Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters
Abstract
:1. Introduction
2. Results
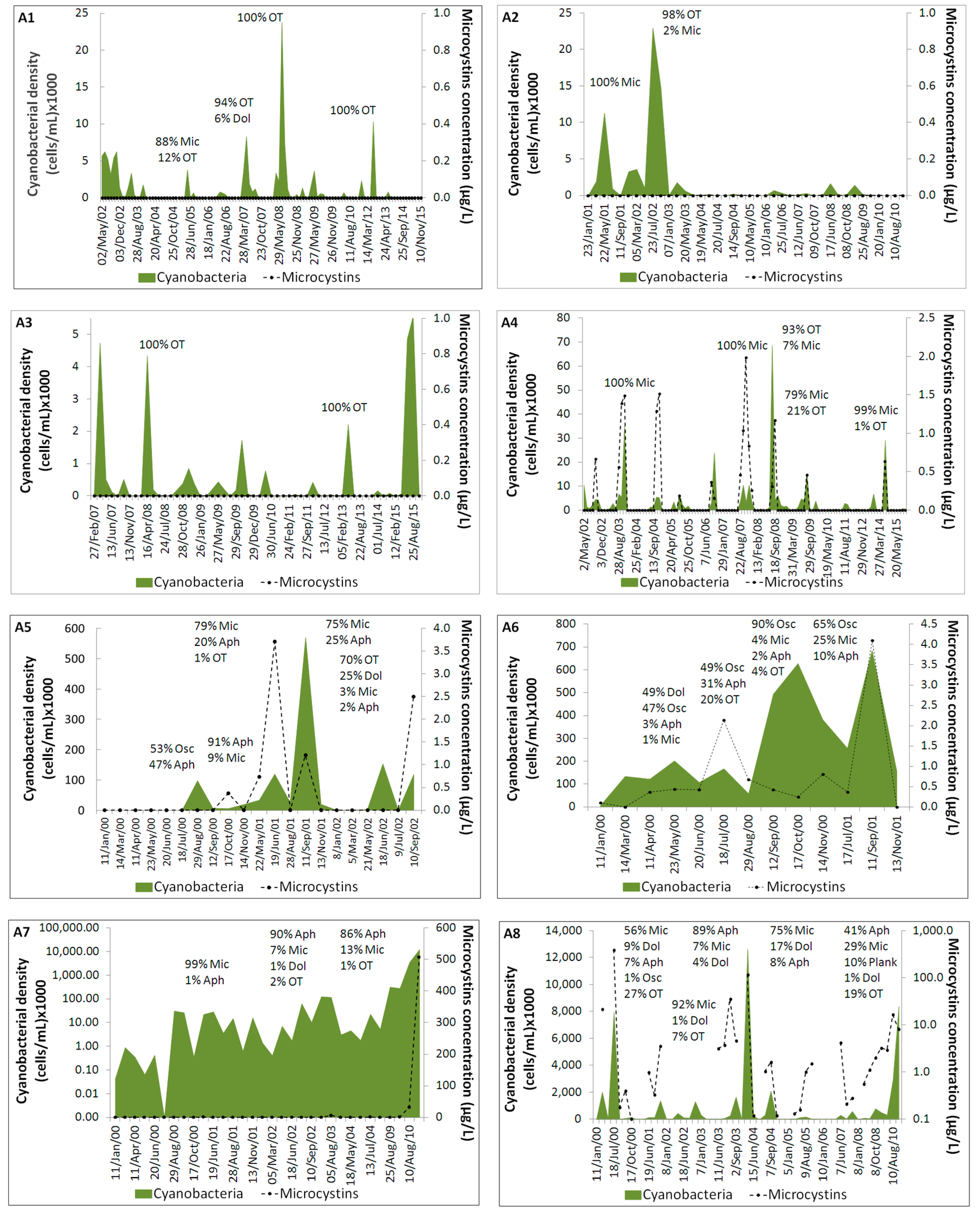
2.1. Cyanobacteria and Microcystins Profiles in the Studied Reservoirs
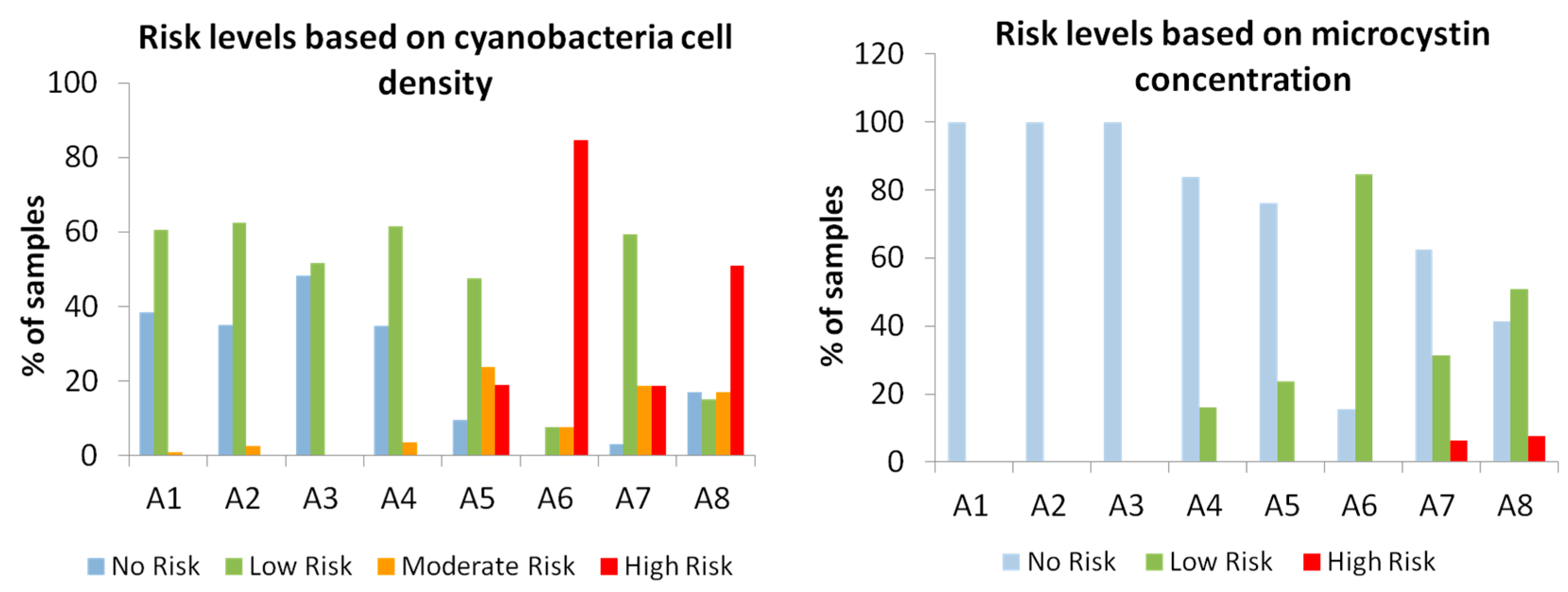
2.2. Risk Levels of Toxic Cyanobacteria Occurrence in the Studied Reservoirs
2.3. Human Exposure Scenarios to Microcystins during Recreational Activities in the Studied Reservoirs
3. Discussion
4. Conclusions
5. Materials and Methods
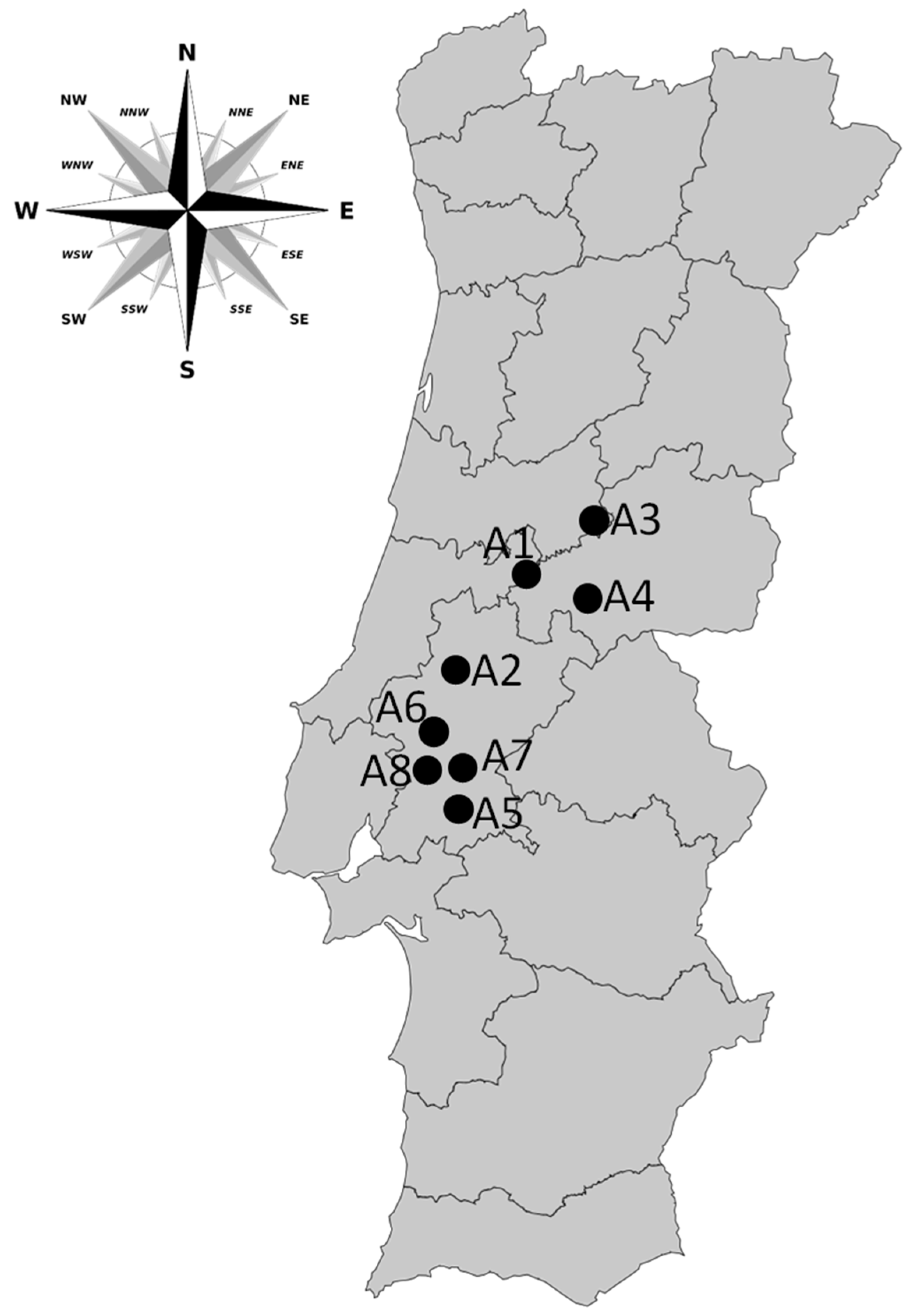
5.1. Study Approach
5.2. Sampling and Sample Processing
5.3. Phytoplankton Analysis
5.4. Quantification of Microcystins in Water Samples
5.5. Risk Levels and Human Exposure Scenarios
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Buratti, F.M.; Manganelli, M.; Vichi, S.; Stefanelli, M.; Scardala, S.; Testai, E.; Funari, E. Cyanotoxins: Producing organisms, occurrence, toxicity, mechanism of action and human health toxicological risk evaluation. Arch. Toxicol. 2017, 91, 1049–1130. [Google Scholar] [CrossRef] [PubMed]
- Antunes, J.T.; Leão, P.N.; Vasconcelos, V.M. Cylindrospermopsis raciborskii: Review of the distribution, phylogeography, and ecophysiology of a global invasive species. Front. Microbiol. 2015, 6, 473. [Google Scholar] [CrossRef] [PubMed]
- Funari, E.; Testai, E. Human health risk assessment related to cyanotoxins exposure. Crit. Rev. Toxicol. 2008, 38, 97–125. [Google Scholar] [CrossRef] [PubMed]
- PNA. Plano Nacional da Água—National Water Plan. Agência Portuguesa do Ambiente, Republica Portuguesa. 2015. Available online: http://www.apambiente.pt/?ref=16&subref=7&sub2ref=9&sub3ref=833 (accessed on 20 July 2016).
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Goblerc, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Vasconcelos, V.; Sivonen, K.; Evans, W.R.; Carmichael, W.W.; Namikoshi, M. Hepatotoxic microcystin diversity in cyanobacterial blooms collected in Portuguese freshwaters. Water Res. 1996, 30, 2377–2384. [Google Scholar] [CrossRef]
- Vasconcelos, V.M. Cyanobacterial toxins in Portugal: Effects on aquatic animals and risk for human health. Braz. J. Med. Biol. Res. 1999, 32, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.; Onodera, H.; Andrinolo, D.; Franca, S.; Araújo, F.; Lagos, N.; Oshima, Y. Co-occurrence of PSP toxins and microcystins in Montargil freshwater reservoir, Portugal. In Harmful Algal Blooms 2000, Proceedings of the Ninth International Conference on Harmful Algal Blooms, Hobart, Australia, 7–11 February 2000; Hallegraeff, G.M., Bolch, C.J., Lewis, R.J., Eds.; IOC, UNESCO: Paris, France, 2001; pp. 108–111. [Google Scholar]
- Dias, E.; Pereira, P.; Franca, S. Production of paralytic shellfish toxins by Aphanizomenon sp. LMECYA 31 (cyanobacteria). J. Phycol. 2002, 38, 705–712. [Google Scholar] [CrossRef]
- Galvão, H.; Reis, M.; Valério, E.; Domingues, R.; Costa, C.; Lourenço, D.; Condinho, S.; Miguel, R.; Barbosa, A.; Gago, C.; et al. Cyanobacterial blooms in natural waters in Southern Portugal: A water management perspective. Aquat. Microbiol. Ecol. 2008, 53, 129–140. [Google Scholar] [CrossRef]
- Vasconcelos, V.; Morais, J.; Vale, M. Microcystins and cyanobacteria trends in a 14 year monitoring of a temperate eutrophic reservoir (Aguieira, Portugal). J. Environ. Monit. 2011, 13, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Churro, C.; Dias, E.; Valério, E. Risk Assessment of Cyanobacteria and Cyanotoxins, the Particularities and Challenges of Planktothrix spp. Monitoring. In Novel Approaches and Their Applications in Risk Assessment; Luo, Y., Ed.; InTech: Rijeka, Croatia, 2012; Chapter 4; pp. 59–84. ISBN 978-9-53-510519-0. [Google Scholar]
- Bellém, F.; Nunes, S.; Morais, M. Cyanobacteria toxicity: Potential public health impact in south Portugal populations. J. Toxicol. Environ. Health Part A 2013, 76, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Decreto-Lei, n.º 306/2007 de 27 de Agosto, Diário da República, I Série, n.º 164, 27 de Agosto de 2007. Available online: https://dre.pt/application/dir/pdf1sdip/2007/08/16400/0574705765.pdf (accessed on 20 July 2016).
- COUNCIL DIRECTIVE 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31998L0083&from=PT (accessed on 20 July 2016).
- Decreto-Lei nº 135/2009 de 3 de Junho, Diário da República, I Série, nº 107 de 3 de Junho. Available online: http://snirh.pt/snirh/_divulgacao/legislacao/site/verlei.php?id=20646 (accessed on 20 July 2016).
- Decreto-Lei nº 113/2012 de 23 de Maio, Diário da República, I Série, nº100 de 23 de Maio. Available online: http://publicos.pt/documento/id177865/decreto-lei-113/2012 (accessed on 20 July 2016).
- DIRECTIVE 2006/7/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 15 February 2006 Concerning the Management of Bathing Water Quality and Repealing Directive 76/160/EEC. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:064:0037:0051:EN:PDF (accessed on 20 July 2016).
- Chorus, I. (Ed.) Current Approaches to Cyanotoxin Risk Assessment, Risk Management and Regulations in Different Countries; Federal Environment Agency: Dessau-Roßlau, Germany, 2012. Available online: http://www.uba.de/uba-infomedien-e/4390.html (accessed on 20 July 2016).
- Ibelings, B.W.; Backer, L.C.; Kardinaal, W.E.A.; Chorus, I. Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 2015, 49, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, V. Cyanobacteria toxins: Diversity and ecological effects. Limnetica 2001, 20, 45–48. Available online: http://www.limnetica.com/Limnetica/Limne20/L20a045_Cyanobacterial_toxins.pdf (accessed on 20 July 2016).
- DGS. Direcção Geral da Saúde. 2011. Available online: http://www.dgs.pt/saude-ambiental/areas-de-intervencao/agua.aspx (accessed on 20 July 2016).
- Valério, E.; Faria, N.; Paulino, S.; Pereira, P. Seasonal variation of phytoplankton and cyanobacteria composition and associated microcystins in six Portuguse freshwater reservoirs. Ann. Limnol. Int. J. Limnol. 2008, 44, 189–196. [Google Scholar] [CrossRef]
- Valério, E.; Chambel, L.; Paulino, S.; Faria, N.; Pereira, P.; Tenreiro, R. Molecular identification, typing and traceability of cyanobacteria from freshwater reservoirs. Microbiology 2009, 155, 642–656. [Google Scholar] [CrossRef] [PubMed]
- Valério, E.; Chambel, L.; Paulino, S.; Faria, N.; Pereira, P.; Tenreiro, R. Multiplex PCR for Detection of Microcystins-Producing Cyanobacteria from Freshwater Samples. Environ. Toxicol. 2010, 25, 251–260. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Algae and cyanobacteria in fresh water. In Guidelines for Safe Recreational Water Environments; Coastal and Fresh Waters; World Health Organization: Geneva, Switzerland, 2003; Volume 1, pp. 136–158. Available online: http://apps.who.int/iris/bitstream/10665/42591/1/9241545801.pdf (accessed on 20 July 2016).
- Vasconcelos, V.M. Isolation of microcystin-LR from a Microcystis (cyanobacteria) waterbloom collected in the drinking water reservoir for Porto, Portugal. J. Environ. Sci. Health A 1993, 28, 2081–2094. [Google Scholar] [CrossRef]
- Vasconcelos, V.M. Toxic cyanobacteria (blue-green algae) in Portuguese freshwaters. Arch. Hydrobiol. 1994, 130, 139–451. Available online: https://sigarra.up.pt/faup/pt//pub_geral.pub_view?pi_pub_base_id=97400 (accessed on 20 July 2016).
- Pereira, P.; Li, R.; Carmichael, W.; Dias, E.; Franca, S. Taxonomy and production of paralytic shellfish toxins by the freswater cyanobacterium Aphanizomenon gracile LMECYA40. Eur. J. Phycol. 2004, 39, 361–368. [Google Scholar] [CrossRef]
- Figueiredo, D.; Reboleira, A.; Antunes, S.; Abrantes, N.; Azeitão, U.; Gonçalves, F.; Pereira, M. The effect of environmental parameters and cyanobacterial blooms on phytoplankton dynamics of a Portuguese temperate lake. Hydrobiologia 2006, 568, 145–157. [Google Scholar] [CrossRef]
- Churro, C.; Pereira, P.; Vasconcelos, V.; Valério, E. Species-specific real-time PCR cell number quantification of the bloom-forming cyanobacterium Planktothrix agardhii. Arch. Microbiol. 2012, 194, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Paulino, S.; Valério, E.; Faria, N.; Fastner, J.; Welker, M.; Tenreiro, R.; Pereira, P. Detection of Planktothrix rubescens (cyanobacteria) associated with microcystin production in a freshwater reservoir. Hydrobiologia 2009, 621, 207–211. [Google Scholar] [CrossRef]
- Vasconcelos, V.; Sivonen, K.; Evans, W.R.; Carmichael, W.W.; Namikoshi, M. Isolation and characterization of microcystins (heptapeptide hepatotoxins) from Portuguese strains of Microcystis aeruginosa Kutz. emed Elekin. Arch. Hydrobiol. 1995, 134, 295–305. [Google Scholar]
- Pereira, P.; Onodera, H.; Andrinolo, D.; Franca, S.; Araújo, F.; Lagos, N.; Oshima, Y. Paralytic shellfish toxins in the freshwater cyanobacterium Aphanizomenon flos-aquae, isolated from Montargil reservoir, Portugal. Toxicon 2000, 38, 1689–1702. [Google Scholar] [CrossRef]
- Ferreira, F.; Solerb, J.; Fidalgoa, M.; Fernández-Vilac, P. PSP toxins from Aphanizomenon flos-aquae (cyanobacteria) collected in the Crestuma-Lever reservoir (Douro river, northern Portugal). Toxicon 2001, 39, 757–761. [Google Scholar] [CrossRef]
- Osswald, J.; Rellán, S.; Gago-Martinez, A.; Vasconcelos, V. Production of anatoxin-a by cyanobacterial strains isolated from Portuguese fresh water systems. Ecotoxicology 2009, 18, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Menezes, C.; Martins, O.; Dias, E. Tratamento de águas para consumo humano: Um episódio de sobrevivência de cianobactérias. Boletim Epidemiológico Observações. 2016, 5 (Suppl. S8), 14–17. Available online: http://repositorio.insa.pt/bitstream/10400.18/4135/3/observacoesNEspecia8–2016_artigo14.pdf (accessed on 20 July 2016).
- Vasconcelos, V. Emergent Marine Toxins in Europe: Is there a new Invasion? Mar. Sci. Res. Dev. 2013, 3, e117. [Google Scholar] [CrossRef]
- Giannuzzi, L.; Sedan, D.; Echenique, R.; Andrinolo, D. An Acute Case of Intoxication with Cyanobacteria and Cyanotoxins in Recreational Water in Salto Grande Dam, Argentina. Mar. Drugs 2011, 9, 2164–2175. [Google Scholar] [CrossRef] [PubMed]
- Vidal, F.; Sedan, D.; D’Agostino, D.; Cavalieri, M.; Mullen, E.; Varela, M.; Flores, C.; Caixach, J.; Andrinolo, D. Recreational Exposure during Algal Bloom in Carrasco Beach, Uruguay: A Liver Failure Case Report. Toxins 2017, 9, 267. [Google Scholar] [CrossRef] [PubMed]
- Zanchett, G.; Oliveira-Filho, E.C. Cyanobacteria and Cyanotoxins: From Impacts on Aquatic Ecosystems and Human Health to Anticarcinogenic Effects. Toxins 2013, 5, 1896–1917. [Google Scholar] [CrossRef] [PubMed]
- Backer, L.; Carmichael, W.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.; Nierenberg, K.; Hill, V.; Kieszak, S.; et al. Recreational Exposure to Low Concentrations of Microcystins during an Algal Bloom in a Small Lake. Mar. Drugs 2008, 6, 389–406. [Google Scholar] [CrossRef]
- Backer, L.C.; McNeel, S.; Barber, T.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.; Nierenberg, K.; Aubel, M.; et al. Recreational exposure to microcystins during algal blooms in two California lakes. Toxicon 2010, 55, 909–921. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Praena, D.; Campos, A.; Azevedo, J.; Neves, J.; Freitas, M.; Guzmán-Guillén, R.; Cameán, A.M.; Renaut, J.; Vasconcelos, V. Exposure of Lycopersicon esculentum to Microcystin-LR: Effects in the Leaf Proteome and Toxin Translocation from Water to Leaves and Fruits. Toxins 2014, 6, 1837–1854. [Google Scholar] [CrossRef] [PubMed]
- Araújo, F. Cianobacterias: Ocurrencias y gestión del riesgo en Portugal de 1993 a 2005. In Jornada Sobre las Cianobacterias tóxicas. Problemas asociados. Seguimiento y control; Madrid, Spain, 2006; Available online: http://ceh-flumen64.cedex.es/Ecosistemas/Jornadas%20Cianos/F.Araujo.pdf (accessed on 20 July 2016).
- CyanoNews. Toxin Suspect in Mass Killing. Cyanonews 1996, 12, 4. Available online: http://wwwcyanosite.bio.purdue.edu/cyanonews/cn122/cn122.pdf (accessed on 20 July 2016).
- Bellém, F. As Cianobactérias Na água e a Morbilidade e Mortalidade na Região do Alentejo. Ph.D. Thesis, University of Évora, Évora, Portugal, 2014. Available online: http://hdl.handle.net/10174/12251 (accessed on 20 July 2016).
- Welles, M.L.; Trainer, V.L.; Smayda, T.J.; Karlson, B.S.O.; Trick, C.G.; Kudela, R.M.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.M.; et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 2015, 49, 68–93. [Google Scholar] [CrossRef] [PubMed]
- Gkelis, S.; Papadimitriou, T.; Zaoutsos, N.; Leonardos, I. Anthropogenic and climate-induced change favors toxic cyanobacteria blooms: Evidence from monitoring a highly eutrophic, urban Mediterranean lake. Harmful Algae 2014, 39, 322–333. [Google Scholar] [CrossRef]
- Vlamis, A; Katikou, P. Human impact in Mediterranean coastal ecosystems and climate change: Emerging toxins. In Climate Change and Marine and Freshwater Toxins; Botana, L., Louzao, C., Natalia, V., Eds.; Walter de Gruyter: Berlin, Germany, 2015; Chapter 8; pp. 239–272. ISBN 978-3-11-033359-6. [Google Scholar]
- Pires, V.; Silva, A.; Mendes, L. Riscos de Secas em Portugal Continental. Territorium 2010, 17, 27–34. [Google Scholar] [CrossRef]
- Portuguese Institute for the Sea and Atmosphere (Instituto Português do Mar e Atmosfera). Available online: http://www.ipma.pt/pt/publicacoes/boletins.jsp?cmbDep=cli&cmbTema=pcl&idDep=cli&idTema=pcl&curAno=-1 (accessed on 25 September 2017).
- Diogo, P. Fontes de Fósforo Total e o Estado Trófico de Albufeiras em Portugal Continental. Master’s Thesis, Faculty of Sciences and Technology of the New University of Lisbon, Lisbon, Portugal, May 2008. Available online: https://run.unl.pt/bitstream/10362/1685/1/Diogo_2008.pdf (accessed on 25 September 2017).
- National Information System on Water Resources (Sistema Nacional de Informação de Recursos Hídricos). Available online: http://snirh.apambiente.pt (accessed on 11 September 2017).
- European Committee for Standardization. Water Quality—Guidance Standard on Enumeration of Phytoplankton Using Inverted Microscopy (Utermöhl Technique); European Standard EN 15204:2006; European Committee for Standardization: Brussels, Belgium, 2006. [Google Scholar]
- Gkelis, S.; Lanaras, T.; Sivonen, K. Cyanobacterial Toxic and Bioactive Peptides in Freshwater Bodies of Greece: Concentrations, Occurrence Patterns, and Implications for Human Health. Mar. Drugs 2015, 13, 6319–6335. [Google Scholar] [CrossRef] [PubMed]
| Reservoir | Samples with Blooms (%) | Samples with Microcystins (%) |
|---|---|---|
| A1 | 30 | 0 |
| A2 | 25 | 0 |
| A3 | 15 | 0 |
| A4 | 44 | 16 |
| A5 | 77 | 24 |
| A6 | 100 | 85 |
| A7 | 69 | 38 |
| A8 | 79 | 58 |
| Cyanobacteria Species | Maximum Cell Density (Cells/mL) | Frequency of Occurrence (% of Samples) | Frequency of Blooming (% of Samples) |
|---|---|---|---|
| Reservoir A1 | |||
| Aphanizomenon sp. | 26 | 1 | 0 |
| Aphanizomenon gracile | 20 | 1 | 0 |
| Aphanothece sp. | 4980 | 11 | 4 |
| Aphanocapsa sp. | 10,314 | 3 | 1 |
| Aphanocapsa holsatica | 410 | 1 | 0 |
| Coelosphaerium sp. | 266 | 1 | 0 |
| Chroococcus sp. | 10 | 1 | 0 |
| CHROOCOCCALES n.i. | 23,820 | 26 | 8 |
| Dolichospermum sp. | 506 | 5 | 0 |
| Gomphosphaeria sp. | 169 | 3 | 0 |
| Gloeothece linearis | 7426 | 1 | 1 |
| Leptolyngbya sp. | 233 | 1 | 0 |
| Merismopedia sp. | 128 | 2 | 0 |
| Microcystis sp. | 1078 | 6 | 0 |
| Microcystis aeruginosa | 3371 | 28 | 2 |
| Microcystis incerta | 210 | 1 | 0 |
| NOSTOCALES n.i. | 77 | 1 | 0 |
| Oscillatoria sp. | 3677 | 2 | 1 |
| OSCILLATORIALES n.i. | 659 | 3 | 0 |
| Planktolyngbya limnetica | 181 | 2 | 0 |
| Pseudanabaena sp. | 4157 | 9 | 1 |
| Pseudanabaena limnetica | 181 | 3 | 0 |
| Synechocystis sp. | 10 | 1 | 0 |
| Woronichinia naegeliana | 572 | 2 | 0 |
| Reservoir A2 | |||
| Aphanothece sp. | 17,408 | 5 | 5 |
| Aphanocapsa sp. | 20 | 3 | 0 |
| CHROOCOCCALES n.i. | 5198 | 30 | 3 |
| Lyngbya sp. | 92 | 3 | 0 |
| Microcystis sp. | 352 | 3 | 0 |
| Microcystis aeruginosa | 11,311 | 23 | 3 |
| Microcystis wesenberghii | 218 | 5 | 0 |
| Oscillatoria sp. | 153 | 5 | 0 |
| Pseudanabaena sp. | 3325 | 10 | 5 |
| OSCILLATORIALES n.i. | 919 | 10 | 0 |
| Reservoir A3 | |||
| Aphanizomenon sp. | 52 | 2 | 0 |
| Aphanizomenon flos-aquae | 14 | 2 | 0 |
| Aphanothece sp. | 271 | 2 | 0 |
| Aphanocapsa sp. | 2129 | 4 | 2 |
| Aphanocapsa incerta | 233 | 2 | 0 |
| Chroococcus sp. | 20 | 2 | 0 |
| CHROOCOCCALES n.i. | 4474 | 18 | 4 |
| Dactylococcopsis fascicularis | 31 | 4 | 0 |
| Microcystis sp. | 511 | 2 | 0 |
| Microcystis aeruginosa | 1736 | 16 | 0 |
| Microcystis wesenberghii | 174 | 2 | 0 |
| Merismopedia sp. | 20 | 2 | 0 |
| Planktothrix agardhii | 48 | 2 | 0 |
| Pseudanabaena sp. | 250 | 5 | 0 |
| Pseudanabaena limnetica | 276 | 9 | 0 |
| Phormidium sp. | 215 | 2 | 0 |
| Snowella sp. | 363 | 2 | 0 |
| Snowella atomus | 5576 | 2 | 2 |
| Snowella litoralis | 4624 | 2 | 2 |
| Reservoir A4 | |||
| Aphanothece sp. | 1098 | 5 | 0 |
| Anathece clathrata | 4876 | 1 | 1 |
| Aphanocapsa sp. | 14,076 | 1 | 1 |
| Aphanocapsa holsatica | 1352 | 1 | 0 |
| Chroococcus sp. | 817 | 2 | 0 |
| CHROOCOCCALES n.i. | 63,177 | 25 | 4 |
| Coelosphaerium sp. | 270 | 1 | 0 |
| Dactylococcopsis fascicularis | 3146 | 4 | 1 |
| Dolichospermum sp. | 312 | 4 | 0 |
| Gomphosphaeria sp. | 409 | 2 | 0 |
| Leptolyngbya sp. | 306 | 1 | 0 |
| Microcystis sp. | 521 | 2 | 0 |
| Microcystis aeruginosa | 35,301 | 29 | 19 |
| Microcystis ichthyoblabe | 28,990 | 5 | 1 |
| Microcystis wesenberghii | 20,429 | 1 | 1 |
| Merismopedia sp. | 61 | 3 | 0 |
| NOSTOCALES n.i. | 358 | 3 | 0 |
| Planktolyngbya limnetica | 405 | 3 | 0 |
| Planktothrix sp. | 521 | 3 | 0 |
| Planktothrix agardhii | 526 | 1 | 0 |
| Planktothrix isothrix | 1181 | 1 | 0 |
| Planktothrix pseudoagardhii | 271 | 1 | 0 |
| Oscillatoria sp. | 255 | 1 | 0 |
| Oscillatoria lacustris | 695 | 1 | 0 |
| Oscillatoria lauterbornii | 100 | 1 | 0 |
| Pseudanabaena sp. | 10,010 | 3 | 0 |
| Pseudanabaena limnetica | 171 | 1 | 0 |
| Pseudanabaena catenata | 210 | 3 | 0 |
| Pseudanabaena redekei | 52 | 1 | 0 |
| Phormidium sp. | 1154 | 3 | 0 |
| Snowella sp. | 603 | 1 | 0 |
| Synechocystis sp. | 112 | 2 | 0 |
| OSCILLATORIALES n.i. | 919 | 4 | 0 |
| Woronichinia naegeliana | 293 | 1 | 0 |
| Reservoir A5 | |||
| Aphanizomenon sp. | 14,632 | 10 | 10 |
| Aphanizomenon flos-aquae | 144,339 | 38 | 33 |
| Aphanothece sp. | 225 | 5 | 0 |
| CHROOCOCCALES n.i. | 2197 | 62 | 5 |
| Dactylococcopsis sp. | 82 | 10 | 0 |
| Dolichospermum sp. | 13,829 | 14 | 10 |
| Dolichospermum flos-aquae | 136 | 5 | 0 |
| Gomphosphaeria sp. | 81,165 | 19 | 10 |
| Microcystis sp. | 2370 | 5 | 5 |
| Microcystis aeruginosa | 426,966 | 43 | 24 |
| Microcystis wesenberghii | 791 | 5 | 0 |
| Merismopedia sp. | 198 | 5 | 0 |
| NOSTOCALES n.i. | 664 | 14 | 0 |
| Oscillatoria sp. | 51,858 | 10 | 5 |
| Pseudanabaena sp. | 1451 | 5 | 0 |
| Reservoir A6 | |||
| Aphanizomenon sp. | 65,307 | 8 | 8 |
| Aphanizomenon flos-aquae | 319,216 | 77 | 69 |
| Aphanizomenon issatschenkoi | 138,820 | 54 | 31 |
| Aphanothece sp. | 4092 | 8 | 8 |
| Coelosphaerium sp. | 14,405 | 8 | 8 |
| CHROOCOCCALES n.i. | 9939 | 31 | 31 |
| Dactylococcopsis sp. | 288 | 15 | 0 |
| Dolichospermum sp. | 14,117 | 23 | 23 |
| Dolichospermum aphanizomenoides | 81,648 | 23 | 23 |
| Dolichospermum spiroides | 2017 | 8 | 8 |
| Gomphosphaeria sp. | 11,524 | 23 | 23 |
| Cylindrospermopsis raciborskii | 124,069 | 15 | 15 |
| Microcystis aeruginosa | 169,156 | 69 | 54 |
| Microcystis incerta | 74,741 | 15 | 15 |
| Merismopedia sp. | 2305 | 23 | 8 |
| Oscillatoria sp. | 451,261 | 77 | 69 |
| Trichodesmium sp. | 2017 | 8 | 8 |
| OSCILLATORIALES n.i. | 60,616 | 15 | 8 |
| Reservoir A7 | |||
| Anabaenopsis sp. | 32 | 3 | 0 |
| Aphanizomenon sp. | 603 | 6 | 0 |
| Aphanizomenon flos-aquae | 50,153 | 38 | 16 |
| Aphanizomenon flos-aquae var. klebahnii | 10,704,801 | 3 | 3 |
| Aphanizomenon yezoense | 251,865 | 6 | 6 |
| Aphanizomenon gracile | 77,835 | 6 | 6 |
| Aphanizomenon issatschenkoi | 47,702 | 6 | 6 |
| Aphanothece sp. | 1596 | 9 | 0 |
| Chroococcus sp. | 613 | 3 | 0 |
| CHROOCOCCALES n.i. | 5585 | 47 | 6 |
| Dactylococcopsis sp. | 41 | 9 | 0 |
| Dolichospermum sp. | 112,615 | 16 | 6 |
| Dolichospermum lemmermannii | 85,802 | 3 | 3 |
| Dolichospermum mendotae | 1618 | 3 | 0 |
| Dolichospermum solitaria | 378 | 6 | 0 |
| Dolichospermum spiroides | 21,450 | 3 | 3 |
| Gomphosphaeria sp. | 24,686 | 25 | 13 |
| Microcystis sp. | 12,492 | 13 | 9 |
| Microcystis aeruginosa | 116,828 | 50 | 19 |
| Microcystis ichthyoblabe | 1,578,141 | 9 | 9 |
| Merismopedia sp. | 2043 | 22 | 3 |
| NOSTOCALES n.i. | 377 | 3 | 0 |
| Planktothrix sp. | 162,104 | 3 | 3 |
| Oscillatoria sp. | 1239 | 9 | 0 |
| Pseudanabaena sp. | 6742 | 9 | 3 |
| Raphidiopsis mediterranea | 16,445 | 3 | 3 |
| Spirulina sp. | 34 | 3 | 0 |
| OSCILLATORIALES n.i. | 807 | 13 | 0 |
| Woronichinia naegeliana | 3,233,912 | 9 | 9 |
| Reservoir A8 | |||
| Anabaenopsis sp. | 39,632 | 2 | 2 |
| Aphanizomenon sp. | 1,297,190 | 11 | 9 |
| Aphanizomenon flos-aquae | 2,027,579 | 42 | 38 |
| Aphanizomenon flos-aquae var. klebahnii | 3,282,942 | 2 | 2 |
| Aphanizomenon gracile | 129,888 | 15 | 15 |
| Aphanizomenon issatschenkoi | 959,142 | 26 | 25 |
| Aphanizomenon aphanizomenoides | 203,146 | 2 | 2 |
| Aphanothece sp. | 11,747 | 6 | 6 |
| CHROOCOCCALES n.i. | 98,876 | 25 | 11 |
| Dactylococcopsis sp. | 6946 | 2 | 2 |
| Dolichospermum sp. | 2,166,292 | 43 | 34 |
| Dolichospermum aphanizomenoides | 413,483 | 4 | 2 |
| Dolichospermum circinalis | 9193 | 8 | 8 |
| Dolichospermum flos-aquae | 652,482 | 4 | 4 |
| Dolichospermum solitaria | 225 | 2 | 0 |
| Dolichospermum spiroides | 22,483 | 8 | 8 |
| Microcystis incerta | 53,236 | 6 | 6 |
| Gomphosphaeria sp. | 2,200,096 | 28 | 25 |
| Merismopedia sp. | 4787 | 6 | 2 |
| Planktothrix sp. | 233,606 | 13 | 13 |
| Planktothrix agardhii | 612,870 | 2 | 2 |
| Planktothrix clathrata | 194,076 | 2 | 2 |
| Planktothrix isothrix | 426,966 | 4 | 4 |
| Oscillatoria sp. | 985,618 | 25 | 23 |
| Pseudanabaena sp. | 1788 | 4 | 0 |
| Pseudanabaena mucicola | 140,960 | 4 | 2 |
| Phormidium sp. | 94,995 | 2 | 2 |
| Raphidiopsis sp. | 2247 | 2 | 2 |
| Raphidiopsis mediterranea | 26,558 | 2 | 2 |
| OSCILLATORIALES n.i. | 130,950 | 11 | 6 |
| Woronichinia naegeliana | 1,395,301 | 8 | 8 |
| Reservoirs | MC Range Detected (µg/L) | Minimum Water Ingestion to Reach TDI (mL) | |
|---|---|---|---|
| Children | Adults | ||
| A1 | - | - | - |
| A2 | - | - | - |
| A3 | - | - | - |
| A4 | 0.16–2 | 201 | 1206 |
| A5 | 0.37–3.7 | 108 | 647 |
| A6 | 0.1–4.1 | 98 | 585 |
| A7 | 0.16–506 | 1 | 5 |
| A8 | 0.2–389 | 1 | 6 |
| Reservoirs | Total Capacity (dam3) | Surface Area (ha) | Mean Depth (m) | Other Uses | Trophic State | Years of Monitoring | Number of Samples |
|---|---|---|---|---|---|---|---|
| A1 | 72,000 | 2023 | 35.6 | Energy production | Mesotrophic | 14 | 109 |
| A2 | 1,095,000 | 3291 | 33.3 | Drinking water supply | Oligotrophic | 10 | 40 |
| Energy production | |||||||
| Flood defense | |||||||
| A3 | 53,700 | 246 | 21.8 | Energy production | Mesotrophic | 9 | 56 |
| A4 | 660 | 12 | 6.4 | Drinking water supply | Mesotrophic | 14 | 112 |
| Flood defense | |||||||
| A5 | ND | ND | ND | ND | ND | 3 | 21 |
| A6 | ND | ND | ND | ND | ND | 2 | 13 |
| A7 | ND | ND | ND | ND | ND | 8 | 32 |
| A8 | 3032 | 124 | 3.8 | Irrigation | Eutrophic | 11 | 53 |
| Risk Level | Criteria Based on Cyanobacterial Density (Cells/mL) | Criteria Based on Microcystin Concentration (µg MC/L) |
|---|---|---|
| No Risk | Not detected | Not detected or not analysed * |
| Low Risk | <20,000 | <20 |
| Moderate Risk | 20,000–100,000 | - |
| High Risk | >100,000 | ≥20 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menezes, C.; Churro, C.; Dias, E. Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters. Toxins 2017, 9, 327. https://doi.org/10.3390/toxins9100327
Menezes C, Churro C, Dias E. Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters. Toxins. 2017; 9(10):327. https://doi.org/10.3390/toxins9100327
Chicago/Turabian StyleMenezes, Carina, Catarina Churro, and Elsa Dias. 2017. "Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters" Toxins 9, no. 10: 327. https://doi.org/10.3390/toxins9100327
APA StyleMenezes, C., Churro, C., & Dias, E. (2017). Risk Levels of Toxic Cyanobacteria in Portuguese Recreational Freshwaters. Toxins, 9(10), 327. https://doi.org/10.3390/toxins9100327








