Non-Hematologic Toxicity of Bortezomib in Multiple Myeloma: The Neuromuscular and Cardiovascular Adverse Effects
Simple Summary
Abstract
1. Introduction
2. Bortezomib: The First-in-Class Proteasome Inhibitor
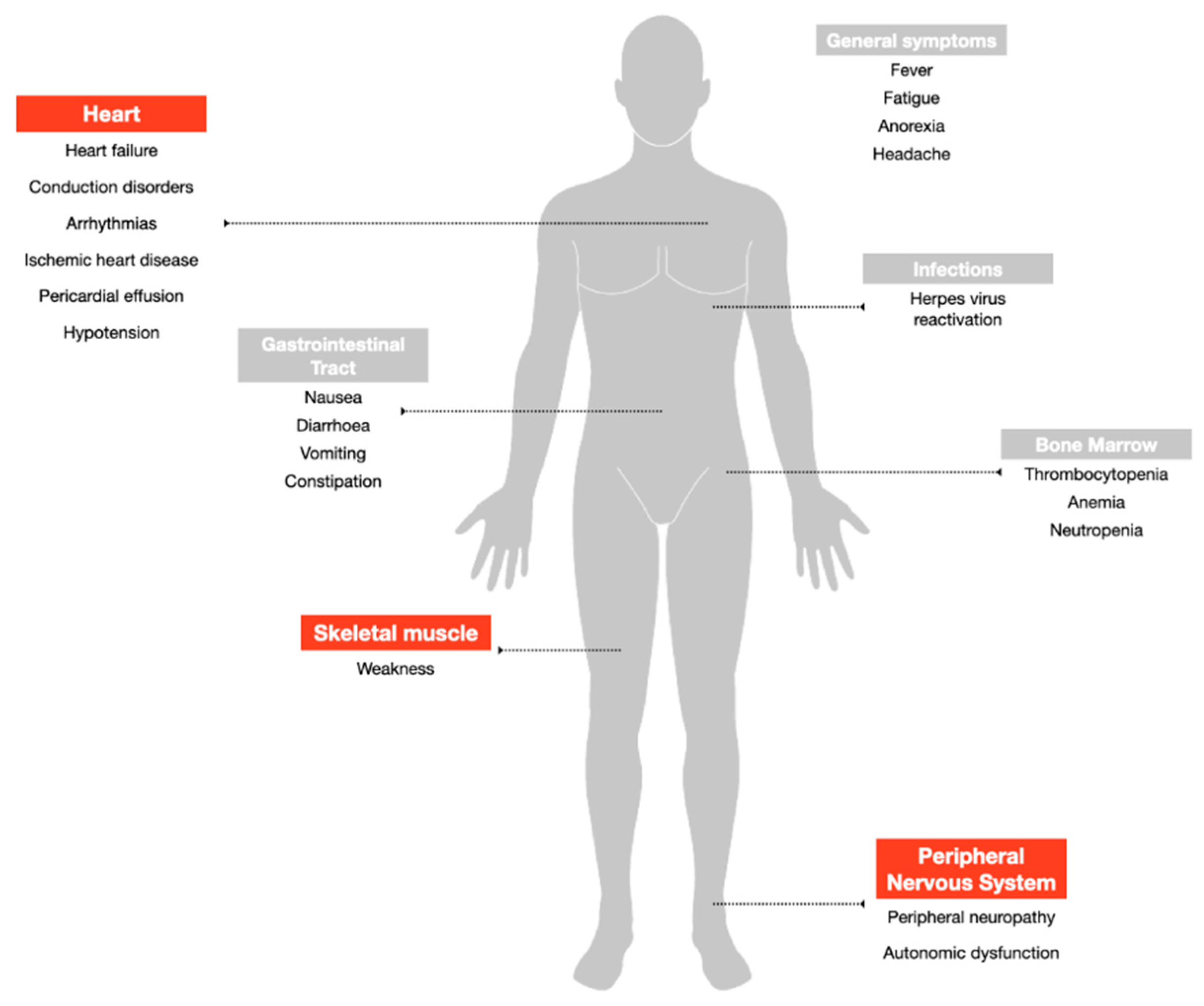
3. Peripheral Neuropathy: The Key Dose-Limiting Toxicity
4. Cardiovascular Toxicity: The Low Rate and Reversible Side Effect
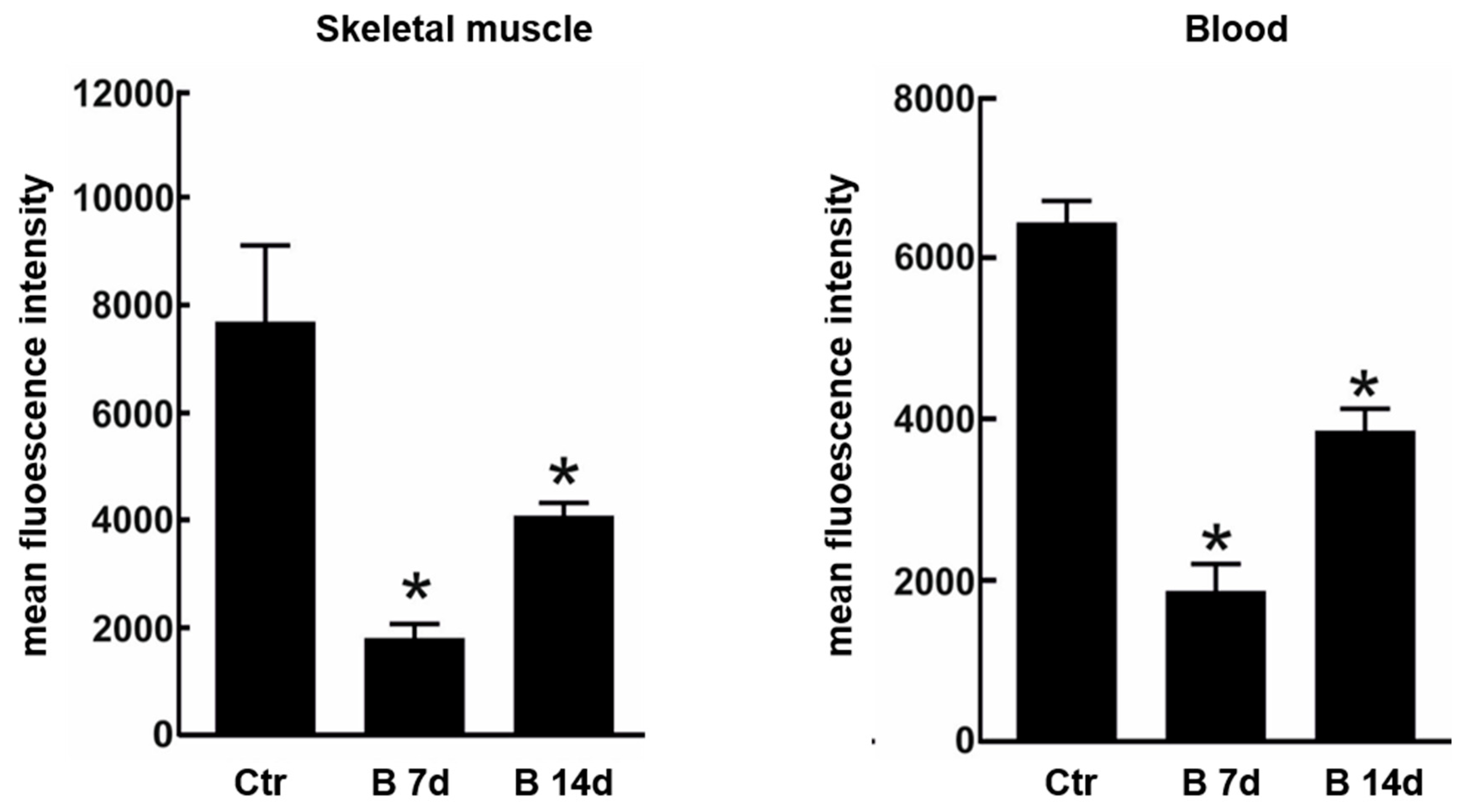
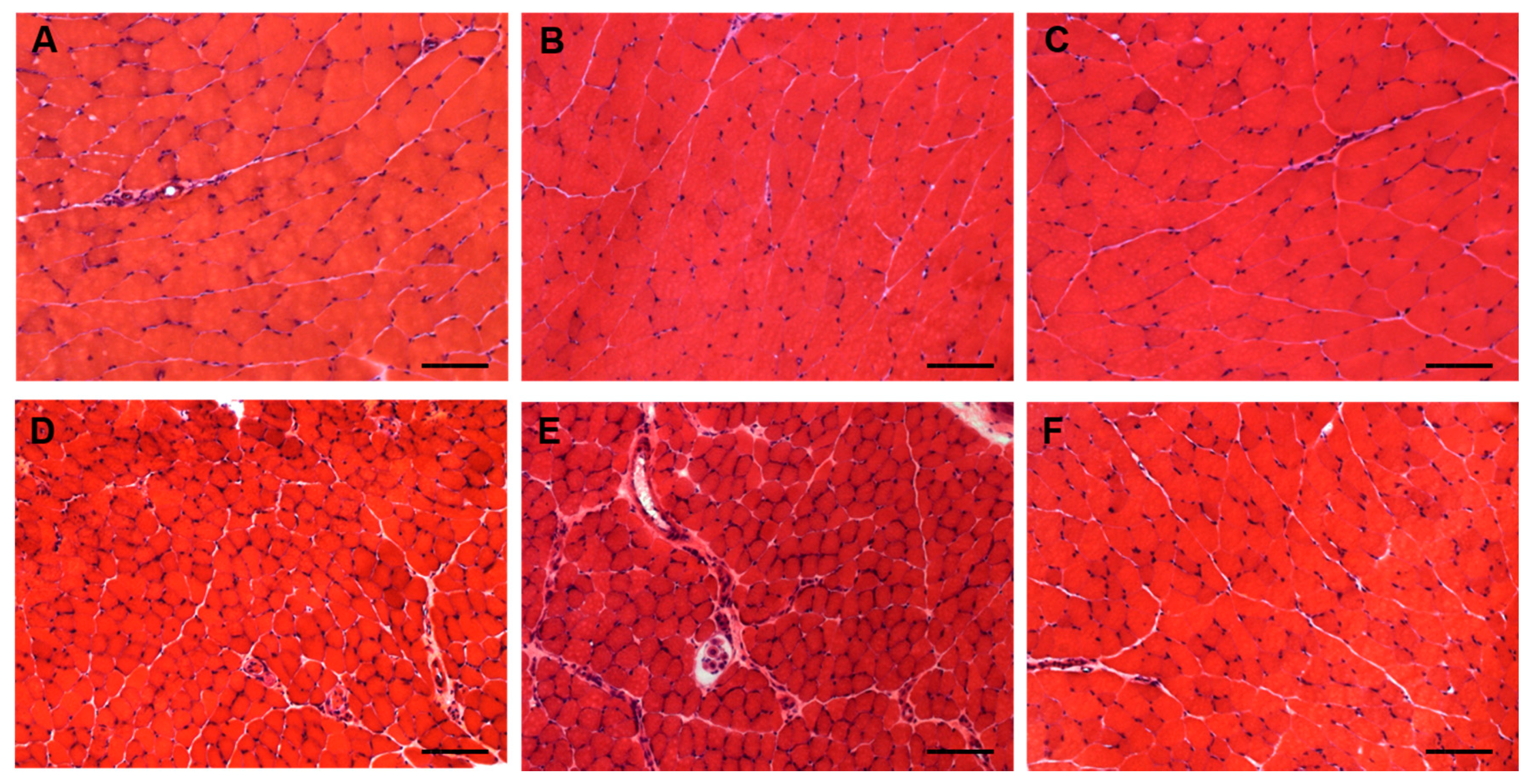
5. Muscle Toxicity: The Neglected Adverse Event
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.-V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef]
- Palumbo, A.; Anderson, K. Multiple Myeloma. N. Engl. J. Med. 2011, 364, 1046–1060. [Google Scholar] [CrossRef] [PubMed]
- Landgren, O.; Kyle, R.A.; Pfeiffer, R.M.; Katzmann, J.A.; Caporaso, N.E.; Hayes, R.B.; Dispenzieri, A.; Kumar, S.; Clark, R.J.; Baris, D.; et al. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: A prospective study. Blood 2009, 113, 5412–5417. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Rajkumar, V.; Kyle, R.A.; van Duin, M.; Sonneveld, P.; Mateos, M.-V.; Gay, F.; Anderson, K.C. Multiple myeloma. Nat. Rev. Dis. Primer 2017, 3, 17046. [Google Scholar] [CrossRef] [PubMed]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. SEER Cancer Statistics Review, 1975–2017; National Cancer Institute: Bethesda, MD, USA, 2020.
- Kumar, S.K.; Dispenzieri, A.; Lacy, M.Q.; Gertz, M.A.; Buadi, F.K.; Pandey, S.; Kapoor, P.; Dingli, D.; Hayman, S.R.; Leung, N.; et al. Continued improvement in survival in multiple myeloma: Changes in early mortality and outcomes in older patients. Leukemia 2014, 28, 1122–1128. [Google Scholar] [CrossRef]
- Gandolfi, S.; Laubach, J.P.; Hideshima, T.; Chauhan, D.; Anderson, K.C.; Richardson, P.G. The proteasome and proteasome inhibitors in multiple myeloma. Cancer Metastasis Rev. 2017, 36, 561–584. [Google Scholar] [CrossRef] [PubMed]
- Bird, S.A.; Boyd, K. Multiple myeloma: An overview of management. Palliat. Care Soc. Pract. 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Pinto, V.; Bergantim, R.; Caires, H.R.; Seca, H.; Guimarães, J.E.; Vasconcelos, M.H. Multiple Myeloma: Available Therapies and Causes of Drug Resistance. Cancers 2020, 12, 407. [Google Scholar] [CrossRef]
- Sherman, D.J.; Li, J. Proteasome Inhibitors: Harnessing Proteostasis to Combat Disease. Molecules 2020, 25, 671. [Google Scholar] [CrossRef]
- Walter, P.; Ron, D. The Unfolded Protein Response: From Stress Pathway to Homeostatic Regulation. Science 2011, 334, 1081–1086. [Google Scholar] [CrossRef]
- Ri, M. Endoplasmic-reticulum stress pathway-associated mechanisms of action of proteasome inhibitors in multiple myeloma. Int. J. Hematol. 2016, 104, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Adams, J. The proteasome: A suitable antineoplastic target. Nat. Rev. Cancer 2004, 4, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Mitsiades, N.; Mitsiades, C.S.; Richardson, P.G.; Poulaki, V.; Tai, Y.-T.; Chauhan, D.; Fanourakis, G.; Gu, X.; Bailey, C.; Joseph, M.; et al. The proteasome inhibitor PS-341 potentiates sensitivity of multiple myeloma cells to conventional chemotherapeutic agents: Therapeutic applications. Blood 2003, 101, 2377–2380. [Google Scholar] [CrossRef] [PubMed]
- Mateos, M.-V.; Ludwig, H.; Bazarbachi, A.; Beksac, M.; Bladé, J.; Boccadoro, M.; Cavo, M.; Delforge, M.; Dimopoulos, M.A.; Facon, T.; et al. Insights on Multiple Myeloma Treatment Strategies. HemaSphere 2019, 3, e163. [Google Scholar] [CrossRef]
- Kumar, S.K.; Callander, N.S.; Hillengass, J.; Liedtke, M.; Baljevic, M.; Campagnaro, E.; Castillo, J.J.; Chandler, J.C.; Cornell, R.F.; Costello, C.; et al. NCCN Guidelines Insights: Multiple Myeloma, Version 1.2020. J. Natl. Compr. Cancer Netw. 2019, 17, 1154–1165. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Kumar, S. Multiple Myeloma: Diagnosis and Treatment. Mayo Clin. Proc. 2016, 91, 101–119. [Google Scholar] [CrossRef]
- Manasanch, E.E.; Orlowski, R.Z. Proteasome inhibitors in cancer therapy. Nat. Rev. Clin. Oncol. 2017, 14, 417–433. [Google Scholar] [CrossRef]
- Groll, M.; Berkers, C.R.; Ploegh, H.L.; Ovaa, H. Crystal Structure of the Boronic Acid-Based Proteasome Inhibitor Bortezomib in Complex with the Yeast 20S Proteasome. Structure 2006, 14, 451–456. [Google Scholar] [CrossRef]
- Barrio, S.; Stühmer, T.; Da-Viá, M.; Barrio-Garcia, C.; Lehners, N.; Besse, A.; Cuenca, I.; Garitano-Trojaola, A.; Fink, S.; Leich, E.; et al. Spectrum and functional validation of PSMB5 mutations in multiple myeloma. Leukemia 2019, 33, 447–456. [Google Scholar] [CrossRef]
- Allmeroth, K.; Horn, M.; Kroef, V.; Miethe, S.; Müller, R.-U.; Denzel, M.S. Bortezomib resistance mutations in PSMB5 determine response to second-generation proteasome inhibitors in multiple myeloma. Leukemia 2020. [Google Scholar] [CrossRef]
- Lü, S.; Wang, J. The resistance mechanisms of proteasome inhibitor bortezomib. Biomark. Res. 2013, 1, 13. [Google Scholar] [CrossRef] [PubMed]
- Oakes, S.A.; Papa, F.R. The Role of Endoplasmic Reticulum Stress in Human Pathology. Annu. Rev. Pathol. Mech. Dis. 2015, 10, 173–194. [Google Scholar] [CrossRef]
- Ito, S. Proteasome Inhibitors for the Treatment of Multiple Myeloma. Cancers 2020, 12, 265. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-W.; Chen, H.; Campbell, R.A.; Bonavida, B.; Berenson, J.R. NF-kB in the pathogenesis and treatment of multiple myeloma. Curr. Opin. Hematol. 2008, 15, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Hideshima, T.; Mitsiades, C.; Akiyama, M.; Hayashi, T.; Chauhan, D.; Richardson, P.; Schlossman, R.; Podar, K.; Munshi, N.C.; Mitsiades, N.; et al. Molecular mechanisms mediating antimyeloma activity of proteasome inhibitor PS-341. Blood 2003, 101, 1530–1534. [Google Scholar] [CrossRef]
- Rajkumar, S.V.; Richardson, P.G.; Hideshima, T.; Anderson, K.C. Proteasome Inhibition As a Novel Therapeutic Target in Human Cancer. J. Clin. Oncol. 2005, 23, 630–639. [Google Scholar] [CrossRef]
- Tan, C.R.C.; Abdul-Majeed, S.; Cael, B.; Barta, S.K. Clinical Pharmacokinetics and Pharmacodynamics of Bortezomib. Clin. Pharmacokinet. 2019, 58, 157–168. [Google Scholar] [CrossRef]
- Richardson, P.G.; Barlogie, B.; Berenson, J.; Singhal, S.; Jagannath, S.; Irwin, D.; Rajkumar, S.V.; Srkalovic, G.; Alsina, M.; Alexanian, R.; et al. A Phase 2 Study of Bortezomib in Relapsed, Refractory Myeloma. N. Engl. J. Med. 2003, 348, 2609–2617. [Google Scholar] [CrossRef]
- Richardson, P.G.; Sonneveld, P.; Schuster, M.W.; Irwin, D.; Stadtmauer, E.A.; Facon, T.; Harousseau, J.-L.; Ben-Yehuda, D.; Lonial, S.; Goldschmidt, H.; et al. Bortezomib or High-Dose Dexamethasone for Relapsed Multiple Myeloma. N. Engl. J. Med. 2005, 352, 2487–2498. [Google Scholar] [CrossRef]
- Argyriou, A.A.; Iconomou, G.; Kalofonos, H.P. Bortezomib-induced peripheral neuropathy in multiple myeloma: A comprehensive review of the literature. Blood 2008, 112, 1593–1599. [Google Scholar] [CrossRef]
- Song, X.; Wilson, K.L.; Kagan, J.; Panjabi, S. Cost of peripheral neuropathy in patients receiving treatment for multiple myeloma: A US administrative claims analysis. Ther. Adv. Hematol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Argyriou, A.A.; Cavaletti, G.; Bruna, J.; Kyritsis, A.P.; Kalofonos, H.P. Bortezomib-induced peripheral neurotoxicity: An update. Arch. Toxicol. 2014, 88, 1669–1679. [Google Scholar] [CrossRef] [PubMed]
- Giannoccaro, M.P.; Donadio, V.; Gomis Pèrez, C.; Borsini, W.; Di Stasi, V.; Liguori, R. Somatic and autonomic small fiber neuropathy induced by bortezomib therapy: An immunofluorescence study. Neurol. Sci. 2011, 32, 361–363. [Google Scholar] [CrossRef] [PubMed]
- Mele, G.; Coppi, M.R.; Melpignano, A.; Quarta, G. Paralytic ileus following “subcutaneous bortezomib” therapy: Focus on the clinical emergency—Report of two cases. Clin. Exp. Med. 2016, 16, 99–101. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.H. Phase II Study of the Proteasome Inhibitor Bortezomib (PS-341) in Patients with Metastatic Neuroendocrine Tumors. Clin. Cancer Res. 2004, 10, 6111–6118. [Google Scholar] [CrossRef][Green Version]
- Stratogianni, A.; Tosch, M.; Schlemmer, H.; Weis, J.; Katona, I.; Isenmann, S.; Haensch, C.A. Bortezomib-induced severe autonomic neuropathy. Clin. Auton. Res. 2012, 22, 199–202. [Google Scholar] [CrossRef]
- Mauermann, M.L.; Blumenreich, M.S.; Dispenzieri, A.; Staff, N.P. A case of peripheral nerve microvasculitis associated with multiple myeloma and bortezomib treatment. Muscle Nerve 2012, 46, 964–970. [Google Scholar] [CrossRef]
- Chaudhry, V.; Cornblath, D.R.; Polydefkis, M.; Ferguson, A.; Borrello, I. Characteristics of bortezomib- and thalidomide-induced peripheral neuropathy. J. Peripher. Nerv. Syst. 2008, 13, 275–282. [Google Scholar] [CrossRef]
- Ravaglia, S.; Corso, A.; Piccolo, G.; Lozza, A.; Alfonsi, E.; Mangiacavalli, S.; Varettoni, M.; Zappasodi, P.; Moglia, A.; Lazzarino, M.; et al. Immune-mediated neuropathies in myeloma patients treated with bortezomib. Clin. Neurophysiol. 2008, 119, 2507–2512. [Google Scholar] [CrossRef]
- Thawani, S.P.; Tanji, K.; De Sousa, E.A.; Weimer, L.H.; Brannagan, T.H. Bortezomib-Associated Demyelinating Neuropathy—Clinical and Pathologic Features. J. Clin. Neuromuscul. Dis. 2015, 16, 202–209. [Google Scholar] [CrossRef]
- Filosto, M.; Rossi, G.; Pelizzari, A.M.; Buzio, S.; Tentorio, M.; Broglio, L.; Mancuso, M.; Rinaldi, M.; Scarpelli, M.; Padovani, A. A high-dose bortezomib neuropathy with sensory ataxia and myelin involvement. J. Neurol. Sci. 2007, 263, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Thomas, V.M.; Mulay, S. Bortezomib-induced motor neuropathy: A case report. J. Oncol. Pharm. Pract. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Pagliuca, A.; Devereux, S.; Mufti, G.J.; Schey, S. Life-threatening motor neurotoxicity in association wih bortezomib. Haematologica 2006, 91, 1001. [Google Scholar] [PubMed]
- Jeter, A.; Kang, Y. Immune modulation therapy in the management of bortezomib-induced peripheral neuropathy. Exp. Hematol. Oncol. 2012, 1, 20. [Google Scholar] [CrossRef]
- Schmitt, S.; Goldschmidt, H.; Storch-Hagenlocher, B.; Pham, M.; Fingerle-Rowson, G.; Ho, A.D.; Neben, K. Inflammatory autoimmune neuropathy, presumably induced by bortezomib, in a patient suffering from multiple myeloma. Int. J. Hematol. 2011, 93, 791–794. [Google Scholar] [CrossRef]
- Saifee, T.A.; Elliott, K.J.; Lunn, M.P.; Blake, J.; Reilly, M.M.; Rabin, N.; Yong, K.L.; D’Sa, S.; Brandner, S. Bortezomib-induced inflammatory neuropathy. J. Peripher. Nerv. Syst. 2010, 15, 366–368. [Google Scholar] [CrossRef]
- Li, T.; Timmins, H.C.; King, T.; Kiernan, M.C.; Goldstein, D.; Park, S.B. Characteristics and risk factors of bortezomib induced peripheral neuropathy: A systematic review of phase III trials. Hematol. Oncol. 2020. [Google Scholar] [CrossRef]
- Velasco, R.; Alberti, P.; Bruna, J.; Psimaras, D.; Argyriou, A.A. Bortezomib and other proteosome inhibitors—Induced peripheral neurotoxicity: From pathogenesis to treatment. J. Peripher. Nerv. Syst. 2019, 24. [Google Scholar] [CrossRef]
- Richardson, P.G.; Briemberg, H.; Jagannath, S.; Wen, P.Y.; Barlogie, B.; Berenson, J.; Singhal, S.; Siegel, D.S.; Irwin, D.; Schuster, M.; et al. Frequency, Characteristics, and Reversibility of Peripheral Neuropathy During Treatment of Advanced Multiple Myeloma With Bortezomib. J. Clin. Oncol. 2006, 24, 3113–3120. [Google Scholar] [CrossRef]
- Knopf, K.B.; Duh, M.S.; Lafeuille, M.-H.; Gravel, J.; Lefebvre, P.; Niculescu, L.; Ba-Mancini, A.; Ma, E.; Shi, H.; Comenzo, R.L. Meta-Analysis of the Efficacy and Safety of Bortezomib Re-Treatment in Patients With Multiple Myeloma. Clin. Lymphoma Myeloma Leuk. 2014, 14, 380–388. [Google Scholar] [CrossRef]
- Vidisheva, A.P.; Wang, J.; Spektor, T.M.; Bitran, J.D.; Lutzky, J.; Tabbara, I.A.; Ye, J.Z.; Ailawadhi, S.; Stampleman, L.V.; Steis, R.G.; et al. Safety of BTZ retreatment for patients with low-grade peripheral neuropathy during the initial treatment. Support. Care Cancer 2017, 25, 3217–3224. [Google Scholar] [CrossRef] [PubMed]
- Bringhen, S.; Larocca, A.; Rossi, D.; Cavalli, M.; Genuardi, M.; Ria, R.; Gentili, S.; Patriarca, F.; Nozzoli, C.; Levi, A.; et al. Efficacy and safety of once-weekly bortezomib in multiple myeloma patients. Blood 2010, 116, 4745–4753. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Chen, J.; Xuan, Z.; Yang, W.; Chen, J. Subcutaneous bortezomib might be standard of care for patients with multiple myeloma: A systematic review and meta-analysis. Drug Des. Devel. Ther. 2019, 13, 1707–1716. [Google Scholar] [CrossRef] [PubMed]
- Mu, S.; Ai, L.; Qin, Y.; Hu, Y. Subcutaneous versus Intravenous Bortezomib Administration for Multiple Myeloma Patients: A Meta-analysis. Curr. Med. Sci. 2018, 38, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Kleber, M.; Engelhardt, M.; Zweegman, S.; Gay, F.; Kastritis, E.; van de Donk, N.W.C.J.; Bruno, B.; Sezer, O.; Broijl, A.; et al. European Myeloma Network Guidelines for the Management of Multiple Myeloma-related Complications. Haematologica 2015, 100, 1254–1266. [Google Scholar] [CrossRef]
- Richardson, P.G.; Delforge, M.; Beksac, M.; Wen, P.; Jongen, J.L.; Sezer, O.; Terpos, E.; Munshi, N.; Palumbo, A.; Rajkumar, S.V.; et al. Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia 2012, 26, 595–608. [Google Scholar] [CrossRef]
- Dimopoulos, M.A.; Mateos, M.-V.; Richardson, P.G.; Schlag, R.; Khuageva, N.K.; Shpilberg, O.; Kropff, M.; Spicka, I.; Palumbo, A.; Wu, K.L.; et al. Risk factors for, and reversibility of, peripheral neuropathy associated with bortezomib-melphalan-prednisone in newly diagnosed patients with multiple myeloma: Subanalysis of the phase 3 VISTA study: Reversibility of PN with VMP in newly diagnosed MM. Eur. J. Haematol. 2011, 86, 23–31. [Google Scholar] [CrossRef]
- Richardson, P.G.; Sonneveld, P.; Schuster, M.W.; Stadtmauer, E.A.; Facon, T.; Harousseau, J.-L.; Ben-Yehuda, D.; Lonial, S.; Goldschmidt, H.; Reece, D.; et al. Reversibility of symptomatic peripheral neuropathy with bortezomib in the phase III APEX trial in relapsed multiple myeloma: Impact of a dose-modification guideline. Br. J. Haematol. 2009, 144, 895–903. [Google Scholar] [CrossRef]
- Boyette-Davis, J.A.; Cata, J.P.; Zhang, H.; Driver, L.C.; Wendelschafer-Crabb, G.; Kennedy, W.R.; Dougherty, P.M. Follow-Up Psychophysical Studies in Bortezomib-Related Chemoneuropathy Patients. J. Pain 2011, 12, 1017–1024. [Google Scholar] [CrossRef]
- Ibrahim, E.Y.; Ehrlich, B.E. Prevention of chemotherapy-induced peripheral neuropathy: A review of recent findings. Crit. Rev. Oncol. Hematol. 2020, 145, 102831. [Google Scholar] [CrossRef]
- Lanzani, F.; Mattavelli, L.; Frigeni, B.; Rossini, F.; Cammarota, S.; Petrò, D.; Jann, S.; Cavaletti, G. Role of a pre-existing neuropathy on the course of bortezomib-induced peripheral neurotoxicity. J. Peripher. Nerv. Syst. 2008, 13, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Badros, A.; Goloubeva, O.; Dalal, J.S.; Can, I.; Thompson, J.; Rapoport, A.P.; Heyman, M.; Akpek, G.; Fenton, R.G. Neurotoxicity of bortezomib therapy in multiple myeloma: A single-center experience and review of the literature. Cancer 2007, 110, 1042–1049. [Google Scholar] [CrossRef] [PubMed]
- Richardson, P.G.; Xie, W.; Mitsiades, C.; Chanan-Khan, A.A.; Lonial, S.; Hassoun, H.; Avigan, D.E.; Oaklander, A.L.; Kuter, D.J.; Wen, P.Y.; et al. Single-Agent Bortezomib in Previously Untreated Multiple Myeloma: Efficacy, Characterization of Peripheral Neuropathy, and Molecular Correlations With Response and Neuropathy. J. Clin. Oncol. 2009, 27, 3518–3525. [Google Scholar] [CrossRef] [PubMed]
- Corso, A.; Mangiacavalli, S.; Varettoni, M.; Pascutto, C.; Zappasodi, P.; Lazzarino, M. Bortezomib-induced peripheral neuropathy in multiple myeloma: A comparison between previously treated and untreated patients. Leuk. Res. 2010, 34, 471–474. [Google Scholar] [CrossRef] [PubMed]
- Mateos, M.-V.; Hernández, J.-M.; Hernández, M.-T.; Gutiérrez, N.-C.; Palomera, L.; Fuertes, M.; Díaz-Mediavilla, J.; Lahuerta, J.-J.; de la Rubia, J.; Terol, M.-J.; et al. Bortezomib plus melphalan and prednisone in elderly untreated patients with multiple myeloma: Results of a multicenter phase 1/2 study. Blood 2006, 108, 2165–2172. [Google Scholar] [CrossRef]
- Casafont, I.; Berciano, M.T.; Lafarga, M. Bortezomib Induces the Formation of Nuclear poly(A) RNA Granules Enriched in Sam68 and PABPN1 in Sensory Ganglia Neurons. Neurotox. Res. 2010, 17, 167–178. [Google Scholar] [CrossRef]
- Carozzi, V.A.; Canta, A.; Oggioni, N.; Sala, B.; Chiorazzi, A.; Meregalli, C.; Bossi, M.; Marmiroli, P.; Cavaletti, G. Neurophysiological and neuropathological characterization of new murine models of chemotherapy-induced chronic peripheral neuropathies. Exp. Neurol. 2010, 226, 301–309. [Google Scholar] [CrossRef]
- Cata, J.P.; Weng, H.-R.; Burton, A.W.; Villareal, H.; Giralt, S.; Dougherty, P.M. Quantitative Sensory Findings in Patients With Bortezomib-Induced Pain. J. Pain 2007, 8, 296–306. [Google Scholar] [CrossRef]
- Terkelsen, A.J.; Karlsson, P.; Lauria, G.; Freeman, R.; Finnerup, N.B.; Jensen, T.S. The diagnostic challenge of small fibre neuropathy: Clinical presentations, evaluations, and causes. Lancet Neurol. 2017, 16, 934–944. [Google Scholar] [CrossRef]
- Meregalli, C.; Canta, A.; Carozzi, V.A.; Chiorazzi, A.; Oggioni, N.; Gilardini, A.; Ceresa, C.; Avezza, F.; Crippa, L.; Marmiroli, P.; et al. Bortezomib-induced painful neuropathy in rats: A behavioral, neurophysiological and pathological study in rats. Eur. J. Pain 2010, 14, 343–350. [Google Scholar] [CrossRef]
- Carozzi, V.A.; Renn, C.L.; Bardini, M.; Fazio, G.; Chiorazzi, A.; Meregalli, C.; Oggioni, N.; Shanks, K.; Quartu, M.; Serra, M.P.; et al. Bortezomib-Induced Painful Peripheral Neuropathy: An Electrophysiological, Behavioral, Morphological and Mechanistic Study in the Mouse. PLoS ONE 2013, 8, e72995. [Google Scholar] [CrossRef] [PubMed]
- Bruna, J.; Udina, E.; Alé, A.; Vilches, J.J.; Vynckier, A.; Monbaliu, J.; Silverman, L.; Navarro, X. Neurophysiological, histological and immunohistochemical characterization of bortezomib-induced neuropathy in mice. Exp. Neurol. 2010, 223, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Bruna, J.; Alé, A.; Velasco, R.; Jaramillo, J.; Navarro, X.; Udina, E. Evaluation of pre-existing neuropathy and bortezomib retreatment as risk factors to develop severe neuropathy in a mouse model. J. Peripher. Nerv. Syst. 2011, 16, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.K.; Jang, S.Y.; Lee, H.K.; Jung, J.; Suh, D.J.; Seo, S.-Y.; Park, H.T. Pathological adaptive responses of Schwann cells to endoplasmic reticulum stress in bortezomib-induced peripheral neuropathy. Glia 2010, 58, 1961–1976. [Google Scholar] [CrossRef] [PubMed]
- Cavaletti, G.; Gilardini, A.; Canta, A.; Rigamonti, L.; Rodriguez-Menendez, V.; Ceresa, C.; Marmiroli, P.; Bossi, M.; Oggioni, N.; D’Incalci, M.; et al. Bortezomib-induced peripheral neurotoxicity: A neurophysiological and pathological study in the rat. Exp. Neurol. 2007, 204, 317–325. [Google Scholar] [CrossRef]
- Gilardini, A.; Avila, R.L.; Oggioni, N.; Rodriguez-Menendez, V.; Bossi, M.; Canta, A.; Cavaletti, G.; Kirschner, D.A. Myelin structure is unaltered in chemotherapy-induced peripheral neuropathy. NeuroToxicology 2012, 33, 1–7. [Google Scholar] [CrossRef]
- Zheng, H.; Xiao, W.H.; Bennett, G.J. Mitotoxicity and bortezomib-induced chronic painful peripheral neuropathy. Exp. Neurol. 2012, 238, 225–234. [Google Scholar] [CrossRef]
- Jannuzzi, A.T.; Arslan, S.; Yilmaz, A.M.; Sari, G.; Beklen, H.; Méndez, L.; Fedorova, M.; Arga, K.Y.; Karademir Yilmaz, B.; Alpertunga, B. Higher proteotoxic stress rather than mitochondrial damage is involved in higher neurotoxicity of bortezomib compared to carfilzomib. Redox Biol. 2020, 32, 101502. [Google Scholar] [CrossRef]
- Landowski, T.H.; Megli, C.J.; Nullmeyer, K.D.; Lynch, R.M.; Dorr, R.T. Mitochondrial-Mediated Disregulation of Ca2+ Is a Critical Determinant of Velcade (PS-341/Bortezomib) Cytotoxicity in Myeloma Cell Lines. Cancer Res. 2005, 65, 3828–3836. [Google Scholar] [CrossRef]
- Sullivan, P.G.; Dragicevic, N.B.; Deng, J.-H.; Bai, Y.; Dimayuga, E.; Ding, Q.; Chen, Q.; Bruce-Keller, A.J.; Keller, J.N. Proteasome Inhibition Alters Neural Mitochondrial Homeostasis and Mitochondria Turnover. J. Biol. Chem. 2004, 279, 20699–20707. [Google Scholar] [CrossRef]
- Nasu, S.; Misawa, S.; Nakaseko, C.; Shibuya, K.; Isose, S.; Sekiguchi, Y.; Mitsuma, S.; Ohmori, S.; Iwai, Y.; Beppu, M.; et al. Bortezomib-induced neuropathy: Axonal membrane depolarization precedes development of neuropathy. Clin. Neurophysiol. 2014, 125, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.H.; Bennett, G.J. Effects of mitochondrial poisons on the neuropathic pain produced by the chemotherapeutic agents, paclitaxel and oxaliplatin. Pain 2012, 153, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.H.; Zheng, H.; Zheng, F.Y.; Nuydens, R.; Meert, T.F.; Bennett, G.J. Mitochondrial abnormality in sensory, but not motor, axons in paclitaxel-evoked painful peripheral neuropathy in the rat. Neuroscience 2011, 199, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Klimaschewski, L.; Hausott, B.; Ingorokva, S.; Pfaller, K. Constitutively expressed catalytic proteasomal subunits are up-regulated during neuronal differentiation and required for axon initiation, elongation and maintenance. J. Neurochem. 2006, 96, 1708–1717. [Google Scholar] [CrossRef]
- Laser, H.; Mack, T.G.A.; Wagner, D.; Coleman, M.P. Proteasome inhibition arrests neurite outgrowth and causes "dying-back" degeneration in primary culture. J. Neurosci. Res. 2003, 74, 906–916. [Google Scholar] [CrossRef]
- Poruchynsky, M.S.; Sackett, D.L.; Robey, R.W.; Ward, Y.; Annunziata, C.; Fojo, T. Proteasome inhibitors increase tubulin polymerization and stabilization in tissue culture cells: A possible mechanism contributing to peripheral neuropathy and cellular toxicity following proteasome inhibition. Cell Cycle 2008, 7, 940–949. [Google Scholar] [CrossRef]
- Meregalli, C.; Chiorazzi, A.; Carozzi, V.A.; Canta, A.; Sala, B.; Colombo, M.; Oggioni, N.; Ceresa, C.; Foudah, D.; La Russa, F.; et al. Evaluation of tubulin polymerization and chronic inhibition of proteasome as citotoxicity mechanisms in bortezomib-induced peripheral neuropathy. Cell Cycle 2014, 13, 612–621. [Google Scholar] [CrossRef]
- Alé, A.; Bruna, J.; Herrando, M.; Navarro, X.; Udina, E. Toxic Effects of Bortezomib on Primary Sensory Neurons and Schwann Cells of Adult Mice. Neurotox. Res. 2015, 27, 430–440. [Google Scholar] [CrossRef]
- Staff, N.P.; Podratz, J.L.; Grassner, L.; Bader, M.; Paz, J.; Knight, A.M.; Loprinzi, C.L.; Trushina, E.; Windebank, A.J. Bortezomib alters microtubule polymerization and axonal transport in rat dorsal root ganglion neurons. NeuroToxicology 2013, 39, 124–131. [Google Scholar] [CrossRef]
- Karademir, B.; Sari, G.; Jannuzzi, A.T.; Musunuri, S.; Wicher, G.; Grune, T.; Mi, J.; Hacioglu-Bay, H.; Forsberg-Nilsson, K.; Bergquist, J.; et al. Proteomic approach for understanding milder neurotoxicity of Carfilzomib against Bortezomib. Sci. Rep. 2018, 8, 16318. [Google Scholar] [CrossRef] [PubMed]
- Palanca, A.; Casafont, I.; Berciano, M.T.; Lafarga, M. Proteasome inhibition induces DNA damage and reorganizes nuclear architecture and protein synthesis machinery in sensory ganglion neurons. Cell. Mol. Life Sci. 2014, 71, 1961–1975. [Google Scholar] [CrossRef] [PubMed]
- Stockstill, K.; Doyle, T.M.; Yan, X.; Chen, Z.; Janes, K.; Little, J.W.; Braden, K.; Lauro, F.; Giancotti, L.A.; Harada, C.M.; et al. Dysregulation of sphingolipid metabolism contributes to bortezomib-induced neuropathic pain. J. Exp. Med. 2018, 215, 1301–1313. [Google Scholar] [CrossRef] [PubMed]
- Mangiacavalli, S.; Corso, A.; De Amici, M.; Varettoni, M.; Alfonsi, E.; Lozza, A.; Lazzarino, M. Emergent T-helper 2 profile with high interleukin-6 levels correlates with the appearance of bortezomib-induced neuropathic pain: Correspondence. Br. J. Haematol. 2010, 149, 916–918. [Google Scholar] [CrossRef]
- Hung, A.L.; Lim, M.; Doshi, T.L. Targeting cytokines for treatment of neuropathic pain. Scand. J. Pain 2017, 17, 287–293. [Google Scholar] [CrossRef]
- Stemkowski, P.L.; Smith, P.A. Sensory Neurons, Ion Channels, Inflammation and the Onset of Neuropathic Pain. Can. J. Neurol. Sci. 2012, 39, 416–435. [Google Scholar] [CrossRef]
- Myers, R.R.; Shubayev, V.I. The ology of neuropathy: An integrative review of the role of neuroinflammation and TNF-α axonal transport in neuropathic pain. J. Peripher. Nerv. Syst. 2011, 16, 277–286. [Google Scholar] [CrossRef]
- Leung, L.; Cahill, C.M. TNF-α and neuropathic pain—A review. J. Neuroinflamm. 2010, 7, 27. [Google Scholar] [CrossRef]
- Alé, A.; Bruna, J.; Morell, M.; van de Velde, H.; Monbaliu, J.; Navarro, X.; Udina, E. Treatment with anti-TNF alpha protects against the neuropathy induced by the proteasome inhibitor bortezomib in a mouse model. Exp. Neurol. 2014, 253, 165–173. [Google Scholar] [CrossRef]
- Zhang, J.; Su, Y.-M.; Li, D.; Cui, Y.; Huang, Z.-Z.; Wei, J.-Y.; Xue, Z.; Pang, R.-P.; Liu, X.-G.; Xin, W.-J. TNF-α-mediated JNK activation in the dorsal root ganglion neurons contributes to Bortezomib-induced peripheral neuropathy. Brain. Behav. Immun. 2014, 38, 185–191. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Zhang, Y.-P.; Zhang, J.; Zhang, S.-B.; Li, D.; Huang, Z.-Z.; Xin, W.-J. The possible involvement of JNK activation in the spinal dorsal horn in bortezomib-induced allodynia: The role of TNF-α and IL-1β. J. Anesth. 2016, 30, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Deng, T.; Shang, Z.; Wang, D.; Xiao, Y. Blocking TRPA1 and TNF-α Signal Improves Bortezomib-Induced Neuropathic Pain. Cell. Physiol. Biochem. 2018, 51, 2098–2110. [Google Scholar] [CrossRef] [PubMed]
- Chiorazzi, A.; Canta, A.; Meregalli, C.; Carozzi, V.; Sala, B.; Oggioni, N.; Monbaliu, J. Antibody Against Tumor Necrosis Factor-α Reduces Bortezomib-induced Allodynia in a Rat Model. Anticancer Res. 2013, 33, 5453–5459. [Google Scholar] [PubMed]
- Hideshima, T.; Richardson, P.; Chauhan, D.; Palombella, V.J.; Elliott, P.J.; Adams, J.; Anderson, K.C. The Proteasome Inhibitor PS-341 Inhibits Growth, Induces Apoptosis, and Overcomes Drug Resistance in Human Multiple Myeloma Cells. Cancer Res. 2001, 61, 3071–3076. [Google Scholar]
- Karin, M. Nuclear factor-κB in cancer development and progression. Nature 2006, 441, 431–436. [Google Scholar] [CrossRef]
- Alé, A.; Bruna, J.; Calls, A.; Karamita, M.; Haralambous, S.; Probert, L.; Navarro, X.; Udina, E. Inhibition of the neuronal NFκB pathway attenuates bortezomib-induced neuropathy in a mouse model. NeuroToxicology 2016, 55, 58–64. [Google Scholar] [CrossRef]
- Cole, D.C.; Frishman, W.H. Cardiovascular Complications of Proteasome Inhibitors Used in Multiple Myeloma. Cardiol. Rev. 2018, 26, 122–129. [Google Scholar] [CrossRef]
- Kistler, K.D.; Kalman, J.; Sahni, G.; Murphy, B.; Werther, W.; Rajangam, K.; Chari, A. Incidence and Risk of Cardiac Events in Patients With Previously Treated Multiple Myeloma Versus Matched Patients Without Multiple Myeloma: An Observational, Retrospective, Cohort Study. Clin. Lymphoma Myeloma Leuk. 2017, 17, 89–96.e3. [Google Scholar] [CrossRef]
- Orciuolo, E.; Buda, G.; Cecconi, N.; Galimberti, S.; Versari, D.; Cervetti, G.; Salvetti, A.; Petrini, M. Unexpected cardiotoxicity in haematological bortezomib treated patients. Br. J. Haematol. 2007, 138, 396–397. [Google Scholar] [CrossRef]
- Gupta, A.; Pandey, A.; Sethi, S. Bortezomib-Induced Congestive Cardiac Failure in a Patient with Multiple Myeloma. Cardiovasc. Toxicol. 2012, 12, 184–187. [Google Scholar] [CrossRef]
- Bockorny, M.; Chakravarty, S.; Schulman, P.; Bockorny, B.; Bona, R. Severe Heart Failure after Bortezomib Treatment in a Patient with Multiple Myeloma: A Case Report and Review of the Literature. Acta Haematol. 2012, 128, 244–247. [Google Scholar] [CrossRef] [PubMed]
- Meseeha, M.G.; Kolade, V.O.; Attia, M.N. Partially reversible bortezomib-induced cardiotoxicity: An unusual cause of acute cardiomyopathy. J. Community Hosp. Intern. Med. Perspect. 2015, 5, 28982. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Voortman, J.; Giaccone, G. Severe reversible cardiac failure after bortezomib treatment combined with chemotherapy in a non-small cell lung cancer patient: A case report. BMC Cancer 2006, 6, 129. [Google Scholar] [CrossRef] [PubMed]
- Honton, B.; Despas, F.; Dumonteil, N.; Rouvellat, C.; Roussel, M.; Carrie, D.; Galinier, M.; Montastruc, J.L.; Pathak, A. Bortezomib and heart failure: Case-report and review of the French Pharmacovigilance database. Fundam. Clin. Pharmacol. 2014, 28, 349–352. [Google Scholar] [CrossRef]
- Hacihanefioglu, A.; Tarkun, P.; Gonullu, E. Acute severe cardiac failure in a myeloma patient due to proteasome inhibitor bortezomib. Int. J. Hematol. 2008, 88, 219–222. [Google Scholar] [CrossRef]
- Jerkins, J. Bortezomib-induced Severe Congestive Heart Failure. Cardiol. Res. 2010. [Google Scholar] [CrossRef]
- Chakraborty, R.; Mukkamalla, S.K.R.; Calderon, N. Bortezomib induced reversible left ventricular systolic dysfunction: A case report and review of literature. BJMP 2013, 6, a631. [Google Scholar]
- Foley, P.W.; Hamilton, M.S.; Leyva, F. Myocardial scarring following chemotherapy for multiple myeloma detected using late gadolinium hyperenhancement cardiovascular magnetic resonance. J. Cardiovasc. Med. 2010, 11, 386–388. [Google Scholar] [CrossRef]
- Subedi, A.; Sharma, L.R.; Shah, B.K. Bortezomib-induced acute congestive heart failure: A case report and review of literature. Ann. Hematol. 2014, 93, 1797–1799. [Google Scholar] [CrossRef]
- Dasanu, C.A. Complete heart block secondary to bortezomib use in multiple myeloma. J. Oncol. Pharm. Pract. 2011, 17, 282–284. [Google Scholar] [CrossRef]
- Lee, W.-S.; Kim, D.-H.; Shin, S.-H.; Woo, S.-I.; Kwan, J.; Park, K.-S.; Park, S.-D.; Yi, H.-G.; Jeon, S.-H. Complete Atrioventricular Block Secondary to Bortezomib Use in Multiple Myeloma. Yonsei Med. J. 2011, 52, 196. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Diwadkar, S.; Patel, A.A.; Fradley, M.G. Bortezomib-Induced Complete Heart Block and Myocardial Scar: The Potential Role of Cardiac Biomarkers in Monitoring Cardiotoxicity. Case Rep. Cardiol. 2016, 2016, 3456287. [Google Scholar] [CrossRef]
- Berenson, J.R.; Matous, J.; Swift, R.A.; Mapes, R.; Morrison, B.; Yeh, H.S. A Phase I/II Study of Arsenic Trioxide/Bortezomib/Ascorbic Acid Combination Therapy for the Treatment of Relapsed or Refractory Multiple Myeloma. Clin. Cancer Res. 2007, 13, 1762–1768. [Google Scholar] [CrossRef] [PubMed]
- Takamatsu, H.; Yamashita, T.; Kotani, T.; Sawazaki, A.; Okumura, H.; Nakao, S. Ischemic heart disease associated with bortezomib treatment combined with dexamethasone in a patient with multiple myeloma. Int. J. Hematol. 2010, 91, 903–906. [Google Scholar] [CrossRef] [PubMed]
- Burkhart, T.; Keith, M.C.L.; Lenneman, C.A.G.; Fernando, R.R. Bortezomib-Induced Cardiac Tamponade in a 49-Year-Old Man. Tex. Heart Inst. J. 2018, 45, 260–263. [Google Scholar] [CrossRef]
- Xiao, Y.; Yin, J.; Wei, J.; Shang, Z. Incidence and Risk of Cardiotoxicity Associated with Bortezomib in the Treatment of Cancer: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e87671. [Google Scholar] [CrossRef]
- Laubach, J.P.; Moslehi, J.J.; Francis, S.A.; San Miguel, J.F.; Sonneveld, P.; Orlowski, R.Z.; Moreau, P.; Rosiñol, L.; Faber, E.A.; Voorhees, P.; et al. A retrospective analysis of 3954 patients in phase 2/3 trials of bortezomib for the treatment of multiple myeloma: Towards providing a benchmark for the cardiac safety profile of proteasome inhibition in multiple myeloma. Br. J. Haematol. 2017, 178, 547–560. [Google Scholar] [CrossRef]
- Cornell, R.F.; Ky, B.; Weiss, B.M.; Dahm, C.N.; Gupta, D.K.; Du, L.; Carver, J.R.; Cohen, A.D.; Engelhardt, B.G.; Garfall, A.L.; et al. Prospective Study of Cardiac Events During Proteasome Inhibitor Therapy for Relapsed Multiple Myeloma. J. Clin. Oncol. 2019, 37, 1946–1955. [Google Scholar] [CrossRef]
- Koulaouzidis, G.; Lyon, A.R. Proteasome Inhibitors as a Potential Cause of Heart Failure. Heart Fail. Clin. 2017, 13, 289–295. [Google Scholar] [CrossRef]
- Shukla, S.K.; Rafiq, K. Proteasome biology and therapeutics in cardiac diseases. Transl. Res. 2019, 205, 64–76. [Google Scholar] [CrossRef]
- Portbury, A.L.; Ronnebaum, S.M.; Zungu, M.; Patterson, C.; Willis, M.S. Back to your heart: Ubiquitin proteasome system-regulated signal transduction. J. Mol. Cell. Cardiol. 2012, 52, 526–537. [Google Scholar] [CrossRef] [PubMed]
- Gilda, J.E.; Gomes, A.V. Proteasome dysfunction in cardiomyopathies: Proteasome dysfunction in cardiomyopathies. J. Physiol. 2017, 595, 4051–4071. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-F.; Wang, X. The role of the proteasome in heart disease. Biochim. Biophys. Acta BBA Gene Regul. Mech. 2011, 1809, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Hasinoff, B.B.; Patel, D.; Wu, X. Molecular Mechanisms of the Cardiotoxicity of the Proteasomal-Targeted Drugs Bortezomib and Carfilzomib. Cardiovasc. Toxicol. 2017, 17, 237–250. [Google Scholar] [CrossRef]
- Pokorna, Z.; Jirkovsky, E.; Hlavackova, M.; Jansova, H.; Jirkovska, A.; Lencova-Popelova, O.; Brazdova, P.; Kubes, J.; Sotakova-Kasparova, D.; Mazurova, Y.; et al. In vitro and in vivo investigation of cardiotoxicity associated with anticancer proteasome inhibitors and their combination with anthracycline. Clin. Sci. 2019, 133, 1827–1844. [Google Scholar] [CrossRef]
- Nowis, D.; Mączewski, M.; Mackiewicz, U.; Kujawa, M.; Ratajska, A.; Wieckowski, M.R.; Wilczyński, G.M.; Malinowska, M.; Bil, J.; Salwa, P.; et al. Cardiotoxicity of the Anticancer Therapeutic Agent Bortezomib. Am. J. Pathol. 2010, 176, 2658–2668. [Google Scholar] [CrossRef]
- Tang, M.; Li, J.; Huang, W.; Su, H.; Liang, Q.; Tian, Z.; Horak, K.M.; Molkentin, J.D.; Wang, X. Proteasome functional insufficiency activates the calcineurin–NFAT pathway in cardiomyocytes and promotes maladaptive remodelling of stressed mouse hearts. Cardiovasc. Res. 2010, 88, 424–433. [Google Scholar] [CrossRef]
- Carrier, L. Too much of a good thing is bad: Proteasome inhibition induces stressed hearts to fail. Cardiovasc. Res. 2010, 88, 389–390. [Google Scholar] [CrossRef]
- Bonuccelli, G.; Sotgia, F.; Capozza, F.; Gazzerro, E.; Minetti, C.; Lisanti, M.P. Localized Treatment with a Novel FDA-Approved Proteasome Inhibitor Blocks the Degradation of Dystrophin and Dystrophin-Associated Proteins in mdx Mice. Cell Cycle 2007, 6, 1242–1248. [Google Scholar] [CrossRef]
- Herrmann, J.; Saguner, A.M.; Versari, D.; Peterson, T.E.; Chade, A.; Olson, M.; Lerman, L.O.; Lerman, A. Chronic Proteasome Inhibition Contributes to Coronary Atherosclerosis. Circ. Res. 2007, 101, 865–874. [Google Scholar] [CrossRef]
- Kisselev, A.F.; Goldberg, A.L. Proteasome inhibitors: From research tools to drug candidates. Chem. Biol. 2001, 8, 739–758. [Google Scholar] [CrossRef]
- Herrmann, J.; Wohlert, C.; Saguner, A.M.; Flores, A.; Nesbitt, L.L.; Chade, A.; Lerman, L.O.; Lerman, A. Primary proteasome inhibition results in cardiac dysfunction. Eur. J. Heart Fail. 2013, 15, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Gavazzoni, M.; Vizzardi, E.; Gorga, E.; Bonadei, I.; Rossi, L.; Belotti, A.; Rossi, G.; Ribolla, R.; Metra, M.; Raddino, R. Mechanism of cardiovascular toxicity by proteasome inhibitors: New paradigm derived from clinical and pre-clinical evidence. Eur. J. Pharmacol. 2018, 828, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Mårtensson, C.U.; Priesnitz, C.; Song, J.; Ellenrieder, L.; Doan, K.N.; Boos, F.; Floerchinger, A.; Zufall, N.; Oeljeklaus, S.; Warscheid, B.; et al. Mitochondrial protein translocation-associated degradation. Nature 2019, 569, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Yui, J.C.; Van Keer, J.; Weiss, B.M.; Waxman, A.J.; Palmer, M.B.; D’Agati, V.D.; Kastritis, E.; Dimopoulos, M.A.; Vij, R.; Bansal, D.; et al. Proteasome inhibitor associated thrombotic microangiopathy: Proteasome inhibitor associated TMA. Am. J. Hematol. 2016, 91, E348–E352. [Google Scholar] [CrossRef] [PubMed]
- Roccaro, A.M.; Hideshima, T.; Raje, N.; Kumar, S.; Ishitsuka, K.; Yasui, H.; Shiraishi, N.; Ribatti, D.; Nico, B.; Vacca, A.; et al. Bortezomib Mediates Antiangiogenesis in Multiple Myeloma via Direct and Indirect Effects on Endothelial Cells. Cancer Res. 2006, 66, 184–191. [Google Scholar] [CrossRef]
- Tamura, D.; Arao, T.; Tanaka, K.; Kaneda, H.; Matsumoto, K.; Kudo, K.; Aomatsu, K.; Fujita, Y.; Watanabe, T.; Saijo, N.; et al. Bortezomib potentially inhibits cellular growth of vascular endothelial cells through suppression of G2/M transition. Cancer Sci. 2010, 101, 1403–1408. [Google Scholar] [CrossRef]
- Wei, Q.; Xia, Y. Proteasome Inhibition Down-regulates Endothelial Nitric-oxide Synthase Phosphorylation and Function. J. Biol. Chem. 2006, 281, 21652–21659. [Google Scholar] [CrossRef]
- Hu, X.-S.; Du, C.-Q.; Yang, L.; Yao, X.-Y.; Hu, S.-J. Proteasome inhibitor MG132 suppresses number and function of endothelial progenitor cells: Involvement of nitric oxide synthase inhibition. Int. J. Mol. Med. 2010, 25. [Google Scholar] [CrossRef]
- Herrmann, J.; Edwards, W.D.; Holmes, D.R.; Shogren, K.L.; Lerman, L.O.; Ciechanover, A.; Lerman, A. Increased ubiquitin immunoreactivity in unstable atherosclerotic plaques associated with acute coronary syndromes. J. Am. Coll. Cardiol. 2002, 40, 1919–1927. [Google Scholar] [CrossRef]
- Versari, D.; Herrmann, J.; Gössl, M.; Mannheim, D.; Sattler, K.; Meyer, F.B.; Lerman, L.O.; Lerman, A. Dysregulation of the Ubiquitin-Proteasome System in Human Carotid Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2132–2139. [Google Scholar] [CrossRef] [PubMed]
- Muchtar, E.; Derudas, D.; Mauermann, M.; Liewluck, T.; Dispenzieri, A.; Kumar, S.K.; Dingli, D.; Lacy, M.Q.; Buadi, F.K.; Hayman, S.R.; et al. Systemic Immunoglobulin Light Chain Amyloidosis–Associated Myopathy: Presentation, Diagnostic Pitfalls, and Outcome. Mayo Clin. Proc. 2016, 91, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Jagannath, S.; Barlogie, B.; Berenson, J.; Siegel, D.; Irwin, D.; Richardson, P.G.; Niesvizky, R.; Alexanian, R.; Limentani, S.A.; Alsina, M.; et al. A phase 2 study of two doses of bortezomib in relapsed or refractory myeloma. Br. J. Haematol. 2004, 127, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Guglielmi, V.; Nowis, D.; Tinelli, M.; Malatesta, M.; Paoli, L.; Marini, M.; Manganotti, P.; Sadowski, R.; Wilczynski, G.M.; Meneghini, V.; et al. Bortezomib-Induced Muscle Toxicity in Multiple Myeloma. J. Neuropathol. Exp. Neurol. 2017, 76, 620–630. [Google Scholar] [CrossRef] [PubMed]
- Arastu-Kapur, S.; Anderl, J.L.; Kraus, M.; Parlati, F.; Shenk, K.D.; Lee, S.J.; Muchamuel, T.; Bennett, M.K.; Driessen, C.; Ball, A.J.; et al. Nonproteasomal Targets of the Proteasome Inhibitors Bortezomib and Carfilzomib: A Link to Clinical Adverse Events. Clin. Cancer Res. 2011, 17, 2734–2743. [Google Scholar] [CrossRef]
- Zammit, P.S. Function of the myogenic regulatory factors Myf5, MyoD, Myogenin and MRF4 in skeletal muscle, satellite cells and regenerative myogenesis. Semin. Cell Dev. Biol. 2017, 72, 19–32. [Google Scholar] [CrossRef]
- Xing, S.S.; Shen, C.C.; Godard, M.P.; Wang, J.J.; Yue, Y.Y.; Yang, S.T.; Zhao, Q.; Zhang, S.B.; Wang, T.X.; Yang, X.L.; et al. Bortezomib inhibits C2C12 growth by inducing cell cycle arrest and apoptosis. Biochem. Biophys. Res. Commun. 2014, 445, 375–380. [Google Scholar] [CrossRef]
- Jiacheng, M.; Kavelaars, A.; Dougherty, P.M.; Heijnen, C.J. Beyond Symptomatic Relief for Chemotherapy-Induced Peripheral Neuopathy:targeting the source. Cancer 2018, 124, 2289–2298. [Google Scholar] [CrossRef]
- Maschio, M.; Zarabla, A.; Maialetti, A.; Marchesi, F.; Giannarelli, D.; Gumenyuk, S.; Pisani, F.; Renzi, D.; Galiè, E.; Mengarelli, A. Prevention of Bortezomib-Related Peripheral Neuropathy with Docosahexaenoic acid and α-Lipoic Acid in Patients with Multiple Myeloma: Preliminary Data. Integr. Cancer Ther. 2018, 17, 1115–1124. [Google Scholar] [CrossRef]
- Callander, N.; Markovina, S.; Eickhoff, J.; Hutson, P.; Campell, T.; Hematti, P.; Go, R.; Hegeman, R.; Longo, W.; Williams, E.; et al. Acetyl-L-carnitine (ALCAR) for the prevention of chemotherapy-induced peripheral neuropathy in patients with relapsed or refractory multiple myeloma treated with bortezomib, doxorubin and low-dose dexamethasone: A study from the Wisconsin Ocology Network. Cancer Chemother. Pharmacol. 2014, 74, 875–882. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pancheri, E.; Guglielmi, V.; Wilczynski, G.M.; Malatesta, M.; Tonin, P.; Tomelleri, G.; Nowis, D.; Vattemi, G. Non-Hematologic Toxicity of Bortezomib in Multiple Myeloma: The Neuromuscular and Cardiovascular Adverse Effects. Cancers 2020, 12, 2540. https://doi.org/10.3390/cancers12092540
Pancheri E, Guglielmi V, Wilczynski GM, Malatesta M, Tonin P, Tomelleri G, Nowis D, Vattemi G. Non-Hematologic Toxicity of Bortezomib in Multiple Myeloma: The Neuromuscular and Cardiovascular Adverse Effects. Cancers. 2020; 12(9):2540. https://doi.org/10.3390/cancers12092540
Chicago/Turabian StylePancheri, Elia, Valeria Guglielmi, Grzegorz M. Wilczynski, Manuela Malatesta, Paola Tonin, Giuliano Tomelleri, Dominika Nowis, and Gaetano Vattemi. 2020. "Non-Hematologic Toxicity of Bortezomib in Multiple Myeloma: The Neuromuscular and Cardiovascular Adverse Effects" Cancers 12, no. 9: 2540. https://doi.org/10.3390/cancers12092540
APA StylePancheri, E., Guglielmi, V., Wilczynski, G. M., Malatesta, M., Tonin, P., Tomelleri, G., Nowis, D., & Vattemi, G. (2020). Non-Hematologic Toxicity of Bortezomib in Multiple Myeloma: The Neuromuscular and Cardiovascular Adverse Effects. Cancers, 12(9), 2540. https://doi.org/10.3390/cancers12092540





