Preclinical Evaluation of the Efficacy of Antivenoms for Snakebite Envenoming: State-of-the-Art and Challenges Ahead
Abstract
:1. Introduction
2. Neutralization of Lethality: The Gold Standard in the Preclinical Assessment of Antivenom Efficacy
3. The Need to Understand the Mechanisms of Toxicity in the Lethality Assay
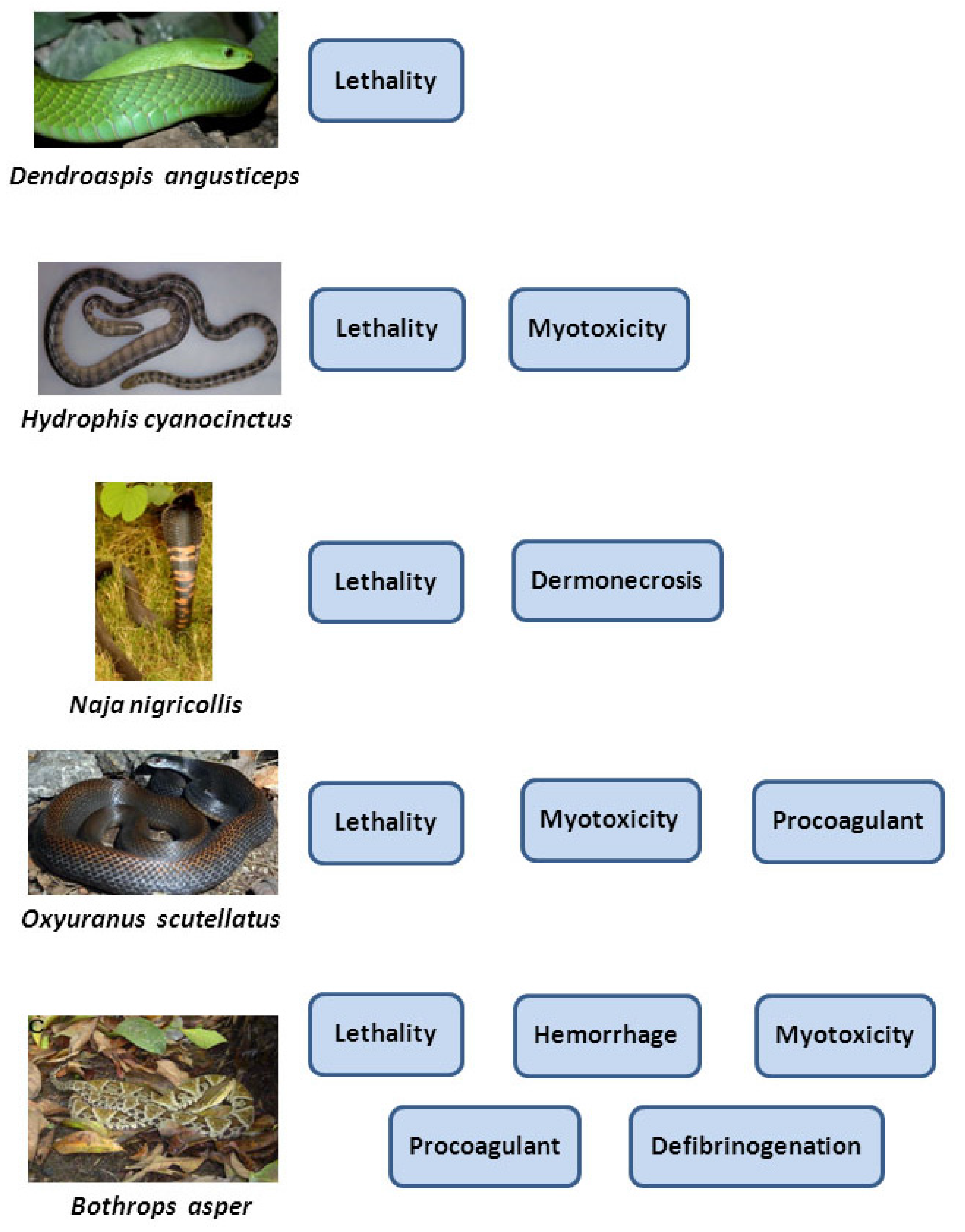
4. The Complex Pathophysiology of Snakebite Envenoming Demands a Wider Approach for the Preclinical Testing of Antivenoms
4.1. Hemorrhagic Activity
4.2. Necrotizing Activity
4.3. In Vitro Coagulant Activity
4.4. Defibrinogenating Activity
4.5. Myotoxic Activity
4.6. Edematogenic Activity
4.7. Neurotoxic Activity
5. Towards Expanding the Set of Laboratory Tests for the Evaluation of Antivenoms
6. The Issue of Minimum Accepted Values of ED50 for Preclinical Neutralization Tests
7. The Impact of Venom Toxicokinetics and Antivenom Pharmacokinetics in the Effectiveness of Antivenoms: Pharmacokinetic-Pharmacodynamic Relationships
8. Implementation of the 3Rs in Antivenom Testing
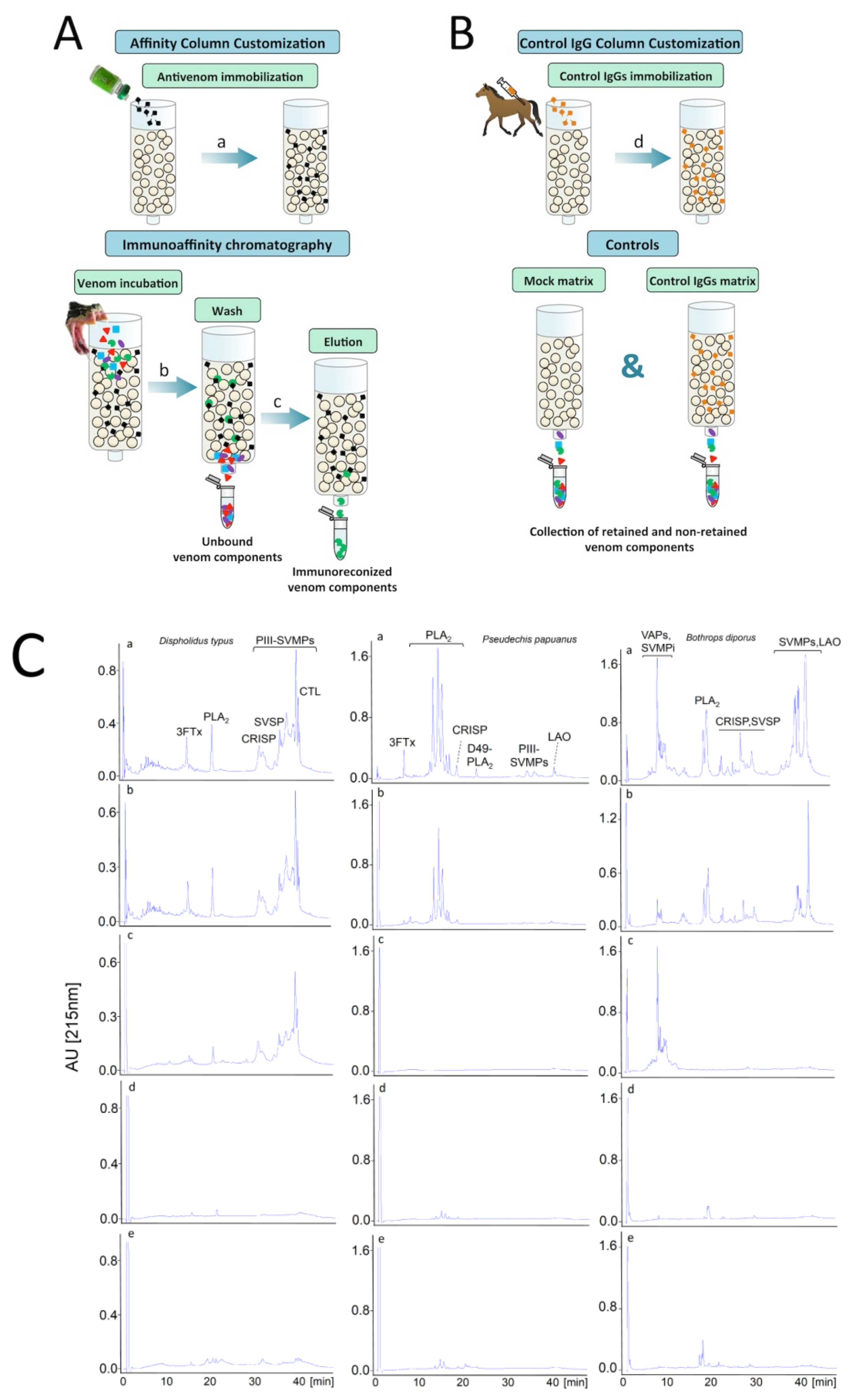
9. Antivenomics: Harnessing the Analytical Potential of Proteomics for the Evaluation of Antivenoms
10. The Need of International Partnerships to Evaluate Antivenoms on a Global Basis and to Strengthen National Regulatory Agencies
11. Preclinical Testing Should Be Followed by Appropriate Clinical Assessment of Efficacy and Safety of Antivenoms
12. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chippaux, J.P. Snake-bites: Appraisal of the global situation. Bull. World Health Organ. 1998, 76, 515–524. [Google Scholar] [PubMed]
- Gutiérrez, J.M.; Theakston, R.D.G.; Warrell, D.A. Confronting the neglected problem of snake bite envenoming: The need for a global partnership. PLoS Med. 2006, 3, e150. [Google Scholar] [CrossRef] [PubMed]
- Kasturiratne, A.; Wickremasinghe, A.R.; de Silva, N.; Gunawardena, N.K.; Pathmeswaran, A.; Premaratna, R.; Savioli, L.; Lalloo, D.G.; de Silva, H.J. The global burden of snakebite: A literature analysis and modelling based on regional estimates of envenoming and deaths. PLoS Med. 2008, 5, e218. [Google Scholar] [CrossRef] [PubMed]
- Calmette, A. Contribution a l’etude du venin des serpents. Immunisation des animaux et traitment de l’envenimation. Ann. Inst. Pasteur 1894, 8, 275–291. [Google Scholar]
- Phisalix, C.; Bertrand, G. Sur la propriete antitoxique du sang des animaux vaccines contre le venin de vipere. C. R. Soc. Biol. 1894, 46, 111–113. [Google Scholar]
- Gutiérrez, J.M.; León, G.; Lomonte, B.; Angulo, Y. Antivenoms for snakebite envenomings. Inflamm. Allergy Drug Targets 2011, 10, 369–380. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Rabies and Envenoming: A Neglected Public Health Issue; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Williams, D.J.; Gutiérrez, J.M.; Calvete, J.J.; Wüster, W.; Ratanabanangkoon, K.; Paiva, O.; Brown, H.I.; Casewell, N.R.; Harrison, R.A.; Rowley, P.D.; et al. Ending the drought: New strategies for improving the flow of affordable, effective antivenoms in Asia and Africa. J. Proteom. 2011, 74, 1735–1767. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M. Improving antivenom availability and accessibility: Science, technology, and beyond. Toxicon 2012, 60, 676–687. [Google Scholar] [CrossRef] [PubMed]
- Chippaux, J.P.; Williams, V.; White, J. Snake venom variability: Methods of study, results and interpretation. Toxicon 1991, 29, 1279–1303. [Google Scholar] [CrossRef]
- Calvete, J.J. Venomics: Integrative venom proteomics and beyond. Biochem. J. 2017, 474, 611–634. [Google Scholar] [CrossRef] [PubMed]
- Alape-Girón, A.; Sanz, L.; Escolano, J.; Flores-Díaz, M.; Madrigal, M.; Sasa, M.; Calvete, J.J. Snake venomics of the lancehead pitviper Bothrops asper: Geographic. Individual, and ontogenetic variations. J. Proteom. Res. 2008, 7, 3556–3571. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Sanz, L.; Cid, P.; de la Torre, P.; Flores-Diaz, M.; dos Santos, M.C.; Borges, A.; Bremo, A.; Angulo, Y.; Lomonte, B.; et al. Snake venomics of the Central American rattlesnake Crotalus simus and the South American Crotalus durissus complex points to neurotoxicity as an adaptive paedomorphic trend along Crotalus dispersal in South America. J. Proteom. Res. 2010, 9, 528–544. [Google Scholar] [CrossRef] [PubMed]
- Durban, J.; Pérez, A.; Sanz, L.; Gómez, A.; Bonilla, F.; Rodríguez, S.; Chacón, D.; Sasa, M.; Angulo, Y.; Gutiérrez, J.M.; et al. Integrated “omics” profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus. BMC Genom. 2013, 14, 234. [Google Scholar] [CrossRef] [PubMed]
- Jackson, T.N.; Koludarov, I.; Ali, S.A.; Dobson, J.; Zdenek, C.N.; Dashevsky, D.; Op den Brouw, B.; Masci, P.P.; Nouwens, A.; Josh, P.; et al. Rapid radiations and the race to redundancy: An investigation of the evolution of Australian elapid snake venoms. Toxins 2016, 8, 309. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.; Alape-Girón, A.; Angulo, Y.; Sanz, L.; Gutiérrez, J.M.; Calvete, J.J.; Lomonte, B. Venomic and antivenomic analyses of the Central American coral snake, Micrurus nigrocinctus (Elapidae). J. Proteom. Res. 2011, 10, 1816–1827. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wagstaff, S.C.; Wüster, W.; Cook, W.; Bolton, F.M.; King, S.I.; Pla, D.; Sanz, L.; Calvete, J.J.; Harrison, R.A. Medically important differences in snake venom composition are dictated by distinct postgenomic mechanisms. Proc. Natl. Acad. Sci. USA 2014, 111, 9205–9210. [Google Scholar] [CrossRef] [PubMed]
- Petras, D.; Heiss, P.; Harrison, R.A.; Süssmuth, R.D.; Calvete, J.J. Top-down venomics of the East African green mamba, Dendroaspis angusticeps, and the black mamba, Dendroaspis polylepis, highlight the complexity of their toxin arsenals. J. Proteom. 2016, 146, 148–164. [Google Scholar] [CrossRef] [PubMed]
- Petras, D.; Sanz, L.; Segura, A.; Herrera, M.; Villalta, M.; Solano, D.; Vargas, M.; León, G.; WArrell, D.A.; Theakston, R.D.G.; et al. Snake venomics of African spitting cobras: Toxin composition and assessment of congeneric cross-reactivity of the pan-African EchiTAb-Plus-ICP antivenom by antivenomics and neutralization approaches. J. Proteom. Res. 2011, 10, 1266–1280. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Rey-Suárez, P.; Fernández, J.; Sasa, M.; Pla, D.; Vargas, N.; Bénard-Valle, M.; Sanz, L.; Corrêa-Netto, C.; Núñez, V.; et al. Venoms of Micrurus coral snakes: Evolutionary trends in compositional patterns emerging from proteomic analyses. Toxicon 2016, 122, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Modahl, C.M.; Mukherjee, A.K.; Mackessy, S.P. An analysis of venom ontogeny and prey-specific toxicity in the Monocled Cobra (Naja kaouthia). Toxicon 2016, 119, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Fernández, J.; Sanz, L.; Angulo, Y.; Sasa, M.; Gutiérrez, J.M.; Calvete, J.J. Venomous snakes of Costa Rica: Biological and medical implications of their venom proteomic profiles analyzed through the strategy of snake venomics. J. Proteom. 2014, 105, 323–339. [Google Scholar] [CrossRef] [PubMed]
- Bdolah, A. Sarafotoxins, the snake venom homologs of endothelins. In Handbook of Venoms and Toxins of Reptiles; CRC Press: Boca Raton, FL, USA, 2010; pp. 303–315. [Google Scholar]
- Junqueira-de-Azevedo, I.L.; Campos, P.F.; Ching, A.T.; Mackessy, S.P. Colubrid venom composition: An-omics perspective. Toxins 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J. Proteomic tools against the neglected pathology of snake bite envenoming. Expert Rev. Proteom. 2011, 8, 739–758. [Google Scholar] [CrossRef] [PubMed]
- Visser, L.E.; Kyei-Faried, S.; Belcher, D.W.; Geelhoed, D.W.; van Leeuwen, J.S.; van Roosmalen, J. Failure of a new antivenom to treat Echis ocellatus snake bite in rural Ghana: The importance of quality surveillance. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Stock, R.P.; Massougbodji, A.; Alagón, A.; Chippaux, J.P. Bringing antivenoms to sub-Saharan Africa. Nat. Biotechnol. 2007, 25, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Lomonte, B.; León, G.; Alape-Girón, A.; Flores-Díaz, M.; Sanz, L.; Angulo, Y.; Calvete, J.J. Snake venomics and antivenomics: Proteomic tolos in the design and control of antivenoms for the treatment of snakebite envenoming. J. Proteom. 2009, 72, 165–182. [Google Scholar] [CrossRef] [PubMed]
- Calmette, A. The treatment of animals poisoned with snake venoms by the injection of antivenomous serum. Br. Med. J. 1896, 1859, 399–400. [Google Scholar] [CrossRef]
- Brazil, V. Dosagem do valor anti-toxico dos serums anti-peçonhentos. In Vital Brazil. Obra Científica Completa; Instituto Vital Brazil: Niteroi, Brazil, 2002; pp. 217–223. [Google Scholar]
- Christensen, P.A. Venom and antivenom potency estimation. Mem. Inst. Butantan 1966, 33, 305–326. [Google Scholar]
- World Health Organization. WHO Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins; World Health Organization: Geneva, Switzerland, 2010; Available online: http://www.who.int/bloodproducts/snake_antivenoms/ snakeantivenomguide/en/ (accessed on 12 May 2017).
- Finney, D.J. Probit Analysis; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- World Health Organization. Progress in the Characterization of Venoms and Standardization of Antivenoms; WHO Offset Publication No. 58; World Health Organization: Geneva, Switzerland, 1981. [Google Scholar]
- Araújo, H.P.; Bourguignon, S.C.; Boller, M.A.; Dias, A.A.; Lucas, E.P.; Santos, I.C.; Delgado, I.F. Potency evaluation of antivenoms in Brazil: The national control laboratory experience between 2000 and 2006. Toxicon 2008, 51, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Bogarín, G.; Morais, J.F.; Yamaguchi, I.K.; Stephano, M.A.; Marcelino, J.R.; Nishikawa, A.K.; Guidolin, R.; Rojas, G.; Higashi, H.G.; Gutiérrez, J.M. Neutralization of crotaline snake venoms from Central and South America by antivenoms produced in Brazil and Costa Rica. Toxicon 2000, 38, 1429–1441. [Google Scholar] [CrossRef]
- Villalta, M.; Pla, D.; Yang, S.L.; Sanz, L.; Segura, A.; Vargas, M.; Chen, P.Y.; Herrera, M.; Estrada, R.; Cheng, Y.F.; et al. Snake venomics and antivenomics of Protobothrops mucrosquamatus and Viridovipera stejnegeri from Taiwan: Keys to understand the variable immune response in horses. J. Proteom. 2012, 75, 5628–5645. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.R.; dos Santos, M.C.; Tambourgi, D.V.; Marques, T.; da Silva, W.D.; Kipnis, T. Susceptibility of different strains of mice to South American rattlesnake (Crotalus durissus terrificus) venom: Correlation between lethal effect and creatine kinase release. Toxicon 1991, 29, 783–786. [Google Scholar] [CrossRef]
- Solano, G.; Segura, A.; Herrera, M.; Gómez, A.; Villalta, M.; Gutiérrez, J.M.; León, G. Study of the design and analytical properties of the lethality neutralization assay used to estimate antivenom potency against Bothrops asper snake venom. Biologicals 2010, 38, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Segura, A.; Castillo, M.C.; Núñez, V.; Yarlequé, A.; Gonçalves, L.R.C.; Villalta, M.; Bonilla, C.; Herrera, M.; Vargas, M.; Fernández, M.; et al. Preclinical assessment of the neutralizing capacity of antivenoms produced in six Latin American countries against medically-relevant Bothrops snake venoms. Toxicon 2010, 56, 980–989. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines for the Production, Control and Regulation of Snake Antivenom Immunoglobulins, 2nd ed.; World Health Organization: Geneva, Switzerland, 2017; In press. [Google Scholar]
- Chaniot, S.; Netter, R. Study of various factors influencing the titration of anti-venom sera. Pathol. Biol. Paris 1971, 19, 991–1001. [Google Scholar] [PubMed]
- Krifi, M.N.; Marrakchi, N.; el Ayeb, M.; Dellagi, K. Effct of some variables on the in vivo determination of scorpion and viper venom toxicities. Biologicals 1998, 26, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Rojas, G.; Cerdas, L. Ability of a polyvalent antivenom to neutralize the venom of Lachesis muta melanocephala, a new Costa Rican subspecies of the bushmaster. Toxicon 1987, 25, 713–720. [Google Scholar] [CrossRef]
- León, G.; Stiles, B.; Alape, A.; Rojas, G.; Gutiérrez, J.M. Comparative study on the ability of IgG and F(ab’)2 antivenoms to neutralize lethal and myotoxic effects induced by Micrurus nigrocinctus (coral snake) venom. Am. J. Trop. Med. Hyg. 1999, 61, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Solano, G.; Pla, D.; Herrera, M.; Segura, Á.; Villalta, M.; Vargas, M.; Sanz, L.; Lomonte, B.; Calvete, J.J.; et al. Assessing the preclinical efficacy of antivenoms: From the lethality neutralization assay to antivenomics. Toxicon 2013, 69, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Warrel, D.A. Snake bite. Lancet 2010, 375, 77–88. [Google Scholar] [CrossRef]
- Loría, G.D.; Rucavado, A.; Kamiguti, A.S.; Theakston, R.D.G.; Fox, J.W.; Alape, A.; Gutiérrez, J.M. Characterization of ‘basparin A’, a prothrombin-activating metalloproteinase, from the venom of the snake Bothrops asper that inhibits platelet aggregation and induces defibrination and thrombosis. Arch. Biochem. Biophys. 2003, 418, 13–24. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Escalante, T.; Rucavado, A. Experimental pathophysiology of systemic alterations induced by Bothrops asper snake venom. Toxicon 2009, 54, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Herrera, M.; Fernández, J.; Vargas, M.; Villalta, M.; Segura, Á.; León, G.; Angulo, Y.; Paiva, O.; Matainaho, T.; Jensen, S.D.; et al. Comparative proteomic analysis of the venom of the taipan snake, Oxyuranus scutellatus, from Papua New Guinea and Australia: Role of neurotoxic and procoagulant effects in venom toxicity. J. Proteom. 2012, 75, 2128–2140. [Google Scholar] [CrossRef] [PubMed]
- Chacón, F.; Oviedo, A.; Escalante, T.; Solano, G.; Rucavado, A.; Gutiérrez, J.M. The lethality test used for estimating the potency of antivenoms against Bothrops asper snake venom: Pathophysiological mechanisms, prophylactic analgesia and a surrogate in vitro assay. Toxicon 2015, 93, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Marques, M.M.; Cupo, P.; Coimbra, T.M.; Hering, S.E.; Rossi, M.A.; Laure, C.J. Myonecrosis, myoglobinuria and acute renal failure induced by South American rattlesnake (Crotalus durissus terrificus) envenomation in Brazil. Toxicon 1985, 23, 631–636. [Google Scholar] [CrossRef]
- White, J. Clinical toxicology of sea snakebites. In Handbook of Clinical Toxicology of Animal Venoms and Poisons; CRC Press: Boca Raton, FL, USA, 1995; pp. 159–170. [Google Scholar]
- Warrell, D.A. Clinical toxicology of snakebite in Africa and the Middle East/Arabian peninsula. In Handbook of Clinical Toxicology of Animal Venoms and Poisons; CRC Press: Boca Raton, FL, USA, 1995; pp. 433–492. [Google Scholar]
- White, J. Envenomation. Prevention and treatment in Australia. In Handbook of Venoms and Toxins of Reptiles; CRC Press: Boca Raton, FL, USA, 2010; pp. 423–451. [Google Scholar]
- Cardoso, J.L.C.; França, F.O.S.; Wen, F.H.; Málaque, C.M.S.; Haddad, V. Animais Peçonhentos no Brasil. Biologia, Clínica e Terapêutica dos Acidentes; Sarvier: São Paulo, Brazil, 2009. [Google Scholar]
- Kondo, H.; Kondo, S.; Ikezawa, H.; Murata, R.; Ohsaka, A. Studies on the quantitative method for determination of hemorrhagic activity of Habu snake venom. Jpn. J. Med. Sci. Biol. 1960, 13, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Theakston, R.D.G.; Reid, H.A. Development of simple standard assay procedures for the characterization of snake venom. Bull. World Health Organ. 1983, 61, 949–956. [Google Scholar] [PubMed]
- Gutiérrez, J.M.; Gené, J.A.; Rojas, G.; Cerdas, L. Neutralization of proteolytic and hemorrhagic activities of Costa Rican snake venoms by a polyvalent antivenom. Toxicon 1985, 23, 887–893. [Google Scholar] [CrossRef]
- Ownby, C.L.; Colberg, T.R.; Odell, G.V. A new method for quantitating hemorrhage induced by rattlesnake venoms: Ability of polyvalent antivenom to neutralize hemorrhagic activity. Toxicon 1984, 22, 227–233. [Google Scholar] [CrossRef]
- Warrell, D.A. Clinical toxicology of snakebite in Asia. In Handbook of Clinical Toxicology of Animal Venoms and Poisons; CRC Press: Boca Raton, FL, USA, 1995; pp. 493–594. [Google Scholar]
- Gutiérrez, J.M.; Rojas, E.; Quesada, L.; León, G.; Núñez, J.; Laing, G.D.; Sasa, M.; Renjifo, J.M.; Nasidi, A.; Warrell, D.A.; et al. Pan-African polyspecific antivenom produced by caprylic acid precipitation of horse IgG: An alternative to the antivenom crisis in Africa. Trans. R. Soc. Trop. Med. Hyg. 2005, 99, 468–475. [Google Scholar] [CrossRef] [PubMed]
- White, J. Snake venoms and coagulopathy. Toxicon 2005, 45, 951–967. [Google Scholar] [CrossRef] [PubMed]
- Gené, J.A.; Roy, A.; Rojas, G.; Gutiérrez, J.M.; Cerdas, L. Comparative study on coagulant, defibrinating, fibrinolytic and fibrinogenolytic activities of Costa Rican crotaline snake venoms and their neutralization by a polyvalent antivenom. Toxicon 1989, 27, 841–848. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Ownby, C.L. Skeletal muscle degeneration induced by venom phospholipases A2: Insights into the mechanisms of local and systemic myotoxicity. Toxicon 2003, 42, 915–931. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-Marques, M.M.; Hering, S.E.; Cupo, P. Acidente crotálico. In Animais Peçonhentos no Brasil. Biologia, Clínica e Terapêutica dos Acidentes; Sarvier: São Paulo, Brazil, 2009; pp. 108–115. [Google Scholar]
- Teixeira, C.F.P.; Zamuner, S.R.; Zuliani, J.P.; Fernandes, C.M.; Cruz-Hoffling, M.A.; Fernandes, I.; Chaves, F.; Gutiérrez, J.M. Neutrophils do not contribute to local tissue damage, but play a key role in skeletal muscle regeneration, in mice injected with Bothrops asper snake venom. Muscle Nerve 2003, 28, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Arroyo, O.; Bolaños, R. Mionecrosis, hemorragia y edema inducidos por el veneno de Bothrops asper en el ratón blanco. Toxicon 1980, 18, 603–610. [Google Scholar] [CrossRef]
- Rojas, E.; Quesada, L.; Arce, V.; Lomonte, B.; Rojas, G.; Gutiérrez, J.M. Neutralization of four Peruvian Bothrops sp snake venoms by polyvalent antivenoms produced in Perú and Costa Rica: Preclinical assessment. Acta Trop. 2005, 93, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Trebien, H.A.; Calixto, J.B. Pharmacological evaluation of rat paw oedema induced by Bothrops jararaca venom. Agents Actions 1989, 26, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Chaves, F.; Barboza, M.; Gutiérrez, J.M. Pharmacological study of edema induced by venom of the snake Bothrops asper (terciopelo) in mice. Toxicon 1995, 33, 31–39. [Google Scholar] [CrossRef]
- Lomonte, B.; Tarkowski, A.; Hanson, L.Å. Host response to Bothrops asper snake venom. Analysis of edema formation, inflammatory cells, and cytokine release in a mouse model. Inflammation 1993, 17, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Yamakawa, M.; Nozaki, M.; Hokama, Z. Fractionation of Sakishima-habu (Trimeresurus elegans) venom and lethal, hemorrhagic and edema-forming activities of the fractions. In Animal, Plant and Microbial Toxins, Volume 1, Biochemistry; Plenum Press: New York, NY, USA, 1976; pp. 97–109. [Google Scholar]
- Harvey, A.L.; Barfaraz, A.; Thomson, E.; Faiz, A.; Preston, S.; Harris, J.B. Screening of snake venoms for neurotoxic and myotoxic effects using simple in vitro preparations from rodents and chicks. Toxicon 1994, 32, 257–265. [Google Scholar] [CrossRef]
- Crachi, M.T.; Hammer, L.W.; Hodgson, W.C. The effects of antivenom on the in vitro neurotoxicity of venoms from the taipans Oxyuranus scutellatus, Oxyuranus microlepidotus and Oxyuranus scutellatus canni. Toxicon 1999, 37, 1771–1778. [Google Scholar] [CrossRef]
- Damico, D.C.; Bueno, L.G.; Rodrigues-Simioni, L.; Marangoni, S.; da Cruz-Hoffling, M.A.; Novello, J.C. Neurotoxic and myotoxic actions from Lachesis muta muta (surucucú) whole venom on the mouse and chick nerve-muscle preparations. Toxicon 2005, 46, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, S.; Fry, B.G.; Hodgson, W.C. Neurotoxic effects of venoms from seven species of Australasian black snakes (Pseudechis): Efficacy of black and tiger snake antivenoms. Clin. Exp. Pharmacol. Physiol. 2005, 32, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.; Hodgson, W.C.; Isbister, G.K. Cross-neutralization of in vitro neurotoxicity of Asian and Australian snake neurotoxins and venoms by different antivenoms. Toxins 2016, 8, 302. [Google Scholar] [CrossRef] [PubMed]
- Herrera, M.; de Collaço, R.C.O.; Villalta, M.; Segura, Á.; Vargas, M.; Wright, C.E.; Paiva, O.K.; Matainaho, T.; Jensen, S.D.; León, G.; et al. Neutralization of the neuromuscular inhibition of venom and taipoxin from the taipan (Oxyuranus scutellatus) by F(ab’)2 and whole IgG antivenoms. Toxicol. Lett. 2016, 241, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Carlson, R.W.; Schaeffer, R.C.; Whigham, H.; Michaels, S.; Russell, F.E.; Weil, M.H. Rattlesnake venom shock in the rat: Development of a method. Am. J. Physiol. 1975, 229, 1668–1674. [Google Scholar] [PubMed]
- Dias, L.; Rodrigues, M.A.; Smaal, A.; Rennó, A.L.; Mello, S.M.; Moreno, H.; Hyslop, S. Cardiovascular responses to Bothrops alternatus (Urutu) snake venom in anesthetized dogs. Cardiovasc. Toxicol. 2012, 12, 243–257. [Google Scholar] [CrossRef] [PubMed]
- Sano-Martins, I.S.; Santoro, M.L.; Castro, S.C.; Fan, H.W.; Cardoso, J.L.C.; Theakston, R.D.G. Platelet aggregation in patients bitten by the Brazilian snake Bothrops jararaca. Thromb. Res. 1997, 87, 183–195. [Google Scholar] [CrossRef]
- Ruha, A.M.; Curry, S.C.; Beuhler, M.; Katz, K.; Brooks, D.E.; Graeme, K.A.; Wallace, K.; Gerkin, R.; Lovecchio, F.; Wax, P.; et al. Initial postmarketing experience with crotalidae polyvalent immune Fab for treatment of rattlesnake envenomation. Ann. Emerg. Med. 2002, 39, 607–615. [Google Scholar] [CrossRef]
- Rucavado, A.; Soto, M.; Escalante, T.; Loría, G.D.; Arni, R.; Gutiérrez, J.M. Thrombocytopenia and platelet hypoaggregation induced by Bothrops asper snake venom. Toxins involved and their contribution to metalloproteinase-induced pulmonary hemorrhage. Thromb. Haemost. 2005, 94, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.L.; Sano-Martins, I.S. Platelet dysfunction during Bothrops jararaca snake envenomation in rabbits. Thromb. Haemost. 2004, 92, 369–383. [Google Scholar] [CrossRef] [PubMed]
- De Morais, I.C.; Torres, A.F.; Pereira, G.J.; Pereira, T.P.; de Menezes, R.R.P.B.; Mello, C.P.; Jorge, A.R.C.; Bindá, A.H.; Toyama, M.H.; Monteiro, H.S.; et al. Bothrops leucurus venom induces nephrotoxicity in the isolated perfused kidney and cultures renal tubular epithelia. Toxicon 2013, 61, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.H.; Tan, N.H.; Sim, S.M.; Fung, S.Y.; Jayalakshmi, P.; Gnanathasan, C.A. Nephrotoxicity of hump-nosed pit viper (Hypnale hypnale) venom in mice is preventable by the paraspecific Hemato polyvalent antivenom (HPA). Toxicon 2012, 60, 1259–1262. [Google Scholar] [CrossRef] [PubMed]
- Resiere, D.; Mégarbane, B.; Valentino, R.; Mehdaoui, H.; Thomas, L. Bothrops lanceolatus bites: Guidelines for severity assessment and emergent management. Toxicon 2010, 2, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Höjer, J.; Tran Hung, H.; Warrell, D.A. Life-threatening hyponatremia after krait bite envenoming—A new syndrome. Clin. Toxicol. Phila. 2010, 48, 956–957. [Google Scholar] [CrossRef] [PubMed]
- Bdolah, A.; Wollberg, Z.; Ambar, I.; Kloog, Y.; Sokolovsky, M.; Kochva, E. Disturbances in the cardiovascular system caused by endothelin and sarafotoxin. Biochem. Pharmacol. 1989, 38, 3145–3146. [Google Scholar] [CrossRef]
- Calvete, J.J.; Arias, A.S.; Rodríguez, Y.; Quesada-Bernat, S.; Sánchez, L.V.; Chippaux, J.P.; Pla, D.; Gutiérrez, J.M. Preclinical evaluation of three polyspecific antivenoms against the venom of Echis ocellatus: Neutralization of toxic activities and antivenomics. Toxicon 2016, 119, 280–288. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Lomonte, B.; Lohse, B.; Fernández, J.; Gutiérrez, J.M. Unveiling the nature of black mamba (Dendroaspis polylepis) venom through venomics and antivenom immunoprofiling: Identificatin of key toxin targets for antivenom development. J. Proteom. 2015, 119, 126–142. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, L.V.; Pla, D.; Herrera, M.; Chippaux, J.P.; Calvete, J.J.; Gutiérrez, J.M. Evaluation of the preclinical efficacy of four antivenoms, distributed in sub-Saharan Africa, to neutralize the venom of the carpet viper, Echis ocellatus, from Mali, Cameroon, and Nigeria. Toxicon 2015, 106, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; León, G.; Lomonte, B. Pharmacokinetic-pharmacodynamic relationships of immunoglobulin therapy for envenomation. Clin. Pharmacokinet. 2003, 42, 721–741. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; León, G.; Rojas, G.; Lomonte, B.; Rucavado, A.; Chaves, F. Neutralization of local tissue damage induced by Bothrops asper (terciopelo) snake venom. Toxicon 1998, 36, 1529–1538. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Ownby, C.L.; Odell, G.V. Pathogenesis of myonecrosis induced by crude venom and a myotoxin of Bothrops asper. Exp. Mol. Pathol. 1984, 40, 367–379. [Google Scholar] [CrossRef]
- Moreira, L.; Gutiérrez, J.M.; Borkow, G.; Ovadia, M. Ultrastructural alterations in mouse capillary blood vessels after experimental injection of venom from the snake Bothrops asper. Exp. Mol. Pathol. 1992, 57, 124–133. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Chaves, F.; Bolaños, R.; Cerdas, L.; Rojas, E.; Arroyo, O.; Portilla, E. Neutralización de los efectos locales del veneno de Bothrops asper por un antiveneno polivalente. Toxicon 1981, 19, 493–500. [Google Scholar] [CrossRef]
- Rivel, M.; Solano, D.; Herrera, M.; Vargas, M.; Villalta, M.; Segura, Á.; Arias, A.S.; León, G.; Gutiérrez, J.M. Pathogenesis of dermonecrosis induced by venom of the spitting cobra, Naja nigricollis: An experimental study in mice. Toxicon 2016, 119, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Abd-Elsalam, M.A. Pharmacokinetics of 125I-labelled IgG, F(ab’)2 and Fab fractions of scorpion and snake antivenins: Merits and potential for therapeutic use. Toxicon 1998, 36, 1523–1528. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Lomonte, B.; León, G.; Rucavado, A.; Chaves, F.; Angulo, Y. Trends in snakebite envenomation therapy: Scientific, technological and public health considerations. Curr. Pharm. Des. 2007, 13, 2935–2950. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L.; Hider, R.C.; Khader, F. Effect of phospholipase A on actions of cobra venom cardiotoxins on erythrocytes and skeletal muscle. Biochim. Biophys. Acta 1983, 728, 215–221. [Google Scholar] [CrossRef]
- Bustillo, S.; Gay, C.C.; García Denegri, M.E.; Ponce-Soto, L.A.; Bal de Kier, J.E.; Acosta, O.; Leiva, L.C. Synergism between baltergin metalloproteinase and Ba SPII RP4 PLA2 from Bothrops alternatus venom on skeletal muscle (C2C12) cells. Toxicon 2012, 59, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Barfaraz, A.; Harvey, A.L. The use of the chick biventer cervicis preparation to assess the protective activity of six international reference antivenoms on the neuromuscular effects of snake venoms in vitro. Toxicon 1994, 32, 267–272. [Google Scholar] [CrossRef]
- Beghini, D.G.; Hernandez-Oliveira, S.; Rodrigues-Simioni, L.; Novello, J.C.; Hyslop, S.; Marangoni, S. Anti-sera raised in rabbits against crotoxin and phospholipase A2 from Crotalus durissus cascavella venom neutralize the neurotoxicity of the venom and crotoxin. Toxicon 2004, 44, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Maria, W.S.; Pacheco, B.G.; Barbosa, C.F.; Velarde, D.T.; Chávez-Olórtegui, C. Determination of the neutralizing potency of horse antibothropic and anticrotalic antivenoms in blood samples collected in filter paper. Toxicon 2001, 39, 1607–1609. [Google Scholar] [CrossRef]
- Rial, A.; Morais, V.; Rossi, S.; Massaldi, H. A new ELISA for determination of potency in snake antivenoms. Toxicon 2006, 48, 462–466. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Gutiérrez, J.M.; Lohse, B.; Rasmussen, A.R.; Fernández, J.; Milbo, C.; Lomonte, B. Snake venomics of the monocled cobra (Naja kaouthia) and investigation of human IgG response against venom toxins. Toxicon 2015, 99, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Laustsen, A.H.; Lohse, B.; Lomonte, B.; Engmark, M.; Gutiérrez, J.M. Selecting key toxins for focused development of elapid snake antivenoms and inhibitors guided by a Toxicity Score. Toxicon 2015, 104, 43–45. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.M.; Lomonte, B. A bright future for integrative venomics. Toxicon 2015, 107, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Sanny, C.G. In vitro evaluation of total venom-antivenin immune complex formation and binding parameters relevant to antivenin protection against venom toxicity and lethality based on size-exclusion high-performance liquid chromatography. Toxicon 2011, 57, 871–881. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, M.A.; Maduwage, K.; Isbister, G.K. Use of immunoturbidimetry to detect venom-antivenom binding using snake venoms. J. Pharmacol. Toxicol. Meth. 2013, 67, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Avila, C.; Rojas, E.; Cerdas, L. An alternative in vitro method for testing the potency of the polyvalent antivenom produced in Costa Rica. Toxicon 1988, 26, 411–413. [Google Scholar] [CrossRef]
- Ownby, C.L.; Fletcher, J.E.; Colberg, T.R. Cardiotoxin 1 from cobra (Naja naja atra) venom causes necrosis of skeletal muscle in vivo. Toxicon 1993, 31, 697–709. [Google Scholar] [CrossRef]
- Lomonte, B.; Angulo, Y.; Rufini, S.; Cho, W.; Giglio, J.R.; Ohno, M.; Daniele, J.J.; Geoghegan, P.; Gutiérrez, J.M. Comparative study of the cytolytic activity of myotoxic phosphlipases A2 on mouse endothelial (tEnd) and skeletal muscle (C2C12) cells in vitro. Toxicon 1999, 37, 145–158. [Google Scholar] [CrossRef]
- Méndez, I.; Gutiérrez, J.M.; Angulo, Y.; Calvete, J.J.; Lomonte, B. Comparative study of the cytolytic activity of snake venoms from African spitting cobras (Naja spp., Elapidae) and its neutralization by a polyspecific antivenom. Toxicon 2011, 58, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Sitprija, V.; Sitprija, S. Renal effects and injury induced by animal toxins. Toxicon 2012, 60, 943–953. [Google Scholar] [CrossRef] [PubMed]
- Escalante, T.; Rucavado, A.; Fox, J.W.; Gutiérrez, J.M. Key events in microvascular damage induced by snake venom hemorrhagic metalloproteinases. J. Proteom. 2011, 74, 1781–1794. [Google Scholar] [CrossRef] [PubMed]
- Sells, P.G.; Richards, A.M.; Laing, G.D.; Theakston, R.D.G. The use of hens’ eggs as an alternative to the conventional in vivo rodent assay for antidotes to haemorrhagic venoms. Toxicon 1997, 35, 1413–1421. [Google Scholar] [CrossRef]
- Barber, C.M.; Madaras, F.; Turnbull, R.K.; Morley, T.; Dunstan, N.; Allen, L.; Kuchel, T.; Mirtschin, P.; Hodgson, W.C. Comparative studies of the venom of a new Taipan species, Oxyuranus temporalis, with other members of its genus. Toxins 2014, 6, 1979–1995. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.; Flecknell, P.; Thomas, A.; Warrell, D.A. On the use of analgesia in experimental toxinology. Toxicon 2013, 64, 36–37. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Herrera, C. The analgesics morphine and tramadol do not alter the acute toxicity induced by Bothrops asper snake venom in mice. Toxicon 2014, 81, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Eichberg, S.; Sanz, L.; Calvete, J.J.; Pla, D. Constructing comprehensive venom proteome reference maps for integrative venomics. Expert Rev. Proteom. 2015, 12, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J.; Juárez, P.; Sanz, L. Snake venomics. Strategy and applications. J. Mass Spectrom. 2007, 42, 1405–1414. [Google Scholar] [CrossRef] [PubMed]
- Calvete, J.J. Snake venomics: From the inventory of toxins to biology. Toxicon 2013, 75, 44–62. [Google Scholar] [CrossRef] [PubMed]
- Lomonte, B.; Escolano, J.; Fernández, J.; Sanz, L.; Angulo, Y.; Gutiérrez, J.M.; Calvete, J.J. Snake venomics and antivenomics of the arboreal neotropical pitvipers Bothriechis lateralis and Bothriechis schlegelii. J. Proteom. Res. 2008, 7, 2445–2457. [Google Scholar] [CrossRef] [PubMed]
- Pla, D.; Gutiérrez, J.M.; Calvete, J.J. Second generation snake antivenomics: Comparing immunoaffinity and immunodepletion protocols. Toxicon 2012, 60, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, J.M.; Lomonte, B.; Sanz, L.; Calvete, J.J.; Pla, D. Immunological profile of antivenoms: Preclinical analysis of the efficacy of a polyspecific antivenom through antivenomics and neutralization assays. J. Proteom. 2014, 105, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Wang, J.; He, Y.; Qu, Y.F.; Lin, L.H.; Ma, X.M.; Ji, X. Proteomic and biochemical analyses of short-tailed viper (Gloydius brevicaudus) venom: Age-related variation and composition-activity correlation. J. Proteom. 2014, 105, 307–322. [Google Scholar] [CrossRef] [PubMed]
- Saviola, A.J.; Pla, D.; Sanz, L.; Castoe, T.A.; Calvete, J.J.; Mackessy, S.P. Comparative venomics of the Prairie Rattlesnake (Crotalus viridis viridis) from Colorado: Identification of a novel pattern of ontogenetic changes in venom composition and assessment of the immunoreactivity of the commercial antivenom CroFab®. J. Proteom. 2015, 121, 28–43. [Google Scholar] [CrossRef] [PubMed]
- Shan, L.L.; Gao, J.F.; Zhang, Y.X.; Shen, S.S.; He, Y.; Wang, J.; Ma, X.M.; Ji, X. Proteomic characterization and comparison of venoms from two elapid snakes (Bungarus multicinctus and Naja atra) from China. J. Proteom. 2016, 138, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Pla, D.; Bande, B.W.; Welton, R.E.; Paiva, O.K.; Sanz, L.; Segura, Á.; Wright, C.; Calvete, J.J.; Gutiérrez, J.M.; Williams, D.J. Proteomics and antivenomics of Papuan black snake (Pseudechis papuanus) venom with analysis of its toxicological profile and the preclinical efficacy of Australian antivenoms. J. Proteom. 2017, 150, 201–215. [Google Scholar] [CrossRef] [PubMed]
- Sintiprungrat, K.; Chausuriya, P.; Watcharatanyatip, K.; Ratanabanangkoon, K. Immunoaffinity chromatography in antivenomics studies: Various parameters that can affect the results. Toxicon 2016, 119, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Pla, D.; Sanz, L.; Whiteley, G.; Wagstaff, S.C.; Harrison, R.A.; Casewell, N.R.; Calvete, J.J. What killed Karl Patterson Schmidt? Combined venom gland transcriptomic, venomic and antivenomic analysis of the South African green tree snake (the boomslang), Dispholidus typus. Biochim. Biophys. Acta 2017, 1861, 814–823. [Google Scholar] [CrossRef] [PubMed]
- Gay, C.; Sanz, L.; Calvete, J.J.; Pla, D. Snake venomics and antivenomics of Bothrops diporus, a medically important pitviper in northeastern Argentina. Toxins 2016, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Otero, R.; Núñez, V.; Osorio, R.G.; Gutiérrez, J.M.; Giraldo, C.A.; Posada, L.E. Ability of six Latin American antivenoms to neutralize the venom of mapaná equis (Bothrops atrox) from Antioquia and Chocó (Colombia). Toxicon 1995, 33, 809–815. [Google Scholar] [CrossRef]
- De Roodt, A.R.; Dolab, J.A.; Fernández, T.; Segre, L.; Hajos, S.E. Cross-reactivity and heterologous neutralization of crotaline antivenoms used in Argentina. Toxicon 1998, 36, 1025–1038. [Google Scholar] [CrossRef]
- Maduwage, K.; Silva, A.; O´Leary, M.A.; Hodgson, W.C.; Isbister, G.K. Efficacy of Indian polyvalent snake antivenoms against Sri Lankan snake venoms: Lethality studies or clinically focused in vitro studies. Sci. Rep. 2016, 6, 26778. [Google Scholar] [CrossRef] [PubMed]
- Isbister, G.K.; O’Leary, M.A.; Hagan, J.; Nichols, K.; Jacoby, T.; Davern, K.; Hodgson, W.C.; Schneider, J.J. Cross-neutralisation of Australian brown snake, taipan and adder venoms by monovalent antivenoms. Vaccine 2010, 28, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.; Gutiérrez, J.M.; Harrison, R.; Warrell, D.A.; White, J.; Winkel, K.D.; Gopalakrishnakone, P. The Global Snake Bite Initiative: An antidote for snake bite. Lancet 2010, 375, 89–91. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez, J.M.; Solano, G.; Pla, D.; Herrera, M.; Segura, Á.; Vargas, M.; Villalta, M.; Sánchez, A.; Sanz, L.; Lomonte, B.; et al. Preclinical Evaluation of the Efficacy of Antivenoms for Snakebite Envenoming: State-of-the-Art and Challenges Ahead. Toxins 2017, 9, 163. https://doi.org/10.3390/toxins9050163
Gutiérrez JM, Solano G, Pla D, Herrera M, Segura Á, Vargas M, Villalta M, Sánchez A, Sanz L, Lomonte B, et al. Preclinical Evaluation of the Efficacy of Antivenoms for Snakebite Envenoming: State-of-the-Art and Challenges Ahead. Toxins. 2017; 9(5):163. https://doi.org/10.3390/toxins9050163
Chicago/Turabian StyleGutiérrez, José María, Gabriela Solano, Davinia Pla, María Herrera, Álvaro Segura, Mariángela Vargas, Mauren Villalta, Andrés Sánchez, Libia Sanz, Bruno Lomonte, and et al. 2017. "Preclinical Evaluation of the Efficacy of Antivenoms for Snakebite Envenoming: State-of-the-Art and Challenges Ahead" Toxins 9, no. 5: 163. https://doi.org/10.3390/toxins9050163
APA StyleGutiérrez, J. M., Solano, G., Pla, D., Herrera, M., Segura, Á., Vargas, M., Villalta, M., Sánchez, A., Sanz, L., Lomonte, B., León, G., & Calvete, J. J. (2017). Preclinical Evaluation of the Efficacy of Antivenoms for Snakebite Envenoming: State-of-the-Art and Challenges Ahead. Toxins, 9(5), 163. https://doi.org/10.3390/toxins9050163