Humoral Response of Buffaloes to a Recombinant Vaccine against Botulism Serotypes C and D
Abstract
:1. Introduction
2. Results
3. Discussion and Conclusions
4. Materials and Methods
4.1. Ethics Statement
4.2. Vaccines
4.3. Vaccination of Buffaloes
4.4. Humoral Immune Response Evaluation
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gil, L.A.F.; da Cunha, C.E.P.; Moreira, G.M.S.G.; Salvarani, F.M.; Assis, R.A.; Lobato, F.C.F.; Mendonça, M.; Dellagostin, O.A.; Conceição, F.R. Production and Evaluation of a Recombinant Chimeric Vaccine against Clostridium botulinum Neurotoxin Types C and D. PLoS ONE 2013, 8, e69692. [Google Scholar] [CrossRef] [PubMed]
- Krüger, M.; Skau, M.; Shehata, A.A.; Schrödl, W. Efficacy of Clostridium botulinum types C and D toxoid vaccination in Danish cows. Anaerobe 2013, 23, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Cunha, C.E.P.; Moreira, G.M.S.G.; Salvarani, F.M.; Neves, M.S.; Lobato, F.C.F.; Dellagostin, O.A.; Conceição, F.R. Vaccination of cattle with a recombinant bivalent toxoid against botulism serotypes C and D. Vaccine 2014, 32, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Moreira, C.; da Cunha, C.E.P.; Moreira, G.M.S.G.; Mendonça, M.; Salvarani, F.M.; Moreira, Â.N.; Conceição, F.R. Protective potential of recombinant non-purified botulinum neurotoxin serotypes C and D. Anaerobe 2016, 40, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Salvarani, F.M.; Otaka, D.Y.; Oliveira, C.M.C.; Reis, A.S.B.; Perdigão, H.H.; Souza, A.E.C.; Brito, M.F.; Barbosa, J.D. Type C waterborne botulism outbreaks in buffaloes (Bubablus bubalis) in the Amazon region. Pesqui. Vet. Bras 2017, in press. [Google Scholar]
- Silva, T.M.D.; Dutra, I.S.; Castro, R.N.; Döbereiner, J. Ocorrência e distribuição de esporos de Clostridium botulinum tipos C e D em áreas de criação de búfalos na Baixada Maranhense. Pesqui. Vet. Bras. 1998, 18, 127–131. [Google Scholar] [CrossRef]
- Moreira, G.M.S.G.; Cunha, C.E.P.; Salvarani, F.M.; Gonçalves, L.A.; Pires, P.S.; Conceição, F.R.; Lobato, F.C.F. Production of recombinant botulism antigens: A review of expression systems. Anaerobe 2014, 28, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Langenegger, J.; Döbereiner, J. Botulismo Enzoótico em búfalos no Maranhão. Pesqui. Vet. Bras. 1988, 8, 37–42. [Google Scholar]
- Brasil. Ministério da Agricultura Pecuária e Abastecimento. In Instrução Normativa n° 23; Diário Ofical da União: Brasília, Brazil, 2002. [Google Scholar]
- FAO Food and Agriculture Organization of the United Nations. Available online: http://faostat.fao.org/ (accessed on 8 August 2017).
- Instituto Brasileiro de Geografia e Estatística. IBGE Produção da Pecuária Municipal 2014. Minist. Planej. Orçamento Gestão 2014, 42, 1–39. [Google Scholar]
- Borghese, A.; Mazzi, M. Buffalo Production and Research; Borghese, A., Ed.; Food and Agriculture Organization: Rome, Italy, 2005; Volume 67, ISBN 3383172740. [Google Scholar]
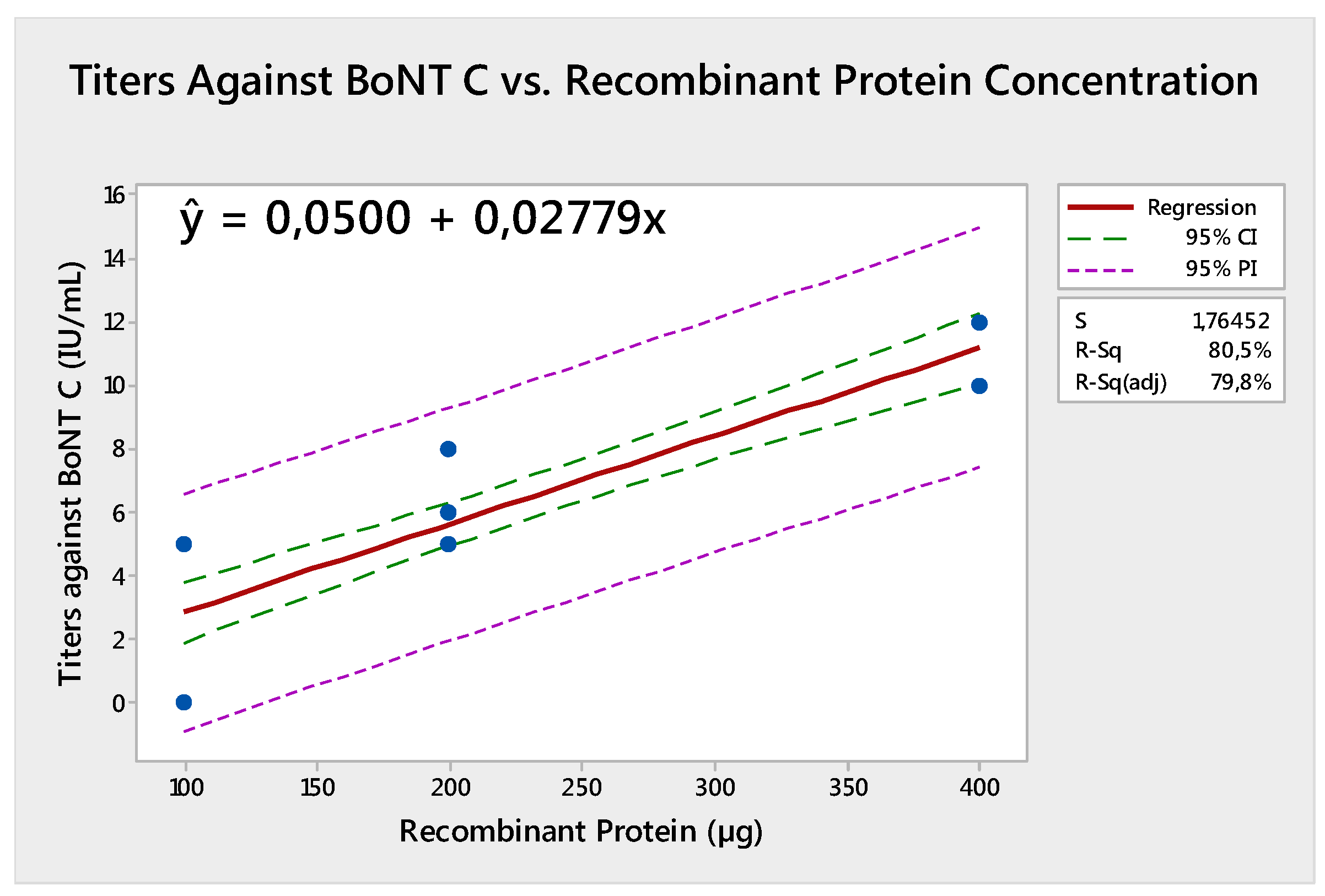
| Vaccine | Vrec100 | Vrec200 | Vrec400 | Vcom | ||||
|---|---|---|---|---|---|---|---|---|
| Repetition | C | D | C | D | C | D | C | D |
| 1 | 0 | 0 | 5 | 4 | 10 | 15 | 5 | 3 |
| 2 | 5 | 2 | 5 | 2 | 10 | 15 | 5 | 2 |
| 3 | 5 | 2 | 8 | 8 | 12 | 15 | 5 | 0 |
| 4 | 0 | 0 | 6 | 8 | 12 | 15 | 6 | 5 |
| 5 | 0 | 0 | 6 | 2 | 12 | 15 | 5 | 0 |
| 6 | 5 | 4 | 6 | 4 | 12 | 15 | 6 | 4 |
| 7 | 0 | 2 | 5 | 10 | 10 | 10 | 5 | 2 |
| 8 | 0 | 2 | 6 | 8 | 10 | 10 | 0 | 5 |
| 9 | 5 | 2 | 8 | 8 | 10 | 12 | 5 | 2 |
| 10 | 5 | 2 | 6 | 8 | 12 | 15 | 5 | 2 |
| Mean titers | 2.5 | 1.6 | 6.1 | 6.2 | 11.0 | 13.7 | 4.7 | 2.5 |
| Seroconversion rate * | 50% | 70% | 100% | 100% | 100% | 100% | 90% | 80% |
| Vaccine | BoNT Mean Titer | |
|---|---|---|
| C | D | |
| Vrec400 | 11.0 a | 13.7 A |
| Vrec200 | 6.1 b | 6.2 B |
| Vcom | 4.7 b,c | 2.5 B,C |
| Vrec100 | 2.5 c | 1.6 C |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otaka, D.Y.; Barbosa, J.D.; Moreira, C., Jr.; Ferreira, M.R.A.; Cunha, C.E.P.; Brito, A.R.S.; Donassolo, R.A.; Moreira, Â.N.; Conceição, F.R.; Salvarani, F.M. Humoral Response of Buffaloes to a Recombinant Vaccine against Botulism Serotypes C and D. Toxins 2017, 9, 297. https://doi.org/10.3390/toxins9100297
Otaka DY, Barbosa JD, Moreira C Jr., Ferreira MRA, Cunha CEP, Brito ARS, Donassolo RA, Moreira ÂN, Conceição FR, Salvarani FM. Humoral Response of Buffaloes to a Recombinant Vaccine against Botulism Serotypes C and D. Toxins. 2017; 9(10):297. https://doi.org/10.3390/toxins9100297
Chicago/Turabian StyleOtaka, Denis Y., José D. Barbosa, Clóvis Moreira, Jr., Marcos R. A. Ferreira, Carlos E. P. Cunha, Antônio R. S. Brito, Rafael A. Donassolo, Ângela N. Moreira, Fabrício R. Conceição, and Felipe M. Salvarani. 2017. "Humoral Response of Buffaloes to a Recombinant Vaccine against Botulism Serotypes C and D" Toxins 9, no. 10: 297. https://doi.org/10.3390/toxins9100297
APA StyleOtaka, D. Y., Barbosa, J. D., Moreira, C., Jr., Ferreira, M. R. A., Cunha, C. E. P., Brito, A. R. S., Donassolo, R. A., Moreira, Â. N., Conceição, F. R., & Salvarani, F. M. (2017). Humoral Response of Buffaloes to a Recombinant Vaccine against Botulism Serotypes C and D. Toxins, 9(10), 297. https://doi.org/10.3390/toxins9100297







