Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review
Abstract
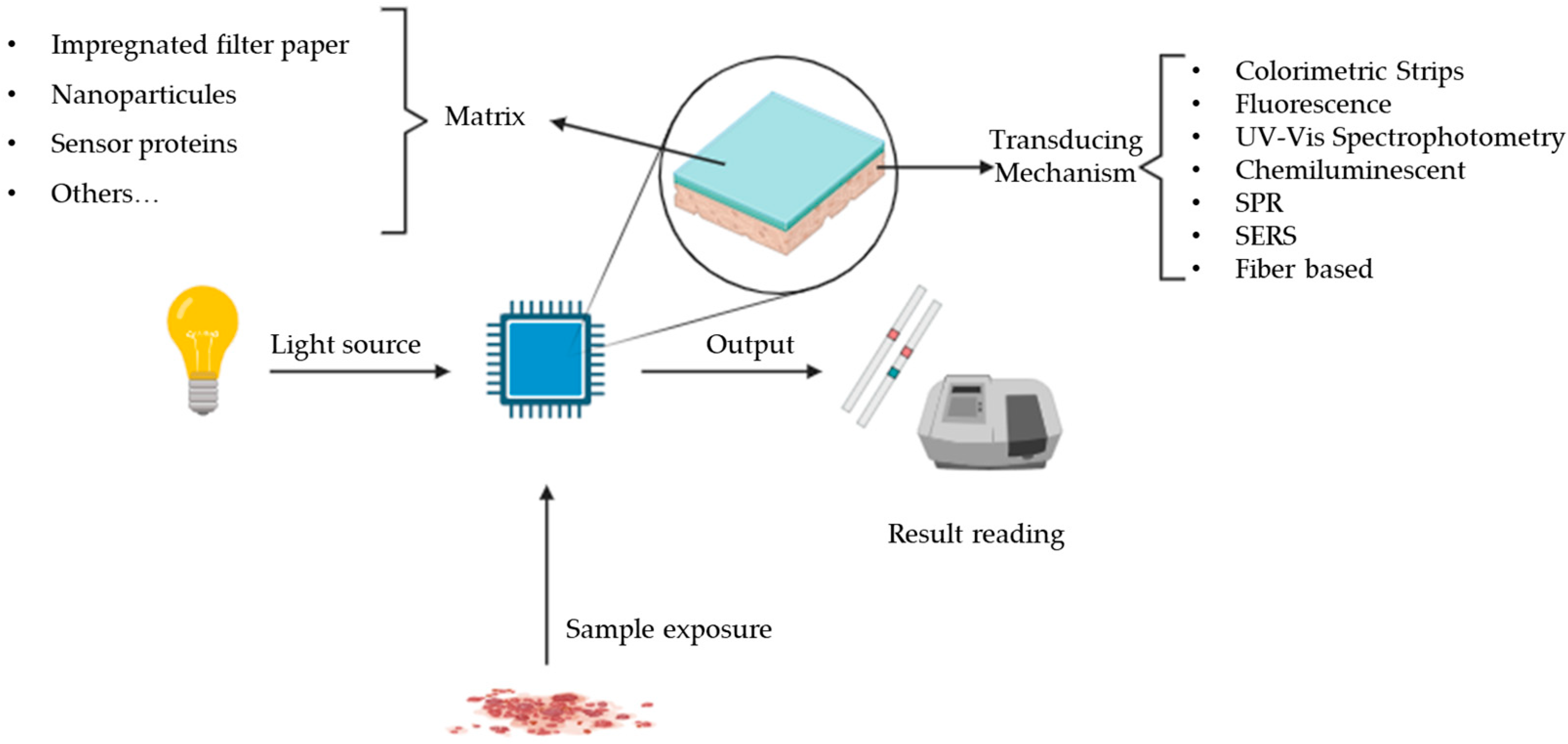
:1. Introduction
2. Classification
2.1. Blood and Derivatives
2.2. Urine
2.3. Oral Fluid
2.4. Sweat
2.5. Hair
2.6. Exhaled Air
2.7. Vapours
2.8. Illicit Drugs in Solid Samples
3. Future Perspectives
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- European Monitoring Center for Drugs and Drug Addiction. European Drug Report 2022: Trends and Developments; European Monitoring Center for Drugs and Drug Addiction: Lisbon, Portugal, 2022; pp. 207–221. [Google Scholar]
- UNODC. Executive summary. In Proceedings of the 2017 Transportation Association of Canada Conference and Exhibition, TAC 2017, St. John’s, NF, Canada, 24–27 September 2017. [Google Scholar]
- Dagar, M.; Yadav, S.; Sai, V.V.R.; Satija, J.; Bhatia, H. Emerging trends in point-of-care sensors for illicit drugs analysis. Talanta 2022, 238, 123048. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, M.; Janani, R.; Deepa, C.; Rajeshkumar, L. Nanotechnology-Enabled Biosensors: A Review of Fundamentals, Design Principles, Materials, and Applications. Biosensors 2023, 13, 40. [Google Scholar] [CrossRef]
- Khorablou, Z.; Shahdost-fard, F.; Razmi, H.; Yola, M.L.; Karimi-Maleh, H. Recent advances in developing optical and electrochemical sensors for analysis of methamphetamine: A review. Chemosphere 2021, 278, 130393. [Google Scholar] [CrossRef] [PubMed]
- Wani, A.K.; Akhtar, N.; Katoch, V.; Shukla, S.; Kadam, U.S.; Hong, J.C. Advancing biological investigations using portable sensors for detection of sensitive samples. Heliyon 2023, 9, e22679. [Google Scholar] [CrossRef]
- Mostafa, I.M.; Meng, C.; Dong, Z.; Lou, B.; Xu, G. Potentiometric sensors for the determination of pharmaceutical drugs. Anal. Sci. 2022, 38, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Razlansari, M.; Ulucan-Karnak, F.; Kahrizi, M.; Mirinejad, S.; Sargazi, S.; Mishra, S.; Rahdar, A.; Díez-Pascual, A.M. Nanobiosensors for detection of opioids: A review of latest advancements. Eur. J. Pharm. Biopharm. 2022, 179, 79–94. [Google Scholar] [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef]
- Prabowo, B.A.; Purwidyantri, A.; Liu, K.C. Surface Plasmon Resonance Optical Sensor: A Review on Light Source Technology. Biosensors 2018, 8, 80. [Google Scholar] [CrossRef]
- Wu, Y.; Feng, J.; Hu, G.; Zhang, E.; Yu, H.H. Colorimetric Sensors for Chemical and Biological Sensing Applications. Sensors 2023, 23, 2749. [Google Scholar] [CrossRef]
- Malik, S.; Singh, J.; Goyat, R.; Saharan, Y.; Chaudhry, V.; Umar, A.; Ibrahim, A.A.; Akbar, S.; Ameen, S.; Baskoutas, S. Nanomaterials-based biosensor and their applications: A review. Heliyon 2023, 9, e19929. [Google Scholar] [CrossRef]
- Munawar, A.; Ong, Y.; Schirhagl, R.; Tahir, M.A.; Khan, W.S.; Bajwa, S.Z. Nanosensors for diagnosis with optical, electric and mechanical transducers. RSC Adv. 2019, 9, 6793–6803. [Google Scholar] [CrossRef]
- De Campos, E.G.; da Costa, B.R.B.; dos Santos, F.S.; Monedeiro, F.; Alves, M.N.R.; Santos Junior, W.J.R.; De Martinis, B.S. Alternative matrices in forensic toxicology: A critical review. Forensic Toxicol. 2022, 40, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, E.; Queiroz, J.A. The role of alternative specimens in toxicological analysis. Biomed. Chromatogr. 2008, 22, 795–821. [Google Scholar] [CrossRef] [PubMed]
- Wilkirson, E.C.; Singampalli, K.L.; Li, J.; Dixit, D.D.; Jiang, X.; Gonzalez, D.H.; Lillehoj, P.B. Affinity-based electrochemical sensors for biomolecular detection in whole blood. Anal. Bioanal. Chem. 2023, 415, 3983–4002. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Chen, X.; Huang, J.; Zhang, X.; Sun, J.; Yang, L. Point-of-care testing of methylamphetamine with a portable optical fiber immunosensor. Anal. Chim. Acta 2022, 1192, 339345. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Wu, S.; Chen, Z.; Jiang, J.; Sun, J. Rapid nanomolar detection of methamphetamine in biofluids via a reagentless electrochemical aptamer-based biosensor. Anal. Chim. Acta 2022, 1207, 339742. [Google Scholar] [CrossRef]
- Karim-Nezhad, G.; Khorablou, Z.; Sadegh, B.; Mahmoudi, T. Electro-Polymerized Poly(L-Arginine) Film as An Efficient Electrode Modifier for Highly Sensitive Determination of Methadone in Real Samples. Anal. Bioanal. Electrochem. 2022, 14, 730–741. [Google Scholar]
- Baghayeri, M.; Nabavi, S.; Hasheminejad, E.; Ebrahimi, V. Introducing an Electrochemical Sensor Based on Two Layers of Ag Nanoparticles Decorated Graphene for Rapid Determination of Methadone in Human Blood Serum. Top. Catal. 2022, 65, 623–632. [Google Scholar] [CrossRef]
- Sanati, A.L.; Faridbod, F. A New Nanostructure Approach based on Pr(OH) 3 /GQD and Imidazolium Ionic Liquid for Voltammetric Analysis of Tramadol. Anal. Bioanal. Electrochem. 2022, 14, 510–522. [Google Scholar]
- Li, C.; Han, D.; Liang, Z.; Han, F.; Fu, W.; Wang, W.; Han, D.; Wang, Y.; Niu, L. Novel electrochemical-surface plasmon resonance (EC-SPR) sensor for amphetamine-type stimulants detection based on molecularly imprinted strategy. Sens. Actuators B Chem. 2022, 369, 132258. [Google Scholar] [CrossRef]
- Dokuzparmak, E.; Brown, K.; Dennany, L. Electrochemiluminescent screening for methamphetamine metabolites. Analyst 2021, 146, 3336–3345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Fu, K.; Zou, F.; Bai, H.; Zhang, G.; Liang, F.; Liu, Q. Highly sensitive electrochemical sensor based on Pt nanoparticles/carbon nanohorns for simultaneous determination of morphine and MDMA in biological samples. Electrochim. Acta 2021, 370, 137803. [Google Scholar] [CrossRef]
- Hassan, S.A.; ElDin, N.B.; Zaazaa, H.E.; Moustafa, A.A.; Mahmoud, A.M. Point-of-care diagnostics for drugs of abuse in biological fluids: Application of a microfabricated disposable copper potentiometric sensor. Microchim. Acta 2020, 187, 491. [Google Scholar] [CrossRef] [PubMed]
- Mynttinen, E.; Wester, N.; Lilius, T.; Kalso, E.; Mikladal, B.; Varjos, I.; Sainio, S.; Jiang, H.; Kauppinen, E.I.; Koskinen, J.; et al. Electrochemical Detection of Oxycodone and Its Main Metabolites with Nafion-Coated Single-Walled Carbon Nanotube Electrodes. Anal. Chem. 2020, 92, 8218–8227. [Google Scholar] [CrossRef]
- Xie, Y.; Lin, J.H.; Chen, L.Y.; Feng, L.; Chen, Z.M.; Zheng, J.X.; Qin, S.N.; Li, G.W.; Salminen, K.; Sun, J.J. Rapid nanomolar detection of ketamine in biofluids based on electrochemical aptamer-based sensor for drugged driving screening within 30 s. Sens. Actuators B Chem. 2023, 390, 133903. [Google Scholar] [CrossRef]
- Huang, L.; Li, T.; Zhang, Y.; Sun, X.; Wang, Y.; Nie, Z. Discrimination of narcotic drugs in human urine based on nanoplasmonics combined with chemometric method. J. Pharm. Biomed. Anal. 2020, 186, 113174. [Google Scholar] [CrossRef]
- González-Hernández, J.; Moya-Alvarado, G.; Alvarado-Gámez, A.L.; Urcuyo, R.; Barquero-Quirós, M.; Arcos-Martínez, M.J. Electrochemical biosensor for quantitative determination of fentanyl based on immobilized cytochrome c on multi-walled carbon nanotubes modified screen-printed carbon electrodes. Microchim. Acta 2022, 189, 483. [Google Scholar] [CrossRef]
- Lin, Y.; Sun, J.; Tang, M.; Zhang, G.; Yu, L.; Zhao, X.; Ai, R.; Yu, H.; Shao, B.; He, Y. Synergistic Recognition-Triggered Charge Transfer Enables Rapid Visual Colorimetric Detection of Fentanyl. Anal. Chem. 2021, 93, 6544–6550. [Google Scholar] [CrossRef]
- Li, C.; Han, D.; Wu, Z.; Liang, Z.; Han, F.; Chen, K.; Fu, W.; Han, D.; Wang, Y.; Niu, L. Polydopamine-based molecularly imprinted electrochemical sensor for the highly selective determination of ecstasy components. Analyst 2022, 147, 3291–3297. [Google Scholar] [CrossRef]
- Du, X.; Gu, H.; Liu, X.; Li, W.; Lv, S.; Ding, H.; Li, L.; Qiu, D.; Hao, H. Selective and reversible chemical sensor for methamphetamine detection using AIEgen and cucurbit[7]uril. Chin. J. Anal. Chem. 2023, 51, 100275. [Google Scholar] [CrossRef]
- Zhang, R.; Ren, Y.; Zhang, Q.; Huang, W.; Bai, H.; Zeng, X. Water-soluble pillar[5]arene-modified graphdiyne functional material and its application towards ultrasensitive and robust electrochemical methylamphetamine determination. New J. Chem. 2022, 46, 20909–20917. [Google Scholar] [CrossRef]
- Cho, S.; Kim, Y. Donor–acceptor Stenhouse adduct formation for the simple and rapid colorimetric detection of amphetamine-type stimulants. Sens. Actuators B Chem. 2022, 355, 131274. [Google Scholar] [CrossRef]
- Soni, S.; Jain, U.; Burke, D.H.; Chauhan, N. A label free, signal off electrochemical aptasensor for amphetamine detection. Surf. Interfaces 2022, 31, 102023. [Google Scholar] [CrossRef]
- Guo, G.; Wang, T.; Ding, X.; Wang, H.; Wu, Q.; Zhang, Z.; Ding, S.; Li, S.; Li, J. Fluorescent lanthanide metal-organic framework for rapid and ultrasensitive detection of methcathinone in human urine. Talanta 2022, 249, 123663. [Google Scholar] [CrossRef] [PubMed]
- Saichanapan, J.; Promsuwan, K.; Saisahas, K.; Soleh, A.; Chang, K.H.; Abdullah, A.F.L.; Limbut, W. Voltammetric Determination of Tramadol Using a Hierarchical Graphene Oxide Nanoplatelets Modified Electrode. J. Electrochem. Soc. 2021, 168, 117512. [Google Scholar] [CrossRef]
- Hu, M.; Han, Q.; Xing, B. Metallic Nanoparticle-Enabled Sensing of a Drug-of-Abuse: An Attempt at Forensic Application. ChemBioChem 2020, 21, 2512–2517. [Google Scholar] [CrossRef]
- Ghorbanizamani, F.; Moulahoum, H.; Guler Celik, E.; Timur, S. Ionic liquid-hydrogel hybrid material for enhanced electron transfer and sensitivity towards electrochemical detection of methamphetamine. J. Mol. Liq. 2022, 361, 119627. [Google Scholar] [CrossRef]
- Yao, W.; Wang, B.; Wu, Y.; Wang, J.; Xu, Z.; Meng, F.; Wang, P. Rapid Determination of Methamphetamine and Cocaine in Saliva by Portable Surface Plasmon Resonance (SPR). Anal. Lett. 2022, 55, 2944–2953. [Google Scholar] [CrossRef]
- Beduk, D.; Beduk, T.; de Oliveira Filho, J.I.; Ait Lahcen, A.; Aldemir, E.; Guler Celik, E.; Salama, K.N.; Timur, S. Smart Multiplex Point-of-Care Platform for Simultaneous Drug Monitoring. ACS Appl. Mater. Interfaces 2023, 15, 37247–37258. [Google Scholar] [CrossRef]
- Parrilla, M.; Joosten, F.; De Wael, K. Enhanced electrochemical detection of illicit drugs in oral fluid by the use of surfactant-mediated solution. Sens. Actuators B Chem. 2021, 348, 130659. [Google Scholar] [CrossRef]
- Sanli, S.; Moulahoum, H.; Ghorbanizamani, F.; Celik, E.G.; Timur, S. Ultrasensitive covalently-linked Aptasensor for cocaine detection based on electrolytes-induced repulsion/attraction of colloids. Biomed. Microdevices 2020, 22, 51. [Google Scholar] [CrossRef] [PubMed]
- Akgönüllü, S.; Battal, D.; Yalcin, M.S.; Yavuz, H.; Denizli, A. Rapid and sensitive detection of synthetic cannabinoids JWH-018, JWH-073 and their metabolites using molecularly imprinted polymer-coated QCM nanosensor in artificial saliva. Microchem. J. 2020, 153, 104454. [Google Scholar] [CrossRef]
- Khorablou, Z.; Shahdost-Fard, F.; Razmi, H. Voltammetric determination of pethidine in biofluids at a carbon cloth electrode modified by carbon selenide nanofilm. Talanta 2022, 239, 123131. [Google Scholar] [CrossRef] [PubMed]
- Garrido, E.; Alfonso, M.; Díaz de Greñu, B.; Lozano-Torres, B.; Parra, M.; Gaviña, P.; Marcos, M.D.; Martínez-Máñez, R.; Sancenón, F. Nanosensor for Sensitive Detection of the New Psychedelic Drug 25I-NBOMe. Chem. A Eur. J. 2020, 26, 2813–2816. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.K.; Sempionatto, J.R.; Li, Z.; Brown, C.; Galdino, N.M.; Shah, R.; Liu, S.; Hubble, L.J.; Bagot, K.; Tapert, S.; et al. Simultaneous detection of salivary Δ9-tetrahydrocannabinol and alcohol using a Wearable Electrochemical Ring Sensor. Talanta 2020, 211, 120757. [Google Scholar] [CrossRef] [PubMed]
- Koh, E.H.; Lee, W.-C.; Choi, Y.-J.; Moon, J.-I.; Jang, J.; Park, S.-G.; Choo, J.; Kim, D.-H.; Jung, H.S. A Wearable Surface-Enhanced Raman Scattering Sensor for Label-Free Molecular Detection. ACS Appl. Mater. Interfaces 2021, 13, 3024–3032. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Tang, Y.; Wu, H.; Wang, Y.; Niu, L.; Li, F. Integrated Aptasensor Array for Sweat Drug Analysis. Anal. Chem. 2022, 94, 7936–7943. [Google Scholar] [CrossRef]
- Guo, J.; Tian, S.; Liu, K.; Guo, J. IoT-Enabled Fluorescence Sensor for Quantitative KET Detection and Anti-Drug Situational Awareness. IEEE Trans. Nanobiosci. 2021, 20, 2–8. [Google Scholar] [CrossRef]
- Fan, L.; Yang, J.; Wu, J.; Li, F.; Yan, W.; Tan, F.; Zhang, M.; Draz, M.S.; Han, H.; Zhang, P. Deeply-dyed nanobead system for rapid lateral flow assay testing of drugs at point-of-care. Sens. Actuators B Chem. 2022, 362, 131829. [Google Scholar] [CrossRef]
- Biswas, R.; Saha, D. Probing volatile liquid through an electrical sensor with up gradation to a breathalyzer for drunken drivers. Appl. Phys. A Mater. Sci. Process. 2020, 126, 313. [Google Scholar] [CrossRef]
- Gusso, S.L.; Prado, L.B.; Ximim Gavim, A.E.; de Deus, J.F.; Foti, L.; Bin Mohd Yusoff, A.R.; da Silva, W.J.; Rodrigues, P.C.; Macedo, A.G. A Disposable and Noncontact Paper Breathalyzer Based on Small Conjugated Molecules/Carbon Nanotubes Electrodes. Phys. Status Solidi Appl. Mater. Sci. 2022, 219, 2100808. [Google Scholar] [CrossRef]
- Hwang, S.I.; Franconi, N.G.; Rothfuss, M.A.; Bocan, K.N.; Bian, L.; White, D.L.; Burkert, S.C.; Euler, R.W.; Sopher, B.J.; Vinay, M.L.; et al. Tetrahydrocannabinol Detection Using Semiconductor-Enriched Single-Walled Carbon Nanotube Chemiresistors. ACS Sensors 2019, 4, 2084–2093. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, F.; Carhart, M.; Andreescu, S. A 3D-Printed Breath Analyzer Incorporating CeO2Nanoparticles for Colorimetric Enzyme-Based Ethanol Sensing. ACS Appl. Nano Mater. 2021, 4, 9361–9369. [Google Scholar] [CrossRef]
- Cali, K.; Persaud, K.C. Modification of an Anopheles gambiae odorant binding protein to create an array of chemical sensors for detection of drugs. Sci. Rep. 2020, 10, 3890. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Keke, L.; Wei, X.; Mingzhu, Y. Micro-interfaces modulation by UV-ozone substrate treatment for MPEA vapor fluorescence detection. Nano Res. 2023, 16, 4055–4060. [Google Scholar] [CrossRef]
- Liu, K.; Shang, C.; Wang, Z.; Qi, Y.; Miao, R.; Liu, K.; Liu, T.; Fang, Y. Non-contact identification and differentiation of illicit drugs using fluorescent films. Nat. Commun. 2018, 9, 1695. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Bunes, B.R.; Wu, N.; Ansari, A.; Rajabali, S.; Zang, L. Sensing methamphetamine with chemiresistive sensors based on polythiophene-blended single-walled carbon nanotubes. Sens. Actuators B Chem. 2018, 255, 1814–1818. [Google Scholar] [CrossRef]
- Liang, T.T.; Kim, D.S.; Yoon, J.W.; Yu, Y.T. Rapid synthesis of rhombohedral In2O3 nanoparticles via a microwave-assisted hydrothermal pathway and their application for conductometric ethanol sensing. Sens. Actuators B Chem. 2021, 346, 130578. [Google Scholar] [CrossRef]
- Parrilla, M.; Felipe Montiel, N.; Van Durme, F.; De Wael, K. Derivatization of amphetamine to allow its electrochemical detection in illicit drug seizures. Sens. Actuators B Chem. 2021, 337, 129819. [Google Scholar] [CrossRef]
- Schram, J.; Parrilla, M.; Slosse, A.; Van Durme, F.; Åberg, J.; Björk, K.; Bijvoets, S.M.; Sap, S.; Heerschop, M.W.J.; De Wael, K. Paraformaldehyde-coated electrochemical sensor for improved on-site detection of amphetamine in street samples. Microchem. J. 2022, 179, 107518. [Google Scholar] [CrossRef]
- Rocha, L.R.; de Cássica Mendonça, J.; Boareto Capelari, T.; Antigo Medeiros, R.; Teixeira Tarley, C.R. Development of a reliable and selective voltammetric method for determination of designer drug 1-(3-chlorophenyl)piperazine (mCPP) using boron-doped diamond electrode and exploiting surfactant-mediated measurements. Sens. Actuators B Chem. 2020, 310, 127812. [Google Scholar] [CrossRef]
- Silva, W.P.; Rocha, R.G.; Arantes, L.C.; Lima, C.D.; Melo, L.M.A.; Munoz, R.A.A.; dos Santos, W.T.P.; Richter, E.M. Development of a simple and rapid screening method for the detection of 1-(3-chlorophenyl)piperazine in forensic samples. Talanta 2021, 233, 122597. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, M.F.M.; Bento, F.; Ipólito, A.J.; de Oliveira, M.F. Development of a Pencil Drawn Paper-based Analytical Device to Detect Lysergic Acid Diethylamide (LSD). J. Forensic Sci. 2020, 65, 2121–2128. [Google Scholar] [CrossRef] [PubMed]
- Barfidokht, A.; Mishra, R.K.; Seenivasan, R.; Liu, S.; Hubble, L.J.; Wang, J.; Hall, D.A. Wearable electrochemical glove-based sensor for rapid and on-site detection of fentanyl. Sens. Actuators B Chem. 2019, 296, 126422. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Novais, A.; Arantes, L.C.; Almeida, E.S.; Rocha, R.G.; Lima, C.D.; de Almeida Melo, L.M.; Richter, E.M.; Munoz, R.A.A.; dos Santos, W.T.P.; da Silva, R.A.B. Fast on-site screening of 3,4-methylenedioxyethylamphetamine (MDEA) in forensic samples using carbon screen-printed electrode and square wave voltammetry. Electrochim. Acta 2022, 403, 139599. [Google Scholar] [CrossRef]
- Florea, A.; Cowen, T.; Piletsky, S.; De Wael, K. Polymer platforms for selective detection of cocaine in street samples adulterated with levamisole. Talanta 2018, 186, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Zeng, J.; Zheng, Z.; Shi, H. Perspective and application of modified electrode material technology in electrochemical voltammetric sensors for analysis and detection of illicit drugs. Sens. Actuators A Phys. 2021, 329, 112821. [Google Scholar] [CrossRef]
- Piraianu, A.-I.; Fulga, A.; Musat, C.L.; Ciobotaru, O.-R.; Poalelungi, D.G.; Stamate, E.; Ciobotaru, O.; Fulga, I. Enhancing the Evidence with Algorithms: How Artificial Intelligence Is Transforming Forensic Medicine. Diagnostics 2023, 13, 2992. [Google Scholar] [CrossRef]
- Kadian, S.; Kumari, P.; Shukla, S.; Narayan, R. Recent advancements in machine learning enabled portable and wearable biosensors. Talanta Open 2023, 8, 100267. [Google Scholar] [CrossRef]
- Ma, X.; Guo, G.; Wu, X.; Wu, Q.; Liu, F.; Zhang, H.; Shi, N.; Guan, Y. Advances in Integration, Wearable Applications, and Artificial Intelligence of Biomedical Microfluidics Systems. Micromachines 2023, 14, 972. [Google Scholar] [CrossRef]
- Singh, K.R.; Nayak, V.; Singh, J.; Singh, R.P. Nano-enabled wearable sensors for the Internet of Things (IoT). Mater. Lett. 2021, 304, 130614. [Google Scholar] [CrossRef]

| Samples | Advantages | Forensic Significance |
|---|---|---|
| Blood and Derivatives | Rich genetic material; precise analysis | Accurate identification, drug detection, toxicological assessments |
| Urine | Non-invasive; accumulates metabolites | Drug screening, substance use monitoring, toxicology evaluations |
| Oral Fluid | Easily collected; reflects recent intake | Detects drugs, alcohol, and toxins; valuable in roadside testing |
| Sweat | Reflects recent exposure; continuous excretion | Identifies recent drug use, complements other sample analyses |
| Hair | Long-term history of substance exposure | Reveals chronic drug use, retrospective analysis of substance use |
| Exhaled Air | Non-invasive; real-time monitoring | Detects volatile substances, aids immediate intoxication assessment |
| Vapours | Captures volatile compounds in immediate vicinity | Identifies substances at crime scenes, aids in forensic analysis |
| Solid Samples | Diverse material sources (e.g., tablets, powder, seized samples); varied composition; convenient handling | Identifies and quantifies drugs, crucial for legal proceedings |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | LOQ | Time of Analysis | Ref. |
|---|---|---|---|---|---|---|---|---|
| Methamphetamine | n.s. | Optical | PCFS | n.s. | 0.5 ng/mL | 1.5 ng/mL | 10 min | [17] |
| Methamphetamine | n.s. | Electrochemical | SWV | Apt-38-MB | 30 nM | n.s | n.s | [18] |
| Methadone | 2 mL | Electrochemical | DPV | P-L-Arg/GCE | 0.032 μM | n.s | n.s | [19] |
| Methadone | 1 mL | Electrochemical | DPV | (Gr/AgNPs)2/ GCE | 0.12 μM | n.s | n.s | [20] |
| Tramadol | n.s. | Electrochemical | CV | 1-M-3-BBr/Pr(OH)3-GQD/CPE | 3.0 × 10−9 [M] | n.s. | n.s | [21] |
| (a) MDA (b) MDMA | n.s. | Optical | SPR | MIPs nanofilm | (a) 57 nM (b) 59 nM | n.s | n.s | [22] |
| Methamphetamine Amphetamine | n.s. | Electrochemical | Electrochemiluminescence | Nafion-[Ru(bpy)3]2+ | 0.2 μM | n.s. | n.s. | [23] |
| (a) Morphine (b) MDMA | 3 mL | Electrochemical | DPV | CNHs-CHI@PtNPs/GCE | (a) 0.02 (b) 0.018 | n.s. | n.s. | [24] |
| Diazepam | n.s. | Electrochemical | Potentiometry | Cu/POT/ISM | 1.2 × 10−7 [M] | n.s. | 11 ± 2 s | [25] |
| Oxycodone | n.s. | Electrochemical | DPV | Nafion/SWCNT | 85 nM | n.s. | n.s. | [26] |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | LOQ | Time of Analysis | Ref. |
|---|---|---|---|---|---|---|---|---|
| Methamphetamine | n.s. | Optical | PCFS | n.s | 0.5 ng/mL | 1.5 ng/mL | 10 min | [17] |
| Methamphetamine | n.s. | Electrochemical | SWV | Apt-38-MB | 50 nM | n.s. | n.s. | [18] |
| Methadone | 2 mL | Electrochemical | DPV | P-L-Arg/GCE | 0.032 μM | n.s. | n.s. | [19] |
| (a) MDA (b) MDMA | n.s. | Optical | SPR | MIPs nanofilm | (a) 57 nM (b) 59 nM | n.s. | n.s. | [22] |
| MethamphetamineAmphetamine | n.s. | Electrochemical | Electrochemiluminescence | Nafion-[Ru(bpy)3]2+ | 0.2 μM | n.s. | n.s. | [23] |
| (a) MDMA (b) Morphine | 3 mL | Electrochemical | DPV | CNHs-CHI@PtNPs/GCE | (a) 0.018 μmol/L (b) 0.02 μmol/L | n.s. | n.s. | [24] |
| Diazepam | n.s. | Electrochemical | POT | Cu/POT/ISM | 1.2 × 10−7 [M] | n.s. | 11 ± 2 s | [25] |
| Ketamine | n.s. | Electrochemical | EAB | Au/Apt/MCH | 10 nM | n.s. | 30 s | [27] |
| (a) Pentobarbitol (b) Caffeine (c) Morphine (d) Remifentanil (e) Fentanyl (f) Ketamine (g) Etomidate (h) Carfentanil (i) Sulfentanyl | n.s. | Optical | PLS-DA | AuNP | n.s. | n.s. | n.s. | [28] |
| Fentanyl | 150 μL | Electrochemical | AdSCV | Cytc/MWCNT/SPC | 0.086 μg/mL (drop) | 2.0 μg/mL | n.s. | [29] |
| Fentanyl | n.s. | Optical | UV–Visspectrophotometery | RB | 0.9 mg/L | n.s. | 6 min | [30] |
| (a) MDA (b) MDMA | n.s. | Electrochemical | DPV | MIP@PDA/Au-E | (a) 37 nM (b) 54 nM | n.s. | n.s. | [31] |
| Methamphetamine | n.s. | Optical | Fluorescence | CB [7] @BBH | 65.2 nM | n.s. | 30 s | [32] |
| Methylamphetamine | n.s. | Electrochemical | DPV | WP5–GDY/GCE | 0.016 μM | n.s. | n.s. | [33] |
| (a) Methamphetamine (b) MDMA | 10 μL | Optical | UV–Vis spectrophotometery | 1/DMAN impregnatedfilter paper | (a) 0.36 μg/mL (b) 0.57 μg/mL | n.s. | 10 min | [34] |
| Amphetamine | n.s. | Electrochemical | DPV | AMP-Apt/AuNFs@Au | 0.51 nM | n.s. | 20 min | [35] |
| Methcathinone | 100 μL | Optical | Fluorescence | Eu-MOF | 0.40 ng/mL | n.s. | n.s. | [36] |
| Tramadol | n.s. | Electrochemical | AdASV | (a) H-GONPs/GCE (b) GONSs/GCE | (a) 0.015 μM (b) n.s. | (a) 0.051 μM (b) n.s. | n.s. | [37] |
| γ-Hydroxybutyric acid (GHB) | n.s. | Optical | UV–Visspectrophotometery | AuNP | n.s. | n.s. | n.s. | [38] |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | LOQ | Time of Analysis | Ref. |
|---|---|---|---|---|---|---|---|---|
| Methamphetamine | n.s. | Optical | PCFS | SA-Bio-HRP nanocomposite | 0.5 ng/mL | 1.5 ng/mL | 10 min | [17] |
| Methamphetamine | n.s. | Electrochemical | EAB | Apt-38-MB | 20 nM | n.s. | n.s. | [18] |
| (a) Methampheta mine (b) Amphetamine (c) para-hydroxy- methamphetamine | n.s. | Electrochemical | Electrochemiluminescence | GCE/Nafion/[Ru(bpy)3]2+ | (a) 10 µM (b) 10 µM (c) 10 µM | n.s. | n.s. | [23] |
| Diazepam | n.s. | Electrochemical | Potentiometry | Cu/POT/ISM | 1.2 × 10−7 M | n.s. | 11 ± 2 s | [25] |
| Ketamine | n.s. | Electrochemical | EAB | Au/APT/MCH | 10 nM | n.s. | 30 s | [27] |
| Tramadol | n.s. | Electrochemical | AdASV | (a) H-GONPs/GCE (b) GONSs/GCE | (a) 0.015 µM (b) n.s. | (a) 0.051 µM (b) n.s. | n.s. | [37] |
| Methamphetamine | n.s. | Electrochemical | Antibody-antigene recognition | SPGE/IG/mAb | 0.72 ng/mL | 2.4 ng/mL | n.s. | [39] |
| (a) Methampheta mine (b) Cocaine | 1 mL | Optical | SPR | MA-BSA/COC-BSA/Au SPR chip | (a) 0.95 ng/mL (b) 3.14 ng/mL | n.s. | n.s. | [40] |
| (a) Cocaine (b) Amphetamine (c) Benzodiazepine | n.s. | Electrochemical | DPV | LSG | (a) 4.3 ng/mL (b) 9.7 ng/mL (c) 9.0 ng/mL | n.s. | n.s. | [41] |
| (a) Cocaine (b) Heroin (c) MDMA (d) Ketamine | n.s. | Electrochemical | SWAdSV | SDS/Graphite SPE | (a) 1.2 µM (b) 2.4 µM (c) 1.0 µM (d) 2.6 µM | n.s. | n.s. | [42] |
| Cocaine | n.s. | Optical | SPR | GNP-Aptamer/NaCl | 0.97 nM | n.s. | <90 min | [43] |
| (a) JWH-018 (b) JWH-073 (c) JWH-018 pentanoic acid (d) JWH-073 butanoic acid | n.s. | Gravimetric | Piezoelectric | SCs-MIP QCM | (a) 0.28 pg/mL (b) 0.3 pg/mL (c) 0.23 pg/mL (d) 0.29 pg/mL | (a) 3.03 pg/mL (b) 3.0 pg/mL (c) 2.4 pg/mL (d) 3.1 pg/mL | n.s. | [44] |
| Pethidine | 2 mL | Electrochemical | DPV | CSe2NF/CC | 19.3 nM | n.s. | n.s. | [45] |
| 25I-NBOMe | n.s. | Optical | Fluorescence | MSNs/Rhodamine B/serotonin derivate/5-HT2A | 0.6 µM | n.s. | n.s. | [46] |
| (a) Tetrahydrocannabinol (b) Ethanol | n.s. | (a) Electrochemical (b) Electrochemical | (a) SWV (b) Amperometry | (a) MWCNT/Carbon (b) PB/AOx/Chitosan | (a) 0.5 µM (b) n.s. | n.s. | 6 min | [47] |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | LOQ | Time of Analysis | Ref |
|---|---|---|---|---|---|---|---|---|
| 2-fluoromethampehtamine | n.s. | Optical | SERS | SFF with AgNW | n.s. | n.s. | n.s. | [48] |
| Cathinone, heroin, cocaine | n.s. | Electrochemical | EAB | (a) Au/APT1/MCH (b) Au/APT2/MCH (c) Au/APT1 + APT2/MCH | (a) 0.32 nM (b) 0.57 nM (c) 0.18 nM | n.s. | n.s. | [49] |
| Compounds | Sample Amount | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD (ng/mL) | Time of Analysis (min) | Ref. |
|---|---|---|---|---|---|---|---|
| Ketamine | n.s. | Optical | Fluorescence | UC NP’s/LFIA | 1 | 5 | [50] |
| Methamphetamine | 5 mg | Optical | Colorimetric Strips | DDNB/LFIA | 8 | 8 | [51] |
| Morphine |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | Time of Analysis | Ref. |
|---|---|---|---|---|---|---|---|
| EtOH | n.s. | Electrical | Amperometry | MQ3/CuO:SnO2 | 230 ng/mL | 8 s | [52] |
| EtOH | n.s. | Electrochemical | Conductometry | SW CNT/PDIC10 | 0.01% BAC | 5 s | [53] |
| Tetrahydrocannabinol | n.s. | Electrochemical | Conductometry | S-SWCNT | 0.163 ng | 15 min | [54] |
| EtOH | 1 L | Optical | Colorimetric strips | ALOx/CeNP’s | 0.001% (v/v) | 15 min | [55] |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | Time of Analysis (s) | Dissociation Constant (μM) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Cocaine Tetrahydrocannabinol Cannabidiol MDMA Heroin Codeine | 0.1 ** L/min | Optical | Fluorescence | AgamOBP1_S82P | n.s. | 10 | 0.48 ± 0.14 0.28 ± 0.07 0.06 ± 0.02 0.08 ± 0.01 0.27 ± 0.17 0.65 ± 0.27 | [56] |
| MPEA | n.s. | Optical | Fluorescence | CP S1a/b/tiophene ring structure on UV-ozone treated quartz plates | S1a 2.59 ng/mL/S1d 0.25 ng/mL | 150 | n.a | [57] |
| Methamphetamine MDMA Ketamine Magu Phenobarbital | n.s. | Optical | Fluorescence | PBI-CB | 5.0 × 105 * 4.0 × 105 * 2.0 × 102 * 2.0 × 105 * 4.0 × 104 * | 60 | n.a | [58] |
| NMPEA | n.s. | Electrochemical | Conductometry | CNT functionalised with a polythiophene derivative | 4 µg/L | 20 | n.a | [59] |
| Ethanol | n.s. | Electrochemical | Conductometry | h-In2O3 NP via the microwave-assisted hydrothermal pathway | 330 µg/L | 37.6 ± 8.26 | n.a | [60] |
| Compounds | Sample Volume | Type of Sensor | Transducing Mechanism | Sensor Matrix | LOD | LOQ | Time of Analysis | Ref. |
|---|---|---|---|---|---|---|---|---|
| (a) Methamphetamine (b) MDMA | 180 μL | Optical | UV–Vis spectrophotometry | 1/DMAN impregnated filter paper | n.s. | n.s. | 5 min | [34] |
| Amphetamine | 80 μL | Electrochemical | SWV | SPEs | 22.2 μM | n.s. | 3 min | [61] |
| Amphetamine | 85 μL | Electrochemical | SWV | PFA-coated SPEs | 0.3 mM | 0.9 mM | 1.5 min | [62] |
| mCPP | 79 μL | Electrochemical | SWV | CPT-BDDE | 1.1 μM | 3.5 μM | n.s. | [63] |
| mCPP | 100 μL | Electrochemical | SWV | C-SPE | 0.1 μM | 0.33 μM | n.s. | [64] |
| LSD | n.s. | Electrochemical | SWV | Paper-based electrodes | 0.38 μM | 1.27 μM | n.s. | [65] |
| Fentanyl | n.s. | Electrochemical | SWV | Ag/AgCl | 10 µM | n.s | n.s. | [66] |
| MDEA | 5 μL | Electrochemical | SWV | C-SPE | 0.03 μM | 0.09 μM | n.s. | [67] |
| Cocaine | n.s. | Electrochemical | SWV | poly(PABA)/GPH-SPE | n.s. | n.s. | 7 min | [68] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosendo, L.M.; Antunes, M.; Simão, A.Y.; Brinca, A.T.; Catarro, G.; Pelixo, R.; Martinho, J.; Pires, B.; Soares, S.; Cascalheira, J.F.; et al. Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review. Micromachines 2023, 14, 2249. https://doi.org/10.3390/mi14122249
Rosendo LM, Antunes M, Simão AY, Brinca AT, Catarro G, Pelixo R, Martinho J, Pires B, Soares S, Cascalheira JF, et al. Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review. Micromachines. 2023; 14(12):2249. https://doi.org/10.3390/mi14122249
Chicago/Turabian StyleRosendo, Luana M., Mónica Antunes, Ana Y. Simão, Ana Teresa Brinca, Gonçalo Catarro, Rodrigo Pelixo, João Martinho, Bruno Pires, Sofia Soares, José Francisco Cascalheira, and et al. 2023. "Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review" Micromachines 14, no. 12: 2249. https://doi.org/10.3390/mi14122249
APA StyleRosendo, L. M., Antunes, M., Simão, A. Y., Brinca, A. T., Catarro, G., Pelixo, R., Martinho, J., Pires, B., Soares, S., Cascalheira, J. F., Passarinha, L., Rosado, T., Barroso, M., & Gallardo, E. (2023). Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review. Micromachines, 14(12), 2249. https://doi.org/10.3390/mi14122249














