Estrogen Receptor Status Oppositely Modifies Breast Cancer Prognosis in BRCA1/BRCA2 Mutation Carriers Versus Non-Carriers
Abstract
1. Introduction
2. Results
2.1. Patient and Tumor Characteristics
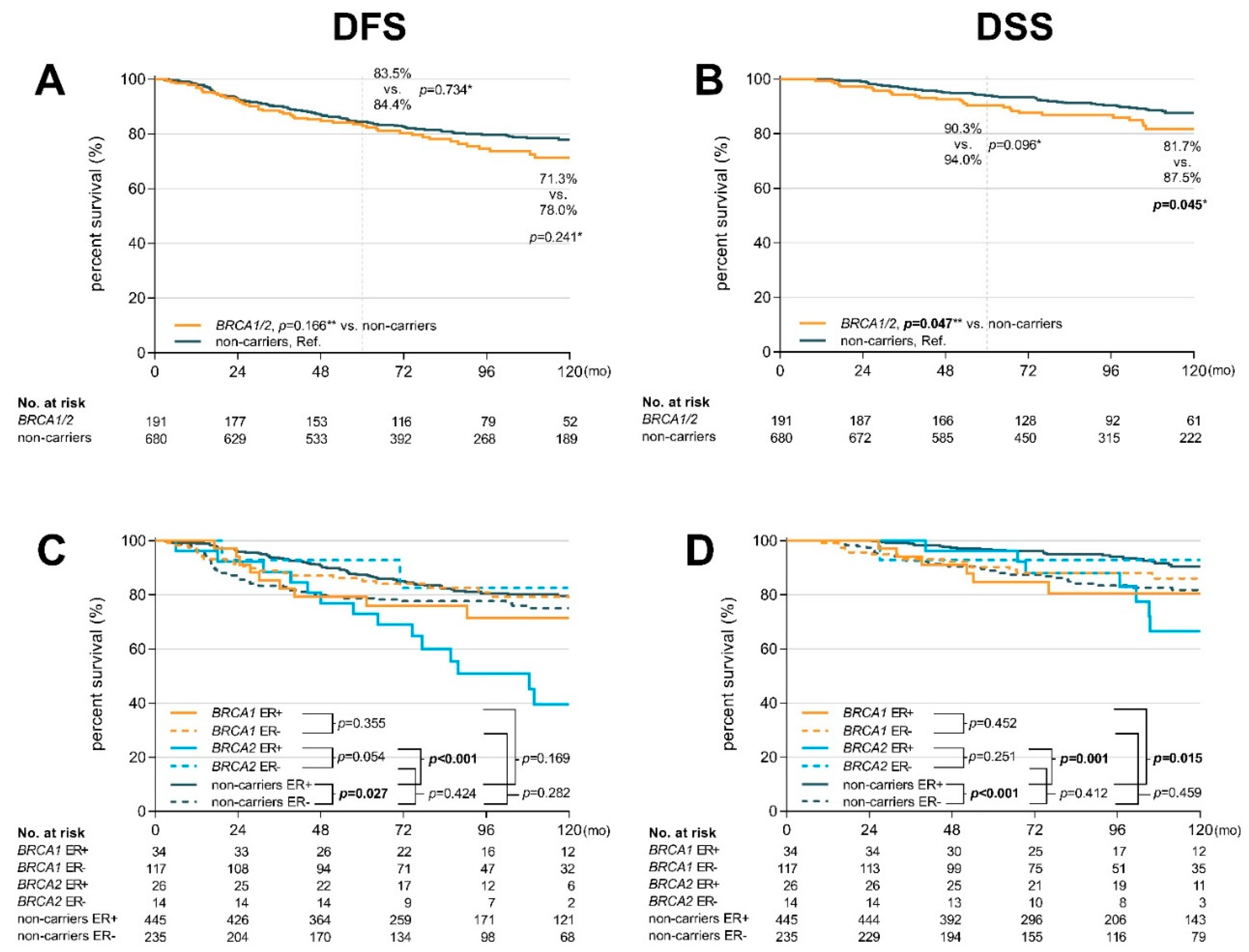
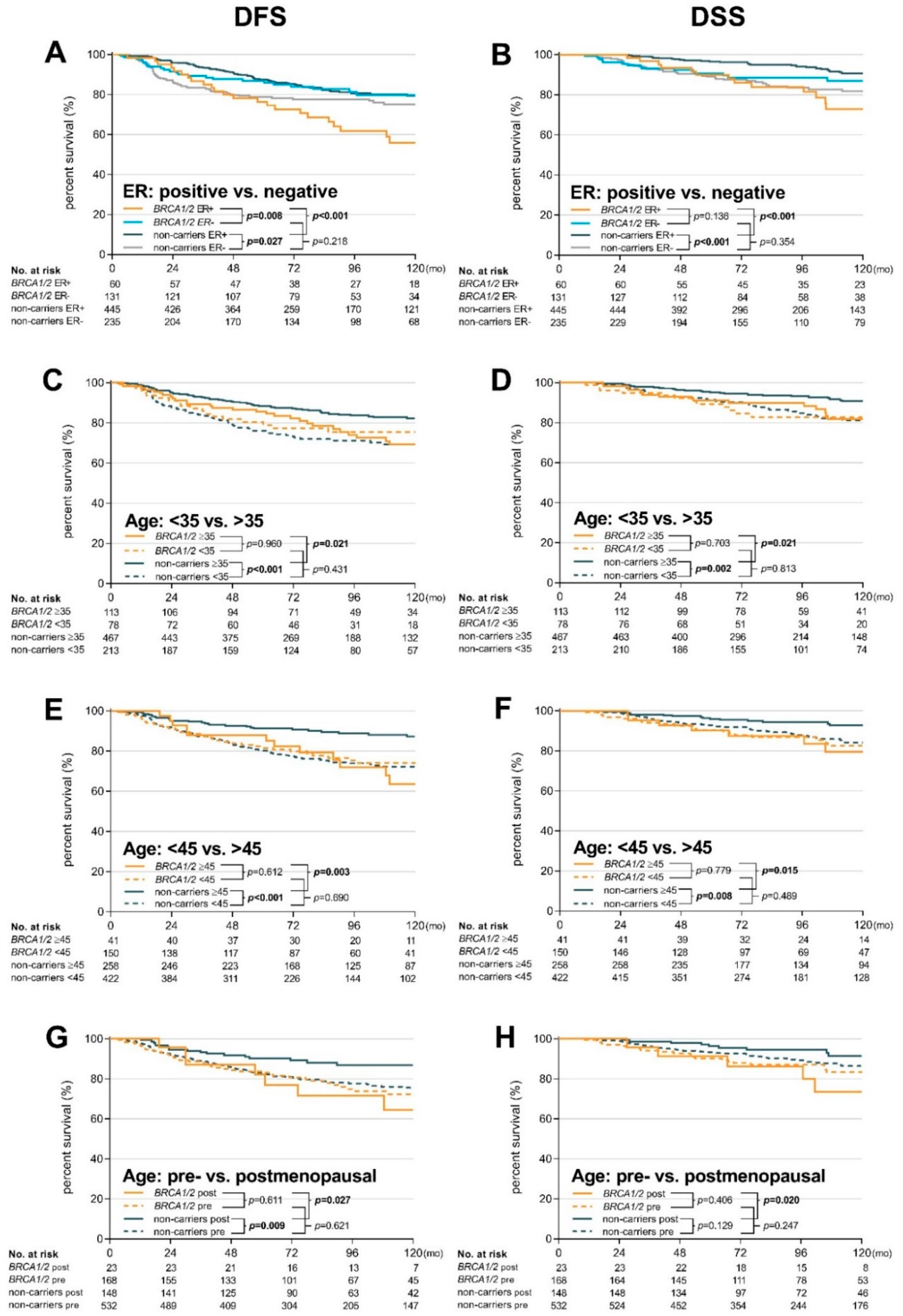
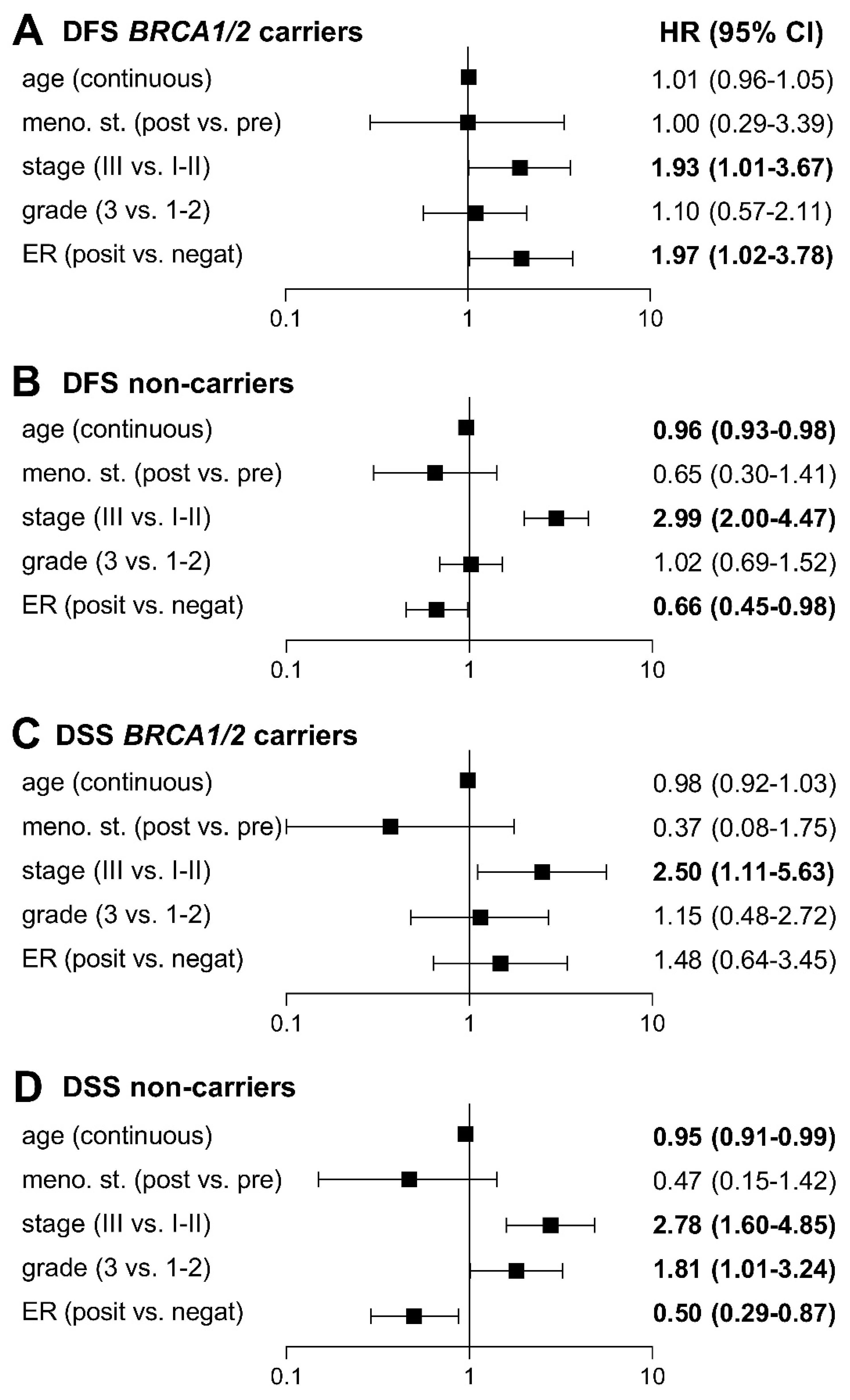
2.2. Prognosis and Long-Term Survival
3. Discussion
4. Materials and Methods
4.1. Patient Characteristics
4.2. Molecular Analysis
4.3. Statistical Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kwon, J.S.; Gutierrez-Barrera, A.M.; Young, D.; Sun, C.C.; Daniels, M.S.; Lu, K.H.; Arun, B. Expanding the criteria for BRCA mutation testing in breast cancer survivors. J. Clin. Oncol. 2010, 28, 4214–4220. [Google Scholar] [CrossRef] [PubMed]
- Begg, C.B.; Haile, R.W.; Borg, A.; Malone, K.E.; Concannon, P.; Thomas, D.C.; Langholz, B.; Bernstein, L.; Olsen, J.H.; Lynch, C.F.; et al. Variation of breast cancer risk among BRCA1/2 carriers. JAMA 2008, 299, 194–201. [Google Scholar] [CrossRef]
- Szabo, C.I.; King, M.C. Population genetics of BRCA1 and BRCA2. Am. J. Hum. Genet. 1997, 60, 1013–1020. [Google Scholar] [PubMed]
- Pohlreich, P.; Zikan, M.; Stribrna, J.; Kleibl, Z.; Janatova, M.; Kotlas, J.; Zidovska, J.; Novotny, J.; Petruzelka, L.; Szabo, C.; et al. High proportion of recurrent germline mutations in the BRCA1 gene in breast and ovarian cancer patients from the Prague area. Breast Cancer Res. 2005, 7, R728–R736. [Google Scholar] [CrossRef] [PubMed]
- King, M.C.; Marks, J.H.; Mandell, J.B. New York Breast Cancer Study G. Breast and ovarian cancer risks due to inherited mutations in BRCA1 and BRCA2. Science 2003, 302, 643–646. [Google Scholar] [CrossRef] [PubMed]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.A.; Mooij, T.M.; Roos-Blom, M.J.; Jervis, S.; van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed]
- Kleibl, Z.; Kristensen, V.N. Women at high risk of breast cancer: Molecular characteristics, clinical presentation and management. Breast 2016, 28, 136–144. [Google Scholar] [CrossRef]
- Parker, J.S.; Mullins, M.; Cheang, M.C.; Leung, S.; Voduc, D.; Vickery, T.; Davies, S.; Fauron, C.; He, X.; Hu, Z.; et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J. Clin. Oncol. 2009, 27, 1160–1167. [Google Scholar] [CrossRef]
- Kennecke, H.; Yerushalmi, R.; Woods, R.; Cheang, M.C.; Voduc, D.; Speers, C.H.; Nielsen, T.O.; Gelmon, K. Metastatic behavior of breast cancer subtypes. J. Clin. Oncol. 2010, 28, 3271–3277. [Google Scholar] [CrossRef]
- Huzarski, T.; Byrski, T.; Gronwald, J.; Górski, B.; Domagala, P.; Cybulski, C.; Oszurek, O.; Szwiec, M.; Gugala, K.; Stawicka, M.; et al. Ten-year survival in patients with BRCA1-negative and BRCA1-positive breast cancer. J. Clin. Oncol. 2013, 31, 3191–3196. [Google Scholar] [CrossRef]
- Spurdle, A.B.; Couch, F.J.; Parsons, M.T.; McGuffog, L.; Barrowdale, D.; Bolla, M.K.; Wang, Q.; Healey, S.; Schmutzler, R.; Wappenschmidt, B.; et al. Refined histopathological predictors of BRCA1 and BRCA2 mutation status: A large-scale analysis of breast cancer characteristics from the BCAC, CIMBA, and ENIGMA consortia. Breast Cancer Res. 2014, 16, 3419. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Meng, H.; Yao, L.; Lv, M.; Bai, J.; Zhang, J.; Wang, L.; Ouyang, T.; Li, J.; Wang, T.; et al. Germline Mutations in Cancer Susceptibility Genes in a Large Series of Unselected Breast Cancer Patients. Clin. Cancer Res. 2017, 23, 6113–6119. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.K.; van den Broek, A.J.; Tollenaar, R.A.; Smit, V.T.; Westenend, P.J.; Brinkhuis, M.; Oosterhuis, W.J.; Wesseling, J.; Janssen-Heijnen, M.L.; Jobsen, J.J.; et al. Breast Cancer Survival of BRCA1/BRCA2 Mutation Carriers in a Hospital-Based Cohort of Young Women. J. Natl. Cancer Inst. 2017, 109, djw329. [Google Scholar] [CrossRef] [PubMed]
- Hicks, D.G.; Short, S.M.; Prescott, N.L.; Tarr, S.M.; Coleman, K.A.; Yoder, B.J.; Crowe, J.P.; Choueiri, T.K.; Dawsonm, A.E.; Budd, G.T.; et al. Breast cancers with brain metastases are more likely to be estrogen receptor negative, express the basal cytokeratin CK5/6, and overexpress HER2 or EGFR. Am. J. Surg. Pathol. 2006, 30, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Lin, N.U.; Vanderplas, A.; Hughes, M.E.; Theriault, R.L.; Edge, S.B.; Wong, Y.N.; Blayney, D.W.; Niland, J.C.; Winer, E.P.; Weeks, J.C. Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network. Cancer 2012, 118, 5463–5472. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, J.G.; Stefansson, O.A.; Johannsson, O.T.; Sigurdsson, H.; Agnarsson, B.A.; Olafsdottir, G.H.; Alexiusdottir, K.K.; Stefansdottir, H.; Mitev, R.M.; Olafsdottir, K.; et al. Oestrogen receptor status, treatment and breast cancer prognosis in Icelandic BRCA2 mutation carriers. Br. J. Cancer 2016, 115, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Van den Broek, A.J.; Schmidt, M.K.; van ‘t Veer, L.J.; Tollenaar, R.A.; van Leeuwen, F.E. Worse breast cancer prognosis of BRCA1/BRCA2 mutation carriers: what’s the evidence? A systematic review with meta-analysis. PLoS One 2015, 10, e0120189. [Google Scholar] [CrossRef] [PubMed]
- Baretta, Z.; Mocellin, S.; Goldin, E.; Olopade, O.I.; Huo, D. Effect of BRCA germline mutations on breast cancer prognosis: A systematic review and meta-analysis. Medicine 2016, 95, e4975. [Google Scholar] [CrossRef]
- Copson, E.R.; Maishman, T.C.; Tapper, W.J.; Cutress, R.I.; Greville-Heygate, S.; Altman, D.G.; Eccles, B.; Gerty, S.; Durcan, L.T.; Jones, L.; et al. Germline BRCA mutation and outcome in young-onset breast cancer (POSH): A prospective cohort study. Lancet Oncol. 2018, 19, 169–180. [Google Scholar] [CrossRef]
- Rennert, G.; Bisland-Naggan, S.; Barnett-Griness, O.; Bar-Joseph, N.; Zhang, S.; Rennert, H.S.; Narod, S.A. Clinical outcomes of breast cancer in carriers of BRCA1 and BRCA2 mutations. N. Engl. J. Med. 2007, 357, 115–123. [Google Scholar] [CrossRef]
- Zhong, Q.; Peng, H.L.; Zhao, X.; Zhang, L.; Hwang, W.T. Effects of BRCA1- and BRCA2-related mutations on ovarian and breast cancer survival: A meta-analysis. Clin. Cancer Res. 2015, 21, 211–220. [Google Scholar] [CrossRef]
- Goodwin, P.J.; Phillips, K.A.; West, D.W.; Ennis, M.; Hopper, J.L.; John, E.M.; O’Malley, F.P.; Milne, R.L.; Andrulis, I.L.; Friedlander, M.L.; et al. Breast cancer prognosis in BRCA1 and BRCA2 mutation carriers: An International Prospective Breast Cancer Family Registry population-based cohort study. J. Clin. Oncol. 2012, 30, 19–26. [Google Scholar] [CrossRef]
- Carey, L.; Winer, E.; Viale, G.; Cameron, D.; Gianni, L. Triple-negative breast cancer: Disease entity or title of convenience? Nat. Rev. Clin. Oncol. 2010, 7, 683–692. [Google Scholar] [CrossRef]
- Sopik, V.; Sun, P.; Narod, S.A. The prognostic effect of estrogen receptor status differs for younger versus older breast cancer patients. Breast Cancer Res. Treat. 2017, 165, 391–402. [Google Scholar] [CrossRef] [PubMed]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Metcalfe, K.; Lynch, H.T.; Foulkes, W.D.; Tung, N.; Olopade, O.I.; Eisen, A.; Lerner-Ellis, J.; Snyder, C.; Kim, S.J.; Sun, P.; et al. Oestrogen receptor status and survival in women with BRCA2-associated breast cancer. Br. J. Cancer 2019, 120, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Lips, E.H.; Debipersad, R.D.; Scheerman, C.E.; Mulder, L.; Sonke, G.S.; van der Kolk, L.E.; Wesseling, J.; Hogervorst, F.B.; Nederlof, P.M. BRCA1-Mutated Estrogen Receptor-Positive Breast Cancer Shows BRCAness, Suggesting Sensitivity to Drugs Targeting Homologous Recombination Deficiency. Clin. Cancer Res. 2017, 23, 1236–1241. [Google Scholar] [CrossRef]
- Tung, N.; Wang, Y.; Collins, L.C.; Kaplan, J.; Li, H.; Gelman, R.; Comander, A.H.; Gallagher, B.; Fetten, K.; Krag, K.; et al. Estrogen receptor positive breast cancers in BRCA1 mutation carriers: Clinical risk factors and pathologic features. Breast Cancer Res. 2010, 12, R12. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bai, F.; Zhang, L.H.; Scott, A.; Li, E.; Pei, X.H. Estrogen promotes estrogen receptor negative BRCA1-deficient tumor initiation and progression. Breast Cancer Res. 2018, 20, 74. [Google Scholar] [CrossRef]
- Baek, H.J.; Kim, S.E.; Choi, E.K.; Kim, J.K.; Shin, D.H.; Park, E.J.; Kim, T.H.; Kim, J.Y.; Kim, K.G.; Deng, C.X.; et al. Inhibition of Estrogen Signaling Reduces the Incidence of BRCA1-associated Mammary Tumor Formation. Int. J. Biol. Sci. 2018, 14, 1755–1768. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.D.; Patil, S.; Dickler, M.N.; Offit, K.; Hudis, C.A.; Robson, M.E. Twenty-one-gene recurrence score assay in BRCA-associated versus sporadic breast cancers: Differences based on germline mutation status. Cancer 2016, 122, 1178–1184. [Google Scholar] [CrossRef]
- Pohlreich, P.; Stribrna, J.; Kleibl, Z.; Zikan, M.; Kalbacova, R.; Petruzelka, L.; Konopasek, B. Mutations of the BRCA1 gene in hereditary breast and ovarian cancer in the Czech Republic. Med. Princ. Pract. 2003, 12, 23–29. [Google Scholar] [CrossRef]
- Pohlreich, P.; Stribrna, J.; Ticha, I.; Soukupova, J.; Kleibl, Z.; Zikan, M.; Zimovjanova, M.; Kotlas, J.; Panczak, A. Predisposing genes in hereditary breast and ovarian cancer in the Czech Republic. Eur. J. Cancer Suppl. 2010, 8, 16. [Google Scholar] [CrossRef]
- Ticha, I.; Kleibl, Z.; Stribrna, J.; Kotlas, J.; Zimovjanova, M.; Mateju, M.; Zikan, M.; Pohlreich, P. Screening for genomic rearrangements in BRCA1 and BRCA2 genes in Czech high-risk breast/ovarian cancer patients: High proportion of population specific alterations in BRCA1 gene. Breast Cancer Res. Treat. 2010, 124, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Soukupova, J.; Zemankova, P.; Lhotova, K.; Janatova, M.; Borecka, M.; Stolarova, L.; Lhota, F.; Foretova, L.; Machackova, E.; Stranecky, V.; et al. Validation of CZECANCA (CZEch CAncer paNel for Clinical Application) for targeted NGS-based analysis of hereditary cancer syndromes. PLoS One 2018, 13, e0195761. [Google Scholar] [CrossRef] [PubMed]
- Lhota, F.; Zemankova, P.; Kleiblova, P.; Soukupova, J.; Vocka, M.; Stranecky, V.; Janatova, M.; Hartmannova, H.; Hodanova, K.; Kmoch, S.; et al. Hereditary truncating mutations of DNA repair and other genes in BRCA1/BRCA2/PALB2-negatively tested breast cancer patients. Clin. Genet. 2016, 90, 324–333. [Google Scholar] [CrossRef] [PubMed]
| BRCA1/2 Carriers (N = 191) | BRCA1 Carriers (N = 151) | BRCA2 Carriers (N = 40) | Non-Carriers (N = 680) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N | %* | p | N | %* | P | N | %* | p | N | %* | p | |
| Median age at diagnosis | ||||||||||||
| year (25–75% percentile) | 37.1 (32.3–43.8) | <0.001 | 36.9 (31.9–42.6) | <0.001 | 38.7 (32.9–50.8) | 0.645 | 40.2 (33.5–49.6) | Ref. | ||||
| Age diagnosis categories (known) | ||||||||||||
| <35 years | 78 | (40.8) | <0.001 | 64 | (42.4)) | <0.001 | 14 | (35.0) | 0.601 | 213 | (31.3) | Ref. |
| 35–44 years | 72 | (37.7) | 58 | (38.4) | 14 | (35.0) | 209 | (30.7) | ||||
| ≥45 years | 41 | (21.5) | 29 | (19.2) | 12 | (30.0) | 258 | (38.0) | ||||
| Menopausal status | ||||||||||||
| Pre | 168 | (88.0) | 0.003 | 136 | (90.1) | <0.001 | 32 | (80.0) | 0.792 | 532 | (78.2) | Ref. |
| post | 23 | (12.0) | 15 | (9.9) | 8 | (20.0) | 148 | (21.8) | ||||
| Primary tumor (T) | ||||||||||||
| T1 (<2 cm) | 71 | (37.2) | 0.001 | 52 | (34.4) | <0.001 | 19 | (47.5) | 0.093 | 355 | (52.2) | Ref. |
| T2 (2–5 cm) | 84 | (44.0) | 73 | (48.3) | 11 | (27.5) | 244 | (35.9) | ||||
| T3 (>5 cm) | 24 | (12.6) | 17 | (11.3) | 7 | (17.5) | 62 | (9.1) | ||||
| T4 | 12 | (6.3) | 9 | (6.0) | 3 | (7.5) | 19 | (2.8) | ||||
| Regional lymphatic node (N) | ||||||||||||
| N0 | 100 | (52.4) | 0.296 | 85 | (56.3) | 0.206 | 15 | (37.5) | 0.077 | 387 | (56.9) | Ref. |
| N1 | 78 | (40.8) | 55 | (36.4) | 23 | (57.5) | 256 | (37.6) | ||||
| N2 | 8 | (4.2) | 6 | (4.0) | 2 | (5.0) | 30 | (4.4) | ||||
| N3 | 5 | (2.6) | 5 | (3.3) | 0 | (0.0) | 7 | (1.0) | ||||
| Tumor stage | ||||||||||||
| I (T1N0–1mi) | 52 | (27.2) | <0.001 | 41 | (27.2) | 0.002 | 11 | (27.5) | 0.022 | 282 | (41.5) | Ref. |
| II (T2–3N0, T1–2N1) | 97 | (50.8) | 79 | (52.3) | 18 | (45.0) | 310 | (45.6) | ||||
| III (T3N1, TXN2–3, T4NX) | 42 | (22.0) | 31 | (20.5) | 11 | (27.5) | 88 | (12.9) | ||||
| Breast tumor morphology | ||||||||||||
| ductal | 168 | (88.0) | 0.023 | 131 | (86.8) | 0.005 | 37 | (92.5) | 0.325 | 574 | (84.4) | Ref. |
| lobular | 6 | (3.1) | 3 | (2.0) | 3 | (7.5) | 52 | (7.6) | ||||
| medullar | 15 | (7.9) | 15 | (9.9) | 0 | (0.0) | 33 | (4.9) | ||||
| other | 2 | (1.0) | 2 | (1.3) | 0 | (0.0) | 21 | (3.1) | ||||
| Grade | ||||||||||||
| low (1) | 7 | (3.7) | <0.001 | 4 | (2.6) | <0.001 | 3 | (7.5) | 0.554 | 91 | (13.4) | Ref. |
| intermediate (2) | 59 | (30.9) | 40 | (26.5) | 19 | (47.5) | 311 | (45.7) | ||||
| high (3) | 125 | (65.4) | 107 | (70.9) | 18 | (45.0) | 278 | (40.9) | ||||
| ER status | ||||||||||||
| positive | 60 | (31.4) | <0.001 | 34 | (22.5) | <0.001 | 26 | (65.0) | >0.99 | 445 | (65.4) | Ref. |
| PR status | ||||||||||||
| positive | 61 | (31.9) | <0.001 | 34 | (22.5) | <0.001 | 27 | (67.5) | 0.616 | 428 | (62.9) | Ref. |
| HER-2 status | ||||||||||||
| positive | 13 | (6.8) | <0.001 | 9 | (6.0) | <0.001 | 4 | (10.0) | 0.052 | 164 | (24.1) | Ref. |
| TNBC | ||||||||||||
| yes | 114 | (59.7) | <0.001 | 105 | (69.5) | <0.001 | 9 | (22.5) | 0.690 | 138 | (20.3) | Ref. |
| Surgery—primary tumor | ||||||||||||
| mastectomy | 91 | (47.6) | 0.774 | 70 | (46.4) | 0.980 | 21 | (52.5) | 0.458 | 316 | (46.5) | Ref. |
| breast-conserving surgery | 100 | (52.4) | 81 | (53.6) | 19 | (47.5) | 364 | (53.5) | ||||
| Surgery—lymphatic nodes | ||||||||||||
| axillary dissection | 149 | (78.0) | 0.051 | 117 | (77.5) | 0.102 | 32 | (80.0) | 0.215 | 482 | (70.9) | Ref. |
| sentinel node biopsy | 42 | (22.0) | 34 | (22.5) | 8 | (20.0) | 198 | (29.1) | ||||
| Radiotherapy | ||||||||||||
| yes | 132 | (69.1) | 0.759 | 108 | (71.5) | 0.391 | 24 | (60.0) | 0.297 | 462 | (67.9) | Ref. |
| Chemotherapy type | ||||||||||||
| Antra + Tax | 122 | (63.9) | <0.001 | 97 | (64.2) | <0.001 | 25 | (62.5) | 0.096 | 308 | (45.3) | Ref. |
| Antra | 50 | (26.2) | 39 | (25.8) | 11 | (27.5) | 195 | (28.7) | ||||
| Other | 7 | (3.7) | 6 | (4.0) | 1 | (2.5) | 32 | (4.7) | ||||
| No chemotherapy | 12 | (6.3) | 9 | (6.0) | 3 | (7.5) | 145 | (21.3) | ||||
| Endocrine therapy ** | ||||||||||||
| TAM monotherapy | 21 | (35.0) | 0.951 | 14 | (41.2) | 0.947 | 7 | (26.9) | 0.745 | 165 | (37.1) | Ref. |
| AI monotherapy | 8 | (13.4) | 4 | (11.8) | 4 | (15.4) | 65 | (14.6) | ||||
| LHRH analogues + TAM | 29 | (48.3) | 15 | (44.1) | 14 | (53.8) | 204 | (45.8) | ||||
| LHRH analogues + AI | 2 | (3.3) | 1 | (2.9) | 1 | (3.9) | 11 | (2.5) | ||||
| Event during follow-up *** | ||||||||||||
| loco-regional recurrence | 5 | (2.6) | 0.036 | 5 | (3.3) | 0.122 | 0 | (0.0) | - | 45 | (6.6) | Ref. |
| distant metastasis | 41 | (21.5) | 0.001 | 25 | (16.6) | 0.150 | 16 | (40.0) | <0.001 | 83 | (12.2) | Ref. |
| second breast cancer | 24 | (12.6) | 0.009 | 34 | (22.5) | <0.001 | 9 | (22.5) | <0.001 | 46 | (6.8) | Ref. |
| second tumors | 7 | (3.7) | 0.322 | 10 | (6.6) | 0.570 | 2 | (5.0) | 0.905 | 37 | (5.4) | Ref. |
| Median of follow-up | ||||||||||||
| median (25–75% percentile) | 8.6 (6.0–12.1) | 0.235 | 8.2 (5.7–11.8) | 0.733 | 9.4 (6.9–13.4) | 0.031 | 8.2 (5.6–11.8) | Ref. | ||||
| Breast cancer related death | ||||||||||||
| yes | 28 | (14.7) | 0.507 | 16 | (10.6) | 0.655 | 12 | (30.0) | <0.001 | 64 | (9.4) | Ref. |
| Disease Free Survival (DFS) Analysis | Disease Specific Survival (DSS) Analysis | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BRCA1/2 Carriers | Non-Carriers | BRCA1/2 Carriers | Non-Carriers | ||||||||||||||||||
| Pts No. | Ev No. | Ev % | HR | 95% CI | p | Pts No. | Ev No. | Ev % | Pts No. | Ev No. | Ev % | HR | 95% CI | p | Pts No. | Ev No. | Ev % | ||||
| All pts | 191 | 46 | 24.1 | 1.29 | 0.90–1.84 | 0.166 | 680 | 128 | 18.8 | Ref. | 191 | 28 | 14.7 | 1.65 | 1.01–2.70 | 0.047 | 680 | 64 | 9.4 | Ref. | |
| Age at diagnosis | <35 | 78 | 18 | 23.1 | 0.82 | 0.50–1.35 | 0.431 | 213 | 60 | 28.2 | Ref. | 78 | 12 | 15.4 | 1.09 | 0.55–2.13 | 0.813 | 213 | 32 | 15.0 | Ref. |
| ≥35 | 113 | 28 | 24.8 | 1.81 | 1.10–2.99 | 0.021 | 467 | 68 | 14.6 | Ref. | 113 | 16 | 14.2 | 2.29 | 1.13–4.65 | 0.021 | 467 | 32 | 6.9 | Ref. | |
| <45 | 150 | 34 | 22.7 | 0.93 | 0.63–1.36 | 0.690 | 422 | 100 | 23.7 | Ref. | 150 | 21 | 14.0 | 1.21 | 0.71–2.05 | 0.489 | 422 | 49 | 11.6 | Ref. | |
| ≥45 | 41 | 12 | 29.3 | 3.98 | 1.62–9.81 | 0.003 | 258 | 28 | 10.9 | Ref. | 41 | 7 | 17.1 | 4.48 | 1.34–15.0 | 0.015 | 258 | 15 | 5.8 | Ref. | |
| Menopausal status | pre | 168 | 39 | 23.2 | 1.10 | 0.76–1.60 | 0.621 | 532 | 119 | 22.4 | Ref. | 168 | 23 | 13.7 | 1.36 | 0.81–2.29 | 0.247 | 532 | 55 | 10.3 | Ref. |
| post | 23 | 7 | 30.4 | 3.72 | 1.16–11.9 | 0.027 | 148 | 18 | 12.2 | Ref. | 23 | 5 | 21.7 | 5.95 | 1.32–26.8 | 0.020 | 148 | 9 | 6.1 | Ref. | |
| Tumor size | T1 | 71 | 11 | 15.5 | 1.23 | 0.61–2.47 | 0.569 | 355 | 45 | 12.7 | Ref. | 71 | 4 | 5.6 | 1.13 | 0.37–3.74 | 0.835 | 355 | 18 | 5.1 | Ref. |
| T2 | 84 | 20 | 23.8 | 1.07 | 0.63–1.81 | 0.802 | 244 | 52 | 21.3 | Ref. | 84 | 12 | 14.3 | 1.17 | 0.58–2.36 | 0.653 | 244 | 29 | 11.9 | Ref. | |
| T3 | 24 | 9 | 37.5 | 0.90 | 0.42–1.92 | 0.787 | 62 | 23 | 37.1 | Ref. | 24 | 7 | 29.2 | 1.30 | 0.50–3.41 | 0.594 | 62 | 13 | 21.0 | Ref. | |
| T4 | 12 | 6 | 50.0 | 1.42 | 0.47–4.29 | 0.530 | 19 | 8 | 42.1 | Ref. | 12 | 5 | 41.7 | 2.22 | 0.57–8.62 | 0.249 | 19 | 4 | 21.1 | Ref. | |
| Nodal status | N0 | 100 | 17 | 17.0 | 1.26 | 0.70–2.26 | 0.435 | 387 | 51 | 13.2 | Ref. | 100 | 11 | 11.0 | 2.55 | 1.06–6.12 | 0.037 | 387 | 19 | 4.9 | Ref. |
| N1 | 78 | 25 | 32.1 | 1.37 | 0.83–2.25 | 0.217 | 256 | 64 | 25.0 | Ref. | 78 | 15 | 19.2 | 1.42 | 0.74–2.72 | 0.285 | 256 | 38 | 14.8 | Ref. | |
| N2 | 8 | 2 | 25.0 | 0.72 | 0.18–2.90 | 0.647 | 30 | 9 | 30.0 | Ref. | 8 | 1 | 12.5 | 0.86 | 0.11–7.02 | 0.890 | 30 | 4 | 13.3 | Ref. | |
| N3 | 5 | 2 | 40.0 | 0.65 | 0.13–3.30 | 0.605 | 7 | 4 | 57.1 | Ref. | 5 | 1 | 20.0 | 0.40 | 0.05–3.07 | 0.382 | 7 | 3 | 42.9 | Ref. | |
| Tumor stage | I | 52 | 7 | 13.5 | 1.42 | 0.57–3.54 | 0.457 | 282 | 28 | 9.9 | Ref. | 52 | 2 | 3.8 | 1.35 | 0.25–7.31 | 0.730 | 282 | 8 | 2.8 | Ref. |
| II | 97 | 23 | 23.7 | 1.09 | 0.67–1.77 | 0.733 | 310 | 65 | 21.0 | Ref. | 97 | 15 | 15.5 | 1.26 | 0.67–2.37 | 0.472 | 310 | 37 | 11.9 | Ref. | |
| III | 42 | 16 | 38.1 | 0.89 | 0.50–1.60 | 0.699 | 88 | 35 | 39.8 | Ref. | 42 | 11 | 26.2 | 1.22 | 0.57–2.63 | 0.608 | 88 | 19 | 21.6 | Ref. | |
| Tumor grade | 1 | 7 | 2 | 28.6 | 9.83 | 0.77–125.4 | 0.079 | 91 | 6 | 6.6 | Ref. | 7 | 1 | 14.3 | - | - | - | 91 | 1 | 1.1 | Ref. |
| 2 | 59 | 16 | 27.1 | 1.44 | 0.78–2.66 | 0.240 | 311 | 59 | 19.0 | Ref. | 59 | 9 | 15.3 | 2.71 | 1.03–7.13 | 0.044 | 311 | 21 | 6.8 | Ref. | |
| 3 | 125 | 28 | 22.4 | 0.96 | 0.61–1.49 | 0.841 | 278 | 63 | 22.7 | Ref. | 125 | 18 | 14.4 | 0.94 | 0.54–1.62 | 0.824 | 278 | 42 | 15.1 | Ref. | |
| ER status | pos | 60 | 23 | 38.3 | 3.14 | 1.69–5.81 | <0.001 | 445 | 74 | 16.6 | Ref. | 60 | 13 | 21.7 | 5.70 | 2.27–14.4 | <0.001 | 445 | 28 | 6.3 | Ref. |
| neg | 131 | 23 | 17.6 | 0.75 | 0.47–1.19 | 0.218 | 235 | 54 | 23.0 | Ref. | 131 | 15 | 11.5 | 0.76 | 0.43–1.35 | 0.354 | 235 | 36 | 15.3 | Ref. | |
| PR status | pos | 62 | 22 | 35.5 | 2.52 | 1.38–4.60 | 0.003 | 428 | 76 | 17.8 | Ref. | 62 | 10 | 16.1 | 2.85 | 1.44–7.14 | 0.026 | 428 | 30 | 7.0 | Ref. |
| neg | 129 | 24 | 18.6 | 0.87 | 0.54–1.40 | 0.566 | 252 | 52 | 20.6 | Ref. | 129 | 18 | 14.0 | 1.02 | 0.57–1.80 | 0.954 | 252 | 34 | 13.5 | Ref. | |
| HER-2 status | pos | 13 | 4 | 30.8 | 1.68 | 0.49–5.78 | 0.413 | 164 | 34 | 20.7 | Ref. | 13 | 2 | 15.4 | 1.44 | 0.27–7.60 | 0.668 | 164 | 19 | 11.6 | Ref. |
| neg | 129 | 24 | 18.6 | 0.87 | 0.54–1.40 | 0.566 | 252 | 52 | 20.6 | Ref. | 129 | 18 | 14.0 | 1.02 | 0.57–1.80 | 0.954 | 252 | 34 | 13.5 | Ref. | |
| TNBC | yes | 114 | 20 | 17.5 | 0.77 | 0.44–1.35 | 0.367 | 138 | 30 | 21.7 | Ref. | 114 | 14 | 12.3 | 0.78 | 0.40–1.53 | 0.475 | 138 | 21 | 15.2 | Ref. |
| no | 77 | 26 | 33.8 | 2.27 | 1.32–3.88 | 0.003 | 542 | 98 | 18.1 | Ref. | 77 | 14 | 18.2 | 2.92 | 1.34–6.38 | 0.007 | 542 | 43 | 7.9 | Ref. | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vocka, M.; Zimovjanova, M.; Bielcikova, Z.; Tesarova, P.; Petruzelka, L.; Mateju, M.; Krizova, L.; Kotlas, J.; Soukupova, J.; Janatova, M.; et al. Estrogen Receptor Status Oppositely Modifies Breast Cancer Prognosis in BRCA1/BRCA2 Mutation Carriers Versus Non-Carriers. Cancers 2019, 11, 738. https://doi.org/10.3390/cancers11060738
Vocka M, Zimovjanova M, Bielcikova Z, Tesarova P, Petruzelka L, Mateju M, Krizova L, Kotlas J, Soukupova J, Janatova M, et al. Estrogen Receptor Status Oppositely Modifies Breast Cancer Prognosis in BRCA1/BRCA2 Mutation Carriers Versus Non-Carriers. Cancers. 2019; 11(6):738. https://doi.org/10.3390/cancers11060738
Chicago/Turabian StyleVocka, Michal, Martina Zimovjanova, Zuzana Bielcikova, Petra Tesarova, Lubos Petruzelka, Martin Mateju, Ludmila Krizova, Jaroslav Kotlas, Jana Soukupova, Marketa Janatova, and et al. 2019. "Estrogen Receptor Status Oppositely Modifies Breast Cancer Prognosis in BRCA1/BRCA2 Mutation Carriers Versus Non-Carriers" Cancers 11, no. 6: 738. https://doi.org/10.3390/cancers11060738
APA StyleVocka, M., Zimovjanova, M., Bielcikova, Z., Tesarova, P., Petruzelka, L., Mateju, M., Krizova, L., Kotlas, J., Soukupova, J., Janatova, M., Zemankova, P., Kleiblova, P., Novotny, J., Konopasek, B., Chodacka, M., Brychta, M., Sochor, M., Smejkalova-Musilova, D., Cmejlova, V., ... Kleibl, Z. (2019). Estrogen Receptor Status Oppositely Modifies Breast Cancer Prognosis in BRCA1/BRCA2 Mutation Carriers Versus Non-Carriers. Cancers, 11(6), 738. https://doi.org/10.3390/cancers11060738





