Abrin Toxicity and Bioavailability after Temperature and pH Treatment
Abstract
:1. Introduction
2. Results
2.1. Abrin Toxin is Heterogeneous
2.2. Cell Free Translation Assay Measurements of Temperature Effects on Abrin Stability
2.3. Cytotoxic Effects of Temperature-Treated Abrin on Vero Cells
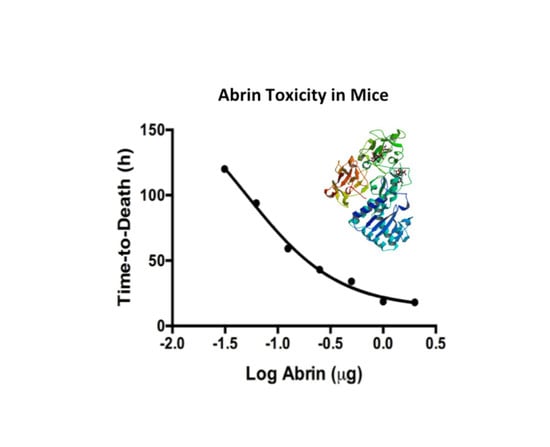
2.4. Mouse Bioassay Determination of Temperature Effects on Abrin Stability and Toxicity
2.5. pH Treatment of Abrin Has No Detrimental Effect on Its Function
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Temperature Stability Assays
4.3. pH Stability Assays
4.4. SDS-PAGE Electrophoresis for Silver Stain
4.5. Abrin In Vitro Cell Free Translation Assay
4.6. Vero Cell Cytotoxicity Assay
4.7. Abrin Intravenous Route (iv) Mouse Bioassay
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Felder, E.; Mossbrugger, I.; Lange, M.; Wolfel, R. Simultaneous detection of ricin and abrin DNA by real-time pcr (qpcr). Toxins 2012, 4, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Zhou, B.; Ma, H.; Carney, C.; Janda, K.D. Selection and characterization of human monoclonal antibodies against abrin by phage display. Bioorg. Med. Chem. Lett. 2007, 17, 5690–5692. [Google Scholar] [CrossRef] [PubMed]
- Reedman, L.; Shih, R.D.; Hung, O. Survival after an intentional ingestion of crushed abrus seeds. West. J. Emerg. Med. 2008, 9, 157–159. [Google Scholar] [PubMed]
- Cheng, J.; Lu, T.H.; Liu, C.L.; Lin, J.Y. A biophysical elucidation for less toxicity of agglutinin than abrin-A from the seeds of abrus precatorius in consequence of crystal structure. J. Biomed. Sci. 2010, 17, 34. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Surolia, A.; Karande, A.A. Ribosome-inactivating protein and apoptosis: Abrin causes cell death via mitochondrial pathway in jurkat cells. Biochem. J. 2004, 377, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, S.; Surendranath, K.; Bora, N.; Surolia, A.; Karande, A.A. Ribosome inactivating proteins and apoptosis. FEBS Lett. 2005, 579, 1324–1331. [Google Scholar] [CrossRef] [PubMed]
- Hegde, R.; Maiti, T.K.; Podder, S.K. Purification and characterization of three toxins and two agglutinins from abrus precatorius seed by using lactamyl-sepharose affinity chromatography. Anal. Biochem. 1991, 194, 101–109. [Google Scholar] [CrossRef]
- Qing, L.T.; Qu, X.L. Cloning, expression of the abrin-a a-chain in escherichia coli and measurement of the biological activities in vitro. Sheng Wu Hua Xue Yu Sheng Wu Wu Li Xue Bao 2002, 34, 405–410. [Google Scholar] [PubMed]
- Lin, J.Y.; Lee, T.C.; Hu, S.T.; Tung, T.C. Isolation of four isotoxic proteins and one agglutinin from jequiriti bean (abrus precatorius). Toxicon 1981, 19, 41–51. [Google Scholar] [PubMed]
- Olsnes, S.; Saltvedt, E.; Pihl, A. Isolation and comparison of galactose-binding lectins from abrus precatorius and ricinus communis. J. Biol. Chem. 1974, 249, 803–810. [Google Scholar] [PubMed]
- Wei, C.H.; Hartman, F.C.; Pfuderer, P.; Yang, W.K. Purification and characterization of two major toxic proteins from seeds of abrus precatorius. J. Biol. Chem. 1974, 249, 3061–3067. [Google Scholar] [PubMed]
- Roy, J.; Som, S.; Sen, A. Isolation, purification, and some properties of a lectin and abrin from abrus precatorius linn. Arch. Biochem. Biophys. 1976, 174, 359–361. [Google Scholar] [CrossRef]
- Lin, J.Y.; Lee, T.C.; Tung, T.C. Isolation of antitumor proteins abrin-A and abrin-B from abrus precatorius. Int. J. Pept. Protein Res. 1978, 12, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, M.S.; Behnke, W.D. A characterization of abrin A from the seeds of the abrus precatorius plant. Biochim. Biophys. Acta 1981, 667, 397–410. [Google Scholar] [CrossRef]
- Garber, E.A. Toxicity and detection of ricin and abrin in beverages. J. Food Prot. 2008, 71, 1875–1883. [Google Scholar] [CrossRef] [PubMed]
- Stirpe, F.; Barbieri, L.; Battelli, M.G.; Soria, M.; Lappi, D.A. Ribosome-inactivating proteins from plants: Present status and future prospects. Biotechnology 1992, 10, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.L.; Tsai, C.C.; Lin, S.C.; Wang, L.I.; Hsu, C.I.; Hwang, M.J.; Lin, J.Y. Primary structure and function analysis of the abrus precatorius agglutinin a chain by site-directed mutagenesis. Pro(199) of amphiphilic alpha-helix H impairs protein synthesis inhibitory activity. J. Biol. Chem. 2000, 275, 1897–1901. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.C.; Zhou, Y.; Jain, R.; Lemire, S.W.; Fox, S.; Sabourin, P.; Barr, J.R. Quantification of l-abrine in human and rat urine: A biomarker for the toxin abrin. J. Anal. Toxicol. 2009, 33, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Gill, D.M. Bacterial toxins: A table of lethal amounts. Microbiol. Rev. 1982, 46, 86–94. [Google Scholar] [PubMed]
- Krupakar, J.; Swaminathan, C.P.; Das, P.K.; Surolia, A.; Podder, S.K. Calorimetric studies on the stability of the ribosome-inactivating protein abrin ii: Effects of pH and ligand binding. Biochem. J. 1999, 338 Pt 2, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Tolleson, W.H.; Jackson, L.S.; Triplett, O.A.; Aluri, B.; Cappozzo, J.; Banaszewski, K.; Chang, C.W.; Nguyen, K.T. Chemical inactivation of protein toxins on food contact surfaces. J. Agric. Food Chem. 2012, 60, 6627–6640. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.S.; Triplett, O.A.; Tolleson, W.H. Influence of yogurt fermentation and refrigerated storage on the stability of protein toxin contaminants. Food Chem. Toxicol. 2015, 80, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Santanche, S.; Bellelli, A.; Brunori, M. The unusual stability of saporin, a candidate for the synthesis of immunotoxins. Biochem. Biophys. Res. Commun. 1997, 234, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Manosroi, J.; von Kleist, S.; Manosroi, A.; Grunert, F. Thermo-stability and antitumor activity on colon cancer cell lines of monoclonal anti-cea antibody-saporin immunotoxin. J. Korean Med. Sci. 1992, 7, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Kayastha, A.M. Thermal stability of phaseolus vulgaris leucoagglutinin: A differential scanning calorimetry study. J. Biochem. Mol. Biol. 2002, 35, 472–475. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Lu, S.; Cheng, L.W.; Rasooly, R.; Carter, J.M. Effect of food matrices on the biological activity of ricin. J. Food Prot. 2008, 71, 2053–2058. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Quinones, B.; Carter, J.M.; Mandrell, R.E. Validation of a cell-free translation assay for detecting shiga toxin 2 in bacterial culture. J. Agric. Food Chem. 2009, 57, 5084–5088. [Google Scholar] [CrossRef] [PubMed]
- Reyes-De-Corcuera, J.I.; Goodrich-Schneider, R.M.; Barringer, S.A.; Landeros-Urbina, M.A. Processing of fruit and vegetable beverages. In Food Processing: Principles and Applications, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar]
- Peng, J.; Tang, J.; Barrett, D.M.; Sablani, S.S.; Anderson, N.; Powers, J.R. Thermal pasteurization of vegetables: Critical factors for process design and effects on quality. Crit. Rev. Food Sci. Nutr. 2015, 57, 2970–2995. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food & Drug Adminstration. Available online: https://www.fda.gov/Food/GuidanceRegulation/default.htm (accessed on 6 October 2017).
- Sandvig, K.; Olsnes, S. Entry of the toxic proteins abrin, modeccin, ricin, and diphtheria toxin into cells. II. Effect of pH, metabolic inhibitors, and ionophores and evidence for toxin penetration from endocytotic vesicles. J. Biol. Chem. 1982, 257, 7504–7513. [Google Scholar] [PubMed]
- Pauly, D.; Worbs, S.; Kirchner, S.; Shatohina, O.; Dorner, M.B.; Dorner, B.G. Real-time cytotoxicity assay for rapid and sensitive detection of ricin from complex matrices. PLoS ONE 2012, 7, e35360. [Google Scholar] [CrossRef] [PubMed]
- Weil, C.S.; Smyth, H.F., Jr.; Nale, T.W. Quest for a suspected industrial carcinogen. AMA Arch. Ind. Hyg. Occup. Med. 1952, 5, 535–547. [Google Scholar] [PubMed]
- Reed, L.J.; Muench, H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938, 27, 493–497. [Google Scholar]
- Fodstad, O.; Johannessen, J.V.; Schjerven, L.; Pihl, A. Toxicity of abrin and ricin in mice and dogs. J. Toxicol. Environ. Health 1979, 5, 1073–1084. [Google Scholar] [CrossRef] [PubMed]
- Goldman, E.R.; Anderson, G.P.; Zabetakis, D.; Walper, S.; Liu, J.L.; Bernstein, R.; Calm, A.; Carney, J.P.; O’Brien, T.W.; Walker, J.L.; et al. Llama-derived single domain antibodies specific for abrus agglutinin. Toxins 2011, 3, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Niyogi, S.K.; Rieders, F. Toxicity studies with fractions from abrus precatorius seed kernels. Toxicon 1969, 7, 211–216. [Google Scholar] [CrossRef]
- Herrera, C.; Tremblay, J.M.; Shoemaker, C.B.; Mantis, N.J. Mechanisms of ricin toxin neutralization revealed through engineered homodimeric and heterodimeric camelid antibodies. J. Biol. Chem. 2015, 290, 27880–27889. [Google Scholar] [CrossRef] [PubMed]


| Treatment | Relative Cytotoxicity (%) |
|---|---|
| DMEM | 0 |
| Abrin | 100 |
| 99 °C | 0 |
| 85 °C | 0 |
| 80 °C | 0 |
| 74 °C | 0 |
| 63 °C | 72 ± 4 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tam, C.C.; Henderson, T.D.; Stanker, L.H.; He, X.; Cheng, L.W. Abrin Toxicity and Bioavailability after Temperature and pH Treatment. Toxins 2017, 9, 320. https://doi.org/10.3390/toxins9100320
Tam CC, Henderson TD, Stanker LH, He X, Cheng LW. Abrin Toxicity and Bioavailability after Temperature and pH Treatment. Toxins. 2017; 9(10):320. https://doi.org/10.3390/toxins9100320
Chicago/Turabian StyleTam, Christina C., Thomas D. Henderson, Larry H. Stanker, Xiaohua He, and Luisa W. Cheng. 2017. "Abrin Toxicity and Bioavailability after Temperature and pH Treatment" Toxins 9, no. 10: 320. https://doi.org/10.3390/toxins9100320
APA StyleTam, C. C., Henderson, T. D., Stanker, L. H., He, X., & Cheng, L. W. (2017). Abrin Toxicity and Bioavailability after Temperature and pH Treatment. Toxins, 9(10), 320. https://doi.org/10.3390/toxins9100320