The Spider Venom Peptide Lycosin-II Has Potent Antimicrobial Activity against Clinically Isolated Bacteria
Abstract
:1. Introduction
2. Results and Discussion
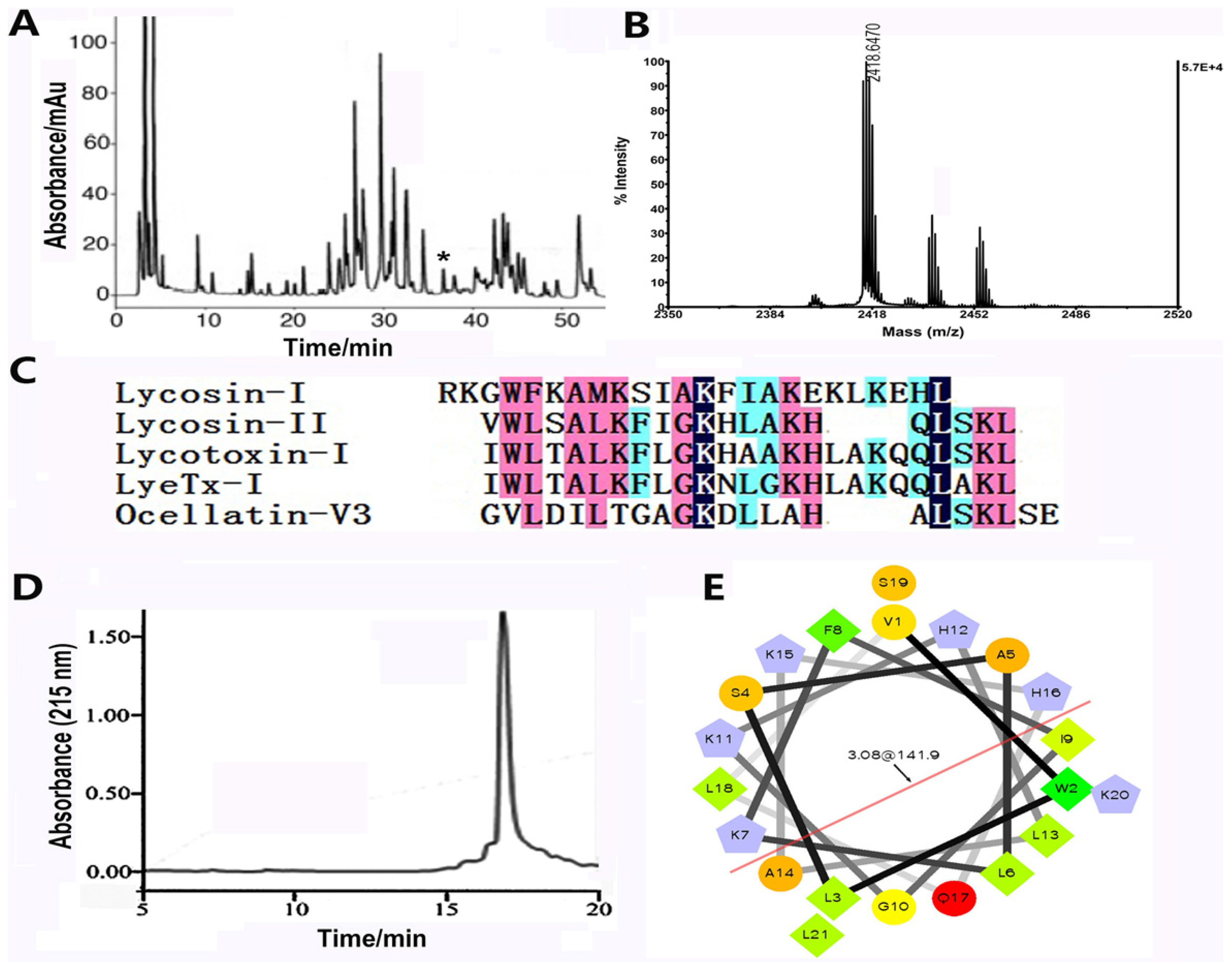
2.1. The Biochemical Properties of Lycosin-II
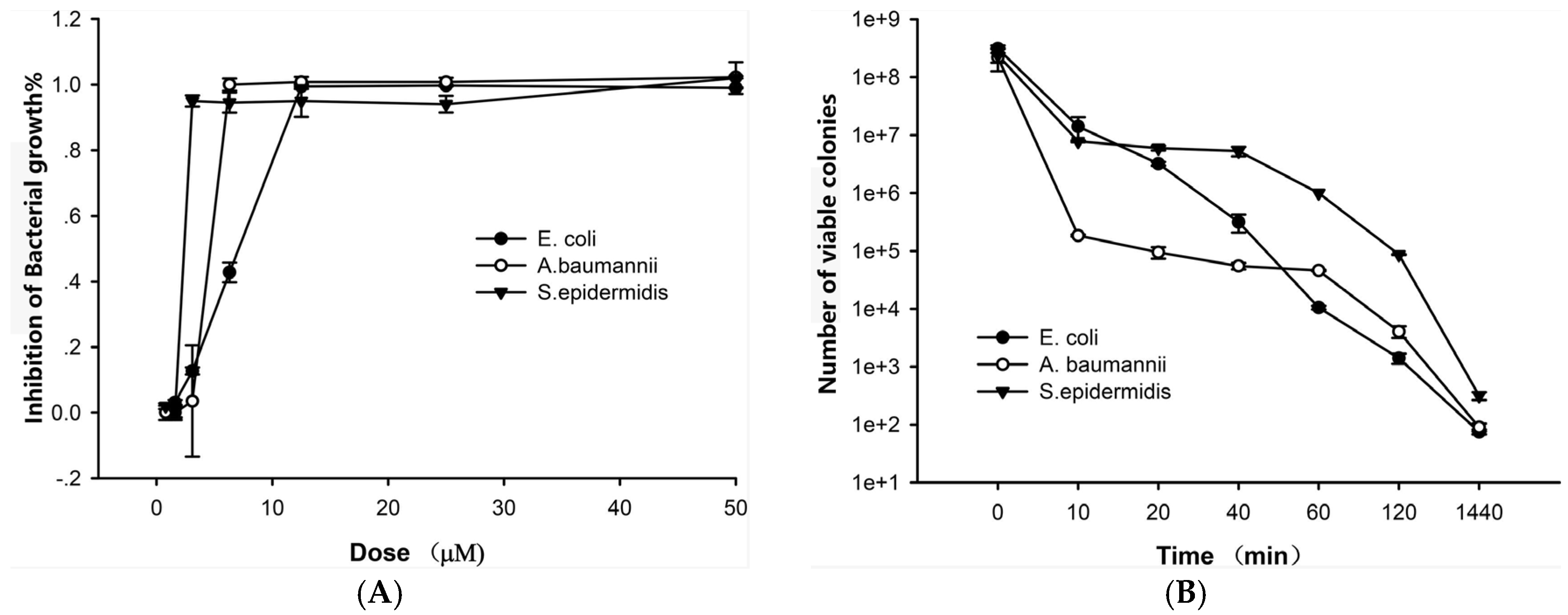
2.2. The Antibacterial Effects of Lycosin-II
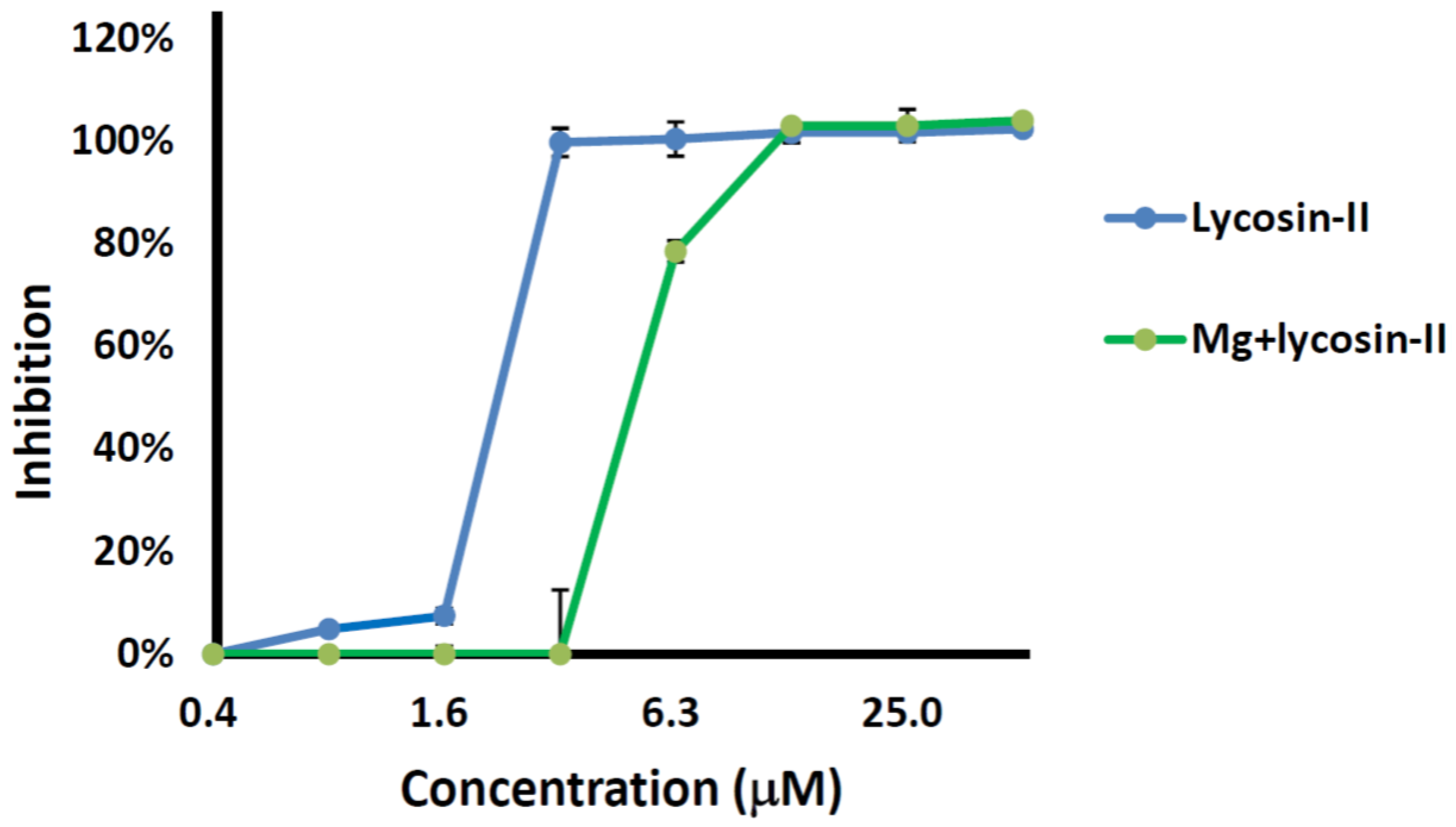
2.3. Competition between Lycosin-II and Mg2+
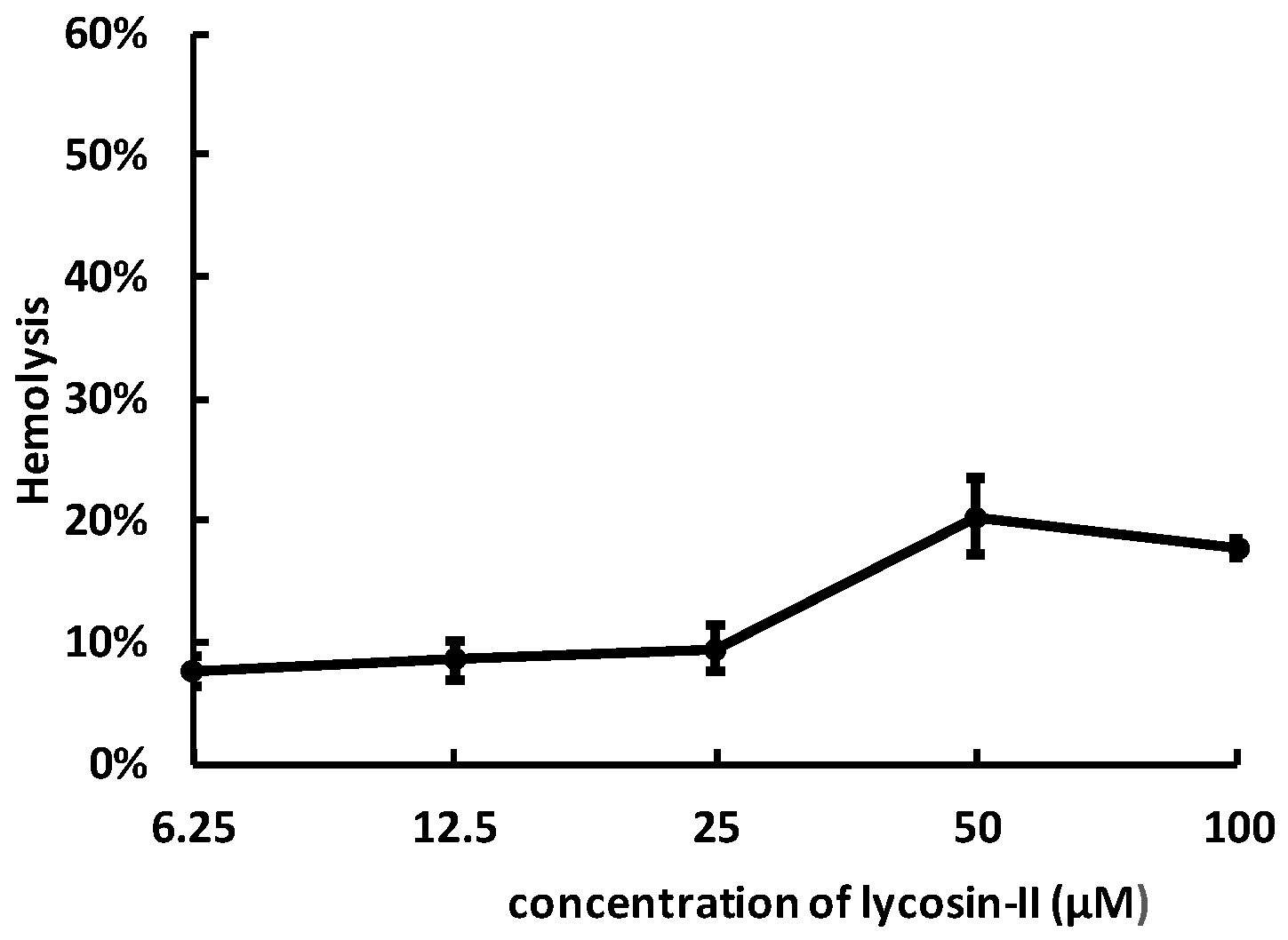
2.4. Lycosin-II Has Low Hemolytic Activity
3. Conclusions
4. Materials and Methods
4.1. Purification, Characterization, and Synthesis of Lycosin-II
4.2. Bacterial Strains
4.3. Minimum Inhibitory Concentration Assay
4.4. Time-Kill Kinetics of Lycosin-II
4.5. Salt Tolerance
4.6. Hemolysis Assay
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brogden, K.A. Antimicrobial peptides: Pore formers or metabolic inhibitors in bacteria? Nat. Rev. Microbiol. 2005, 3, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Narayana, J.L.; Chen, J.Y. Antimicrobial peptides: Possible anti-infective agents. Peptides 2015, 72, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Li, X.; Wang, Z. Apd3: The antimicrobial peptide database as a tool for research and education. Nucl. Acids Res. 2016, 44, D1087–D1093. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Mishra, B.; Lau, K.; Lushnikova, T.; Golla, R.; Wang, X. Antimicrobial peptides in 2014. Pharmaceuticals 2015, 8, 123–150. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Hall, K.N.; Aguilar, M.I. Antimicrobial peptide structure and mechanism of action: A focus on the role of membrane structure. Curr. Top. Med. Chem. 2016, 16, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.L.; Hancock, R.E. Cationic host defense (antimicrobial) peptides. Curr. Opin. Immunol. 2006, 18, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Hilchie, A.L.; Wuerth, K.; Hancock, R.E. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat. Chem. Biol. 2013, 9, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Sato, H.; Feix, J.B. Peptide-membrane interactions and mechanisms of membrane destruction by amphipathic alpha-helical antimicrobial peptides. Biochim. Biophys. Acta 2006, 1758, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Shai, Y. Mode of action of membrane active antimicrobial peptides. Biopolymers 2002, 66, 236–248. [Google Scholar] [CrossRef] [PubMed]
- King, G.F.; Hardy, M.C. Spider-venom peptides: Structure, pharmacology, and potential for control of insect pests. Annu. Rev. Entomol. 2013, 58, 475–496. [Google Scholar] [CrossRef] [PubMed]
- Pineda, S.S.; Undheim, E.A.; Rupasinghe, D.B.; Ikonomopoulou, M.P.; King, G.F. Spider venomics: Implications for drug discovery. Future Med. Chem. 2014, 6, 1699–1714. [Google Scholar] [CrossRef] [PubMed]
- Saez, N.J.; Senff, S.; Jensen, J.E.; Er, S.Y.; Herzig, V.; Rash, L.D.; King, G.F. Spider-venom peptides as therapeutics. Toxins 2010, 2, 2851–2871. [Google Scholar] [CrossRef] [PubMed]
- Dubovskii, P.V.; Vassilevski, A.A.; Kozlov, S.A.; Feofanov, A.V.; Grishin, E.V.; Efremov, R.G. Latarcins: Versatile spider venom peptides. Cell. Mol. Life Sci. 2015, 72, 4501–4522. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.M.; Verly, R.M.; Pilo-Veloso, D.; Maria, M.; de Carvalho, M.A.; de Cisalpino, P.S.; Soares, B.M.; Diniz, C.G.; Farias, L.M.; Moreira, D.F.; et al. LyeTx I, a potent antimicrobial peptide from the venom of the spider lycosa erythrognatha. Amino Acids 2010, 39, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Polyansky, A.A.; Vassilevski, A.A.; Volynsky, P.E.; Vorontsova, O.V.; Samsonova, O.V.; Egorova, N.S.; Krylov, N.A.; Feofanov, A.V.; Arseniev, A.S.; Grishin, E.V.; et al. N-terminal amphipathic helix as a trigger of hemolytic activity in antimicrobial peptides: A case study in latarcins. FEBS Lett. 2009, 583, 2425–2428. [Google Scholar] [CrossRef] [PubMed]
- Kuhn-Nentwig, L.; Sheynis, T.; Kolusheva, S.; Nentwig, W.; Jelinek, R. N-terminal aromatic residues closely impact the cytolytic activity of cupiennin 1a, a major spider venom peptide. Toxicon 2013, 75, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Pukala, T.L.; Doyle, J.R.; Llewellyn, L.E.; Kuhn-Nentwig, L.; Apponyi, M.A.; Separovic, F.; Bowie, J.H. Cupiennin 1a, an antimicrobial peptide from the venom of the neotropical wandering spider cupiennius salei, also inhibits the formation of nitric oxide by neuronal nitric oxide synthase. FEBS J. 2007, 274, 1778–1784. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Corzo, G. The effect of binding of spider-derived antimicrobial peptides, oxyopinins, on lipid membranes. Biochim. Biophys. Acta 2006, 1758, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, S.A.; Vassilevski, A.A.; Feofanov, A.V.; Surovoy, A.Y.; Karpunin, D.V.; Grishin, E.V. Latarcins, antimicrobial and cytolytic peptides from the venom of the spider Lachesana tarabaevi (zodariidae) that exemplify biomolecular diversity. J. Biol. Chem. 2006, 281, 20983–20992. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Adams, M.E. Lycotoxins, antimicrobial peptides from venom of the wolf spider lycosa carolinensis. J. Biol. Chem. 1998, 273, 2059–2066. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Deng, M.; Xiang, J.; Ma, H.; Hu, W.; Zhao, Y.; Li, D.W.; Liang, S. A novel spider peptide toxin suppresses tumor growth through dual signaling pathways. Curr. Mol. Med. 2012, 12, 1350–1360. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Ding, X.; Meng, S.; Liu, C.; Wang, H.; Xia, L.; Liu, Z.; Liang, S. Antimicrobial potential of lycosin-I, a cationic and amphiphilic peptide from the venom of the spider Lycosa singoriensis. Curr. Mol. Med. 2013, 13, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Y.J.; Liu, Y.Y.; Li, H.; Guo, L.X.; Liu, Z.H.; Shi, X.L.; Hu, M. In vitro potential of lycosin-I as an alternative antimicrobial drug for treatment of multidrug-resistant Acinetobacter baumannii infections. Antimicrob. Agents Chemother. 2014, 58, 6999–7002. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.H.; Qian, W.; Li, J.; Zhang, Y.; Liang, S. Biochemical and pharmacological study of venom of the wolf spider Lycosa singoriensis. J. Venom. Anim Toxins 2009, 15, 79–92. [Google Scholar] [CrossRef]
- King, J.D.; Leprince, J.; Vaudry, H.; Coquet, L.; Jouenne, T.; Conlon, J.M. Purification and characterization of antimicrobial peptides from the caribbean frog, leptodactylus validus (anura: Leptodactylidae). Peptides 2008, 29, 1287–1292. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.; Sahl, H.G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 2006, 24, 1551–1557. [Google Scholar] [CrossRef] [PubMed]
- Spellberg, B.; Bonomo, R.A. “Airborne assault”: A new dimension in Acinetobacter baumannii transmission. Crit. Care Med. 2013, 41, 2042–2044. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Chen, C.L.; Wu, S.R.; Huang, C.W.; Chiu, C.H. Risk factors and outcome analysis of Acinetobacter baumannii complex bacteremia in critical patients. Crit. Care Med. 2014, 42, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Hagihara, M.; Housman, S.T.; Nicolau, D.P.; Kuti, J.L. In vitro pharmacodynamics of polymyxin B and tigecycline alone and in combination against carbapenem-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2014, 58, 874–879. [Google Scholar] [CrossRef] [PubMed]
| Bacterial strains | MIC (μM) |
|---|---|
| Escherichia coli | 12.5 |
| Klebsiella pneumoniae | 50 |
| Streptococcus pyogenes | 50 |
| Staphylococcus saprophyticus | 3.1 |
| Staphylococcus epidermidis | 3.1 |
| Pseudomonas aeruginosa | 12.5 |
| Viridans Streptococci | 3.1 |
| Staphylococcus aureus (three strains) | 3.1 |
| Acinetobacter baumannii (three strains) | 3.1–6.3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Wang, L.; Yang, H.; Xiao, H.; Farooq, A.; Liu, Z.; Hu, M.; Shi, X. The Spider Venom Peptide Lycosin-II Has Potent Antimicrobial Activity against Clinically Isolated Bacteria. Toxins 2016, 8, 119. https://doi.org/10.3390/toxins8050119
Wang Y, Wang L, Yang H, Xiao H, Farooq A, Liu Z, Hu M, Shi X. The Spider Venom Peptide Lycosin-II Has Potent Antimicrobial Activity against Clinically Isolated Bacteria. Toxins. 2016; 8(5):119. https://doi.org/10.3390/toxins8050119
Chicago/Turabian StyleWang, Yongjun, Ling Wang, Huali Yang, Haoliang Xiao, Athar Farooq, Zhonghua Liu, Min Hu, and Xiaoliu Shi. 2016. "The Spider Venom Peptide Lycosin-II Has Potent Antimicrobial Activity against Clinically Isolated Bacteria" Toxins 8, no. 5: 119. https://doi.org/10.3390/toxins8050119
APA StyleWang, Y., Wang, L., Yang, H., Xiao, H., Farooq, A., Liu, Z., Hu, M., & Shi, X. (2016). The Spider Venom Peptide Lycosin-II Has Potent Antimicrobial Activity against Clinically Isolated Bacteria. Toxins, 8(5), 119. https://doi.org/10.3390/toxins8050119






