MicroRNAs Deregulated in Intraductal Papillary Mucinous Neoplasm Converge on Actin Cytoskeleton-Related Pathways That Are Maintained in Pancreatic Ductal Adenocarcinoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Clinical Samples
2.2. RNA Extraction
2.3. MiRNome and Transcriptome Data Obtention
2.4. Integrative Analysis
2.5. Analysis of RNA Expression by qRT-PCR
2.6. In Situ Hybridization and Immunohistochemistry
2.7. Cell Culture
2.8. Retrovirus Production and HPDE Infection for Stable Overexpression of miR-181a
2.9. Fluorescence Imaging
2.10. Statistical Analysis
3. Results
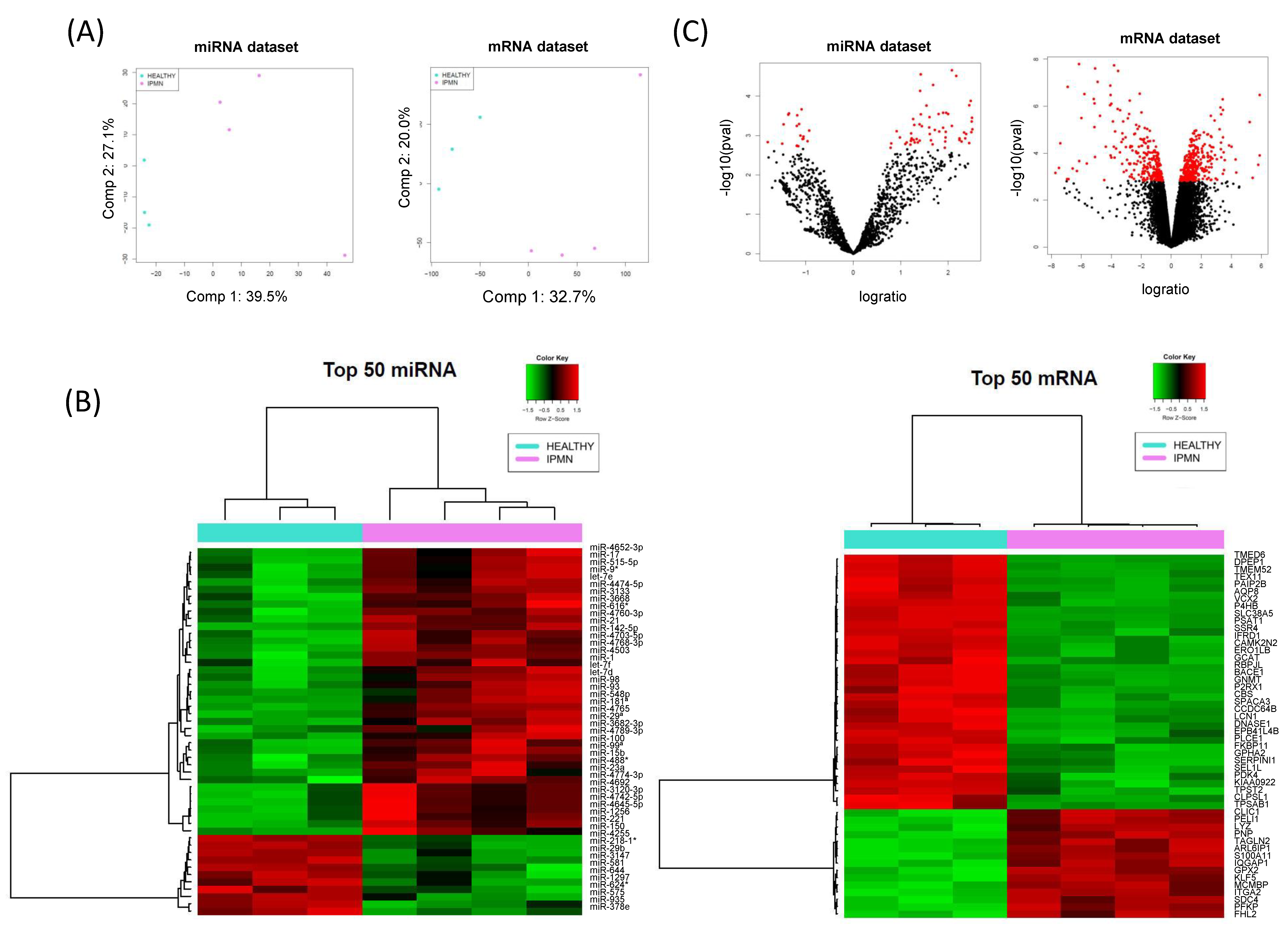
3.1. IPMN miRNome and Transcriptome Exploration
3.2. MiRComb Filtering of Potential miRNA-mRNA Pairs Occurring in IPMN
3.3. Contribution of miRNA-mRNA IPMN Interactions in PDAC
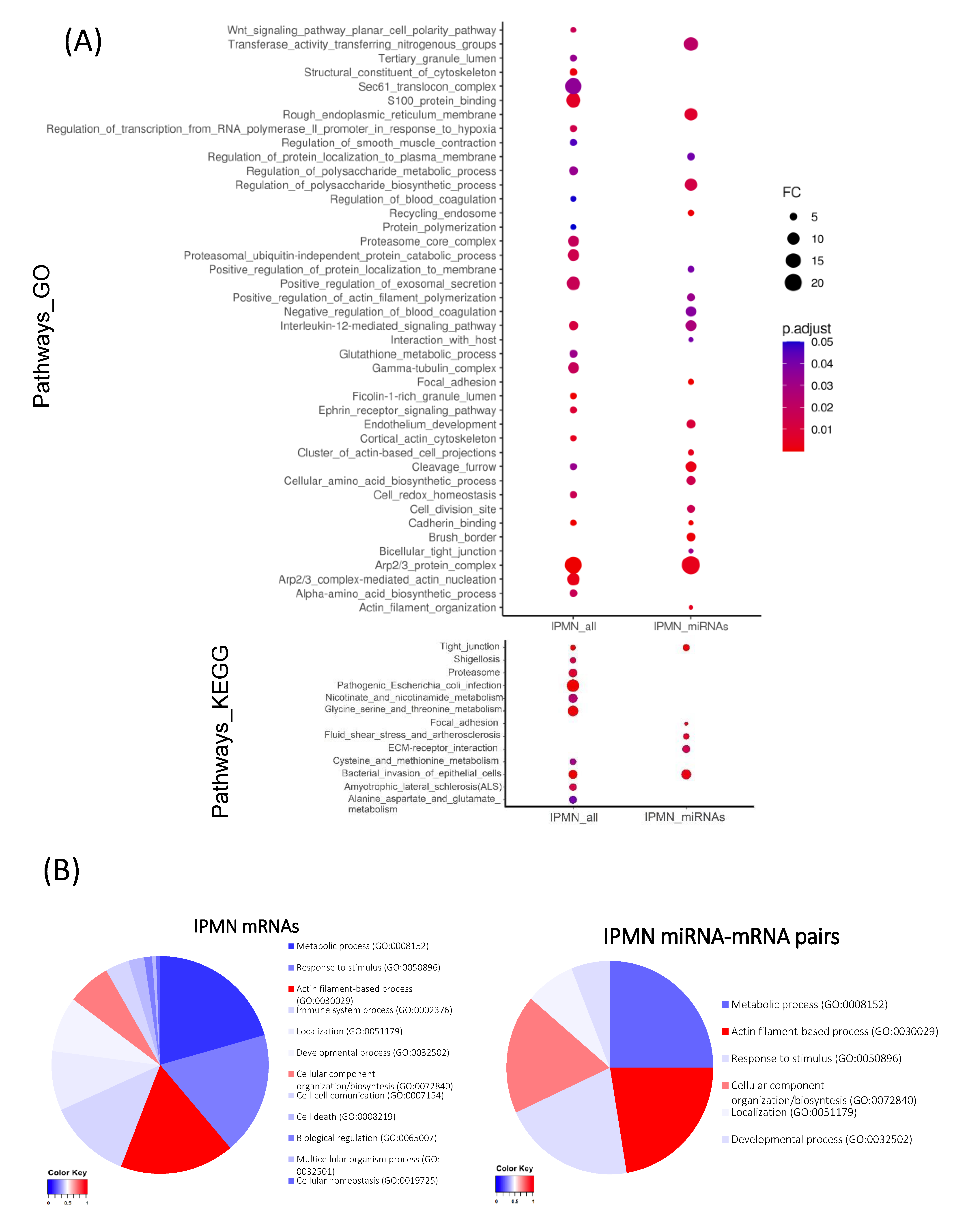
3.4. In-Silico Functional Analysis of miRNA-mRNA Pairs
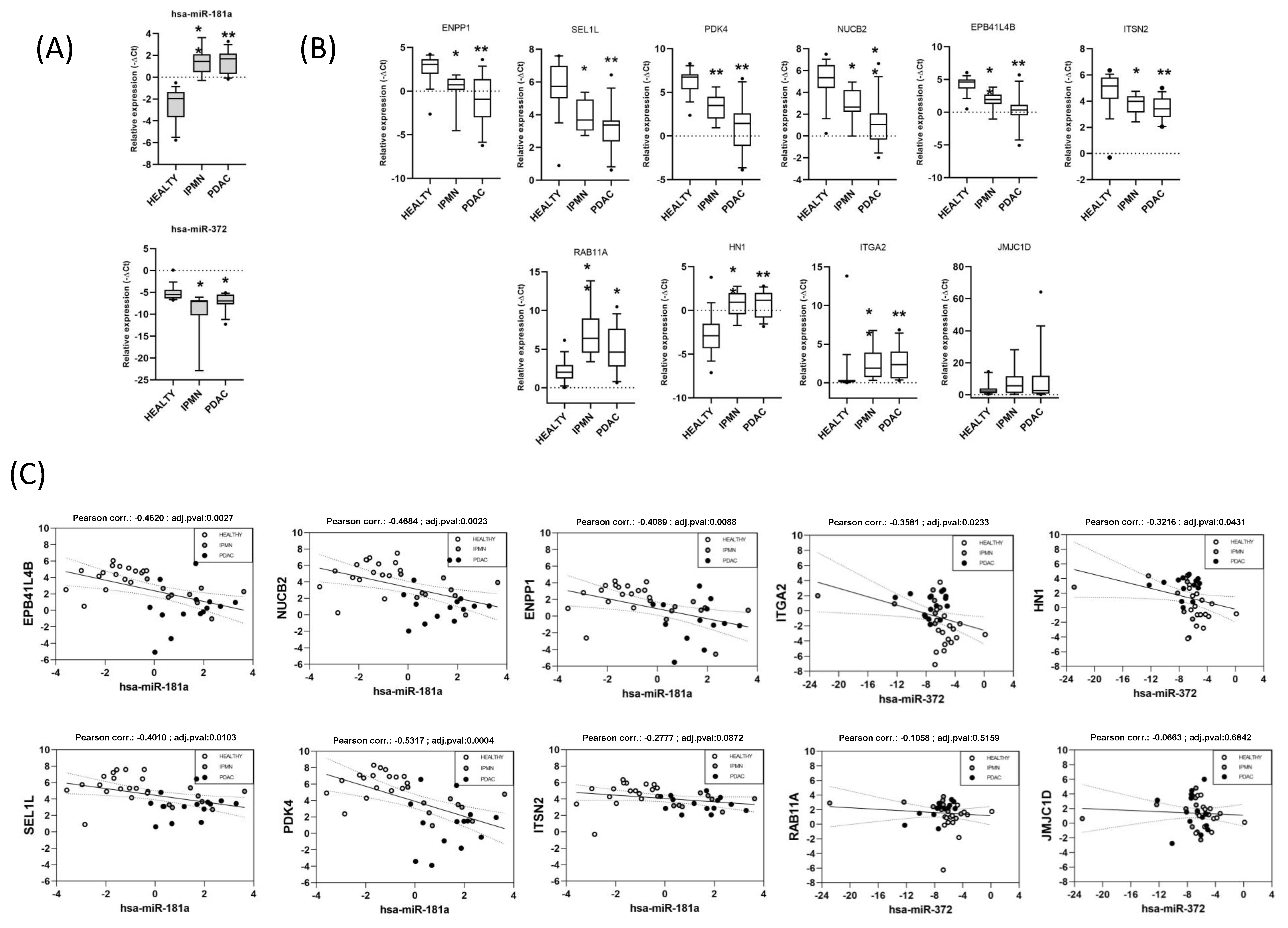
3.5. Validation of Functionally Relevant miRNA-mRNA Negative Correlations for miR-181a and miR-372 by RT-qPCR in an Independent Cohort
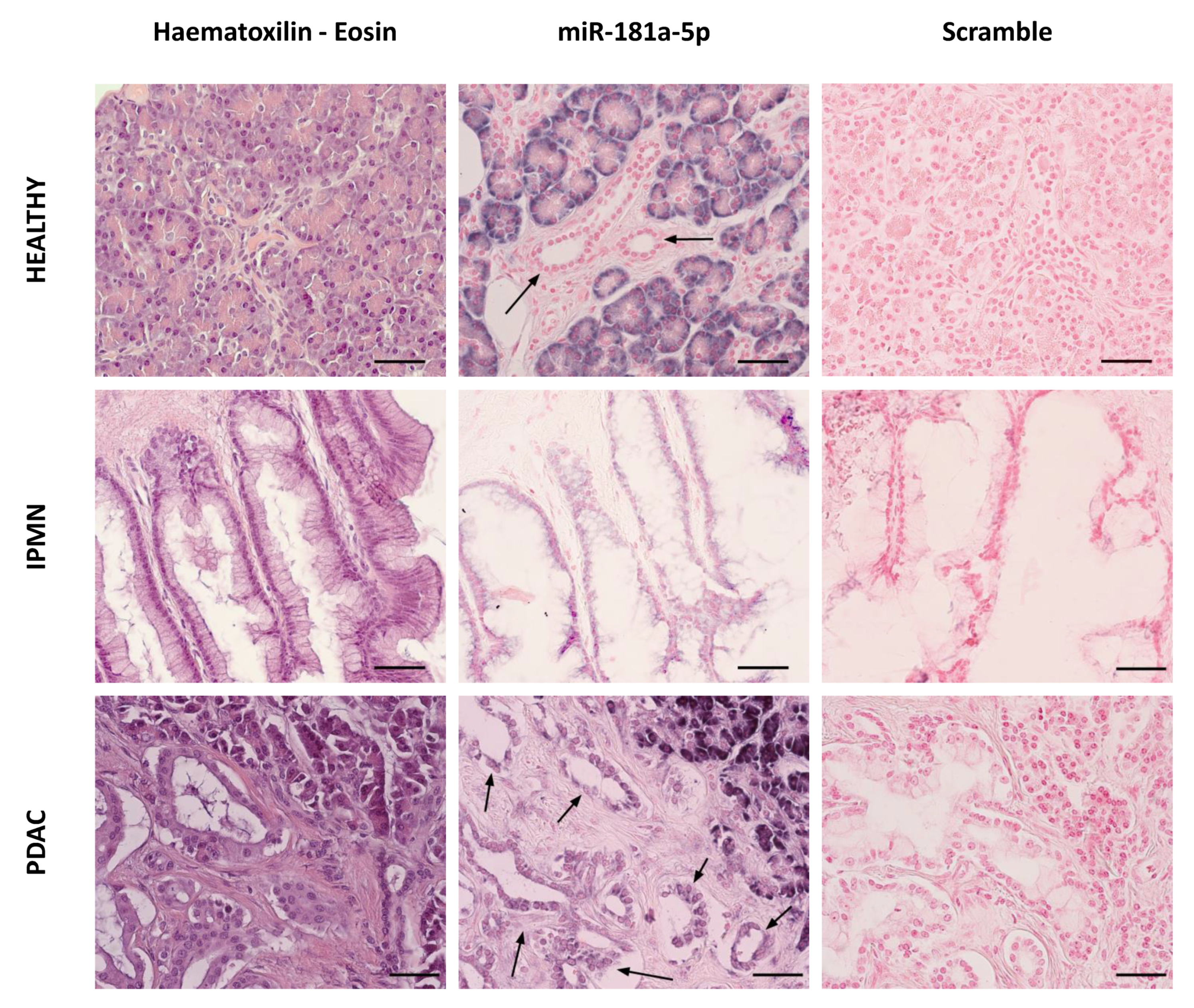
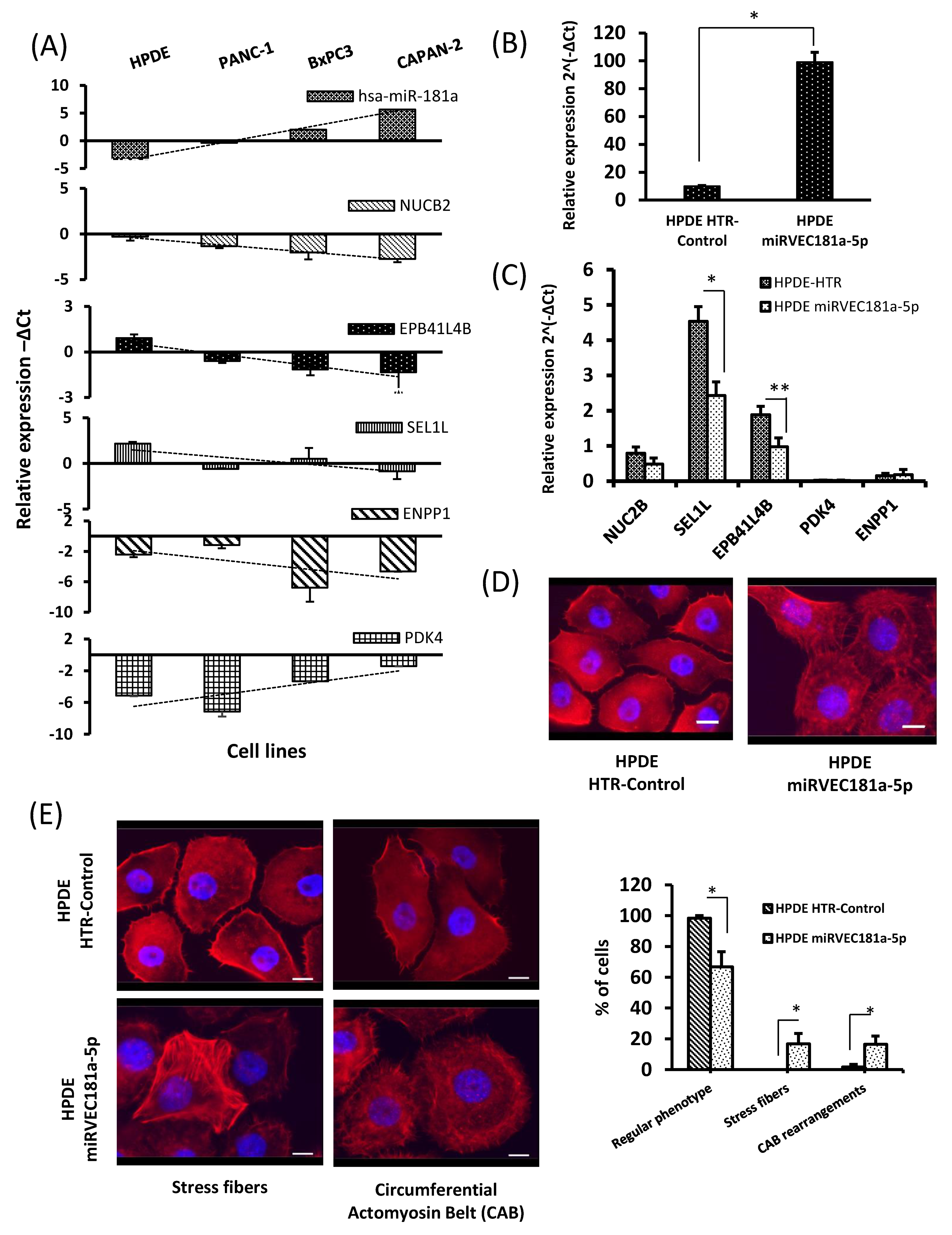
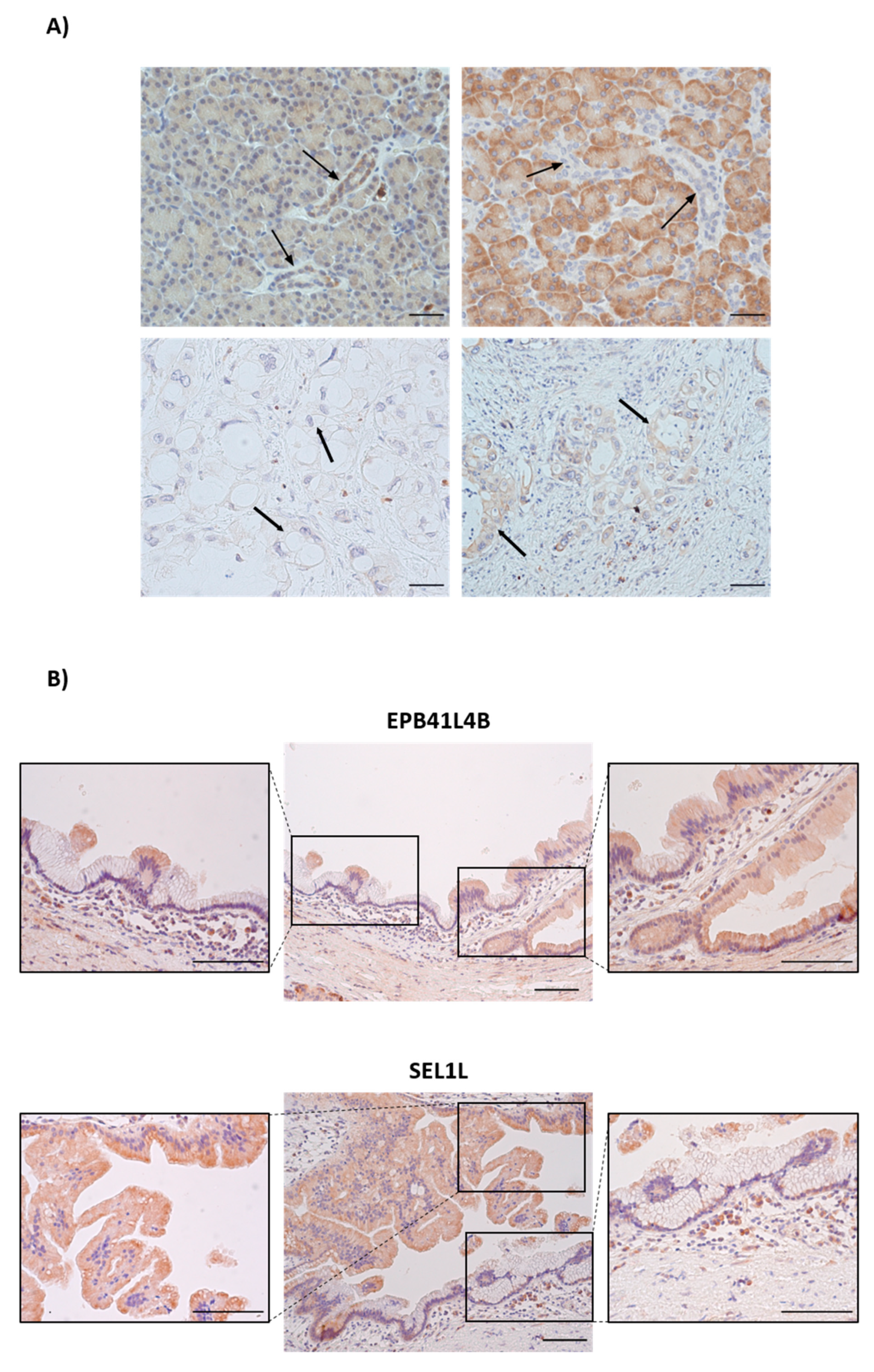
3.6. MiR-181a Staining Is Increased in Pancreatic Ductal Cells from IPMN and PDAC
3.7. Negative Correlation between miR-181a Expression and Its Potential Targets in Pancreatic Cancer Cell Lines
3.8. Pancreatic Ductal Cells Overexpressing miR-181a Show Alterations of the Actin Cytoskeleton Organization
3.9. Immunohistochemical Staining of EPB41L4B and SEL1L in Healthy, IPMN and PDAC Tissues
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA A Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021. [Google Scholar] [CrossRef] [PubMed]
- Singhi, A.D.; Koay, E.J.; Chari, S.T.; Maitra, A. Early Detection of Pancreatic Cancer: Opportunities and Challenges. Gastroenterology 2019, 156, 2024–2040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kopp, J.L.; Dubois, C.L.; Schaeffer, D.F.; Samani, A.; Taghizadeh, F.; Cowan, R.W.; Rhim, A.D.; Stiles, B.L.; Valasek, M.; Sander, M. Loss of Pten and Activation of Kras Synergistically Induce Formation of Intraductal Papillary Mucinous Neoplasia from Pancreatic Ductal Cells in Mice. Gastroenterology 2018, 154, 1509–1523.e5. [Google Scholar] [CrossRef] [PubMed]
- Basturk, O.; Hong, S.-M.; Wood, L.D.; Adsay, N.V.; Albores-Saavedra, J.; Biankin, A.V.; Brosens, L.A.; Fukushima, N.; Goggins, M.; Hruban, R.H.; et al. A Revised Classification System and Recommendations From the Baltimore Consensus Meeting for Neoplastic Precursor Lesions in the Pancreas. Am. J. Surg. Pathol. 2015, 39, 1730–1741. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.G.; Guthrie, V.B.; Braxton, A.M.; Zheng, L.; Wang, P.; Song, Q.; Griffin, J.F.; Chianchiano, P.E.; Hosoda, W.; Niknafs, N.; et al. Intraductal Papillary Mucinous Neoplasms Arise From Multiple Independent Clones, Each With Distinct Mutations. Gastroenterology 2019, 157, 1123–1137.e22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedman, R.C.; Farh, K.K.-H.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2008, 19, 92–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tesfaye, A.A.; Azmi, A.S.; Philip, P.A. miRNA and Gene Expression in Pancreatic Ductal Adenocarcinoma. Am. J. Pathol. 2019, 189, 58–70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vila-Navarro, E.; Vila-Casadesús, M.; Moreira, L.; Duran-Sanchon, S.; Sinha, R.; Ginés, À.; Fernández-Esparrach, G.; Miquel, R.; Cuatrecasas, M.; Castells, A.; et al. MicroRNAs for detection of pancreatic neoplasia: Biomarker discovery by next-generation sequencing and validation in 2 independent cohorts. Ann. Surg. 2017, 265, 1226–1234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Permuth-Wey, J.; Chen, Y.A.; Fisher, K.; McCarthy, S.; Qu, X.; Lloyd, M.C.; Kasprzak, A.; Fournier, M.; Williams, V.L.; Ghia, K.M.; et al. A Genome-Wide Investigation of MicroRNA Expression Identifies Biologically-Meaningful MicroRNAs That Distinguish between High-Risk and Low-Risk Intraductal Papillary Mucinous Neoplasms of the Pancreas. PLoS ONE 2015, 10, e0116869. [Google Scholar] [CrossRef]
- Vila-Navarro, E.; Duran-Sanchon, S.; Vila-Casadesús, M.; Moreira, L.; Ginès, À.; Cuatrecasas, M.; Lozano, J.J.; Bujanda, L.; Castells, A.; Gironella, M. Novel Circulating miRNA Signatures for Early Detection of Pancreatic Neoplasia. Clin. Transl. Gastroenterology 2019, 10, e00029. [Google Scholar] [CrossRef]
- Wilk, G.; Braun, R. Integrative analysis reveals disrupted pathways regulated by microRNAs in cancer. Nucleic Acids Res. 2017, 46, 1089–1101. [Google Scholar] [CrossRef] [Green Version]
- Vila-Casadesús, M.; Gironella, M.; Lozano, J.J. MiRComb: An R Package to Analyse miRNA-mRNA Interactions. Examples across Five Digestive Cancers. PLoS ONE 2016, 11, e0151127. [Google Scholar] [CrossRef]
- Vila-Casadesús, M.; Vila-Navarro, E.; Raimondi, G.; Fillat, C.; Castells, A.; Lozano, J.J.; Gironella, M. Deciphering microRNA targets in pancreatic cancer using miRComb R package. Oncotarget 2018, 9, 6499–6517. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.-G.; Han, Y.; He, Q.-Y. clusterProfiler: An R Package for Comparing Biological Themes Among Gene Clusters. OMICS J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef]
- Sun, S.; Shi, G.; Han, X.; Francisco, A.B.; Ji, Y.; Mendonça, N.; Liu, X.; Locasale, J.W.; Simpson, K.W.; Duhamel, G.E.; et al. Sel1L is indispensable for mammalian endoplasmic reticulum-associated degradation, endoplasmic reticulum homeostasis, and survival. Proc. Natl. Acad. Sci. USA 2014, 111, E582–E591. [Google Scholar] [CrossRef] [Green Version]
- Rezaee, N.; Barbon, C.; Zaki, A.; He, J.; Salman, B.; Hruban, R.H.; Cameron, J.L.; Herman, J.M.; Ahuja, N.; Lennon, A.M.; et al. Intraductal papillary mucinous neoplasm (IPMN) with high-grade dysplasia is a risk factor for the subsequent development of pancreatic ductal adenocarcinoma. HPB 2016, 18, 236–246. [Google Scholar] [CrossRef] [Green Version]
- Shindo, K.; Aishima, S.; Ohuchida, K.; Fujino, M.; Mizuuchi, Y.; Hattori, M.; Ohtsuka, T.; Tokunaga, S.; Mizumoto, K.; Tanaka, M.; et al. Podoplanin expression in the cyst wall correlates with the progression of intraductal papillary mucinous neoplasm. Virchows Archiv. 2014, 465, 265–273. [Google Scholar] [CrossRef]
- Ong, M.S.; Deng, S.; Halim, C.E.; Cai, W.; Tan, T.Z.; Huang, R.Y.-J.; Sethi, G.; Hooi, S.C.; Kumar, A.P.; Yap, C.T. Cytoskeletal Proteins in Cancer and Intracellular Stress: A Therapeutic Perspective. Cancers 2020, 12, 238. [Google Scholar] [CrossRef] [Green Version]
- Norris, V.; Amar, P.; Legent, G.; Ripoll, C.; Thellier, M.; Ovádi, J. Sensor potency of the moonlighting enzyme-decorated cytoskeleton: The cytoskeleton as a metabolic sensor. BMC Biochem. 2013, 14, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Nakajima, H.; Tanoue, T. The circumferential actomyosin belt in epithelial cells is regulated by the Lulu2-p114RhoGEF system. Small GTPases 2012, 3, 91–96. [Google Scholar] [CrossRef] [Green Version]
- Diaferia, G.R.; Cirulli, V.; Biunno, I. SEL1L Regulates Adhesion, Proliferation and Secretion of Insulin by Affecting Integrin Signaling. PLoS ONE 2013, 8, e79458. [Google Scholar] [CrossRef]
- Su, H.-Y.; Waldron, R.T.; Gong, R.; Ramanujan, V.K.; Pandol, S.J.; Lugea, A. The Unfolded Protein Response Plays a Predominant Homeostatic Role in Response to Mitochondrial Stress in Pancreatic Stellate Cells. PLoS ONE 2016, 11, e0148999. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Chen, J.; Wang, J.; Amos, C.; Killary, A.M.; Sen, S.; Wei, C.; Frazier, M.L. Putative tumor suppressor gene SEL1L was downregulated by aberrantly upregulated hsa-mir-155 in human pancreatic ductal adenocarcinoma. Mol. Carcinog. 2014, 53, 711–721. [Google Scholar] [CrossRef] [Green Version]
- Valencia, K.; Erice, O.; Kostyrko, K.; Hausmann, S.; Guruceaga, E.; Thathireddy, A.; Flores, N.M.; Sayles, L.C.; Lee, A.G.; Fragoso, R.; et al. The Mir181ab1 cluster promotes KRAS-driven oncogenesis and progression in lung and pancreas. J. Clin. Investig. 2020, 130, 1879–1895. [Google Scholar] [CrossRef]
- Kang, H.; Ma, D.; Zhang, J.; Zhao, J.; Yang, M. MicroRNA-18a induces epithelial-mesenchymal transition like cancer stem cell phenotype via regulating RKIP pathway in pancreatic cancer. Ann. Transl. Med. 2020, 8, 433. [Google Scholar] [CrossRef]
- Liu, J.; Xu, D.; Wang, Q.; Zheng, D.; Jiang, X.; Xu, L. LPS Induced miR-181a Promotes Pancreatic Cancer Cell Migration via Targeting PTEN and MAP2K4. Dig. Dis. Sci. 2014, 59, 1452–1460. [Google Scholar] [CrossRef]
- Meijer, L.L.; Garajová, I.; Caparello, C.; Le Large, T.Y.S.; Frampton, A.E.; Vasile, E.; Funel, N.; Kazemier, G.; Giovannetti, E. Plasma miR-181a-5p Downregulation Predicts Response and Improved Survival After FOLFIRINOX in Pancreatic Ductal Adenocarcinoma. Ann. Surg. 2020, 271, 1137–1147. [Google Scholar] [CrossRef]
- Vila-Navarro, E.; Fernandez-Castañer, E.; Rovira-Rigau, M.; Raimondi, G.; Vila-Casadesus, M.; Lozano, J.J.; Soubeyran, P.; Iovanna, J.; Castells, A.; Fillat, C.; et al. MiR-93 is related to poor prognosis in pancreatic cancer and promotes tumor progression by targeting microtubule dynamics. Oncogenesis 2020, 9, 43. [Google Scholar] [CrossRef]
- Hausser, J.; Syed, A.P.; Selevsek, N.; Van Nimwegen, E.; Jaskiewicz, L.; Aebersold, R.; Zavolan, M. Timescales and bottlenecks in miRNA-dependent gene regulation. Mol. Syst. Biol. 2013, 9, 711. [Google Scholar] [CrossRef] [PubMed]
- McCall, P.M.; MacKintosh, F.C.; Kovar, D.R.; Gardel, M.L. Cofilin drives rapid turnover and fluidization of entangled F-actin. Proc. Natl. Acad. Sci. USA 2019, 116, 12629–12637. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bleicher, P.; Sciortino, A.; Bausch, A.R. The dynamics of actin network turnover is self-organized by a growth-depletion feedback. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, R.H.; Macdonald, M.M.; Murphy, G.R.; Thorn, B.; Roberts, T.K. Modelling of protein turnover provides insight for metabolic demands on those specific amino acids utilised at disproportionately faster rates than other amino acids. Amino Acids 2019, 51, 945–959. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, M.E.; Perret, V.; Eng, L.F. Metabolic studies in vitro of the CNS cytoskeletal proteins: Synthesis and degradation. Neurochem. Res. 1984, 9, 1493–1507. [Google Scholar] [CrossRef]


| miRNA | mRNA | Cor | Adj.pval | FC.miRNA | FC.mRNA | Dat.Sum |
|---|---|---|---|---|---|---|
| hsa-miR-1297 | RAB21 | −1.00 | 5.14 × 10−3 | −2.27 | 2.03 | 2 |
| hsa-miR-29b | MORF4L1 | −1.00 | 5.14 × 10−3 | −2.21 | 2.22 | 2 |
| hsa-miR-29b | SUB1 | −0.99 | 1.08 × 10−2 | −2.21 | 2.06 | 2 |
| hsa-miR-181a | TPST2 | −0.99 | 1.08 × 10−2 | 5.67 | −34.93 | 2 |
| hsa-miR-1297 | DDX24 | −0.99 | 1.08 × 10−2 | −2.27 | 2.39 | 1 |
| hsa-miR-21 | KLB | −0.99 | 1.08 × 10−2 | 5.45 | −1.70 | 1 |
| hsa-miR-181a | VCX2 | −0.99 | 1.08 × 10−2 | 5.67 | −30.12 | 1 |
| hsa-miR-144 | CEBPA | −0.98 | 1.11 × 10−2 | 2.64 | −3.33 | 1 |
| hsa-miR-21 | ERO1LB | −0.98 | 1.11 × 10−2 | 5.45 | −11.93 | 1 |
| hsa-miR-144 | HERPUD1 | −0.98 | 1.11 × 10−2 | 2.64 | −3.70 | 1 |
| hsa-miR-29a | UHRF1BP1 | −0.98 | 1.11 × 10−2 | 3.29 | −2.26 | 1 |
| hsa-miR-29b | S100A16 | −0.98 | 1.11 × 10−2 | −2.21 | 11.23 | 1 |
| hsa-miR-181a | PDK4 | −0.98 | 1.11 × 10−2 | 5.67 | −9.62 | 2 |
| hsa-miR-21 | VGLL2 | −0.98 | 1.15 × 10−2 | 5.45 | −1.52 | 1 |
| hsa-miR-4760-3p | TRHDE | −0.98 | 1.15 × 10−2 | 4.22 | −5.57 | 1 |
| hsa-miR-144 | FAM129A | −0.98 | 1.16 × 10−2 | 2.64 | −9.69 | 1 |
| hsa-miR-4255 | YWHAB | −0.97 | 1.16 × 10−2 | −1.90 | 2.53 | 1 |
| hsa-miR-1297 | SETX | −0.97 | 1.16 × 10−2 | −2.27 | 1.89 | 1 |
| hsa-miR-29b | ZNF207 | −0.97 | 1.16 × 10−2 | −2.21 | 1.84 | 1 |
| hsa-miR-29b | ARPC5 | −0.97 | 1.16 × 10−2 | −2.21 | 2.24 | 1 |
| hsa-miR-29b | HNRNPF | −0.97 | 1.16 × 10−2 | −2.21 | 2.78 | 2 |
| hsa-miR-181a | SLC25A25 | −0.97 | 1.16 × 10−2 | 5.67 | −1.90 | 2 |
| hsa-miR-29b | CTNNB1 | −0.97 | 1.16 × 10−2 | −2.21 | 1.96 | 1 |
| hsa-miR-1297 | EIF1B | −0.97 | 1.16 × 10−2 | −2.27 | 1.74 | 1 |
| hsa-miR-935 | DENND6A | −0.97 | 1.18 × 10−2 | −2.77 | 2.04 | 1 |
| hsa-miR-3666 | SUB1 | −0.97 | 1.22 × 10−2 | −3.49 | 2.06 | 1 |
| hsa-miR-221 | HDLBP | −0.97 | 1.22 × 10−2 | 3.85 | −2.50 | 1 |
| hsa-miR-1 | SYBU | −0.97 | 1.22 × 10−2 | 2.35 | −7.97 | 1 |
| hsa-miR-29b | TOPORS | −0.97 | 1.22 × 10−2 | −2.21 | 1.72 | 1 |
| hsa-miR-29b | LPAR6 | −0.97 | 1.22 × 10−2 | −2.21 | 5.41 | 1 |
| hsa-miR-181a | IFRD1 | −0.97 | 1.22 × 10−2 | 5.67 | −6.83 | 1 |
| hsa-miR-181a | VGLL2 | −0.97 | 1.22 × 10−2 | 5.67 | −1.52 | 1 |
| hsa-miR-15b | CYP46A1 | −0.97 | 1.22 × 10−2 | 2.91 | −1.59 | 1 |
| hsa-miR-181a | ERO1LB | −0.97 | 1.22 × 10−2 | 5.67 | −11.93 | 1 |
| hsa-miR-378e | CPD | −0.97 | 1.22 × 10−2 | −2.28 | 4.56 | 1 |
| hsa-miR-606 | SEL1L | −0.97 | 1.22 × 10−2 | 1.74 | −5.33 | 1 |
| hsa-miR-1297 | ATP2C1 | −0.97 | 1.22 × 10−2 | −2.27 | 1.95 | 1 |
| hsa-miR-372 | CLIC1 | −0.97 | 1.22 × 10−2 | −1.95 | 4.52 | 1 |
| hsa-miR-3133 | CRELD2 | −0.97 | 1.22 × 10−2 | 5.56 | −3.67 | 1 |
| hsa-miR-21 | IFRD1 | −0.97 | 1.22 × 10−2 | 5.45 | −6.83 | 1 |
| hsa-miR-1297 | BHLHE40 | −0.96 | 1.23 × 10−2 | −2.27 | 5.95 | 2 |
| hsa-miR-181a | TMED6 | −0.96 | 1.23 × 10−2 | 5.67 | −71.29 | 1 |
| hsa-miR-181a | RNF144A | −0.96 | 1.23 × 10−2 | 5.67 | −3.31 | 1 |
| hsa-miR-144 | VLDLR | −0.96 | 1.23 × 10−2 | 2.64 | −5.62 | 2 |
| hsa-miR-29b | MCMBP | −0.96 | 1.23 × 10−2 | −2.21 | 2.73 | 1 |
| miRNA | # Targets | Cum% | Targets (Top20) |
|---|---|---|---|
| hsa-miR-181a | 46 | 24.33 | VCX2, PDK4, SLC25A25, IFRD1, VGLL2, ERO1LB, TMED6, RNF144A, AKAP7, GPR155, PSAT1, LMO3, CYP46A1, CBFA2T3, FKBP11, NUCB2, EPB41L4B, SERPINI1, SEL1L, LYG2 |
| hsa-miR-23a | 32 | 31.74 | ZNF503, SLC43A1, SYBU, MCFD2, ARHGAP18, KIAA0922, KPNA7, SEC63, GATM, SEL1L, LRIG1, ACAT1, SH3BGR, PDCD4, PDK4, ASNS, GCAT, HRASLS5, KLF11, SLC5A10 |
| hsa-miR-372 | 29 | 33.33 | CLIC1, ATP2C1, CAPNS1, MYL12B, JMJD1C, SPTLC2, FRMD4B, ITGA2, SQRDL, TAGLN2, LAMC2, MYO6, TOX3, CPD, PLS1, RFK, PGRMC1, PFKP, CAMK2N1, MYO1D |
| hsa-miR-93 | 28 | 33.86 | KIAA0922, VLDLR, FAM129A, PTGER4, KCNJ16, LMO3, CAMK2N2, ERO1LB, CDH4, ATXN7L2, ERRFI1, PAIP2B, KLB, EPB41L4B, IFRD1, KLF11, GUCA1C, SLC16A10, MUM1L1, KPNA7 |
| hsa-miR-21 | 24 | 35.98 | KLB, ERO1LB, VGLL2, IFRD1, FOXO3, HERPUD1, PAIP2B, LMO3, HRASLS5, SLC16A10, BTG2, DPP10, RNF144A, RUNDC3A, ITSN2, ACAT1, AKAP7, SERPINI1, DPY19L2P2, SEC63 |
| hsa-let-7e | 21 | 37.57 | SLC5A2, KIAA0922, CBFA2T3, CYP46A1, SDK1, ATXN7L2, SPACA3, HDLBP, TRHDE, HRASLS5, SIDT2, FKBP11, TPST2, P2RX1, TTN, LRIG1, RAB40C, KPNA7, BTG2, SLC8A2 |
| hsa-miR-217 | 20 | 38.01 | ARPC2, CAP1, G3BP2, FHL2, PFKP, SAT1, CXCL2, EZR, ADAM9, YWHAZ, RAC1, DPH3, SEP7, TOX3, SEP10, KLF5, ARL6IP1, RAB11A, ITGB5, ARF6 |
| hsa-miR-144 | 20 | 38.01 | HERPUD1, FAM129A, VLDLR, NUCB2, PTGER4, TTN, PDK4, VGLL2, KLB, LMO3, DNASE1, GPR155, KLF11, LOC729020, SEL1L, STC2, DPP10, ITSN2, TIMM8A, PPP2R2D |
| hsa-miR-98 | 18 | 39.07 | KIAA0922, CBFA2T3, EPB41L4B, CYP46A1, SLC5A2, TTN, SDK1, TRHDE, INPP5J, HRASLS5, P2RX1, IFT20, RPH3AL, HDLBP, KPNA7, LRIG1, SLC8A2, RAB40C |
| hsa-miR-1 | 16 | 40.3 | SYBU, ARHGAP18, TRHDE, CYP46A1, KRTAP5-11, KLF11, PABPC3, EPB41L4B, PDCD4, MT1H, FKBP11, LRRC29, SFTPC, KLF15, IFRD1, SEC63 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandez-Castañer, E.; Vila-Casadesus, M.; Vila-Navarro, E.; Parra, C.; Lozano, J.J.; Castells, A.; Gironella, M. MicroRNAs Deregulated in Intraductal Papillary Mucinous Neoplasm Converge on Actin Cytoskeleton-Related Pathways That Are Maintained in Pancreatic Ductal Adenocarcinoma. Cancers 2021, 13, 2369. https://doi.org/10.3390/cancers13102369
Fernandez-Castañer E, Vila-Casadesus M, Vila-Navarro E, Parra C, Lozano JJ, Castells A, Gironella M. MicroRNAs Deregulated in Intraductal Papillary Mucinous Neoplasm Converge on Actin Cytoskeleton-Related Pathways That Are Maintained in Pancreatic Ductal Adenocarcinoma. Cancers. 2021; 13(10):2369. https://doi.org/10.3390/cancers13102369
Chicago/Turabian StyleFernandez-Castañer, Elena, Maria Vila-Casadesus, Elena Vila-Navarro, Carolina Parra, Juan Jose Lozano, Antoni Castells, and Meritxell Gironella. 2021. "MicroRNAs Deregulated in Intraductal Papillary Mucinous Neoplasm Converge on Actin Cytoskeleton-Related Pathways That Are Maintained in Pancreatic Ductal Adenocarcinoma" Cancers 13, no. 10: 2369. https://doi.org/10.3390/cancers13102369
APA StyleFernandez-Castañer, E., Vila-Casadesus, M., Vila-Navarro, E., Parra, C., Lozano, J. J., Castells, A., & Gironella, M. (2021). MicroRNAs Deregulated in Intraductal Papillary Mucinous Neoplasm Converge on Actin Cytoskeleton-Related Pathways That Are Maintained in Pancreatic Ductal Adenocarcinoma. Cancers, 13(10), 2369. https://doi.org/10.3390/cancers13102369






