Chronic Stability of Local Field Potentials Using Amorphous Silicon Carbide Microelectrode Arrays Implanted in the Rat Motor Cortex
Abstract
1. Introduction
2. Materials and Methods
2.1. Devices
2.2. Surgical Implantation of Neural Devices
2.3. Recording
2.4. Electrochemical Assessment
2.5. Data Processing
2.6. Statistical Methods
2.7. Immunohistochemistry
3. Results
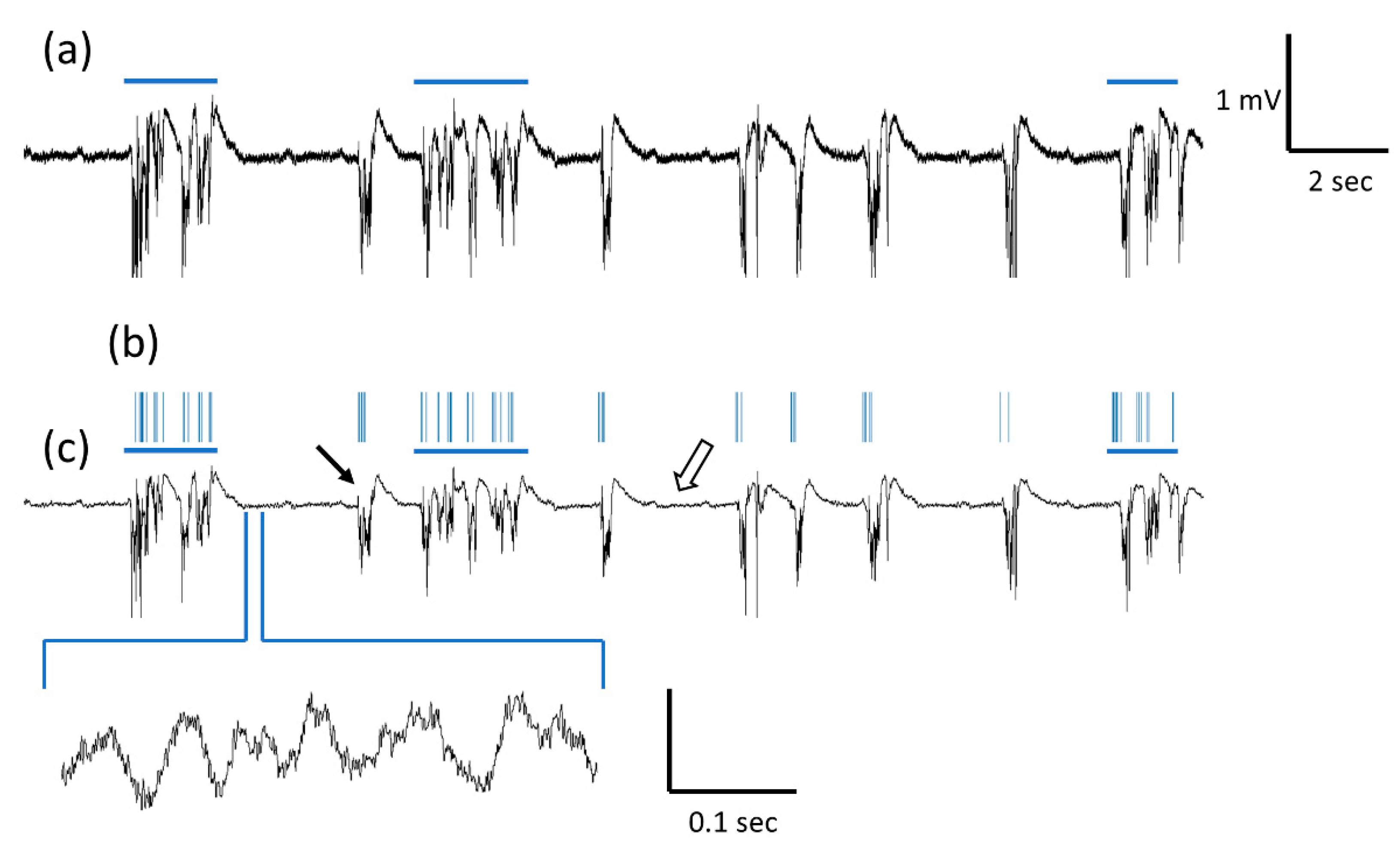
3.1. LFP Recording and Spectrogram
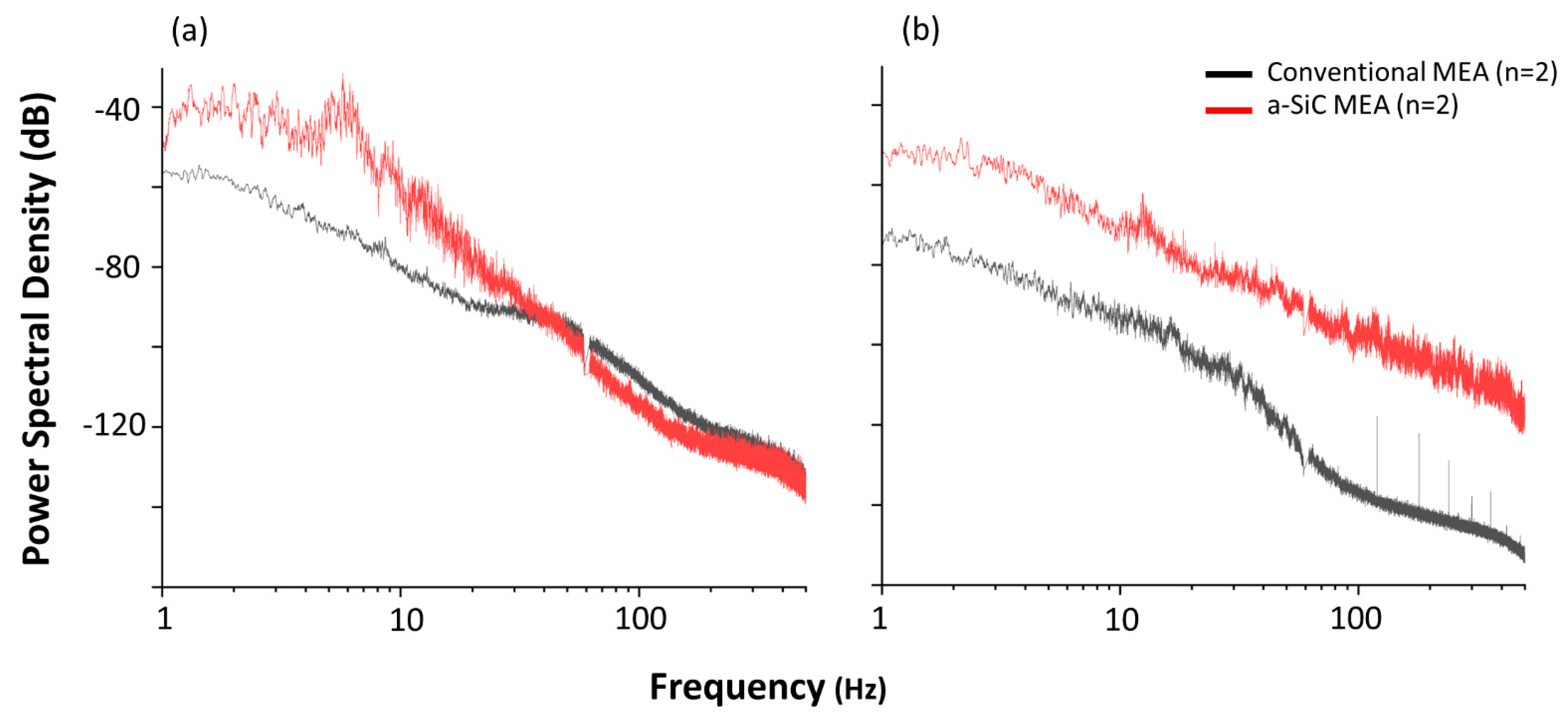
3.2. Power Spectral Density
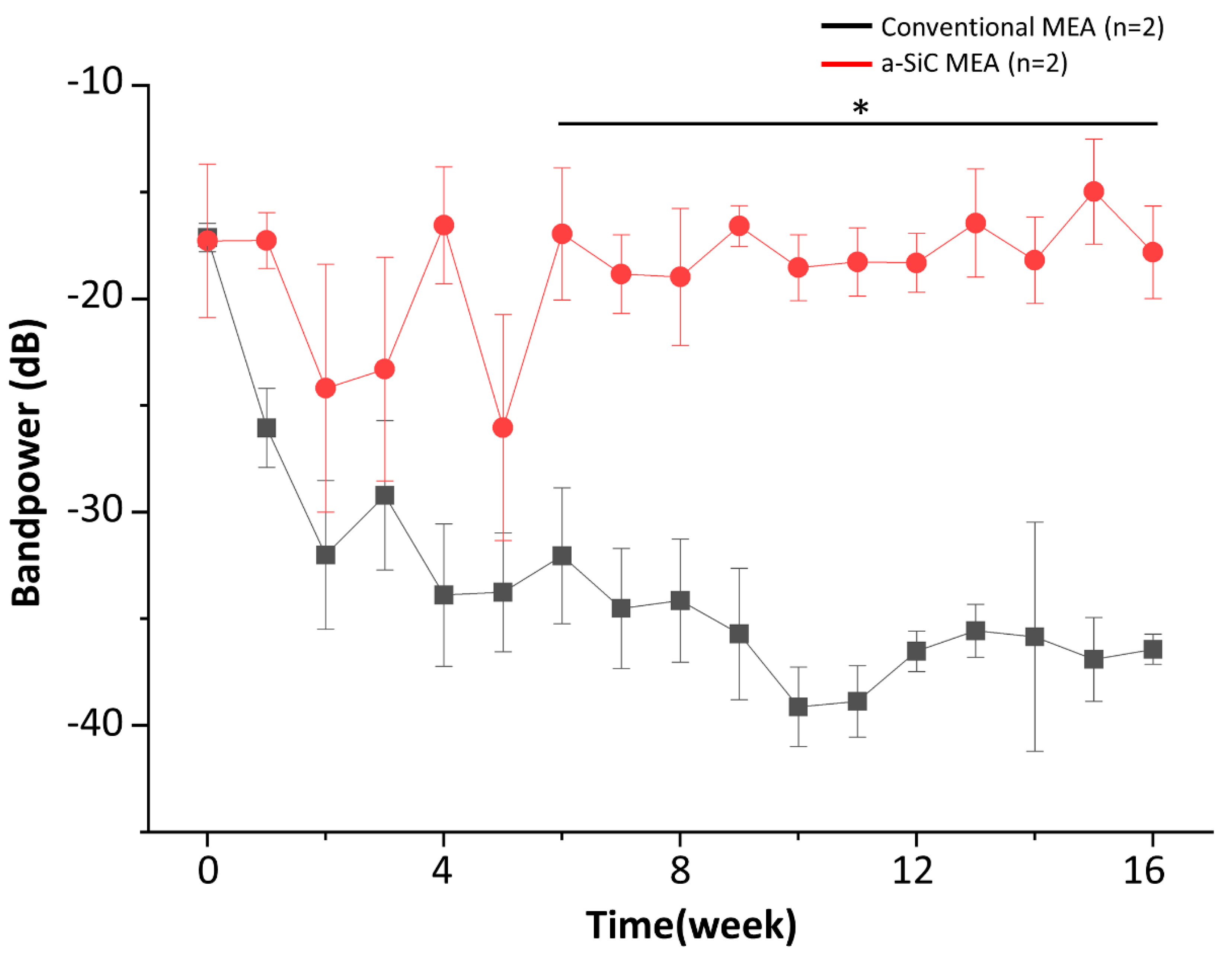
3.3. Bandpower
3.4. Awake LFP Recordings
3.5. Immunohistochemistry
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lebedev, M.A.; Nicolelis, M.A. Brain-machine interfaces: Past, present and future. Trends Neurosci. 2006, 29, 536–546. [Google Scholar] [CrossRef]
- Schwartz, A.B.; Cui, X.T.; Weber, D.J.; Moran, D.W. Brain-controlled interfaces: Movement restoration with neural prosthetics. Neuron 2006, 52, 205–220. [Google Scholar] [CrossRef]
- Andersen, R.A.; Musallam, S.; Pesaran, B. Selecting the signals for a brain-machine interface. Curr. Opin. Neurobiol. 2004, 14, 720–726. [Google Scholar] [CrossRef]
- Rapeaux, A.B.; Constandinou, T.G. Implantable brain machine interfaces: First-in-human studies, technology challenges and trends. Curr. Opin. Biotechnol. 2021, 72, 102–111. [Google Scholar] [CrossRef]
- Slutzky, M.W. Brain-Machine Interfaces: Powerful Tools for Clinical Treatment and Neuroscientific Investigations. Neuroscientist 2019, 25, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Polikov, V.S.; Tresco, P.A.; Reichert, W.M. Response of brain tissue to chronically implanted neural electrodes. J. Neurosci. Methods 2005, 148, 1–18. [Google Scholar] [CrossRef]
- Ferguson, M.; Sharma, D.; Ross, D.; Zhao, F. A Critical Review of Microelectrode Arrays and Strategies for Improving Neural Interfaces. Adv. Healthc. Mater. 2019, 8, e1900558. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T.D.; Jaquins-Gerstl, A.S.; Vazquez, A.L.; Michael, A.C.; Cui, X.T. Brain tissue responses to neural implants impact signal sensitivity and intervention strategies. ACS Chem. Neurosci. 2015, 6, 48–67. [Google Scholar] [CrossRef] [PubMed]
- Freire, M.A.; Morya, E.; Faber, J.; Santos, J.R.; Guimaraes, J.S.; Lemos, N.A.; Sameshima, K.; Pereira, A.; Ribeiro, S.; Nicolelis, M.A. Comprehensive analysis of tissue preservation and recording quality from chronic multielectrode implants. PLoS ONE 2011, 6, e27554. [Google Scholar] [CrossRef] [PubMed]
- Luan, L.; Robinson, J.T.; Aazhang, B.; Chi, T.; Yang, K.; Li, X.; Rathore, H.; Singer, A.; Yellapantula, S.; Fan, Y.; et al. Recent Advances in Electrical Neural Interface Engineering: Minimal Invasiveness, Longevity, and Scalability. Neuron 2020, 108, 302–321. [Google Scholar] [CrossRef]
- Usoro, J.O.; Sturgill, B.S.; Musselman, K.C.; Capadona, J.R.; Pancrazio, J.J. Intracortical Microelectrode Array Unit Yield under Chronic Conditions: A Comparative Evaluation. Micromachines 2021, 12, 972. [Google Scholar] [CrossRef]
- Scheid, M.R.; Flint, R.D.; Wright, Z.A.; Slutzky, M.W. Long-term, stable behavior of local field potentials during brain machine interface use. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2013, 2013, 307–310. [Google Scholar] [CrossRef]
- Andersen, R.A.; Hwang, E.J.; Mulliken, G.H. Cognitive neural prosthetics. Annu. Rev. Psychol. 2010, 61, 169–190. [Google Scholar] [CrossRef]
- Ray, S. Challenges in the quantification and interpretation of spike-LFP relationships. Curr. Opin. Neurobiol. 2015, 31, 111–118. [Google Scholar] [CrossRef]
- Einevoll, G.T.; Kayser, C.; Logothetis, N.K.; Panzeri, S. Modelling and analysis of local field potentials for studying the function of cortical circuits. Nat. Rev. Neurosci. 2013, 14, 770–785. [Google Scholar] [CrossRef] [PubMed]
- Waldert, S.; Pistohl, T.; Braun, C.; Ball, T.; Aertsen, A.; Mehring, C. A review on directional information in neural signals for brain-machine interfaces. J. Physiol. Paris. 2009, 103, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Prakash, S.S.; Mayo, J.P.; Ray, S. Decoding of attentional state using local field potentials. Curr. Opin. Neurobiol. 2022, 76, 102589. [Google Scholar] [CrossRef] [PubMed]
- Kajikawa, Y.; Schroeder, C.E. How local is the local field potential? Neuron 2011, 72, 847–858. [Google Scholar] [CrossRef]
- Buzsaki, G.; Anastassiou, C.A.; Koch, C. The origin of extracellular fields and currents--EEG, ECoG, LFP and spikes. Nat. Rev. Neurosci. 2012, 13, 407–420. [Google Scholar] [CrossRef]
- Friston, K.J.; Bastos, A.M.; Pinotsis, D.; Litvak, V. LFP and oscillations-what do they tell us? Curr. Opin. Neurobiol. 2015, 31, 1–6. [Google Scholar] [CrossRef]
- Abrego, A.M.; Khan, W.; Wright, C.E.; Islam, M.R.; Ghajar, M.H.; Bai, X.; Tandon, N.; Seymour, J.P. Sensing local field potentials with a directional and scalable depth electrode array. J. Neural Eng. 2023, 20, 016041. [Google Scholar] [CrossRef] [PubMed]
- Mirfathollahi, A.; Ghodrati, M.T.; Shalchyan, V.; Daliri, M.R. Decoding locomotion speed and slope from local field potentials of rat motor cortex. Comput. Methods Programs Biomed. 2022, 223, 106961. [Google Scholar] [CrossRef]
- Huang, Y.; Shen, X.; Zhang, X.; Chen, S.; Wang, Y. Decoding Reward Information from Local Field Potential and Spikes in Medial Prefrontal Cortex of Rats. In Proceedings of the IEEE International Conference on Systems, Man, and Cybernetics, Toronto, ON, Canada, 11–14 October 2020. [Google Scholar]
- Perge, J.A.; Zhang, S.; Malik, W.Q.; Homer, M.L.; Cash, S.; Friehs, G.; Eskandar, E.N.; Donoghue, J.P.; Hochberg, L.R. Reliability of directional information in unsorted spikes and local field potentials recorded in human motor cortex. J. Neural Eng. 2014, 11, 046007. [Google Scholar] [CrossRef]
- Paulk, A.C.; Kfir, Y.; Khanna, A.R.; Mustroph, M.L.; Trautmann, E.M.; Soper, D.J.; Stavisky, S.D.; Welkenhuysen, M.; Dutta, B.; Shenoy, K.V.; et al. Large-scale neural recordings with single neuron resolution using Neuropixels probes in human cortex. Nat. Neurosci. 2022, 25, 252–263. [Google Scholar] [CrossRef] [PubMed]
- Pancrazio, J.J.; Cogan, S.F. Editorial for the Special Issue on Neural Electrodes: Design and Applications. Micromachines 2019, 10, 466. [Google Scholar] [CrossRef]
- Gulino, M.; Kim, D.; Pane, S.; Santos, S.D.; Pego, A.P. Tissue Response to Neural Implants: The Use of Model Systems Toward New Design Solutions of Implantable Microelectrodes. Front. Neurosci. 2019, 13, 689. [Google Scholar] [CrossRef]
- Seymour, J.P.; Kipke, D.R. Fabrication of polymer neural probes with sub-cellular features for reduced tissue encapsulation. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2006, 2006, 4606–4609. [Google Scholar] [CrossRef]
- Skousen, J.L.; Merriam, S.M.; Srivannavit, O.; Perlin, G.; Wise, K.D.; Tresco, P.A. Reducing surface area while maintaining implant penetrating profile lowers the brain foreign body response to chronically implanted planar silicon microelectrode arrays. Prog. Brain Res. 2011, 194, 167–180. [Google Scholar] [CrossRef]
- Sommakia, S.; Lee, H.C.; Gaire, J.; Otto, K.J. Materials approaches for modulating neural tissue responses to implanted microelectrodes through mechanical and biochemical means. Curr. Opin. Solid. State Mater. Sci. 2014, 18, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Knaack, G.L.; McHail, D.G.; Borda, G.; Koo, B.; Peixoto, N.; Cogan, S.F.; Dumas, T.C.; Pancrazio, J.J. In vivo Characterization of Amorphous Silicon Carbide As a Biomaterial for Chronic Neural Interfaces. Front. Neurosci. 2016, 10, 301. [Google Scholar] [CrossRef]
- Cogan, S.F.; Edell, D.J.; Guzelian, A.A.; Liu, Y.P.; Edell, R. Plasma-enhanced chemical vapor deposited silicon carbide as an implantable dielectric coating. J. Biomed. Mater. Res. A 2003, 67, 856–867. [Google Scholar] [CrossRef]
- Ghazavi, A.; Gonzalez-Gonzalez, M.A.; Romero-Ortega, M.I.; Cogan, S.F. Intraneural ultramicroelectrode arrays for function-specific interfacing to the vagus nerve. Biosens. Bioelectron. 2020, 170, 112608. [Google Scholar] [CrossRef] [PubMed]
- Deku, F. Neural interfaces based on amorphous silicon carbide ultramicroelectrode arrays. Bioelectron. Med. 2018, 1, 185–200. [Google Scholar] [CrossRef]
- Deku, F.; Frewin, C.L.; Stiller, A.; Cohen, Y.; Aqeel, S.; Joshi-Imre, A.; Black, B.; Gardner, T.J.; Pancrazio, J.J.; Cogan, S.F. Amorphous Silicon Carbide Platform for Next Generation Penetrating Neural Interface Designs. Micromachines 2018, 9, 480. [Google Scholar] [CrossRef] [PubMed]
- Black, B.J.; Kanneganti, A.; Joshi-Imre, A.; Rihani, R.; Chakraborty, B.; Abbott, J.; Pancrazio, J.J.; Cogan, S.F. Chronic recording and electrochemical performance of Utah microelectrode arrays implanted in rat motor cortex. J. Neurophysiol. 2018, 120, 2083–2090. [Google Scholar] [CrossRef] [PubMed]
- Joshi-Imre, A.; Black, B.J.; Abbott, J.; Kanneganti, A.; Rihani, R.; Chakraborty, B.; Danda, V.R.; Maeng, J.; Sharma, R.; Rieth, L.; et al. Chronic recording and electrochemical performance of amorphous silicon carbide-coated Utah electrode arrays implanted in rat motor cortex. J. Neural Eng. 2019, 16, 046006. [Google Scholar] [CrossRef]
- Huang, S.H.; Shmoel, N.; Jankowski, M.M.; Erez, H.; Sharon, A.; Abu-Salah, W.; Nelken, I.; Weiss, A.; Spira, M.E. Immunohistological and Ultrastructural Study of the Inflammatory Response to Perforated Polyimide Cortical Implants: Mechanisms Underlying Deterioration of Electrophysiological Recording Quality. Front. Neurosci. 2020, 14, 926. [Google Scholar] [CrossRef]
- Abbott, J.R.; Geramifard, N.; Hernandez-Reynoso, A.G.; Blum, K.P.; Wu, Y.; Miller, L.E.; Cogan, S.F. Force Insertion and Acute Electrochemical Measurements of Amorphous Silicon Carbide Microelectrode Arrays into Nonhuman Primate Motor Cortex. In Proceedings of the Neuroscience 2022, San Diego, CA, USA, 12–16 November 2022. [Google Scholar]
- Deku, F.; Cohen, Y.; Joshi-Imre, A.; Kanneganti, A.; Gardner, T.J.; Cogan, S.F. Amorphous silicon carbide ultramicroelectrode arrays for neural stimulation and recording. J. Neural Eng. 2018, 15, 016007. [Google Scholar] [CrossRef]
- Geramifard, N.; Dousti, B.; Nguyen, C.; Abbott, J.; Cogan, S.F.; Varner, V.D. Insertion mechanics of amorphous SiC ultra-micro scale neural probes. J. Neural Eng. 2022, 19, 026033. [Google Scholar] [CrossRef]
- Frederick, R.A.; Meliane, I.Y.; Joshi-Imre, A.; Troyk, P.R.; Cogan, S.F. Activated iridium oxide film (AIROF) electrodes for neural tissue stimulation. J. Neural Eng. 2020, 17, 056001. [Google Scholar] [CrossRef]
- Stiller, A.M.; Usoro, J.; Frewin, C.L.; Danda, V.R.; Ecker, M.; Joshi-Imre, A.; Musselman, K.C.; Voit, W.; Modi, R.; Pancrazio, J.J.; et al. Chronic Intracortical Recording and Electrochemical Stability of Thiol-ene/Acrylate Shape Memory Polymer Electrode Arrays. Micromachines 2018, 9, 500. [Google Scholar] [CrossRef] [PubMed]
- Barrese, J.C.; Aceros, J.; Donoghue, J.P. Scanning electron microscopy of chronically implanted intracortical microelectrode arrays in non-human primates. J. Neural Eng. 2016, 13, 026003. [Google Scholar] [CrossRef] [PubMed]
- Barrese, J.C.; Rao, N.; Paroo, K.; Triebwasser, C.; Vargas-Irwin, C.; Franquemont, L.; Donoghue, J.P. Failure mode analysis of silicon-based intracortical microelectrode arrays in non-human primates. J. Neural Eng. 2013, 10, 066014. [Google Scholar] [CrossRef]
- Cogan, S.F. Neural Stimulation and Recording Electrodes. Annu. Rev. Biomed. Eng. 2008, 10, 275–309. [Google Scholar] [CrossRef]
- Usoro, J.O. Chronic stability of local field potentials from standard and modified Blackrock microelectrode arrays implanted in the rat motor cortex. Biomed. Phys. Eng. Express 2019, 5, 065017. [Google Scholar] [CrossRef]
- Bokil, H.; Andrews, P.; Kulkarni, J.E.; Mehta, S.; Mitra, P.P. Chronux: A platform for analyzing neural signals. J. Neurosci. Methods 2010, 192, 146–151. [Google Scholar] [CrossRef]
- Aksenov, D.P.; Miller, M.J.; Dixon, C.J.; Wyrwicz, A.M. The effect of sevoflurane and isoflurane anesthesia on single unit and local field potentials. Exp. Brain Res. 2019, 237, 1521–1529. [Google Scholar] [CrossRef]
- Priyanka, A.; Abhang, B.W.G.; Suresh, M. Technical Aspects of Brain Rhythms and Speech Parameters. In Introduction to EEG- and Speech-Based Emotion Recognition; Academic Press: Cambridge, MA, USA, 2016; pp. 16–50. [Google Scholar]
- Patel, P.R.; Zhang, H.; Robbins, M.T.; Nofar, J.B.; Marshall, S.P.; Kobylarek, M.J.; Kozai, T.D.; Kotov, N.A.; Chestek, C.A. Chronic in vivo stability assessment of carbon fiber microelectrode arrays. J. Neural Eng. 2016, 13, 066002. [Google Scholar] [CrossRef] [PubMed]
- Welle, E.J.; Patel, P.R.; Woods, J.E.; Petrossians, A.; Della Valle, E.; Vega-Medina, A.; Richie, J.M.; Cai, D.; Weiland, J.D.; Chestek, C.A. Ultra-small carbon fiber electrode recording site optimization and improved in vivo chronic recording yield. J. Neural Eng. 2020, 17, 026037. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.H.; Won, S.M.; Orsborn, A.L.; Yu, K.J.; Trumpis, M.; Bent, B.; Wang, C.; Xue, Y.; Min, S.; Woods, V.; et al. Development of a neural interface for high-definition, long-term recording in rodents and nonhuman primates. Sci. Transl. Med. 2020, 12, eaay4682. [Google Scholar] [CrossRef] [PubMed]
- Stiller, A.M.; Black, B.J.; Kung, C.; Ashok, A.; Cogan, S.F.; Varner, V.D.; Pancrazio, J.J. A Meta-Analysis of Intracortical Device Stiffness and Its Correlation with Histological Outcomes. Micromachines 2018, 9, 443. [Google Scholar] [CrossRef]
- Cody, P.A.; Eles, J.R.; Lagenaur, C.F.; Kozai, T.D.Y.; Cui, X.T. Unique electrophysiological and impedance signatures between encapsulation types: An analysis of biological Utah array failure and benefit of a biomimetic coating in a rat model. Biomaterials 2018, 161, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Potter, K.A.; Buck, A.C.; Self, W.K.; Capadona, J.R. Stab injury and device implantation within the brain results in inversely multiphasic neuroinflammatory and neurodegenerative responses. J. Neural Eng. 2012, 9, 046020. [Google Scholar] [CrossRef]
- Wellman, S.M.; Li, L.; Yaxiaer, Y.; McNamara, I.; Kozai, T.D.Y. Revealing Spatial and Temporal Patterns of Cell Death, Glial Proliferation, and Blood-Brain Barrier Dysfunction Around Implanted Intracortical Neural Interfaces. Front. Neurosci. 2019, 13, 493. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Wellman, S.M.; Yaxiaer, Y.; Eles, J.R.; Kozai, T.D. In vivo spatiotemporal patterns of oligodendrocyte and myelin damage at the neural electrode interface. Biomaterials 2021, 268, 120526. [Google Scholar] [CrossRef] [PubMed]
- Crunelli, V.; Hughes, S.W. The slow (<1 Hz) rhythm of non-REM sleep: A dialogue between three cardinal oscillators. Nat. Neurosci. 2010, 13, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Amzica, F.; Steriade, M. The K-complex: Its slow (<1-Hz) rhythmicity and relation to delta waves. Neurology 1997, 49, 952–959. [Google Scholar] [CrossRef]
- Wei, X.; Luan, L.; Zhao, Z.; Li, X.; Zhu, H.; Potnis, O.; Xie, C. Nanofabricated Ultraflexible Electrode Arrays for High-Density Intracortical Recording. Adv. Sci. 2018, 5, 1700625. [Google Scholar] [CrossRef]
- Tseng, A.A.; Kuan, C.; Chen, C.D.; Ma, K.J. Electron beam lithography in nanoscale fabrication: Recent development. IEEE Trans. Electron. Packag. Manuf. 2003, 26, 141–149. [Google Scholar] [CrossRef]
- Yoon, H.-W.; Shin, S.-M.; Kwon, S.-Y.; Cho, H.-M.; Kim, S.-G.; Hong, M.-P. One-Step Etching Characteristics of ITO/Ag/ITO Multilayered Electrode in High-Density and High-Electron-Temperature Plasma. Materials 2021, 14, 2025. [Google Scholar] [CrossRef]
- Kishimoto, K.-I.K.; Matsumoto, T.-C.H.; Okada, N.; Sumihiro, N.; Gomi, H. Stability and Application to Multilevel Metallization of Fluorine-Doped Silicon Oxide by High-Density Plasma Chemical Vapor Deposition. Jpn. Journal. Appl. Phys. 2000, 39, 1091. [Google Scholar] [CrossRef]
- Usoro, J.O.; Dogra, K.; Abbott, J.R.; Radhakrishna, R.; Cogan, S.F.; Pancrazio, J.J.; Patnaik, S.S. Influence of Implantation Depth on the Performance of Intracortical Probe Recording Sites. Micromachines 2021, 12, 1158. [Google Scholar] [CrossRef] [PubMed]


| Band | Frequencies (Hz) |
|---|---|
| Total LFP band | 1–500 |
| Delta | 1–4 |
| Theta | 4–8 |
| Alpha | 8–13 |
| Beta | 13–31 |
| Low gamma | 31–59 |
| High gamma | 61–100 |
| Band | Week 0 | Week 7 | Week 16 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| a-SiC | Conventional | p | a-SiC | Conventional | p | a-SiC | Conventional | p | |
| Total LFP (dB) | −20.7 ± 3.53 | −19.9 ± 2.86 | ns | −21.1 ± 3.2 | −34.1 ± 2.89 | * | −17.8 ± 2.17 | −36.4 ± 0.71 | * |
| Delta (dB) | −22.1 ± 3.53 | −22.3 ± 3.82 | ns | −19 ± 2.53 | −40.7 ± 3.84 | * | −19.9 ± 2.43 | −43.6 ± 1.58 | * |
| Theta | −29.2 ± 4.08 | −28.2 ± 3.08 | ns | −27.7 ± 4.6 | −43.3 ± 3.84 | * | −25.5 ± 2.15 | −45.6 ± 1.09 | * |
| Alpha (dB) | −33.5 ± 4.07 | −32.3 ± 3.21 | ns | −30.6 ± 5.13 | −44.2 ± 3.3 | * | −30.3 ± 1.17 | −46.7 ± 1.22 | * |
| Beta (dB) | −34.0 ± 3.33 | −34.9 ± 2.35 | ns | −31.8 ± 2.45 | −42.4 ± 3.12 | * | −30.3 ± 1.18 | −45.2 ± 1.4 | * |
| Low gamma (dB) | −42.2 ± 3.16 | −38.8 ± 2.11 | ns | −37.6 ± 2.17 | −46.3 ± 2.59 | * | −36.8 ± 0.7 | −50.3 ± 1.09 | * |
| High gamma (dB) | −50.7 ± 2.67 | −45.6 ± 1.06 | ns | −45.7 ± 1.41 | −51.9 ± 1.65 | * | −44.7 ± 1.25 | −54.3 ± 0.76 | * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeakle, E.N.; Abbott, J.R.; Usoro, J.O.; Wu, Y.; Haghighi, P.; Radhakrishna, R.; Sturgill, B.S.; Nakajima, S.; Thai, T.T.D.; Pancrazio, J.J.; et al. Chronic Stability of Local Field Potentials Using Amorphous Silicon Carbide Microelectrode Arrays Implanted in the Rat Motor Cortex. Micromachines 2023, 14, 680. https://doi.org/10.3390/mi14030680
Jeakle EN, Abbott JR, Usoro JO, Wu Y, Haghighi P, Radhakrishna R, Sturgill BS, Nakajima S, Thai TTD, Pancrazio JJ, et al. Chronic Stability of Local Field Potentials Using Amorphous Silicon Carbide Microelectrode Arrays Implanted in the Rat Motor Cortex. Micromachines. 2023; 14(3):680. https://doi.org/10.3390/mi14030680
Chicago/Turabian StyleJeakle, Eleanor N., Justin R. Abbott, Joshua O. Usoro, Yupeng Wu, Pegah Haghighi, Rahul Radhakrishna, Brandon S. Sturgill, Shido Nakajima, Teresa T. D. Thai, Joseph J. Pancrazio, and et al. 2023. "Chronic Stability of Local Field Potentials Using Amorphous Silicon Carbide Microelectrode Arrays Implanted in the Rat Motor Cortex" Micromachines 14, no. 3: 680. https://doi.org/10.3390/mi14030680
APA StyleJeakle, E. N., Abbott, J. R., Usoro, J. O., Wu, Y., Haghighi, P., Radhakrishna, R., Sturgill, B. S., Nakajima, S., Thai, T. T. D., Pancrazio, J. J., Cogan, S. F., & Hernandez-Reynoso, A. G. (2023). Chronic Stability of Local Field Potentials Using Amorphous Silicon Carbide Microelectrode Arrays Implanted in the Rat Motor Cortex. Micromachines, 14(3), 680. https://doi.org/10.3390/mi14030680







