A Phase II Study of Glembatumumab Vedotin for Metastatic Uveal Melanoma
Abstract
1. Significance
2. Background
3. Methods
3.1. Study Design
3.2. Patient Selection
3.3. Dosing Regimen
3.4. Response Criteria
3.5. Toxicity Capture
3.6. Statistical Analysis
3.7. Correlative Analysis
3.8. Immunohistochemistry
4. Results
4.1. Demographics
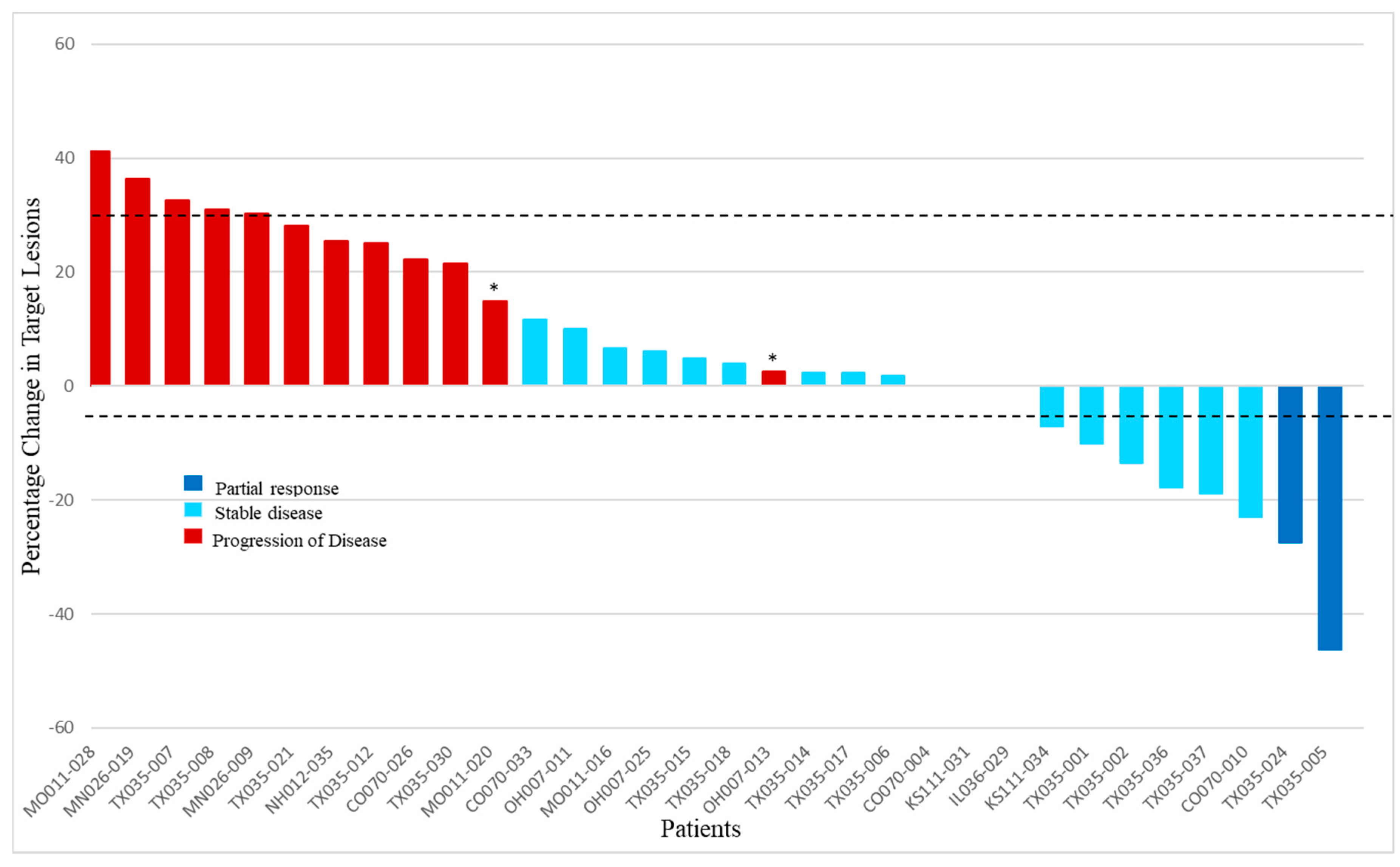
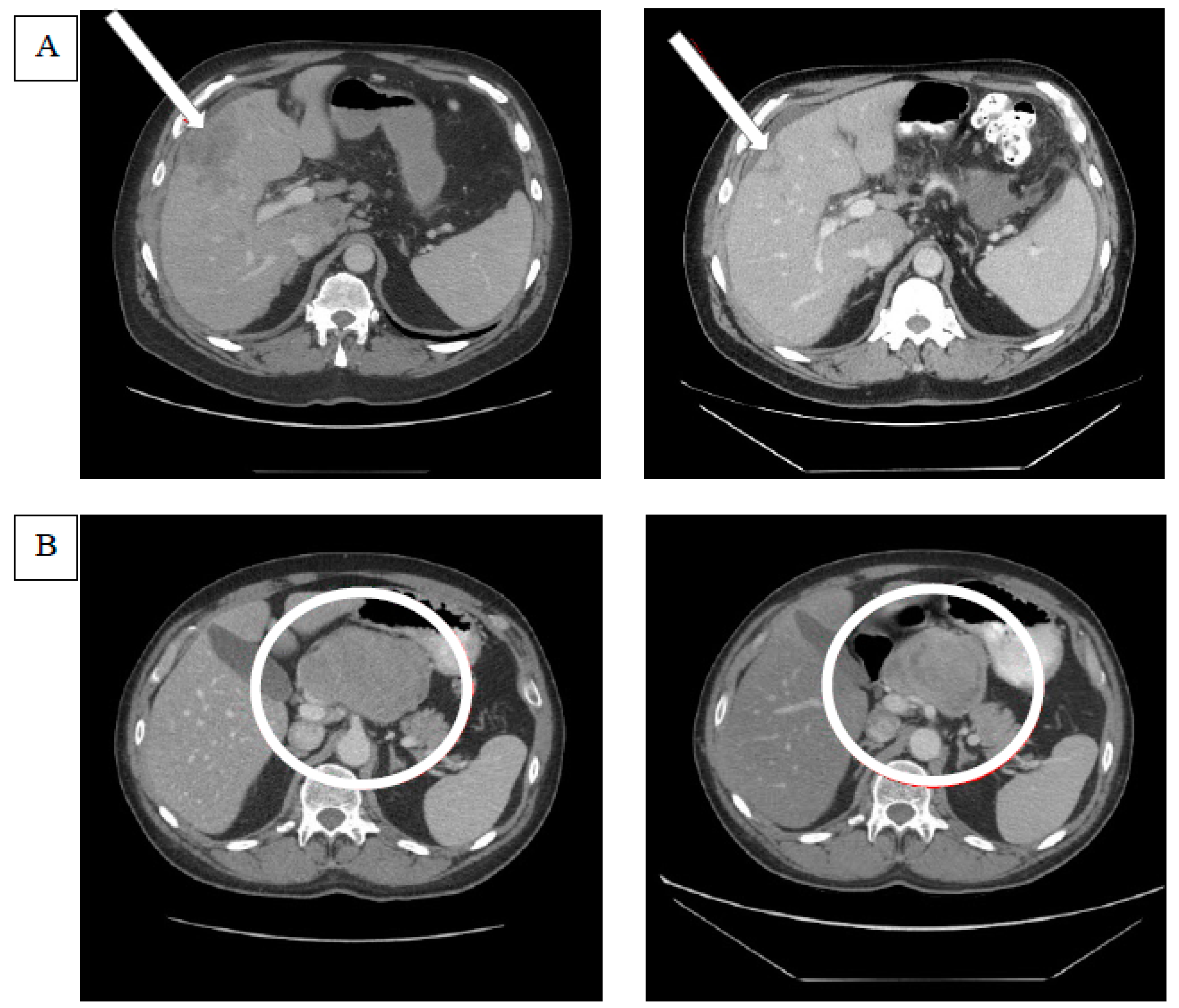
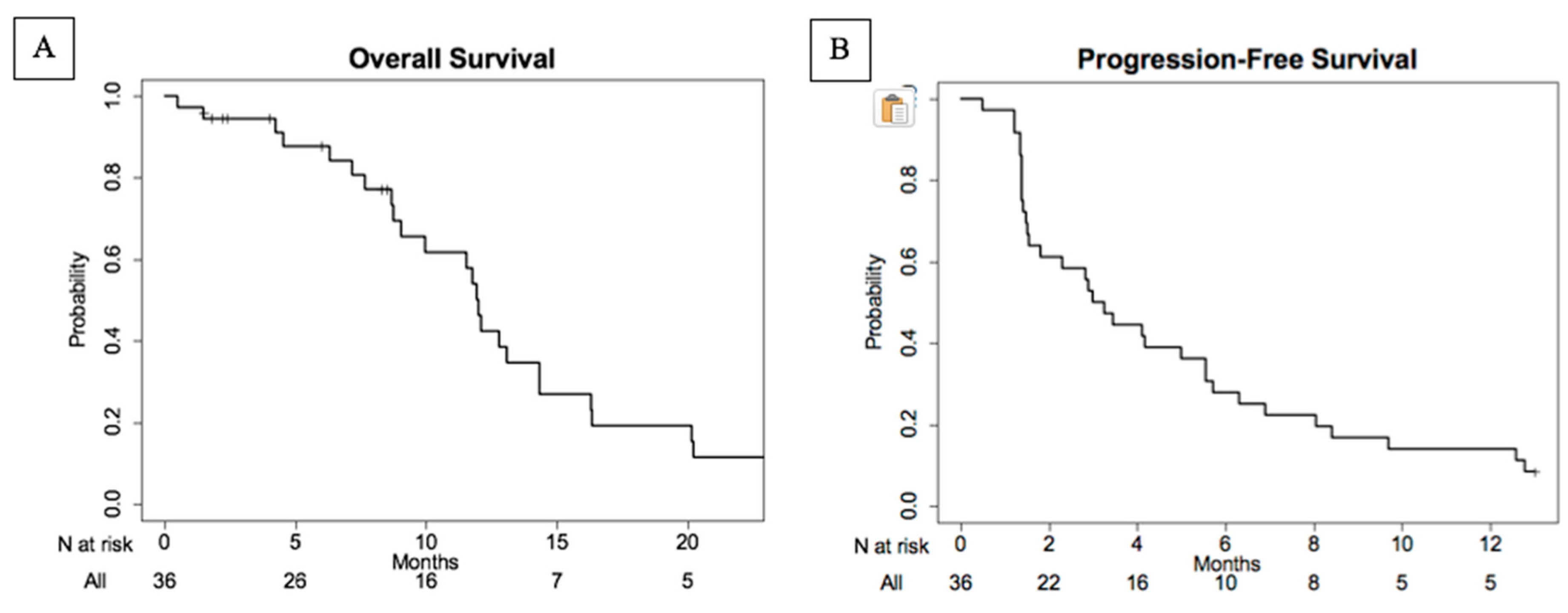
4.2. Efficacy
4.3. Adverse Events
4.4. Gpnmb Tissue Expression
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Singh, A.D.; Turell, M.E.; Topham, A.K. Uveal melanoma: Trends in incidence, treatment, and survival. Ophthalmology 2011, 118, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, C.C.; Wu, X.C.; Jemal, A.; Martin, H.J.; Roche, L.M.; Chen, V.W. Incidence of noncutaneous melanomas in the U.S. Cancer 2005, 103, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Diener-West, M.; Reynolds, S.M.; Agugliaro, D.J.; Caldwell, R.; Cumming, K.; Earle, J.D.; Hawkins, B.S.; Hayman, J.A.; Jaiyesimi, I.; Jampol, L.M.; et al. Development of metastatic disease after enrollment in the COMS trials for treatment of choroidal melanoma: Collaborative Ocular Melanoma Study Group Report No. 26. Arch. Ophthalmol. 2005, 123, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Manson, D.K.; Marr, B.P.; Carvajal, R.D. Treatment of uveal melanoma: Where are we now? Ther. Adv. Med. Oncol. 2018, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Luke, J.J.; Triozzi, P.L.; McKenna, K.C.; Van Meir, E.G.; Gershenwald, J.E.; Bastian, B.C.; Gutkind, J.S.; Bowcock, A.M.; Streicher, H.Z.; Patel, P.M.; et al. Biology of advanced uveal melanoma and next steps for clinical therapeutics. Pigment Cell Melanoma Res. 2015, 28, 135–147. [Google Scholar] [CrossRef]
- Park, J.J.; Diefenbach, R.J.; Joshua, A.M.; Kefford, R.F.; Carlino, M.S.; Rizos, H. Oncogenic signaling in uveal melanoma. Pigment Cell Melanoma Res. 2018, 31, 661–672. [Google Scholar] [CrossRef]
- Algazi, A.P.; Tsai, K.K.; Shoushtari, A.N.; Munhoz, R.R.; Eroglu, Z.; Piulats, J.M.; Ott, P.A.; Johnson, D.B.; Hwang, J.; Daud, A.I.; et al. Clinical outcomes in metastatic uveal melanoma treated with PD-1 and PD-L1 antibodies. Cancer 2016, 122, 3344–3353. [Google Scholar] [CrossRef]
- Javed, A.; Arguello, D.; Johnston, C.; Gatalica, Z.; Terai, M.; Weight, R.M.; Orloff, M.; Mastrangelo, M.J.; Sato, T. PD-L1 expression in tumor metastasis is different between uveal melanoma and cutaneous melanoma. Immunotherapy 2017, 9, 1323–1330. [Google Scholar] [CrossRef]
- Qin, Y.; Petaccia de Macedo, M.; Reuben, A.; Forget, M.A.; Haymaker, C.; Bernatchez, C.; Spencer, C.N.; Gopalakrishnan, V.; Reddy, S.; Cooper, Z.A.; et al. Parallel profiling of immune infiltrate subsets in uveal melanoma versus cutaneous melanoma unveils similarities and differences: A pilot study. Oncoimmunology 2017, 6, e1321187. [Google Scholar] [CrossRef]
- Pan, H.; Lu, L.; Cui, J.; Yang, Y.; Wang, Z.; Fan, X. Immunological analyses reveal an immune subtype of uveal melanoma with a poor prognosis. Aging (Albany NY) 2020, 12, 1446–1464. [Google Scholar] [CrossRef]
- Kuk, D.; Shoushtari, A.N.; Barker, C.A.; Panageas, K.S.; Munhoz, R.R.; Momtaz, P.; Ariyan, C.E.; Brady, M.S.; Coit, D.G.; Bogatch, K.; et al. Prognosis of Mucosal, Uveal, Acral, Nonacral Cutaneous, and Unknown Primary Melanoma From the Time of First Metastasis. Oncologist 2016, 21, 848–854. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.D.; Esmaeli, B.; Soheili, A.; Simantov, R.; Gombos, D.S.; Bedikian, A.Y.; Hwu, P. GPNMB expression in uveal melanoma: A potential for targeted therapy. Melanoma Res. 2010, 20, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Naumovski, L.; Junutula, J.R. Glembatumumab vedotin, a conjugate of an anti-glycoprotein non-metastatic melanoma protein B mAb and monomethyl auristatin E for the treatment of melanoma and breast cancer. Curr. Opin. Mol. Ther. 2010, 12, 248–257. [Google Scholar] [PubMed]
- Pollack, V.A.; Alvarez, E.; Tse, K.F.; Torgov, M.Y.; Xie, S.; Shenoy, S.G.; MacDougall, J.R.; Arrol, S.; Zhong, H.; Gerwien, R.W.; et al. Treatment parameters modulating regression of human melanoma xenografts by an antibody-drug conjugate (CR011-vcMMAE) targeting GPNMB. Cancer Chemother. Pharmacol. 2007, 60, 423–435. [Google Scholar] [CrossRef]
- Qian, X.; Mills, E.; Torgov, M.; LaRochelle, W.J.; Jeffers, M. Pharmacologically enhanced expression of GPNMB increases the sensitivity of melanoma cells to the CR011-vcMMAE antibody-drug conjugate. Mol. Oncol. 2008, 2, 81–93. [Google Scholar] [CrossRef]
- Tse, K.F.; Jeffers, M.; Pollack, V.A.; McCabe, D.A.; Shadish, M.L.; Khramtsov, N.V.; Hackett, C.S.; Shenoy, S.G.; Kuang, B.; Boldog, F.L.; et al. CR011, a fully human monoclonal antibody-auristatin E conjugate, for the treatment of melanoma. Clin. Cancer Res. 2006, 12, 1373–1382. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Ott, P.A.; Hamid, O.; Pavlick, A.C.; Kluger, H.; Kim, K.B.; Boasberg, P.D.; Simantov, R.; Crowley, E.; Green, J.A.; Hawthorne, T.; et al. Phase I/II study of the antibody-drug conjugate glembatumumab vedotin in patients with advanced melanoma. J. Clin. Oncol. 2014, 32, 3659–3666. [Google Scholar] [CrossRef]
- Yardley, D.A.; Weaver, R.; Melisko, M.E.; Saleh, M.N.; Arena, F.P.; Forero, A.; Cigler, T.; Stopeck, A.; Citrin, D.; Oliff, I.; et al. EMERGE: A Randomized Phase II Study of the Antibody-Drug Conjugate Glembatumumab Vedotin in Advanced Glycoprotein NMB-Expressing Breast Cancer. J. Clin. Oncol. 2015, 33, 1609–1619. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Piperno-Neumann, S.; Kapiteijn, E.; Chapman, P.B.; Frank, S.; Joshua, A.M.; Piulats, J.M.; Wolter, P.; Cocquyt, V.; Chmielowski, B.; et al. Selumetinib in Combination With Dacarbazine in Patients With Metastatic Uveal Melanoma: A Phase III, Multicenter, Randomized Trial (SUMIT). J. Clin. Oncol. 2018, 36, 1232–1239. [Google Scholar] [CrossRef]
- Lane, A.M.; Kim, I.K.; Gragoudas, E.S. Survival Rates in Patients After Treatment for Metastasis From Uveal Melanoma. JAMA Ophthalmol. 2018, 136, 981–986. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | No. of Patients | % |
|---|---|---|
| Age in years | ||
| Median | 62 | |
| Range | 29–79 | |
| Gender | ||
| Male | 18 | 51 |
| Female | 17 | 49 |
| Performance Status (ECOG) | ||
| 0 | 26 | 74 |
| 1 | 9 | 26 |
| M-Stage * | ||
| M1a | 16 | 46 |
| M1b | 12 | 34 |
| M1c | 7 | 20 |
| Sites of Metastasis ** | ||
| Liver only | 17 | 48 |
| Extrahepatic only | 3 | 9 |
| Both liver and extra- | 15 | 43 |
| Hepatic | ||
| Extrahepatic sites | ||
| Lung | 14 | 40 |
| Lymph node | 9 | 26 |
| Bones | 6 | 17 |
| Peritoneum/Soft tissue | 5 | 17 |
| Subcutaneous | 2 | 14 |
| Muscle | 1 each | 6 |
| All other sites *** | 3 |
| Response | No. | % |
|---|---|---|
| Evaluable for response | (n = 35) | |
| Complete Response (CR) | 0 | 0 |
| Partial Response (PR) | 2 | 6 |
| Stable Disease (SD) | 18 | 51 |
| Disease Progression | 14 | 40 |
| Unknown * | 1 | 3 |
| Disease Control Rate (CR + PR + SD) | 20 | 57 |
| Stable Disease > 100 days | 12 | 34 |
| Duration of response, days | ||
| Median | 263 | |
| Range | 149–377 | |
| Duration of stable disease, days | ||
| Median | 147 | |
| Range | 82–426 | |
| Adverse Event | Any Severity | CTCAE Grade 3 | CTCAE Grade 4 | |||
|---|---|---|---|---|---|---|
| No. | % | No. | % | No. | % | |
| Hematologic | ||||||
| Leukopenia | 24 | 69 | 1 | 3 | ||
| Neutropenia | 21 | 60 | 12 | 34 | 1 | 3 |
| Anemia | 14 | 40 | 5 | 14 | ||
| Lymphopenia | 6 | 17 | ||||
| Thrombocytopenia | 5 | 14 | ||||
| Gastrointestinal | ||||||
| Elevated ALT/AST | 22 | 63 | 1 | 3 | ||
| Nausea | 18 | 51 | 1 | 3 | ||
| Diarrhea | 11 | 31 | 1 | 3 | ||
| Elevated ALP | 9 | 26 | ||||
| Constipation | 8 | 23 | 1 | 3 | ||
| Mucositis, oral | 6 | 17 | ||||
| Vomiting | 5 | 14 | 2 | 6 | ||
| Hyperbilirubinemia | 4 | 11 | ||||
| Abdominal pain | 2 | 6 | ||||
| Dry mouth | 2 | 6 | ||||
| Dyspepsia | 2 | 6 | ||||
| Oral pain | 2 | 6 | ||||
| Skin and Subcutaneous | ||||||
| Alopecia | 28 | 80 | ||||
| Rash, maculopapular | 19 | 54 | 2 | 6 | ||
| Pruritus | 18 | 51 | ||||
| Dry skin | 3 | 9 | ||||
| Skin hypopigmentation | 3 | 9 | ||||
| Rash, acneiform | 2 | 6 | ||||
| General | ||||||
| Fatigue | 20 | 57 | 1 | 3 | ||
| Pain | 6 | 17 | ||||
| Chills | 2 | 6 | ||||
| Flu like symptoms | 2 | 6 | ||||
| Localized edema | 2 | 6 | ||||
| Weight loss | 2 | 6 | 1 | 3 | ||
| Nervous System | ||||||
| Peripheral Neuropathy | 15 | 43 | ||||
| Dysgeusia | 8 | 23 | ||||
| Headache | 3 | 9 | ||||
| Dizziness | 2 | 6 | ||||
| Musculoskeletal | ||||||
| Arthralgia | 9 | 26 | ||||
| Myalgia | 6 | 17 | ||||
| Pain in extremity | 3 | 9 | ||||
| Generalized muscle weakness | 2 | 6 | ||||
| Respiratory | ||||||
| Dyspnea | 5 | 14 | ||||
| Renal and Electrolytes | ||||||
| Hyponatremia | 5 | 14 | ||||
| Hypophosphatemia | 4 | 11 | 2 | 6 | ||
| Hypokalemia | 3 | 9 | 1 | 3 | ||
| Hyperkalemia | 2 | 6 | ||||
| Metabolism and Nutrition | ||||||
| Anorexia | 13 | 37 | ||||
| Hypoalbuminemia | 3 | 9 | ||||
| Hyperglycemia | 2 | 6 | ||||
| Vascular | ||||||
| Hot flashes | 3 | 9 | ||||
| Hypotension | 3 | 9 | 1 | 3 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hasanov, M.; Rioth, M.J.; Kendra, K.; Hernandez-Aya, L.; Joseph, R.W.; Williamson, S.; Chandra, S.; Shirai, K.; Turner, C.D.; Lewis, K.; et al. A Phase II Study of Glembatumumab Vedotin for Metastatic Uveal Melanoma. Cancers 2020, 12, 2270. https://doi.org/10.3390/cancers12082270
Hasanov M, Rioth MJ, Kendra K, Hernandez-Aya L, Joseph RW, Williamson S, Chandra S, Shirai K, Turner CD, Lewis K, et al. A Phase II Study of Glembatumumab Vedotin for Metastatic Uveal Melanoma. Cancers. 2020; 12(8):2270. https://doi.org/10.3390/cancers12082270
Chicago/Turabian StyleHasanov, Merve, Matthew J. Rioth, Kari Kendra, Leonel Hernandez-Aya, Richard W. Joseph, Stephen Williamson, Sunandana Chandra, Keisuke Shirai, Christopher D. Turner, Karl Lewis, and et al. 2020. "A Phase II Study of Glembatumumab Vedotin for Metastatic Uveal Melanoma" Cancers 12, no. 8: 2270. https://doi.org/10.3390/cancers12082270
APA StyleHasanov, M., Rioth, M. J., Kendra, K., Hernandez-Aya, L., Joseph, R. W., Williamson, S., Chandra, S., Shirai, K., Turner, C. D., Lewis, K., Crowley, E., Moscow, J., Carter, B., & Patel, S. (2020). A Phase II Study of Glembatumumab Vedotin for Metastatic Uveal Melanoma. Cancers, 12(8), 2270. https://doi.org/10.3390/cancers12082270





