Diagnosis and Treatment of Bone Metastases in Breast Cancer: Radiotherapy, Local Approach and Systemic Therapy in a Guide for Clinicians
Abstract
1. Introduction
2. Diagnostic Imaging for Bone Metastasis from Breast Cancer
2.1. Morphologic Imaging
2.2. Functional Imaging
2.3. Diagnostic Imaging for Treatment Planning of Radiotherapy
2.4. Biopsy on Bone Metastasis: When Imaging Is Not Enough
3. Radiotherapy Treatments Options and New Drugs
4. Co-Adjuvant Systemic Therapies and Focal Alternatives to Radiotherapy
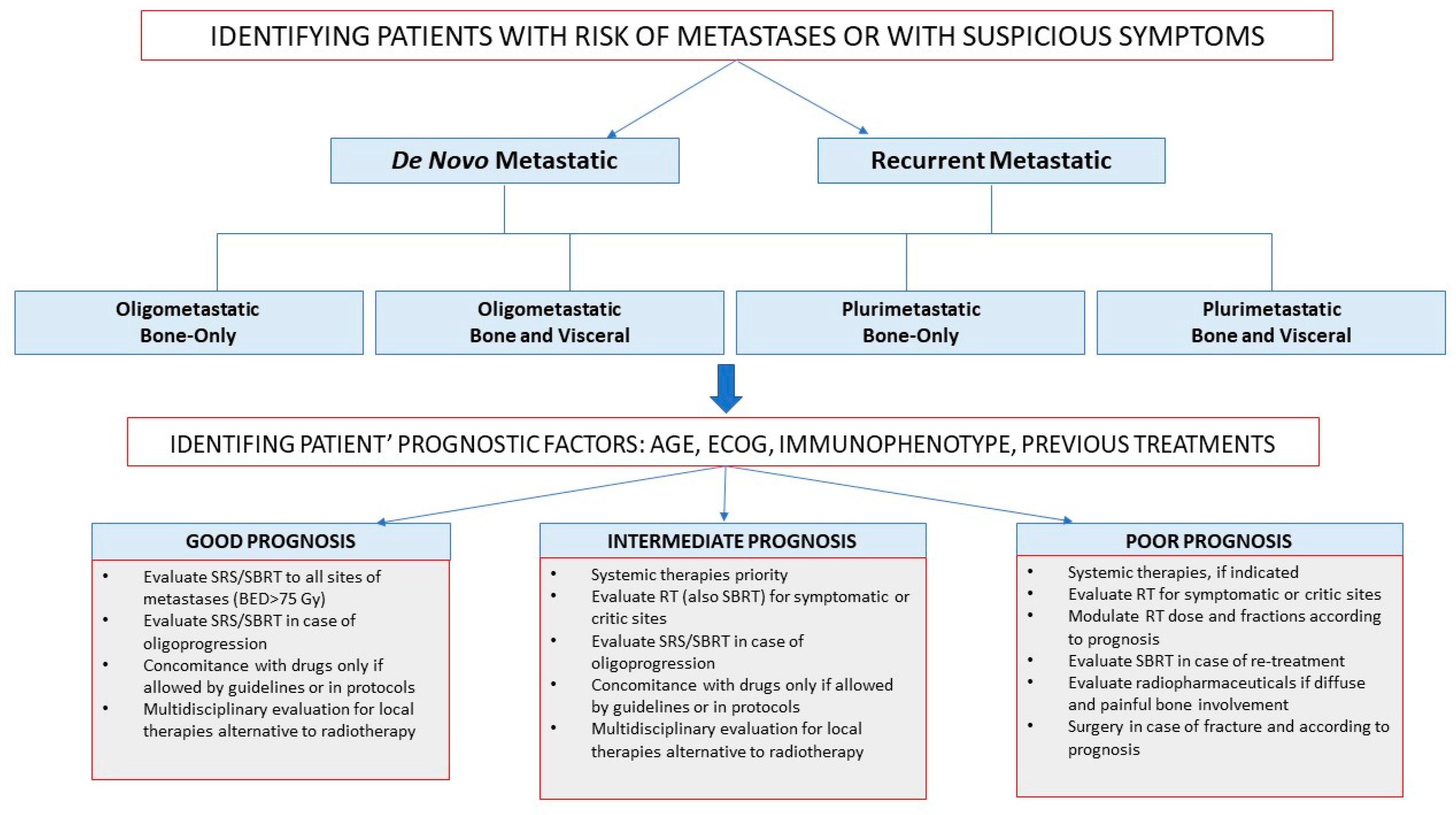
5. Implication for Clinicians
- De novo or recurrent metastatic breast cancer: based on time of metastases presentation;
- Oligometastatic or plurimetastatic breast cancer: based on the presence of five metastatic sites or more;
- Bone-only or visceral metastatic breast cancer: based on parenchymal involvement.
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Andre, F.; Slimane, K.; Bachelot, T.; Dunant, A.; Namer, M.; Barrelier, A.; Kabbaj, O.; Spano, J.F.; Marsiglia, H.; Rouzier, R. Breast Cancer with Synchronous Metastases: Trends in Survival during a 14-Year Period. JCO 2004, 22, 3302–3308. [Google Scholar] [CrossRef]
- Sledge, G.W. Curing Metastatic Breast Cancer. JOP 2016, 12, 6–10. [Google Scholar] [CrossRef]
- American Cancer Society. Survival Rates for Breast Cancer. Available online: https://www.cancer.org/cancer/breast-cancer/understanding-a-breast-cancer-diagnosis/breast-cancer-survival-rates.html (accessed on 20 April 2020).
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Primers 2019, 5, 66. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, R.K.; Wade, S.W.; Reich, A.; Pirolli, M.; Liede, A.; Lyman, G.H. Incidence of bone metastases in patients with solid tumors: Analysis of oncology electronic medical records in the United States. BMC Cancer 2018, 18, 44. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, J.; Zhu, S.; Wu, J.; Chen, C.; Liu, Q.; Wei, W.; Zhang, Y.; Sun, S. Breast cancer subtypes predict the preferential site of distant metastases: A SEER based study. Oncotarget 2017, 8, 27990. [Google Scholar] [CrossRef]
- Molnár, I.A.; Molnár, B.Á.; Vízkeleti, L.; Fekete, K.; Tamás, J.; Deák, P.; Szundi, C.; Székely, B.; Moldvay, J.; Vári-Kakas, S. Breast carcinoma subtypes show different patterns of metastatic behavior. Virchows Arch. 2017, 470, 275–283. [Google Scholar] [CrossRef]
- Guckenberger, M.; Lievens, Y.; Bouma, A.B.; Collette, L.; Dekker, A.; deSouza, N.M.; Dingemans, A.C.; Fournier, B.; Hurkmans, C.; Lecouvet, F.E. Characterisation and classification of oligometastatic disease: A European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. Lancet Oncol. 2020, 21, e18–e28. [Google Scholar] [CrossRef]
- Eisenhauer, E.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M. New Response Evaluation Criteria in Solid Tumours: Revised RECIST Guideline (Version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Mundy, G. Mechanisms of Bone Metastasis. Cancer 1997, 80, 1546–1556. [Google Scholar] [CrossRef]
- D’Oronzo, S.; Coleman, R.; Brown, J.; Silvestris, F. Metastatic bone disease: Pathogenesis and therapeutic options. J. Bone Oncol. 2019, 15, 100205. [Google Scholar] [CrossRef] [PubMed]
- Vakaet, L.A.L.; Boterberg, T. Pain control by ionizing radiation of bone metastasis. Int. J. Dev. Biol. 2004, 48, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Manfrida, S.; Masiello, V.; Cellini, F.; Adducci, E.; Polidori, L.; Longo, S.; Cannelli, G.; Balducci, M.; Rossi, M.; Valentini, V. IMproved MAnagement (IM-MA study) in cancer-related pain: The value of a joint approach by an integrated team of radiotherapist and anesthetist. Support Care Cancer 2019, 27, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Turner, N.C.; Bondarenko, I.; Ro, J.; Im, S.A.; Masuda, N.; Colleoni, M.; DeMichele, A.; Loi, S.; Verma, S. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): Final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 2016, 17, 425–439. [Google Scholar] [CrossRef] [PubMed]
- Ghezzi, P.; Magnanini, S.; Rinaldini, M.; Berardi, F.; Di Biagio, G.; Testare, F.; Tavoni, N.; Schittulli, F.; D’Amico, C.; Pedicini, T.; et al. Impact of Follow-up Testing on Survival and Health-Related Quality of Life in Breast Cancer Patients A Multicenter Randomized Controlled Trial. JAMA 1994, 271, 1587–1592. [Google Scholar] [CrossRef] [PubMed]
- Rosselli Del Turco, M.; Palli, D.; Cariddi, A.; Ciatto, S.; Pacini, P.; Distante, V. Intensive Diagnostic Follow-up After Treatment of Primary Breast Cancer. A Randomized Trial. JAMA 1994, 271, 1593–1597. [Google Scholar] [CrossRef]
- Milano, M.T.; Katz, A.W.; Zhang, H.; Huggins, C.F.; Aujla, K.S.; Okunieff, P. Oligometastatic breast cancer treated with hypofractionated stereotactic radiotherapy: Some patients survive longer than a decade. Radiother. Oncol. 2019, 131, 45–51. [Google Scholar] [CrossRef]
- Kobayashi, T.; Ichiba, T.; Sakuyama, T.; Arakawa, Y.; Nagasaki, E.; Aiba, K.; Nogi, H.; Kawase, K.; Takeyama, H.; Toriumi, Y. Possible clinical cure of metastatic breast cancer: Lessons from our 30-year experience with oligometastatic breast cancer patients and literature review. Breast Cancer 2012, 19, 218–237. [Google Scholar] [CrossRef]
- Mundy, G. Metastasis to bone: Causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2002, 2, 584–593. [Google Scholar] [CrossRef]
- Chow, E.; Wu, J.; Hoskin, P.; Coia, L.; Bentzen, S.; Blitzer, P. International consensus on palliative radiotherapy endpoints for future clinical trials in bone metastases. Radiother. Oncol. 2002, 64, 275–280. [Google Scholar] [CrossRef]
- Chow, E.; Hoskin, P.; Mitera, G.; Zeng, L.; Lutz, S.; Roos, D.; Hahn, C.; van der Linden, Y.; Hartsell, W.; Kumar, E. Update of the International Consensus on Palliative Radiotherapy Endpoints for Future Clinical Trials in Bone Metastases. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1730–1737. [Google Scholar] [CrossRef]
- Zeng, K.L.; Tseng, C.-L.; Soliman, H.; Weiss, Y.; Sahgal, A.; Myrehaug, S. Stereotactic Body Radiotherapy (SBRT) for Oligometastatic Spine Metastases: An Overview. Front. Oncol. 2019, 9, 337. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.S.; Erban, J.K. Timing of Metastasis in Breast Cancer. N. Engl. J. Med 2017, 376, 2486–2488. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, H.; Obradović, M.M.S.; Hoffmann, M.; Harper, K.L.; Sosa, M.S.; Werner-Klein, M.; Nanduri, L.K.; Werno, C.; Ehrl, C.; Maneck, M. Early dissemination seeds metastasis in breast cancer. Nature 2016, 540, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Harper, K.; Sosa, M.S.; Entenberg, D.; Hosseini, H.; Cheung, J.F.; Nobre, R.; Avivar-Valderas, A.; Nagi, C.; Girnius, N.; Davis, R.J. Mechanism of early dissemination and metastasis in Her2+ mammary cancer. Nature 2016, 540, 588–592. [Google Scholar] [CrossRef]
- Ghanem, N.; Uhl, M.; Brink, I.; Schäfer, O.; Kelly, T.; Moser, E.; Langer, M. Diagnostic value of MRI in comparison to scintigraphy, PET, MS-CT and PET/CT for the detection of metastases of bone. Eur. J. Radiol. 2005, 55, 41–55. [Google Scholar] [CrossRef]
- O’Sullivan, G.J. Imaging of bone metastasis: An update. WJR 2015, 7, 202. [Google Scholar] [CrossRef]
- Di Gioia, D.; Stieber, P.; Schmidt, G.P.; Nagel, D.; Heinemann, V.; Baur-Melnyk, A. Early detection of metastatic disease in asymptomatic breast cancer patients with whole-body imaging and defined tumour marker increase. Br. J. Cancer 2015, 112, 809–818. [Google Scholar] [CrossRef]
- Cook, G.; Goh, V. Molecular imaging of bone metastases and their response to therapy. J. Nucl. Med. 2020, 61, 799–806. [Google Scholar] [CrossRef]
- Stecco, A.; Trisoglio, A.; Soligo, E.; Berardo, S.; Sukhovei, L.; Carriero, A. Whole-Body MRI with Diffusion-Weighted Imaging in Bone Metastases: A Narrative Review. Diagnostics 2018, 8, 45. [Google Scholar] [CrossRef]
- Costelloe, C.M.; Rohren, E.M.; Madewell, J.E.; Hamaoka, T.; Theriault, R.L.; Yu, T.K.; Lewis, V.O.; Ma, J.; Stafford, R.J.; Tari, A. Imaging bone metastases in breast cancer: Techniques and recommendations for diagnosis. Lancet Oncol. 2009, 10, 9. [Google Scholar] [CrossRef]
- Taralli, S.; Lorusso, M.; Scolozzi, V.; Masiello, V.; Marazzi, F.; Calcagni, M.L. Response evaluation with 18F-FDG PET/CT in metastatic breast cancer patients treated with Palbociclib: First experience in clinical practice. Ann. Nucl. Med. 2019, 33, 193–200. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) Breast Cancer, version 2; National Comprehensive Cancer Network: Plymouth Meeting, PA, USA, 2020. [Google Scholar]
- Yang, H.-L.; Liu, T.; Wang, X.-M.; Xu, Y.; Deng, S.-M. Diagnosis of bone metastases: A meta-analysis comparing 18FDG PET, CT, MRI and bone scintigraphy. Eur. Radiol. 2011, 21, 2604–2617. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.L.; Kransdorf, M.J.; Schweitzer, M.E.; Murphey, M.D.; Babb, J.S. Can Sarcoidosis and Metastatic Bone Lesions Be Reliably Differentiated on Routine MRI? Am. J. Roentgenol. 2012, 198, 1387–1393. [Google Scholar] [CrossRef] [PubMed]
- Soussan, M.; Augier, A.; Brillet, P.-Y.; Weinmann, P.; Valeyre, D. Functional Imaging in Extrapulmonary Sarcoidosis: FDG-PET/CT and MR Features. Clin. Nucl. Med. 2014, 39, e146–e159. [Google Scholar] [CrossRef] [PubMed]
- Foerster, R.; Cho, B.C.J.; Fahim, D.K.; Gerszten, P.C.; Flickinger, J.C.; Grills, I.S.; Jawad, M.S.; Kersh, C.R.; Létourneau, D.; Mantel, F. Histopathological Findings After Reirradiation Compared to First Irradiation of Spinal Bone Metastases with Stereotactic Body Radiotherapy: A Cohort Study. Neurosurgery 2019, 84, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Alongi, F.; Arcangeli, S.; Filippi, A.R.; Ricardi, U.; Scorsetti, M. Review and Uses of Stereotactic Body Radiation Therapy for Oligometastases. Oncologist 2012, 17, 1100–1107. [Google Scholar] [CrossRef]
- Costa, S.; Reagan, M.R. Therapeutic Irradiation: Consequences for Bone and Bone Marrow Adipose Tissue. Front. Endocrinol. 2019, 10, 587. [Google Scholar] [CrossRef]
- Goblirsch, M.; Mathews, W.; Lynch, C.; Alaei, P.; Gerbi, B.J.; Mantyh, P.W.; Clohisy, D.R. Radiation Treatment Decreases Bone Cancer Pain, Osteolysis and Tumor Size. Radiat. Res. 2004, 161, 228–234. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Z.; Wu, A.; Nie, J.; Pei, H.; Hu, W.; Wang, B.; Shang, P.; Li, B.; Zhou, G. Differences in responses to X-ray exposure between osteoclast and osteoblast cells. J. Radiat. Res. 2017, 58, 791–802. [Google Scholar] [CrossRef]
- Steverink, J.; Willems, S.M.; Philippens, M.E.P.; Kasperts, N.; Eppinga, W.S.C.; Versteeg, A.L.; van der Velden, J.M.; Faruqi, S.; Sahgal, A. Early Tissue Effects of Stereotactic Body Radiation Therapy for Spinal Metastases. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 1254–1258. [Google Scholar] [CrossRef]
- Arriagada, R.; Mouriesse, H.; Sarrazin, D.; Clark, R.; Deboer, G. Analysis of tumor parameters, tumor dose and local control: The experience of the Gustave-Roussy Institute and the Princess Margaret Hospital. Int. J. Radiat. Oncol. Biol. Phys. 1985, 11, 1751–1757. [Google Scholar] [CrossRef]
- Gerszten, P.C.; Burton, S.A.; Welch, W.C.; Brufsky, A.M.; Lembersky, B.C.; Ozhasoglu, C.; Vogel, W.J. Single-fraction radiosurgery for the treatment of spinal breast metastases. Cancer 2005, 104, 2244–2254. [Google Scholar] [CrossRef] [PubMed]
- Kwapisz, D. Oligometastatic breast cancer. Breast Cancer 2019, 26, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Chow, E.; Harris, K.; Fan, G.; Tsao, M.; Sze, W.M. Palliative Radiotherapy Trials for Bone Metastases: A Systematic Review. JCO 2007, 25, 1423–1436. [Google Scholar] [CrossRef]
- Rades, D.; Fehlauer, F.; Stalpers, L.J.; Wildfang, I.; Zschenker, O.; Schild, S.E.; Schmoll, H.J.; Karstens, J.H.; Alberti, W. A prospective evaluation of two radiotherapy schedules with 10 versus 20 fractions for the treatment of metastatic spinal cord compression: Final results of a multicenter study. Cancer 2004, 101, 2687–2692. [Google Scholar] [CrossRef]
- De Vin, T.; Engels, B.; Gevaert, T.; Storme, G.; De Ridder, M. Stereotactic radiotherapy for oligometastatic cancer: A prognostic model for survival. Ann. Oncol. 2014, 25, 467–471. [Google Scholar] [CrossRef]
- Tree, A.C.; Khoo, V.S.; Eeles, R.A.; Ahmed, M.; Dearnaley, D.P.; Hawkins, M.A.; Huddart, R.A.; Nutting, C.M.; Ostler, P.J.; van As, N.J. Stereotactic body radiotherapy for oligometastases. Lancet Oncol. 2013, 14, e28–e37. [Google Scholar] [CrossRef]
- Cellini, F.; Manfrida, S.; Deodato, F.; Cilla, S.; Maranzano, E.; Pergolizzi, S.; Arcidiacono, F.; Di Franco, R.; Pastore, F.; Muto, M. Pain REduction with bone metastases STereotactic radiotherapy (PREST): A phase III randomized multicentric trial. Trials 2019, 20, 609. [Google Scholar] [CrossRef]
- Yoo, G.S.; Yu, J.I.; Park, W.; Huh, S.J.; Choi, D.H. Prognostic factors in breast cancer with extracranial oligometastases and the appropriate role of radiation therapy. Radiat. Oncol. J. 2015, 33, 301. [Google Scholar] [CrossRef]
- Trovo, M.; Furlan, C.; Polesel, J.; Fiorica, F.; Arcangeli, S.; Giaj-Levra, N.; Alongi, F.; Del Conte, A.; Militello, L.; Muraro, E. Radical radiation therapy for oligometastatic breast cancer: Results of a prospective phase II trial. Radiother. Oncol. 2018, 126, 177–180. [Google Scholar] [CrossRef]
- Kam, T.Y.; Chan, O.S.H.; Hung, A.W.M.; Yeung, R.M.W. Utilization of stereotactic ablative radiotherapy in oligometastatic & oligoprogressive skeletal metastases: Results and pattern of failure. Asia-Pac. J. Clin. Oncol. 2019, 15, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, A.; Bishop, A.J.; Narang, S.; Allen, P.K.; Li, J.; McAleer, M.F.; Tatsui, C.E.; Rhines, L.D.; Amini, B.; Wang, X.A. Outcomes after Hypofractionated Dose-Escalation using a Simultaneous Integrated Boost Technique for Treatment of Spine Metastases Not Amenable to Stereotactic Radiosurgery. Pract. Radiat. Oncol. 2019, 9, e142–e148. [Google Scholar] [CrossRef] [PubMed]
- Mizumoto, M.; Harada, H.; Asakura, H.; Hashimoto, T.; Furutani, K.; Hashii, H.; Takagi, T.; Katagiri, H.; Takahashi, M.; Nishimura, T. Prognostic factors and a scoring system for survival after radiotherapy for metastases to the spinal column: A review of 544 patients at Shizuoka Cancer Center Hospital. Cancer 2008, 113, 2816–2822. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.C.; Watson, S.P.; Pitroda, S.P.; Son, C.H.; Das, L.C.; Stack, M.E.; Uppal, A.; Oshima, G.; Khodarev, N.N.; Salama, J.K. Clinical and molecular markers of long-term survival after oligometastasis-directed stereotactic body radiotherapy (SBRT): Survival after SBRT for Oligometastases. Cancer 2016, 122, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Theobald, S.; Rougier, P.; Ducreux, M.; Lusinchi, A.; Bardet, E.; Eymard, J.C.; Conroy, T.; Francois, E.; Seitz, J.F. Simultaneous high-dose external irradiation and daily cisplatin in unresectable, non-metastatic adenocarcinoma of the pancreas: A phase I–II study. Radiother. Oncol. 1997, 45, 129–132. [Google Scholar] [CrossRef]
- Bonet, M.; García, V.; Farré, N.; Algara, M.; Farrús, B.; Fernandez, J.; Reyes, V.; Eraso, A.; Álvarez, A.; Cambra, M.J. Radiation Therapy for Bone-Only Metastases in Breast Cancer Patients: A GOCO Survey of Current Clinical Practice. Rep. Pract. Oncol. Radiother. 2020, 25, 113–116. [Google Scholar] [CrossRef]
- Cardoso, F.; Senkus, E.; Costa, A.; Papadopoulos, E.; Aapro, M.; André, F.; Harbeck, N.; Aguilar Lopez, B.; Barrios, C.H.; Bergh, J. 4th ESO–ESMO International Consensus Guidelines for Advanced Breast Cancer (ABC 4). Ann. Oncol. 2018, 29, 1634–1657. [Google Scholar] [CrossRef]
- Radaideh, S.M.; Sledge, G.W. Taxane vs. taxane: Is the duel at an end? A commentary on a phase-III trial of doxorubicin and docetaxel versus doxorubicin and paclitaxel in metastatic breast cancer: Results of the ERASME 3 study. Breast Cancer Res. Treat. 2008, 111, 203–208. [Google Scholar] [CrossRef]
- Sledge, G.W.; Neuberg, D.; Bernardo, P.; Ingle, J.N.; Martino, S.; Rowinsky, E.K.; Wood, W.C. Phase III Trial of Doxorubicin, Paclitaxel, and the Combination of Doxorubicin and Paclitaxel as Front-Line Chemotherapy for Metastatic Breast Cancer: An Intergroup Trial (E1193). JCO 2003, 21, 588–592. [Google Scholar] [CrossRef]
- Cortes, J.; O’Shaughnessy, J.; Loesch, D.; Blum, J.L.; Vahdat, L.T.; Petrakova, K.; Chollet, P.; Manikas, A.; Diéras, V.; Delozier, T. Eribulin monotherapy versus treatment of physician’s choice in patients with metastatic breast cancer (EMBRACE): A phase 3 open-label randomised study. Lancet 2011, 377, 914–923. [Google Scholar] [CrossRef]
- Gradishar, W.J.; Krasnojon, D.; Cheporov, S.; Makhson, A.N.; Manikhas, G.M.; Clawson, A.; Bhar, P. Significantly Longer Progression-Free Survival With nab -Paclitaxel Compared with Docetaxel as First-Line Therapy for Metastatic Breast Cancer. JCO 2009, 27, 3611–3619. [Google Scholar] [CrossRef] [PubMed]
- Albain, K.S.; Nag, S.; Calderillo-Ruiz, G. Global phase III study of gemcitabine plus paclitaxel (GT) vs. paclitaxel (T) as frontline therapy for metastatic breast cancer (MBC): First report of overall survival. J. Clin. Oncol. 2004, 22, 510. [Google Scholar] [CrossRef]
- Yardley, D.A.; Brufsky, A.; Coleman, R.E.; Conte, P.F.; Cortes, J.; Glück, S.; Nabholtz, J.M.; O’Shaughnessy, J.; Beck, R.M.; Ko, A. Phase II/III weekly nab-paclitaxel plus gemcitabine or carboplatin versus gemcitabine/carboplatin as first-line treatment of patients with metastatic triple-negative breast cancer (the tnAcity study): Study protocol for a randomized controlled trial. Trials 2015, 16, 575. [Google Scholar] [CrossRef] [PubMed]
- Piccart, M.; Hortobagyi, G.N.; Campone, M.; Pritchard, K.I.; Lebrun, F.; Ito, Y.; Noguchi, S.; Perez, A.; Rugo, H.S.; Deleu, I. Everolimus plus exemestane for hormone-receptor-positive, human epidermal growth factor receptor-2-negative advanced breast cancer: Overall survival results from BOLERO-2. Ann. Oncol. 2014, 25, 2357–2362. [Google Scholar] [CrossRef] [PubMed]
- André, F.; Ciruelos, E.; Rubovszky, G.; Campone, M.; Loibl, S.; Rugo, H.S.; Iwata, H.; Conte, P.; Mayer, I.A.; Kaufman, B. Alpelisib for PIK3CA -Mutated, Hormone Receptor–Positive Advanced Breast Cancer. N. Engl. J. Med. 2019, 380, 1929–1940. [Google Scholar] [CrossRef]
- Finn, R.S.; Martin, M.; Rugo, H.S.; Jones, S.; Im, S.A.; Gelmon, K.; Harbeck, N.; Lipatov, O.N.; Walshe, J.M.; Moulder, S. Palbociclib and Letrozole in Advanced Breast Cancer. N. Engl. J. Med. 2016, 375, 1925–1936. [Google Scholar] [CrossRef]
- Goetz, M.P.; Toi, M.; Campone, M.; Sohn, J.; Paluch-Shimon, S.; Huober, J.; Park, I.H.; Trédan, O.; Chen, S.C.; Manso, L. MONARCH 3: Abemaciclib as Initial Therapy for Advanced Breast Cancer. JCO 2017, 35, 3638–3646. [Google Scholar] [CrossRef]
- Tripathy, D.; Im, S.A.; Colleoni, M.; Franke, F.; Bardia, A.; Harbeck, N.; Hurvitz, S.A.; Chow, L.; Sohn, J.; Lee, K.S. Ribociclib plus endocrine therapy for premenopausal women with hormone-receptor-positive, advanced breast cancer (MONALEESA-7): A randomised phase 3 trial. Lancet Oncol. 2018, 19, 904–915. [Google Scholar] [CrossRef]
- Figura, N.B.; Potluri, T.K.; Mohammadi, H.; Oliver, D.E.; Arrington, J.A.; Robinson, T.J.; Etame, A.B.; Tran, N.D.; Liu, J.K.; Soliman, H. CDK 4/6 inhibitors and stereotactic radiation in the management of hormone receptor positive breast cancer brain metastases. J. Neurooncol. 2019, 144, 583–589. [Google Scholar] [CrossRef]
- Messer, J.A.; Ekinci, E.; Patel, T.A.; Teh, B.S. Enhanced dermatologic toxicity following concurrent treatment with palbociclib and radiation therapy: A case report. Rep. Pract. Oncol. Radiother. 2019, 24, 276–280. [Google Scholar] [CrossRef]
- Chowdhary, M.; Sen, N.; Chowdhary, A.; Usha, L.; Cobleigh, M.A.; Wang, D.; Patel, K.R.; Barry, P.N.; Rao, R.D. Safety and Efficacy of Palbociclib and Radiation Therapy in Patients with Metastatic Breast Cancer: Initial Results of a Novel Combination. Adv. Radiat. Oncol. 2019, 4, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Ippolito, E.; Greco, C.; Silipigni, S.; Dell’Aquila, E.; Petrianni, G.M.; Tonini, G.; Fiore, M.; D’Angelillo, R.M.; Ramella, S. Concurrent radiotherapy with palbociclib or ribociclib for metastatic breast cancer patients: Preliminary assessment of toxicity. Breast 2019, 46, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Liang, K.; Lu, Y.; Jin, W. Sensitization of breast cancer cells to radiation by trastuzumab. Mol. Cancer Ther. 2003, 2, 1113–1120. [Google Scholar]
- Guo, G.; Wang, T.; Gao, Q.; Tamae, D.; Wong, P.; Chen, T.; Chen, W.C.; Shively, J.E.; Wong, J.Y. Expression of ErbB2 enhances radiation-induced NF-κB activation. Oncogene 2004, 23, 535–545. [Google Scholar] [CrossRef] [PubMed]
- Pietras, R.; Poen, J.; Gallardo, D.; Wongvipat, P.; Lee, H.; Slamon, D. Monoclonal antibody to HER-2/neureceptor modulates repair of radiation-induced DNA damage and enhances radiosensitivity of human breast cancer cells overexpressing this oncogene. Cancer Res. 1999, 59, 1347–1355. [Google Scholar] [PubMed]
- Hou, J.; Zhou, Z.; Chen, X.; Zhao, R.; Yang, Z.; Wei, N.; Ni, Q.; Feng, Y.; Yu, X.; Ma, J.; et al. HER2 reduces breast cancer radiosensitivity by activating focal adhesion kinase in vitro and in vivo. Oncotarget 2016, 7, 45186–45198. [Google Scholar] [CrossRef] [PubMed]
- Lazaro, G.; Smith, C.; Goddard, L.; Jordan, N.; McClelland, R.; Barrett-Lee, P.; Nicholson, R.I.; Hiscox, S. Targeting focal adhesion kinase in ER+/HER2+ breast cancer improves trastuzumab response. Endocr. Relat. Cancer 2013, 20, 691–704. [Google Scholar] [CrossRef]
- Kim, J.-S.; Kim, H.A.; Seong, M.K.; Seol, H.; Oh, J.S.; Kim, E.K.; Chang, J.W.; Hwang, S.G.; Noh, W.C. STAT3-survivin signaling mediates a poor response to radiotherapy in HER2-positive breast cancers. Oncotarget 2016, 7, 7055–7065. [Google Scholar] [CrossRef]
- Sato, S.; Kajiyama, Y.; Sugano, M.; Iwanuma, Y.; Sonoue, H.; Matsumoto, T.; Sasai, K.; Tsurumaru, M. Monoclonal antibody to HER-2/neu receptor enhances radiosensitivity of esophageal cancer cell lines expressing HER-2/neu oncoprotein. Int. J. Radiat. Oncol. Biol. Phys. 2005, 61, 203–211. [Google Scholar] [CrossRef]
- Uno, M.; Otsuki, T.; Kurebayashi, J.; Sakaguchi, H.; Isozaki, Y.; Ueki, A.; Yata, K.; Fujii, T.; Hiratsuka, J.; Akisada, T. Anti-HER2-antibody enhances irradiation-induced growth inhibition in head and neck carcinoma. Int. J. Cancer 2001, 94, 474–479. [Google Scholar] [CrossRef]
- Bese, N.S.; Umay, C.; Serdengecti, S.; Kepil, N.; Sut, N.; Altug, T.; Ober, A. The impact of trastuzumab on radiation-induced pulmonary fibrosis: Results of an experimental study. Med. Oncol. 2010, 27, 1415–1419. [Google Scholar] [CrossRef] [PubMed]
- Sambade, M.J.; Kimple, R.J.; Camp, J.T.; Peters, E.; Livasy, C.A.; Sartor, C.I.; Shields, J.M. Lapatinib in Combination with Radiation Diminishes Tumor Regrowth in HER2+ and Basal-Like/EGFR+ Breast Tumor Xenografts. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 575–581. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sambade, M.J.; Camp, J.T.; Kimple, R.J.; Sartor, C.I.; Shields, J.M. Mechanism of lapatinib-mediated radiosensitization of breast cancer cells is primarily by inhibition of the Raf > MEK > ERK mitogen-activated protein kinase cascade and radiosensitization of lapatinib-resistant cells restored by direct inhibition of MEK. Radiother. Oncol. 2009, 93, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.R.; Yang, H.C.; Savariar, E.N.; Aguilera, J.; Crisp, J.L.; Jones, K.A.; Whitney, M.A.; Lippman, S.M.; Cohen, E.E.; Tsien, R.Y. Anti-tubulin drugs conjugated to anti-ErbB antibodies selectively radiosensitize. Nat. Commun. 2016, 7, 13019. [Google Scholar] [CrossRef]
- Kimple, R.J.; Horton, J.K.; Livasy, C.A.; Shields, J.M.; Lawrence, J.A.; Chiu, W.M.; Ivanova, A.; Ollila, D.W.; Carey, L.A.; Halle, J.S. Phase I Study and Biomarker Analysis of Lapatinib and Concurrent Radiation for Locally Advanced Breast Cancer. Oncologist 2012, 17, 1496. [Google Scholar] [CrossRef]
- Krop, I.E.; Suter, T.M.; Dang, C.T.; Dirix, L.; Romieu, G.; Zamagni, C.; Citron, M.L.; Campone, M.; Xu, N.; Smitt, M. Feasibility and Cardiac Safety of Trastuzumab Emtansine After Anthracycline-Based Chemotherapy As (neo)Adjuvant Therapy for Human Epidermal Growth Factor Receptor 2–Positive Early-Stage Breast Cancer. JCO 2015, 33, 1136–1142. [Google Scholar] [CrossRef]
- Yu, M.L.X. The role of poly(ADP-ribosyl)ation in DNA damage response and cancer chemotherapy. Oncogene 2015, 34, 3349–3356. [Google Scholar] [CrossRef]
- Scarpa, E.S.; Fabrizio, G.; di Girolamo, M. A role of intracellular mono-ADP-ribosylation in cancer biology. FEBS J. 2013, 280, 3551–3562. [Google Scholar] [CrossRef]
- Dulaney, C.; Marcrom, S.; Stanley, J.; Yang, E.S. Poly(ADP-ribose) Polymerase Activity and Inhibition in Cancer. In Seminars in Cell & Developmental Biology; Academic Press: Cambridge, MA, USA, 2017; pp. 144–153. [Google Scholar]
- Lesueur, P.; Chevalier, F.; Austry, J.B.; Waissi, W.; Burckel, H.; Noël, G.; Habrand, J.L.; Saintigny, Y.; Joly, F. Poly-(ADP-ribose)-polymerase inhibitors as radiosensitizers: A systematic review of pre-clinical and clinical human studies. Oncotarget 2017, 8, 69105–69124. [Google Scholar] [CrossRef]
- McLachlan, J.; George, A.; Banerjee, S. The Current Status of PARP Inhibitors in Ovarian Cancer. Tumori J. 2016, 102, 433–440. [Google Scholar] [CrossRef]
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.A.; Shaw Wright, G. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. N. Engl. J. Med. 2018, 379, 2108–2121. [Google Scholar] [CrossRef] [PubMed]
- Rubini, G.; Nicoletti, A.; Rubini, D.; Asabella, A.N. Radiometabolic Treatment of Bone-Metastasizing Cancer: From 186Rhenium to 223Radium. Cancer Biother. Radiopharm. 2014, 29, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fuster, D.; Herranz, D.; Vidal-Sicart, S.; Muñoz, M.; Conill, C.; Mateos, J.J.; Martín, F.; Pons, F. Usefulness of strontium-89 for bone pain palliation in metastatic breast cancer patients. Nucl. Med. Commun. 2000, 21, 623–626. [Google Scholar] [CrossRef]
- Lam, M.G.E.H.; de Klerk, J.M.H.; van Rijk, P.P. 186Re-HEDP for metastatic bone pain in breast cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2004, 31, S162–S170. [Google Scholar] [CrossRef] [PubMed]
- Palmedo, H.; Bender, H.; Dierke-Dzierzon, C.; Carl, U. Pain Palliation With rhenium-186 HEDP in Breast Cancer Patients With Disseminated Bone Metastases. Clin. Nucl. Med. 1999, 24, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, S.; Larsen, R.; Fossa, S. First Clinical Experience With Alpha-Emitting radium-223 in the Treatment of Skeletal Metastases. Clin. Cancer Res. 2005, 11, 4451–4459. [Google Scholar] [CrossRef]
- Coleman, R.; Aksnes, A.K.; Naume, B.; Garcia, C.; Jerusalem, G.; Piccart, M.; Vobecky, N.; Thuresson, M.; Flamen, P. A phase IIa, nonrandomized study of radium-223 dichloride in advanced breast cancer patients with bone-dominant disease. Breast Cancer Res. Treat. 2014, 145, 411–418. [Google Scholar] [CrossRef]
- Coleman, R.; Brown, J.; Rathbone, E.; Flanagan, L.; Reid, A.; Kendall, J.; Howell, S.; Twelves, C.; Palmieri, C.; Anand, A. CApecitabine plus Radium-223 (XofigoTM) in breast cancer patients with BONe metastases (CARBON): Study protocol for a phase IB/IIA randomised controlled trial. Trials 2020, 21, 89. [Google Scholar] [CrossRef]
- Burke, M.; Atkins, A.; Kiss, A.; Akens, M.; Yee, A.; Whyne, C. The impact of metastasis on the mineral phase of vertebral bone tissue. J. Mech. Behav. Biomed. Mater. 2017, 69, 75–84. [Google Scholar] [CrossRef]
- Manabe, J.; Kawaguchi, N.; Matsumoto, S.; Tanizawa, T. Surgical treatment of bone metastasis: Indications and outcomes. Int. J. Clin. Oncol. 2004, 10, 103–111. [Google Scholar] [CrossRef]
- Durr, H.; Muller, P.; Lenz, T. Surgical Treatment of Bone Metastases in Patients with Breast Cancer. Clin. Orthop. Relat. Res. 2002, 396, 191–196. [Google Scholar]
- Szendrői, M.; Antal, I.; Szendrői, A.; Lazáry, Á.; Varga, P.P. Diagnostic algorithm, prognostic factors and surgical treatment of metastatic cancer diseases of the long bones and spine. EFORT Open Rev. 2017, 2, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Chiras, J.; Shotar, E.; Cormier, E.; Clarençon, F. Interventional radiology in bone metastases. Eur. J. Cancer Care 2017, 26, e12741. [Google Scholar] [CrossRef] [PubMed]
- Colleoni, M.; O’Neill, A.; Goldhirsch, A.; Gelber, R.D.; Bonetti, M.; Thürlimann, B.; Price, K.N.; Castiglione-Gertsch, M.; Coates, A.S.; Lindtner, J. Identifying Breast Cancer Patients at High Risk for Bone Metastases. JCO 2000, 18, 3925–3935. [Google Scholar] [CrossRef]
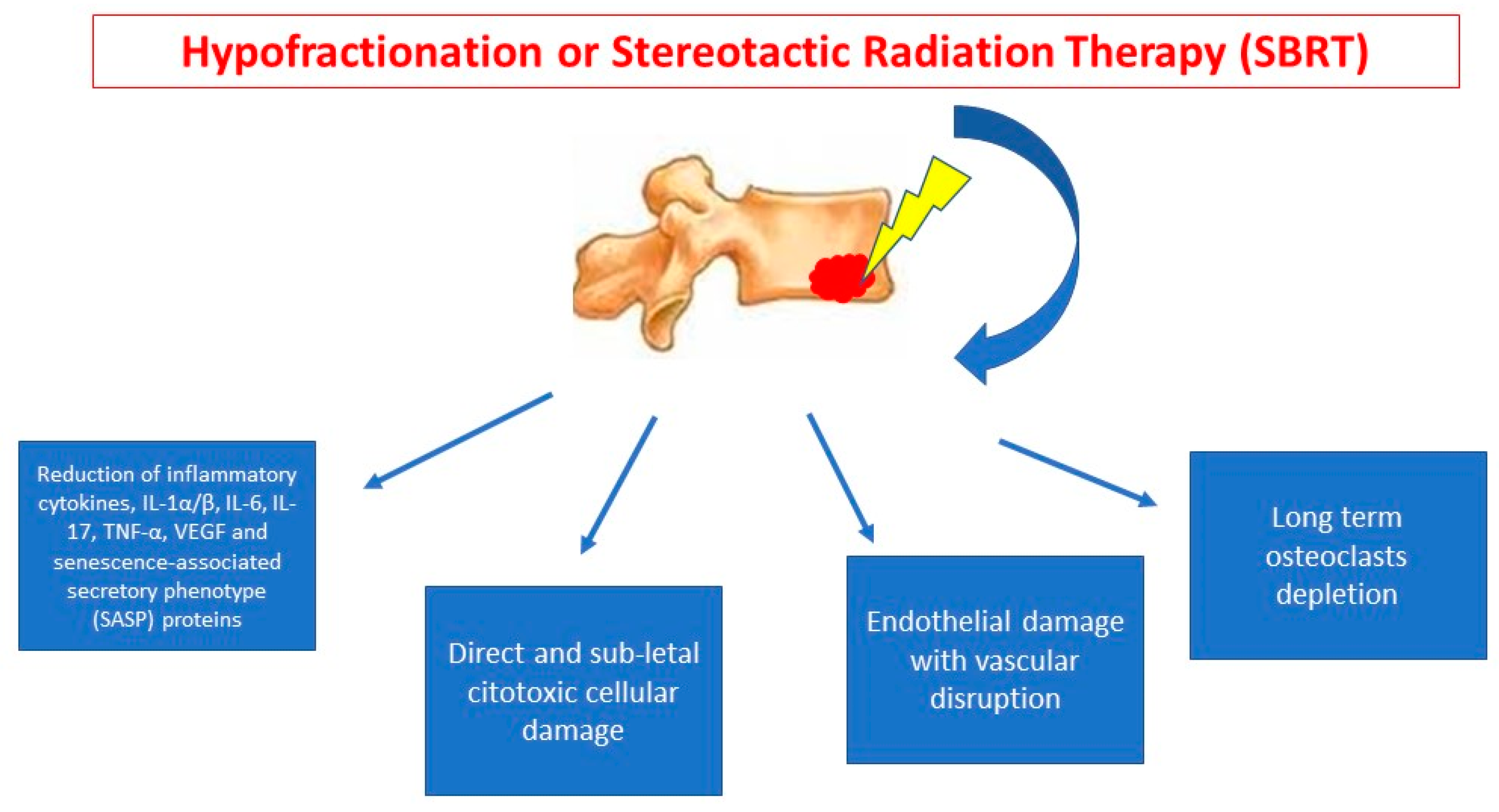
| (a) Dose and Volumes for Palliative Radiotherapy on Bone Metastasis | |||
| Dose | Volume | Outcome | Reference |
| 8 Gy 1 Fr | Bone compartment +/− soft-tissue invasion | Symptom control (pain, neurological impairment) Preferable in case of poor expectation of retreatments | Chow E. 2002 [20] Chow E. 2007 [46] Chow E. 2012 [21] |
| 20 Gy 5 Fr | Bone compartment +/− soft-tissue invasion | Symptom control (pain, neurological impairment) | Chow E. 2002 [20] Chow E. 2007 [46] Chow E. 2012 [21] |
| 30 Gy 10 Fr | Bone compartment +/− soft-tissue invasion | Symptom control (pain, neurological impairment) After surgical stabilization | Rades D, 2004 [47] |
| (b) Dose and Volumes for Radical Radiotherapy on Bone Metastasis | |||
| Dose | Volume | Outcome | Reference |
| EQD2 of 57.3 Gy [38.3–70] BED 60 Gy (obtained) | Bone lesion + margin (mm) | 5-year OS 83% (BO vs. no-BO p = 0.002) 10-year OS 75% (BO vs. no-BO p = 0.002) FFWM (BO vs. no-BO p 0.018) | Milano MT, 2019 [17] |
| BED > 50 Gy | Bone lesion + margin (mm) | 3-year DPFS 36.8% 5-year LC 66.1% 5-year OS 49% univariate Analysis: Higher RT dose (p = 0.002) Whole Lesion RT (p = 0.007) | Yoo GS, 2015 [51] |
| 30–45 Gy 3 Fr | Bone lesion + margin (mm) | 1-year PFS 75% 2-year PFS 53% 2-year LC 97% 2-year OS 95% | Trovò M, 2017 [52] |
| 15–22.5 Gy 1 Fr | Bone lesion + margin (mm) | 15-month pain control 96% | Gerszten PC, 2005 [44] |
| 35 Gy (spinal) 50 Gy (no spinal) 5 Fr | Bone lesion + margin (mm) | 1-year LC 91.2% PFS 10.1 months OS 37.3 months | Kam TY, 2019 [53] |
| 40 Gy (GTV) 30 Gy (WV) 10 Fr | Bone lesion + margin (mm) Whole vertebra (WV) | 1-year LC 93% OS 58% | Farooqi A, 2018 [54] |
| Reference | Setting | Intervention | Radiotherapy Dose/Volumes | Primary Endpoints |
|---|---|---|---|---|
| CLEAR, Jeong J, NCT03750396 | Oligometastatic breast cancer recurrence (>12 months) All site of metastases | Surgery or radiotherapy or radiofrequency on metastasis | Total radiation dose and fractions are various according to metastatic lesions (57–97.5 Gy/6–10 Fraction) | PFS |
| NRG Oncology, NCT02364557 | Limited MBC | SBRT +/− Surgery | Radiosurgery in 1, 3 or 5 fractions (according to discretion of physician) | PFS OS |
| STEREO-SEIN, NCT02089100 | De novo Oligometastatic Breast Cancer, excluding triple negative subtype | SBRT | SBRT with radical intent to all sites of metastases | PFS |
| MSKCC, NCT03808337 | Metastatic NSCLC or TNBC | SBRT concurrently to systemic therapy | SBRT with a minimum BED of 48 Gy to all sites | PFS |
| NCI, NCT00182793 | Stage IIIb-IV BC | RT on primary site or on site of metastasis (oligometastatic), High-dose chemotherapy, autologous stem cells transplant | Tomotherapy on site of disease with standard fractionation | 5-year Relapse-Free-Survival 5-year Overall, Survival-Rate |
| CIMER, NCT04220476 | Oligometastatic, Luminal BC | SBRT (Immune-SBRT every 48 h) | SBRT every 48 h, to all sites of metastases 50GY in 5 fractions | ORR PFS OS |
| MSKCC NCT03808662 | Oligoprogressive NSCLC or TNBC | SBRT | SBRT 9–10 Gy × 3 or 10 Gy × 5 fractions given every other day to all sites | PFS |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marazzi, F.; Orlandi, A.; Manfrida, S.; Masiello, V.; Di Leone, A.; Massaccesi, M.; Moschella, F.; Franceschini, G.; Bria, E.; Gambacorta, M.A.; et al. Diagnosis and Treatment of Bone Metastases in Breast Cancer: Radiotherapy, Local Approach and Systemic Therapy in a Guide for Clinicians. Cancers 2020, 12, 2390. https://doi.org/10.3390/cancers12092390
Marazzi F, Orlandi A, Manfrida S, Masiello V, Di Leone A, Massaccesi M, Moschella F, Franceschini G, Bria E, Gambacorta MA, et al. Diagnosis and Treatment of Bone Metastases in Breast Cancer: Radiotherapy, Local Approach and Systemic Therapy in a Guide for Clinicians. Cancers. 2020; 12(9):2390. https://doi.org/10.3390/cancers12092390
Chicago/Turabian StyleMarazzi, Fabio, Armando Orlandi, Stefania Manfrida, Valeria Masiello, Alba Di Leone, Mariangela Massaccesi, Francesca Moschella, Gianluca Franceschini, Emilio Bria, Maria Antonietta Gambacorta, and et al. 2020. "Diagnosis and Treatment of Bone Metastases in Breast Cancer: Radiotherapy, Local Approach and Systemic Therapy in a Guide for Clinicians" Cancers 12, no. 9: 2390. https://doi.org/10.3390/cancers12092390
APA StyleMarazzi, F., Orlandi, A., Manfrida, S., Masiello, V., Di Leone, A., Massaccesi, M., Moschella, F., Franceschini, G., Bria, E., Gambacorta, M. A., Masetti, R., Tortora, G., & Valentini, V. (2020). Diagnosis and Treatment of Bone Metastases in Breast Cancer: Radiotherapy, Local Approach and Systemic Therapy in a Guide for Clinicians. Cancers, 12(9), 2390. https://doi.org/10.3390/cancers12092390













