Generation of Non-Small Cell Lung Cancer Patient-Derived Xenografts to Study Intratumor Heterogeneity
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Recruitment
2.2. PDX Generation
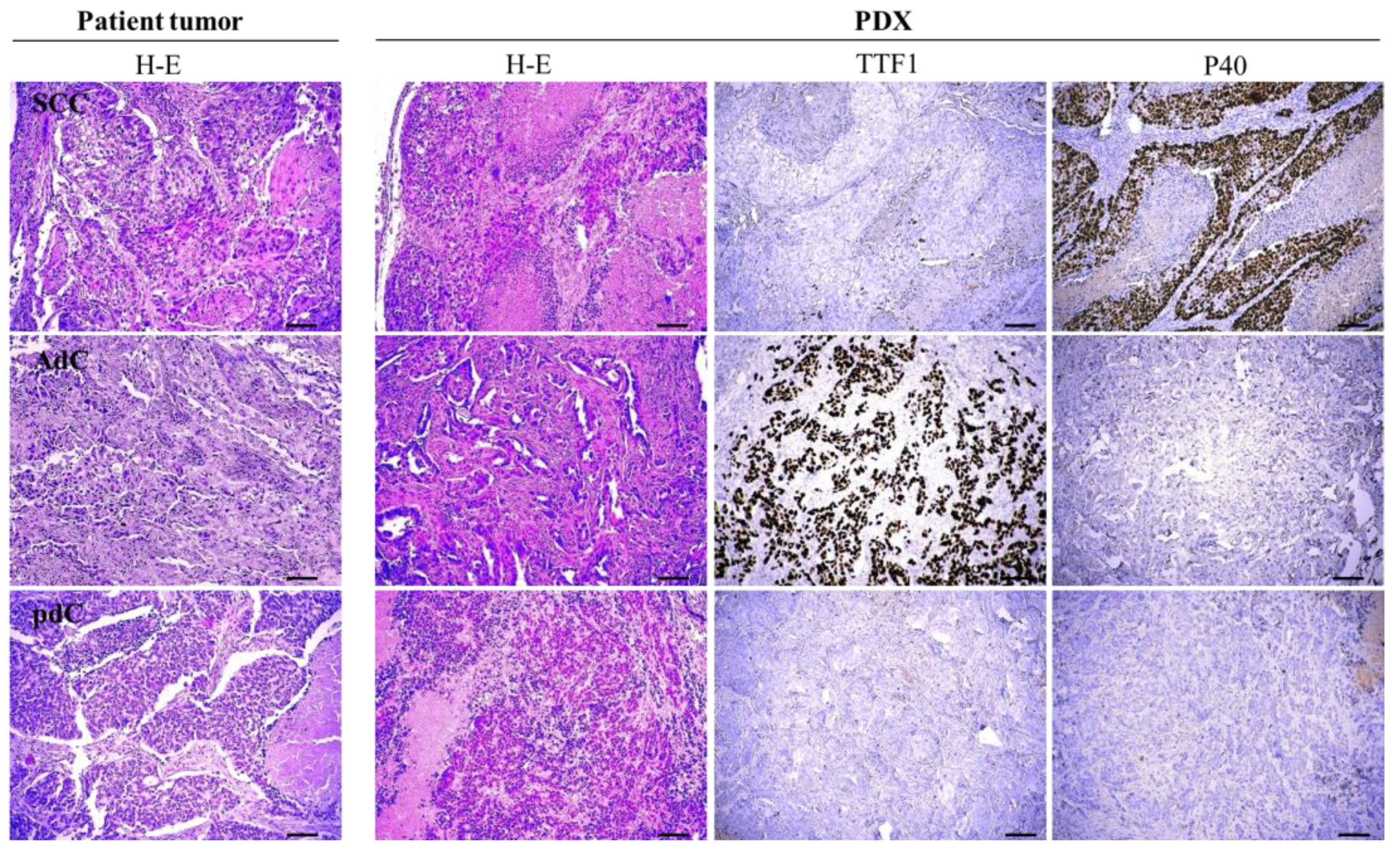
2.3. Histopathological Examination
2.4. DNA/RNA Extraction
2.5. Library Preparation and Sequencing
2.6. Primary and PDX Tumor Comparison
2.7. CTC Enrichment for Molecular Analysis
2.8. Gene Expression Analysis of CTCs by RT-qPCR
2.9. Statistical Analysis
3. Results
3.1. Establishment of NSCLC PDXs
3.2. Clinical Characteristics and Grafting Efficiency
3.3. Correlation between the Presence of CTCs and Grafting
3.4. Molecular Comparison of PDX and Primary Tumors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Postmus, P.E.; Kerr, K.M.; Oudkerk, M.; Senan, S.; Waller, D.A.; Vansteenkiste, J.; Escriu, C.; Peters, S. Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv1–iv21. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, V.M.L.; Carvalho, L. Heterogeneity in Lung Cancer. Pathobiology 2018, 85, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Marino, F.Z.; Bianco, R.; Accardo, M.; Ronchi, A.; Cozzolino, I.; Morgillo, F.; Rossi, G.; Franco, R. Molecular heterogeneity in lung cancer: From mechanisms of origin to clinical implications. Int. J. Med. Sci. 2019, 16, 981–989. [Google Scholar] [CrossRef] [PubMed]
- Fichtner, I.; Rolff, J.; Soong, R.; Hoffmann, J.; Hammer, S.; Sommer, A.; Becker, M.; Merk, J. Establishment of Patient-Derived Non-Small Cell Lung Cancer Xenografts as Models for the Identification of Predictive Biomarkers. Clin. Cancer Res. 2008, 14, 6456–6468. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-C.; Zhang, J.; Li, M.; Huang, X.-S.; Yang, X.-N.; Zhong, W.-Z.; Xie, L.; Zhang, L.; Zhou, M.; Gavine, P.; et al. Establishment of patient-derived non-small cell lung cancer xenograft models with genetic aberrations within EGFR, KRAS and FGFR1: Useful tools for preclinical studies of targeted therapies. J. Transl. Med. 2013, 11, 168. [Google Scholar] [CrossRef] [PubMed]
- Goto, T. Patient-Derived Tumor Xenograft Models: Toward the Establishment of Precision Cancer Medicine. J. Pers. Med. 2020, 10, 64. [Google Scholar] [CrossRef]
- Kita, K.; Fukuda, K.; Takahashi, H.; Tanimoto, A.; Nishiyama, A.; Arai, S.; Takeuchi, S.; Yamashita, K.; Ohtsubo, K.; Otani, S.; et al. Patient-derived xenograft models of non-small cell lung cancer for evaluating targeted drug sensitivity and resistance. Cancer Sci. 2019, 110, 3215–3224. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Guan, J.; English, J.C.; Flint, J.; Yee, J.; Evans, K.; Murray, N.; Macaulay, C.; Ng, R.T.; Gout, P.W.; et al. Patient-Derived First Generation Xenografts of Non–Small Cell Lung Cancers: Promising Tools for Predicting Drug Responses for Personalized Chemotherapy. Clin. Cancer Res. 2010, 16, 1442–1451. [Google Scholar] [CrossRef] [PubMed]
- Russo, M.V.; Efaversani, A.; Egatti, S.; Ericca, D.; Gobbo, A.E.; Eferrero, S.; Epalleschi, A.; Evaira, V.; Ebosari, S. A New Mouse Avatar Model of Non-Small Cell Lung Cancer. Front. Oncol. 2015, 5, 52. [Google Scholar] [CrossRef]
- Ilie, M.; Nunes, M.; Blot, L.; Hofman, V.; Long-Mira, E.; Butori, C.; Selva, E.; Merino-Trigo, A.; Vénissac, N.; Mouroux, J.; et al. Setting up a wide panel of patient-derived tumor xenografts of non-small cell lung cancer by improving the preanalytical steps. Cancer Med. 2014, 4, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.W.; Lee, J.-I.; Lee, S.J.; Cho, H.J.; Song, H.J.; Jeong, D.E.; Seo, Y.J.; Shin, S.; Joung, J.-G.; Kwon, Y.-J.; et al. Patient-Derived Xenografts from Non–Small Cell Lung Cancer Brain Metastases Are Valuable Translational Platforms for the Development of Personalized Targeted Therapy. Clin. Cancer Res. 2015, 21, 1172–1182. [Google Scholar] [CrossRef] [PubMed]
- Morgan, K.M.; Riedlinger, G.M.; Rosenfeld, J.; Ganesan, S.; Pine, S.R. Patient-Derived Xenograft Models of Non-Small Cell Lung Cancer and Their Potential Utility in Personalized Medicine. Front. Oncol. 2017, 7, 2. [Google Scholar] [CrossRef]
- Jung, H.-Y.; Kim, T.H.; Lee, J.-E.; Kim, H.K.; Cho, J.H.; Choi, Y.S.; Shin, S.; Lee, S.-H.; Rhee, H.; Lee, H.K.; et al. PDX models of human lung squamous cell carcinoma: Consideration of factors in preclinical and co-clinical applications. J. Transl. Med. 2020, 18, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hao, C.; Wang, L.; Peng, S.; Cao, M.; Li, H.; Hu, J.; Huang, X.; Liu, W.; Zhang, H.; Wu, S.; et al. Gene mutations in primary tumors and corresponding patient-derived xenografts derived from non-small cell lung cancer. Cancer Lett. 2015, 357, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Coughlan, A.M.; Harmon, C.; Whelan, S.; O’Brien, E.C.; O’Reilly, V.P.; Crotty, P.; Kelly, P.; Ryan, M.; Hickey, F.B.; O’Farrelly, C.; et al. Myeloid Engraftment in Humanized Mice: Impact of Granulocyte-Colony Stimulating Factor Treatment and Transgenic Mouse Strain. Stem Cells Dev. 2016, 25, 530–541. [Google Scholar] [CrossRef] [PubMed]
- Shultz, L.D.; Lyons, B.L.; Burzenski, L.M.; Gott, B.; Chen, X.; Chaleff, S.; Kotb, M.; Gillies, S.D.; King, M.; Mangada, J.; et al. Human Lymphoid and Myeloid Cell Development in NOD/LtSz-scid IL2R γ Null Mice Engrafted with Mobilized Human Hemopoietic Stem Cells. J. Immunol. 2005, 174, 6477–6489. [Google Scholar] [CrossRef]
- Rampias, T.; Karagiannis, D.; Avgeris, M.; Polyzos, A.; Kokkalis, A.; Kanaki, Z.; Kousidou, E.; Tzetis, M.; Kanavakis, E.; Stravodimos, K.; et al. The lysine-specific methyltransferase KMT 2C/ MLL 3 regulates DNA repair components in cancer. EMBO Rep. 2019, 20, e46821. [Google Scholar] [CrossRef] [PubMed]
- Paraskevopoulou, V.; Bonis, V.; Dionellis, V.S.; Paschalidis, N.; Melissa, P.; Chavdoula, E.; Vasilaki, E.; Pateras, I.S.; Klinakis, A. Notch controls urothelial integrity in the mouse bladder. JCI Insight 2020, 5, e133232. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.S.; Quinlan, A.R. Mosdepth: Quick coverage calculation for genomes and exomes. Bioinformatics 2018, 34, 867–868. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
- Li, H. A statistical framework for SNP calling, mutation discovery, association mapping and population genetical parameter estimation from sequencing data. Bioinformatics 2011, 27, 2987–2993. [Google Scholar] [CrossRef]
- Van der Auwera, G.A.; O’Connor, B.D. Genomics in the Cloud: Using Docker, GATK, and WDL in Terra, 1st ed.; O’Reilly Media, Inc.: Sebastopol, CA, USA, 2020. [Google Scholar]
- Wang, K.; Li, M.; Hakonarson, H. ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef] [PubMed]
- Porras, T.B.; Kaur, P.; Ring, A.; Schechter, N.; Lang, J.E. Challenges in using liquid biopsies for gene expression profiling. Oncotarget 2018, 9, 7036–7053. [Google Scholar] [CrossRef] [PubMed]
- Chudziak, J.; Burt, D.J.; Mohan, S.; Rothwell, D.G.; Mesquita, B.; Antonello, J.; Dalby, S.; Ayub, M.; Priest, L.; Carter, L.; et al. Clinical evaluation of a novel microfluidic device for epitope-independent enrichment of circulating tumour cells in patients with small cell lung cancer. Analyst 2015, 141, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Zavridou, M.; Mastoraki, S.; Strati, A.; Koutsodontis, G.; Klinakis, A.; Psyrri, A.; Lianidou, E. Direct comparison of size-dependent versus EpCAM-dependent CTC enrichment at the gene expression and DNA methylation level in head and neck squamous cell carcinoma. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Markou, A.; Tzanikou, E.; Strati, A.; Zavridou, M.; Mastoraki, S.; Bournakis, E.; Lianidou, E. PIM-1 Is Overexpressed at a High Frequency in Circulating Tumor Cells from Metastatic Castration-Resistant Prostate Cancer Patients. Cancers 2020, 12, 1188. [Google Scholar] [CrossRef] [PubMed]
- Ntzifa, A.; Strati, A.; Kallergi, G.; Kotsakis, A.; Georgoulias, V.; Lianidou, E. Gene expression in circulating tumor cells reveals a dynamic role of EMT and PD-L1 during osimertinib treatment in NSCLC patients. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Nhung, N.V.; Mirejovský, P.; Mirejovský, T.; Melínová, L. Cytokeratins and lung carcinomas. Ceskoslovenska Patol. 1999, 35, 80–84. [Google Scholar]
- Bugalho, A.; Martins, C.; Dias, S.S.; Nunes, G.; Silva, Z.; Correia, M.; Gomes, M.J.M.; Videira, P.A. Cytokeratin 19, Carcinoembryonic Antigen, and Epithelial Cell Adhesion Molecule Detect Lung Cancer Lymph Node Metastasis in Endobronchial Ultrasound-Guided Transbronchial Aspiration Samples. Clin. Lung Cancer 2013, 14, 704–712. [Google Scholar] [CrossRef]
- Rosell, R.; Karachaliou, N.; Morales-Espinosa, D.; Costa, C.; Molina, M.A.; Sansano, I.; Gasco, A.; Viteri, S.; Massuti, B.; Wei, J.; et al. Adaptive resistance to targeted therapies in cancer. Transl. Lung Cancer Res. 2013, 2, 152–159. [Google Scholar]
- Nagano, T.; Tachihara, M.; Nishimura, Y. Mechanism of Resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors and a Potential Treatment Strategy. Cells 2018, 7, 212. [Google Scholar] [CrossRef] [PubMed]
- Karachaliou, N.; Codony-Servat, J.; Bracht, J.W.P.; Ito, M.; Filipska, M.; Pedraz, C.; Chaib, I.; Bertran-Alamillo, J.; Cardona, A.F.; Molina, M.A.; et al. Characterising acquired resistance to erlotinib in non-small cell lung cancer patients. Expert Rev. Respir. Med. 2019, 13, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Westover, D.; Zugazagoitia, J.; Cho, B.; Lovly, C.; Paz-Ares, L. Mechanisms of acquired resistance to first- and second-generation EGFR tyrosine kinase inhibitors. Ann. Oncol. 2018, 29, i10–i19. [Google Scholar] [CrossRef]
- Constant, S.; Huang, S.; Wisniewski, L.; Mas, L.W.A.C. Advanced Human In vitro Models for the Discovery and Development of Lung Cancer Therapies. In Drug Discovery and Development—From Molecules to Medicine; Vallisuta, O., Olimat, S., Eds.; IntechOpen: Rijeka, Croatia, 2015. [Google Scholar]
- Pompili, L.; Porru, M.; Caruso, C.; Biroccio, A.; Leonetti, C. Patient-derived xenografts: A relevant preclinical model for drug development. J. Exp. Clin. Cancer Res. 2016, 35, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gristina, V.; Malapelle, U.; Galvano, A.; Pisapia, P.; Pepe, F.; Rolfo, C.; Tortorici, S.; Bazan, V.; Troncone, G.; Russo, A. The significance of epidermal growth factor receptor uncommon mutations in non-small cell lung cancer: A systematic review and critical appraisal. Cancer Treat. Rev. 2020, 85, 101994. [Google Scholar] [CrossRef] [PubMed]
- Nacchio, M.; Sgariglia, R.; Gristina, V.; Pisapia, P.; Pepe, F.; De Luca, C.; Migliatico, I.; Clery, E.; Greco, L.; Vigliar, E.; et al. KRAS mutations testing in non-small cell lung cancer: The role of Liquid biopsy in the basal setting. J. Thorac. Dis. 2020, 12, 3836–3843. [Google Scholar] [CrossRef] [PubMed]
- Canon, J.; Rex, K.; Saiki, A.Y.; Mohr, C.; Cooke, K.; Bagal, D.; Gaida, K.; Holt, T.; Knutson, C.G.; Koppada, N.; et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature 2019, 575, 217–223. [Google Scholar] [CrossRef]
- Hallin, J.; Engstrom, L.D.; Hargis, L.; Calinisan, A.; Aranda, R.; Briere, D.M.; Sudhakar, N.; Bowcut, V.; Baer, B.R.; Ballard, J.A.; et al. The KRASG12C Inhibitor MRTX849 Provides Insight toward Therapeutic Susceptibility of KRAS-Mutant Cancers in Mouse Models and Patients. Cancer Discov. 2020, 10, 54–71. [Google Scholar] [CrossRef]
- Dunnett-Kane, V.; Nicola, P.; Blackhall, F.; Lindsay, C. Mechanisms of Resistance to KRASG12C Inhibitors. Cancers 2021, 13, 151. [Google Scholar] [CrossRef]
- Schliekelman, M.J.; Taguchi, A.; Zhu, J.; Dai, X.; Rodriguez, J.; Celiktas, M.; Zhang, Q.; Chin, A.; Wong, C.-H.; Wang, H.; et al. Molecular Portraits of Epithelial, Mesenchymal, and Hybrid States in Lung Adenocarcinoma and Their Relevance to Survival. Cancer Res. 2015, 75, 1789–1800. [Google Scholar] [CrossRef]
- Menz, A.; Weitbrecht, T.; Gorbokon, N.; Büscheck, F.; Luebke, A.M.; Kluth, M.; Hube-Magg, C.; Hinsch, A.; Höflmayer, D.; Weidemann, S.; et al. Diagnostic and prognostic impact of cytokeratin 18 expression in human tumors: A tissue microarray study on 11,952 tumors. Mol. Med. 2021, 27, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Cespedes, M. The role of LKB1 in lung cancer. Fam. Cancer 2011, 10, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Mogi, A.; Kuwano, H. TP53 Mutations in Nonsmall Cell Lung Cancer. J. Biomed. Biotechnol. 2011, 2011, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Zhang, X.; Murakami, Y.; Hirohashi, S.; Xu, H.-J.; Hu, S.-X.; Benedict, W.F.; Sekiya, T. Deletion of three distinct regions on chromosome 13q in human non-small-cell lung cancer. Int. J. Cancer 1997, 74, 45–49. [Google Scholar] [CrossRef]
- Oren, M.; Rotter, V. Mutant p53 Gain-of-Function in Cancer. Cold Spring Harb. Perspect. Biol. 2009, 2, a001107. [Google Scholar] [CrossRef]
- Klinakis, A.; Rampias, T. TP53 mutational landscape of metastatic head and neck cancer reveals patterns of mutation selection. EBioMedicine 2020, 58, 102905. [Google Scholar] [CrossRef]
| Patient Characteristics | PDX YES (n = 31) | PDX NO (n = 21) | p-Value |
|---|---|---|---|
| Categorical Variables | n (%) | n (%) | |
| Gender | 0.141 a | ||
| Male | 23 (74.2%) | 11 (52.4%) | |
| Female | 8 (25.8%) | 10 (47.6%) | |
| Age | 0.090 a | ||
| <68y b | 10 (34.5%) | 12 (60.0%) | |
| ≥68y | 19 (65.5%) | 8 (40.0%) | |
| Smoking status | 0.497 c | ||
| Current | 19 (61.3%) | 10 (47.6%) | |
| Previous | 6 (19.4%) | 7 (33.3%) | |
| Never | 6 (19.4%) | 4 (19.0%) | |
| Histological type | 0.100 c | ||
| Adenocarcinoma | 10 (32.3%) | 13 (61.9%) | |
| Squamous | 17 (54.8%) | 7 (33.3%) | |
| Large cell or Sarcomatous | 4 (12.9%) | 1 (4.8%) | |
| Clinical stage | 0.040 c | ||
| I | 13 (41.9%) | 10 (47.6%) | |
| II | 6 (19.4%) | 9 (42.9%) | |
| IIIA | 12 (38.7%) | 2 (9.5%) | |
| Tumor stage | 0.038 c | ||
| pT1 | 5 (16.1%) | 9 (42.9%) | |
| pT2 | 15 (48.4%) | 7 (33.3%) | |
| pT3 | 5 (16.1%) | 5 (23.8%) | |
| pT4 | 6 (19.4%) | 0 (0%) | |
| Continuous variables | Median (min–max) | Median (min–max) | |
| Tumor volume (cm3) | 0.070 d | ||
| 33.7 (0.48–572.0) | 14.7 (2.14–245.0) | ||
| Age (years) | 0.030 d | ||
| 71 (56–81) | 64 (40–75) |
| PDX | Total | p Value a | |||
|---|---|---|---|---|---|
| No (−) | Yes (+) | ||||
| KRT18 | Neg | 21 | 19 | 37 | p = 0.009 |
| Pos | 0 | 7 | 7 | ||
| Total | 21 | 26 | 47 | ||
| KRT19 | Neg | 16 | 19 | 34 | p = 1.000 |
| Pos | 5 | 7 | 12 | ||
| Total | 21 | 26 | 47 | ||
| KRT8 | Neg | 17 | 22 | 39 | p = 1.000 |
| Pos | 4 | 4 | 8 | ||
| Total | 21 | 26 | 47 | ||
| Epithelial markers | Neg | 14 | 14 | 28 | p = 0.377 |
| Pos | 7 | 12 | 19 | ||
| Total | 21 | 26 | 47 | ||
| PDX | Total | p Value a | |||
|---|---|---|---|---|---|
| No (−) | Yes (+) | ||||
| VIM | Neg | 11 | 9 | 20 | p = 0.231 |
| Pos | 10 | 17 | 27 | ||
| Total | 21 | 26 | 47 | ||
| TWIST1 | Neg | 18 | 17 | 35 | p = 0.179 |
| Pos | 3 | 9 | 12 | ||
| Total | 21 | 26 | 47 | ||
| EMT markers (VIM & TWIST1) | Neg | 8 | 5 | 13 | p = 0.107 |
| Pos | 13 | 21 | 34 | ||
| Total | 21 | 26 | 47 | ||
| CODE | TYPE | GENETIC ALTERATIONS | TUMOR CELLULARITY (%) | VAFPRIM.TUMOR | VAFPDX |
|---|---|---|---|---|---|
| 105 | LUSC | TP53:c.469G>T:p.V157F KMT2C:c.918T>G:p.Y306* | 90 | 0.057 0.059 | 1 0.062 |
| 110 | LUSC | TP53:c.413C>T:p.A138V ALK:c.2712T>A:p.H904Q | 85 | 0.432 0.36 | 0.952 0.191 |
| 112 | PLEIO/LUAD | CTNNB1:c.98C>T:p.S33F TP53:c.811G>T:p.E271* STK11:c.487G>T:p.G163C KDR:c.2761delinsAT:p.F921Ifs*13 KDR:c.2757C>G:p.C919W | 80 | 0.49 0.436 0.356 0.316 0.317 | 0.868 0.93 0.895 0.83 0.831 |
| 302 | LUSC | KIT:2508G>T:p.M836I EGFR:c.1150A>T:p.T384S, MEN1:c.184A>G:p.T62A, MET:c.1241A>G:p.D414G | 70 | 0.28 0.392 0.325 0.64 | 0.973 0.993 0.644 0.995 |
| 512 | LUSC | TP53:c.845G>C:p.R282P CDKN2A:c.316_317T:p.V106Cfs*39 PDGFRA:c.1102G>C:p.E368Q | 75 | 0.336 0.375 0.4 | 0.999 0.998 0.995 |
| 517 | LUSC | RB1:c.852delinsTA:p.I285Nfs*2 | 75 | 0.249 | 0.998 |
| 518 | LUSC | TP53:c.659A>G:p.Y220C | 80 | 0.304 | 0.996 |
| 519 | LCNEC | RB1:c.1953_1954T:p.V654Cfs*3 | 80 | 0.732 | 0.998 |
| 531 | LUSC | TP53:c.747G>T:p.R249S CDKN2A:c.262G>T:p.E88* PIK3CA:c.1633G>A:p.E545K | 80 | 0.574 0.416 0.251 | 0.998 0.991 0.337 |
| 546 | LUAD | KRAS:c.35G>A:p.G12D ATM:c.8851G>A:p.V2951I | 85 | 0.342 0.371 | 0.296 0.997 |
| 551 | LUSC | MTOR c.1333A>G:p.R445G CDKN2A:c.61G>C:p.A21P | 65 | 0.686 0.295 | 0.994 0.966 |
| 568 | LUAD | TP53:c.880G>T:p.E294* KRAS:c.34G>T:p.G12C | 65 | 0.09 0.16 | 0.63 0.62 |
| 569 | LUSC | NF1:c.4662-1G>T TP53:c.994-2A>T; SMAD4:c.1326G>T:p.Q442H | 80 | 0.301 0.274 0.145 | 0.984 0.991 0.953 |
| 571 | LUSC | CHEK2: c.1114C>T:p.Q372* TP53:c.994-2A>T BRCA2:c.3668A>G:p.H1223R NOTCH1:c.2058_2059T:p.C687Afs*84 | 75 | 0.294 0.287 0.59 0.279 | 0.362 0.952 0.959 0.951 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanaki, Z.; Voutsina, A.; Markou, A.; Pateras, I.S.; Potaris, K.; Avgeris, M.; Makrythanasis, P.; Athanasiadis, E.I.; Vamvakaris, I.; Patsea, E.; et al. Generation of Non-Small Cell Lung Cancer Patient-Derived Xenografts to Study Intratumor Heterogeneity. Cancers 2021, 13, 2446. https://doi.org/10.3390/cancers13102446
Kanaki Z, Voutsina A, Markou A, Pateras IS, Potaris K, Avgeris M, Makrythanasis P, Athanasiadis EI, Vamvakaris I, Patsea E, et al. Generation of Non-Small Cell Lung Cancer Patient-Derived Xenografts to Study Intratumor Heterogeneity. Cancers. 2021; 13(10):2446. https://doi.org/10.3390/cancers13102446
Chicago/Turabian StyleKanaki, Zoi, Alexandra Voutsina, Athina Markou, Ioannis S. Pateras, Konstantinos Potaris, Margaritis Avgeris, Periklis Makrythanasis, Emmanouil I. Athanasiadis, Ioannis Vamvakaris, Eleni Patsea, and et al. 2021. "Generation of Non-Small Cell Lung Cancer Patient-Derived Xenografts to Study Intratumor Heterogeneity" Cancers 13, no. 10: 2446. https://doi.org/10.3390/cancers13102446
APA StyleKanaki, Z., Voutsina, A., Markou, A., Pateras, I. S., Potaris, K., Avgeris, M., Makrythanasis, P., Athanasiadis, E. I., Vamvakaris, I., Patsea, E., Vachlas, K., Lianidou, E., Georgoulias, V., Kotsakis, A., & Klinakis, A. (2021). Generation of Non-Small Cell Lung Cancer Patient-Derived Xenografts to Study Intratumor Heterogeneity. Cancers, 13(10), 2446. https://doi.org/10.3390/cancers13102446











