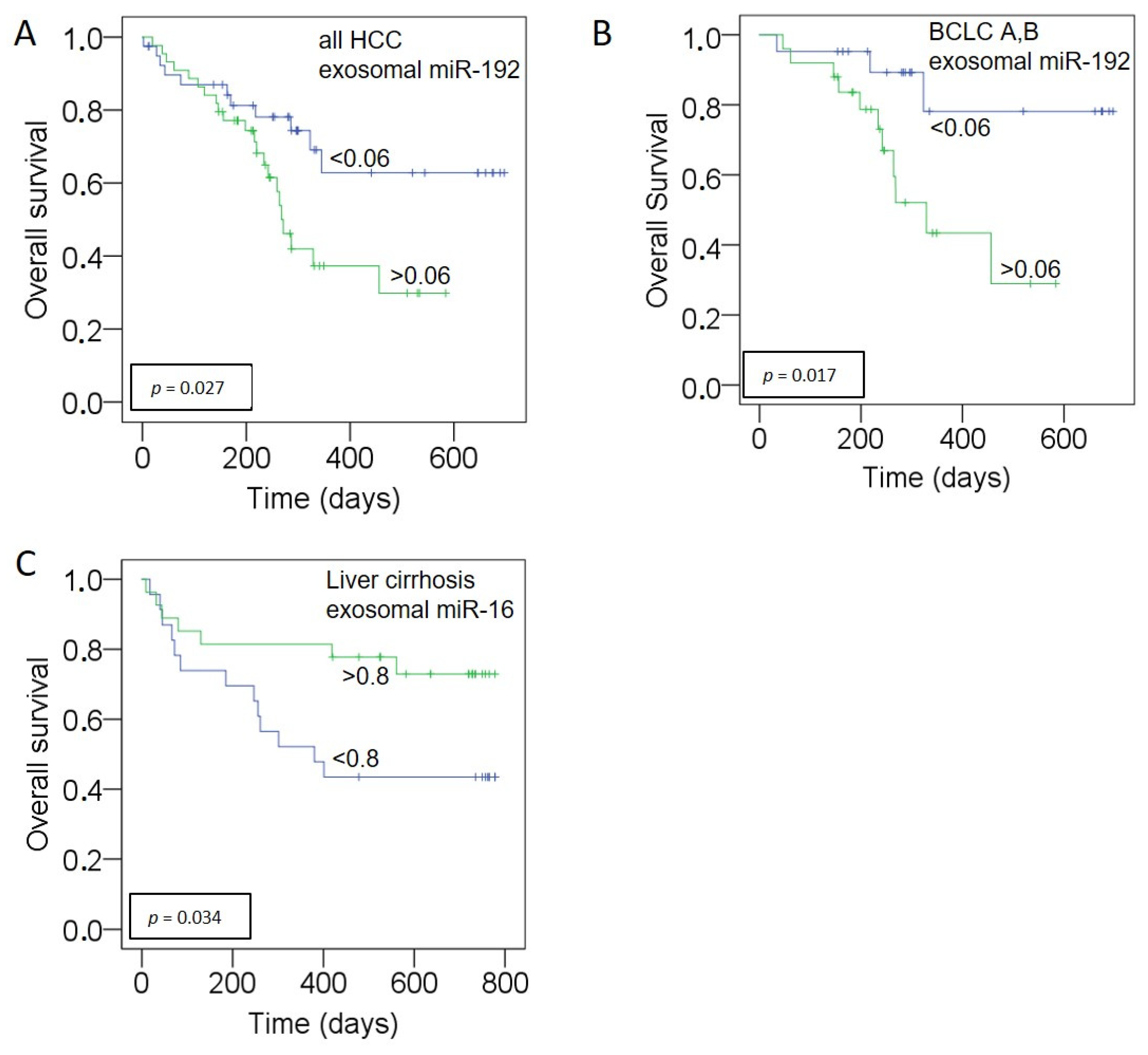
3.5. Prognostic Relevance of miR-192, miR-146a for HCC and miR-16 for Liver Cirrhosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, H.; Naghavi, M.; Allen, C.; Barber, R.M.; Bhutta, Z.A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; Coates, M.M.; et al. Mortality and Causes of Death Collaborators. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Global Burden of DiseaseStudy 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Sangiovanni, A.; Prati, G.M.; Fasani, P.; Ronchi, G.; Romeo, R.; Manini, M.; Del Ninno, E.; Morabito, A.; Colombo, M. The natural history of compensated cirrhosis due to hepatitis C virus: A 17-year cohort study of 214 patients. Hepatology 2006, 43, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Kirstein, M.M.; Schweitzer, N.; Winter, T.; Lappas, K.; Graen, N.; Kunstmann, I.; Voigtländer, T.; Reineke-Plaaß, T.; Manns, M.P.; Lehner, F.; et al. Patterns and challenges of treatment sequencing in patients with hepatocellular carcinoma - experience from a referral center. J. Gastroenterol. Hepatol. 2017, 32, 1730–1738. [Google Scholar] [CrossRef] [PubMed]
- Kanda, M.; Tateishi, R.; Yoshida, H.; Sato, T.; Masuzaki, R.; Ohki, T.; Imamura, J.; Goto, T.; Yoshida, H.; Hamamura, K.; et al. Extrahepatic metastasis of hepatocellular carcinoma: Incidence and risk factors. Liver Int. 2008, 28, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Okuda, K.; Ohtsuki, T.; Obata, H.; Tomimatsu, M.; Okazaki, N.; Hasegawa, H.; Nakajima, Y.; Ohnishi, K. Natural history of hepatocellular carcinoma and prognosis in relation to treatment. Study of 850 patients. Cancer 1985, 56, 918–928. [Google Scholar] [CrossRef]
- The Cancer of the Liver Italian Program [CLIP] Investigators. Prospective validation of the CLIP score: A new prognostic system for patients with cirrhosis and hepatocellular carcinoma. Hepatology 2000, 31, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Levy, I.; Sherman, M.; Liver Cancer Study Group of the University of Toronto. Staging of hepatocellular carcinoma: Assessment of the CLIP, Okuda, and Child-Pugh staging systems in a cohort of 257 patients in Toronto. Gut 2002, 50, 881–885. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Brú, C.; Bruix, J. Prognosis of hepatocellular carcinoma: The BCLC staging classification. Semin. Liver Dis. 1999, 19, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; De Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment [RESORCE]: A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Zhu, A.X.; Kang, Y.K.; Yen, C.J.; Finn, R.S.; Galle, P.R.; Llovet, J.M.; Assenat, E.; Brandi, G.; Pracht, M.; Lim, H.Y.; et al. Ramucirumab after sorafenib in patients with advanced hepatocellular carcinoma and increased a-fetoprotein concentrations [REACH-2]: A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2019, 20, 282–296. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, H.; Nishida, N.; Calin, G.A.; Pantel, K. Clinical relevance of circulating cell-free microRNAs in cancer. Nat. Rev. Clin. Oncol. 2014, 11, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Azmi, A.S.; Bao, B.; Sarkar, F.H. Exosomes in cancer development, metastasis, and drug resistance: A comprehensive review. Cancer Metastasis Rev. 2013, 32, 623–642. [Google Scholar] [CrossRef] [PubMed]
- Vilstrup, H.; Amodio, P.; Bajaj, J.; Cordoba, J.; Ferenci, P.; Mullen, K.D.; Weissenborn, K.; Wong, P. Hepatic encephalopathy in chronic liver disease: 2014 Practice Guideline by the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver. Hepatology 2014, 60, 715–735. [Google Scholar] [CrossRef]
- Angeli, P.; Ginès, P.; Wong, F.; Bernardi, M.; Boyer, T.D.; Gerbes, A.; Moreau, R.; Jalan, R.; Sarin, S.K.; Piano, S.; et al. Diagnosis and management of acute kidney injury in patients with cirrhosis: Revised consensus recommendations of the International Club of Ascites. J. Hepatol. 2015, 62, 968–974. [Google Scholar] [CrossRef] [PubMed]
- The European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J. Hepatol. 2018, 69, 406–460. [Google Scholar] [CrossRef]
- The European Association for the Study of the Liver. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236. [Google Scholar] [CrossRef]
- Kirschner, M.B.; Kao, S.C.; Edelman, J.J.; Armstrong, N.J.; Vallely, M.P.; van Zandwijk, N.; Reid, G. Haemolysis during sample preparation alters microRNA content of plasma. PLoS ONE 2011, 6, e24145. [Google Scholar] [CrossRef]
- Stevic, I.; Muller, V.; Weber, K.; Fasching, P.A.; Karn, T.; Marme, F.; Schem, C.; Stickeler, E.; Denkert, C.; van Mackelenbergh, M.; et al. Specific microRNA signatures in exosomes of triple-negative and HER2-positive breast cancer patients undergoing neoadjuvant therapy within the GeparSixto trial. BMC Med. 2018, 16, 179. [Google Scholar] [CrossRef] [PubMed]
- Eichelser, C.; Stuckrath, I.; Muller, V.; Milde-Langosch, K.; Wikman, H.; Pantel, K.; Schwarzenbach, H. Increased serum levels of circulating exosomal microRNA-373 in receptor-negative breast cancer patients. Oncotarget 2014, 5, 9650–9663. [Google Scholar] [CrossRef] [PubMed]
- Stuckrath, I.; Rack, B.; Janni, W.; Jager, B.; Pantel, K.; Schwarzenbach, H. Aberrant plasma levels of circulating miR-16, miR-107, miR-130a and miR-146a are associated with lymph node metastasis and receptor status of breast cancer patients. Oncotarget 2015, 6, 13387–13401. [Google Scholar] [CrossRef]
- Muller, V.; Gade, S.; Steinbach, B.; Loibl, S.; von Minckwitz, G.; Untch, M.; Schwedler, K.; Lübbe, K.; Schem, C.; Fasching, P.A.; et al. Changes in serum levels of miR-21, miR-210, and miR-373 in HER2-positive breast cancer patients undergoing neoadjuvant therapy: A translational research project within the Geparquinto trial. Breast Cancer Res. Treat. 2014, 147, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Eichelser, C.; Flesch-Janys, D.; Chang-Claude, J.; Pantel, K.; Schwarzenbach, H. Deregulated serum concentrations of circulating cell-free microRNAs miR-17, miR-34a, miR-155, and miR-373 in human breast cancer development and progression. Clin. Chem. 2013, 59, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Kanda, T.; Yokosuka, O.; Kato, N.; Matsuoka, S.; Moriyama, M. Exosomes and Hepatocellular Carcinoma: From Bench to Bedside. Int. J. Mol. Sci. 2019, 20, 1406. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.; Zhao, Y.; Wang, X.; Qin, L.; Hu, R. Development and validation of serum exosomal microRNAs as diagnostic and prognostic biomarkers for hepatocellular carcinoma. J. Cell. Biochem. 2019, 120, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Tiberio, P.; Callari, M.; Angeloni, V.; Daidone, M.G.; Appierto, V. Challenges in Using Circulating miRNAs as Cancer Biomarkers. Biomed. Res. Int. 2015, 2015, 731479. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.T.; Hasan, A.M.; Liu, R.B.; Zhang, Z.C.; Zhang, X.; Wang, J.; Wang, H.Y.; Wang, F.; Shao, J.Y. Serum microRNA profiles as prognostic biomarkers for HBV-positive hepatocellular carcinoma. Oncotarget 2016, 7, 45637–45648. [Google Scholar] [CrossRef] [PubMed]
- Suehiro, T.; Miyaaki, H.; Kanda, Y.; Shibata, H.; Honda, T.; Ozawa, E.; Miuma, S.; Taura, N.; Nakao, K. Serum exosomal microRNA-122 and microRNA-21 as predictive biomarkers in transarterial chemoembolization-treated hepatocellular carcinoma patients. Oncol. Lett. 2018, 16, 3267–3273. [Google Scholar] [CrossRef]
- Wu, W.L.; Wang, W.Y.; Yao, W.Q.; Li, G.D. Suppressive effects of microRNA-16 on the proliferation, invasion and metastasis of hepatocellular carcinoma cells. Int. J. Mol. Med. 2015, 36, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Su, X.F.; Li, N.; Meng, F.L.; Chu, Y.L.; Li, T.; Gao, X.Z. MiR-16 inhibits hepatocellular carcinoma progression by targeting FEAT through NF-kappaB signaling pathway. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 10274–10282. [Google Scholar] [PubMed]
- Kim, S.S.; Nam, J.S.; Cho, H.J.; Won, J.H.; Kim, J.W.; Ji, J.H.; Yang, M.J.; Park, J.H.; Noh, C.K.; Shin, S.J.; et al. Plasma micoRNA-122 as a predictive marker for treatment response following transarterial chemoembolization in patients with hepatocellular carcinoma. J. Gastroenterol. Hepatol. 2017, 32, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, H.; Machado da Silva, A.; Calin, G.; Pantel, K. Data Normalization Strategies for MicroRNA Quantification. Clin. Chem. 2015, 61, 1333–1342. [Google Scholar] [CrossRef]
- Ge, W.; Yu, D.C.; Li, Q.G.; Chen, X.; Zhang, C.Y.; Ding, Y.T. Expression of Serum miR-16, let-7f, and miR-21 in Patients with Hepatocellular Carcinoma and their Clinical Significances. Clin. Lab. 2014, 60, 427–434. [Google Scholar] [CrossRef]
- Qu, K.Z.; Zhang, K.; Li, H.; Afdhal, N.H.; Albitar, M. Circulating microRNAs as biomarkers for hepatocellular carcinoma. J. Clin. Gastroenterol. 2011, 45, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Han, C.Y.; Kim, J.Y.; Cho, S.S.; Kim, Y.S.; Koo, J.H.; Lee, J.M.; Lim, S.C.; Kang, K.W.; Kim, J.S.; et al. Galpha12 overexpression induced by miR-16 dysregulation contributes to liver fibrosis by promoting autophagy in hepatic stellate cells. J. Hepatol. 2018, 68, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, X.; Li, T.; Wang, L.; Wu, X.; Liu, J.; Xu, Y.; Wei, W. Multiple roles of microRNA-146a in immune responses and hepatocellular carcinoma. Oncol. Lett. 2019, 18, 5033–5042. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Pan, Q.; Zhang, X.; Kong, L.Q.; Fan, J.; Dai, Z.; Wang, L.; Yang, X.R.; Hu, J.; Wan, J.L.; et al. MiR-146a enhances angiogenic activity of endothelial cells in hepatocellular carcinoma by promoting PDGFRA expression. Carcinogenesis 2013, 34, 2071–2079. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Han, Q.; Hou, Z.; Zhang, C.; Zhang, J. MiR-146a negatively regulates NK cell functions via STAT1 signaling. Cell. Mol. Immunol. 2017, 14, 712–720. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Han, Q.; Xu, D.; Zheng, B.; Zhao, X.; Zhang, J. SALL4-mediated upregulation of exosomal miR-146a-5p drives T-cell exhaustion by M2 tumor-associated macrophages in HCC. Oncoimmunology 2019, 8, 1601479. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.Y.; Lin, B.D.; Li, B.R. Evaluation of the diagnostic value of alpha-l-fucosidase, alpha-fetoprotein and thymidine kinase 1 with ROC and logistic regression for hepatocellular carcinoma. FEBS Open Bio 2015, 5, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Kannan, M.; Jayamohan, S.; Moorthy, R.K.; Chabattula, S.C.; Ganeshan, M.; Arockiam, A.J.V. AEG-1/miR-221 Axis Cooperatively Regulates the Progression of Hepatocellular Carcinoma by Targeting PTEN/PI3K/AKT Signaling Pathway. Int. J. Mol. Sci. 2019, 20, 5526. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Jiang, J.; Badawi, M.; Schmittgen, T.D. MiR-221 regulates CD44 in hepatocellular carcinoma through the PI3K-AKT-mTOR pathway. Biochem. Biophys. Res. Commun. 2017, 487, 709–715. [Google Scholar] [CrossRef]
- Huang, S.; Zhou, D.; Li, Y.X.; Ming, Z.Y.; Li, K.Z.; Wu, G.B.; Chen, C.; Zhao, Y.N. In vivo and in vitro effects of microRNA-221 on hepatocellular carcinoma development and progression through the JAK-STAT3 signaling pathway by targeting SOCS3. J. Cell. Physiol. 2019, 234, 3500–3514. [Google Scholar] [CrossRef] [PubMed]
- Sohn, W.; Kim, J.; Kang, S.H.; Yang, S.R.; Cho, J.Y.; Cho, H.C.; Shim, S.G.; Paik, Y.H. Serum exosomal microRNAs as novel biomarkers for hepatocellular carcinoma. Exp. Mol. Med. 2015, 47, e184. [Google Scholar] [CrossRef]
- Waidmann, O.; Köberle, V.; Brunner, F.; Zeuzem, S.; Piiper, A.; Kronenberger, B. Serum microRNA-122 predicts survival in patients with liver cirrhosis. PLoS ONE 2012, 7, e45652. [Google Scholar] [CrossRef]

| Characteristics | HCC Patients | LC Patients | |
|---|---|---|---|
| Gender (n; %) | Male | 20 (83.3) | 22 (59.5) |
| Etiology of LC | Alcoholic | 9 (39.1) | 20 (54.1) |
| Non-alcoholic | 14 (60.9) | 17 (45.9) | |
| MELD | ≤20 | 24 (100) | 25 (67.6) |
| >20 | 0 | 12 (32.4) | |
| CPS | A | 9 (37.5) | 6 (16.2) |
| B | 11 (45.8) | 14 (37.8) | |
| C | 4 (16.7) | 17 (46.0) | |
| History of Decompensation | |||
| Ascites | 9 (37.5) | 27 (73) | |
| HE | 0 | 9 (24.3) | |
| HRS | 0 | 9 (24.3) | |
| Variceal bleeding | 0 | 3 (8.1) | |
| Tumor Characteristics | |||
| BCLC | A | 5 (20.9) | n.a. |
| B | 10 (41.6) | n.a. | |
| C | 9 (37.5) | n.a. | |
| Distant metastases (n) | 5 (20.8) | n.a. | |
| Tumor nodules (n) | 2 (1–5) | n.a. | |
| Sum of largest diameter (cm) | 7 (3–16) | n.a. | |
| Baseline Blood Results | |||
| WBC (109/L) | 6.2 (2.1–14.7) | 7.4 (1.4–21.0) | |
| Platelets (109/L) | 138 (33–298) | 117 (11–354) | |
| Albumin (g/L) | 31 (19–39) | 22 (10–34) | |
| Bilirubin (mg/dL) | 1.3 (0.2–6.1) | 5.3 (0.5–30) | |
| Creatinine (mg/dL)) | 1.0 (0.5–1.6) | 1.5 (0.4–5.0) | |
| GFR (ml/min) | 81 (41–119) | 65 (12–126) | |
| AST (U/L) | 67 (21–172) | 91 (21–472) | |
| ALT (U/L) | 50 (18–108) | 73 (13–767) | |
| γGT (U/L) | 290 (40–1378) | 184 (27–1063) | |
| CRP (mg/L) | 22 (5–131) | 32 (5–151) | |
| AFP (kU/L) | 2698 (2–23082) | 8 (2–50) | |
| INR | 1.3 (1.0–1.9) | 1.4 (1.0–2.3) | |
| Healthy Individuals (n = 20) | |||
| Gender (n, %) | Male: 12 (60) |
| Characteristics | HCC Patients | LC Patients | |
|---|---|---|---|
| Gender (n; %) | Male | 74 (86) | 33 (64.7) |
| Etiology of LC | Alcoholic | 41 (47.7) | 25 (49) |
| Non-alcoholic: | 45 (52.3) | 26 (51) | |
| NAFLD | 17 (19.8) | 3 (5.9) | |
| HBV | 7 (8.1) | 3 (5.9) | |
| HCV | 2 (2.3) | 2 (3.9) | |
| PSC | 0 | 2 (3.9) | |
| Others * | 19 (22.1) | 10 (19.6) | |
| MELD | ≤20 | 70 (96) | 35 (71.4) |
| >20 | 3 (4.1) | 14 (28.6) | |
| CPS | A | 39 (45.3) | 11 (21.6) |
| B | 29 (33.7) | 20 (39.2) | |
| C | 18 (20.9) | 20 (39.2) | |
| History of Decompensation | |||
| Ascites | 22 (25.9) | 34 (66.7) | |
| HE | 2 (2.4) | 13 (25.5) | |
| HRS | 1 (1.2) | 9 (17.6) | |
| Variceal bleeding | 2 (2.4) | 6 (11.8) | |
| Total | 23 (27.1) | 38 (74.5) | |
| Tumor Characteristics | |||
| BCLC | A | 7 (8) | n.a. |
| B | 39 (38) | n.a. | |
| C | 40 (54) | n.a. | |
| Distant metastases (n) | 19 (22) | n.a. | |
| Tumor nodules (n) | 2 (1–6) | n.a. | |
| Sum of largest diameter (cm) | 7 (1–26) | n.a. | |
| Progressive disease | 38 (46.3) | n.a. | |
| Overall survial | alive/dead | 51/33 | n.a. |
| Baseline Blood Results | |||
| WBC (109/L) | 6.3 (1.4–14.7) | 6.8 (1.1–21.0) | |
| Platelets (109/L) | 155 (33–555) | 120 (11-354) | |
| Albumin (g/L) | 30 (15–43) | 24 (10–42) | |
| Bilirubin (mg/dL) | 1.5 (0.2–11.5) | 4.7 (0.2–30) | |
| Creatinine (mg/dL) | 1.2 (0.5–9.0) | 1.3 (0.4–5.0) | |
| GFR (ml/min) | 75 (6–119) | 75 (12–151) | |
| AST (U/L) | 92 (11–2017) | 83 (21–472) | |
| ALT (U/L) | 56 (16–810) | 67 (13–767) | |
| γGT (U/L) | 262 (40–1421) | 179 (14–1063) | |
| CRP (mg/L) | 20 (5–161) | 27 (5–151) | |
| AFP (kU/L) | 11.961 (2–373.358) | 8.4 (1.5–49.6) | |
| INR | 1.3 (0.9–11.0) | 1.4 (1.0–2.3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fründt, T.; Krause, L.; Hussey, E.; Steinbach, B.; Köhler, D.; von Felden, J.; Schulze, K.; Lohse, A.W.; Wege, H.; Schwarzenbach, H. Diagnostic and Prognostic Value of miR-16, miR-146a, miR-192 and miR-221 in Exosomes of Hepatocellular Carcinoma and Liver Cirrhosis Patients. Cancers 2021, 13, 2484. https://doi.org/10.3390/cancers13102484
Fründt T, Krause L, Hussey E, Steinbach B, Köhler D, von Felden J, Schulze K, Lohse AW, Wege H, Schwarzenbach H. Diagnostic and Prognostic Value of miR-16, miR-146a, miR-192 and miR-221 in Exosomes of Hepatocellular Carcinoma and Liver Cirrhosis Patients. Cancers. 2021; 13(10):2484. https://doi.org/10.3390/cancers13102484
Chicago/Turabian StyleFründt, Thorben, Linda Krause, Elaine Hussey, Bettina Steinbach, Daniel Köhler, Johann von Felden, Kornelius Schulze, Ansgar W. Lohse, Henning Wege, and Heidi Schwarzenbach. 2021. "Diagnostic and Prognostic Value of miR-16, miR-146a, miR-192 and miR-221 in Exosomes of Hepatocellular Carcinoma and Liver Cirrhosis Patients" Cancers 13, no. 10: 2484. https://doi.org/10.3390/cancers13102484
APA StyleFründt, T., Krause, L., Hussey, E., Steinbach, B., Köhler, D., von Felden, J., Schulze, K., Lohse, A. W., Wege, H., & Schwarzenbach, H. (2021). Diagnostic and Prognostic Value of miR-16, miR-146a, miR-192 and miR-221 in Exosomes of Hepatocellular Carcinoma and Liver Cirrhosis Patients. Cancers, 13(10), 2484. https://doi.org/10.3390/cancers13102484






