Peptide Toxins in Solitary Wasp Venoms
Abstract
:1. Introduction
2. Pompilidotoxins
3. Antimicrobial and Cytolytic Peptides
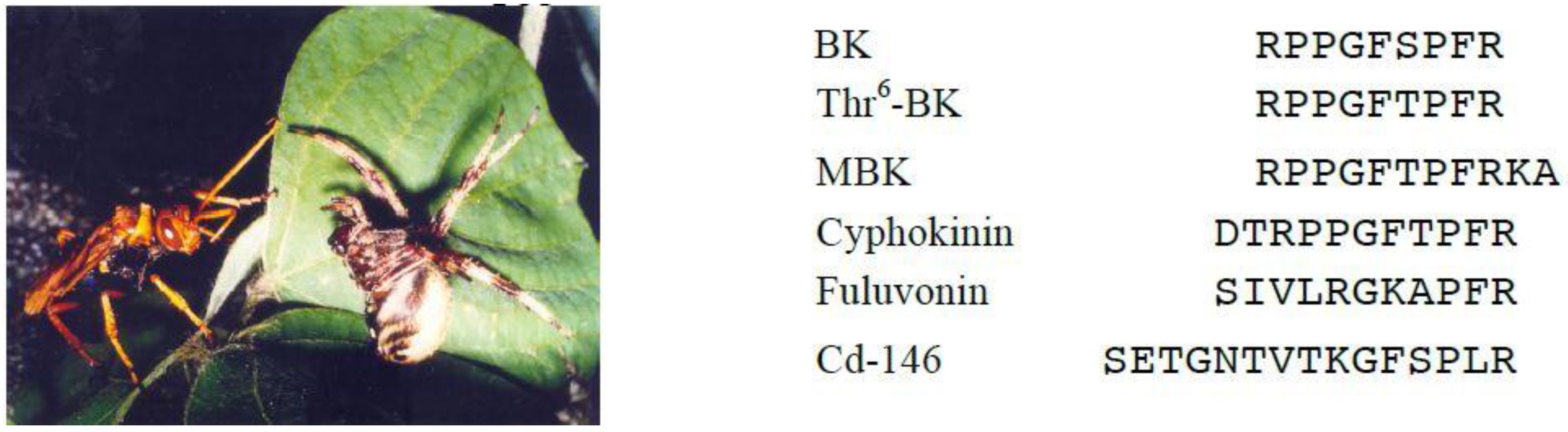
4. Bradykinin-Related Peptides
5. FMRFamide-Related Neuropeptides
6. Other Peptide Toxins
7. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Banks, B.E.C.; Shipolini, R.A. Chemistry and pharmacology of honey-bee venom. In Venoms of the Hymenoptera: Biochemical, Pharmacological and Behavioural Aspects; Piek, T., Ed.; Academic Press: London, UK, 1986; pp. 330–416. [Google Scholar]
- Nakajima, T. Pharmacological biochemistry of vespid venoms. In Venoms of the Hymenoptera: Biochemical, Pharmacological and Behavioural Aspects; Piek, T., Ed.; Academic Press: London, UK, 1986; pp. 309–327. [Google Scholar]
- Piek, T.; Spanjer, W. Chemistry and pharmacology of solitary wasp venoms. In Venoms of the Hymenoptera: Biochemical, Pharmacological and Behavioural Aspects; Piek, T., Ed.; Academic Press: London, UK, 1986; pp. 161–307. [Google Scholar]
- Yasuhara, T.; Mantel, P.; Nakajima, T.; Piek, T. Two kinins isolated from an extract of the venom reservoirs of the solitary wasp Megascolia flavifrons. Toxicon 1987, 25, 527–535. [Google Scholar] [CrossRef]
- Piek, T.; Hue, S.; Mantel, P.; Nakajima, T.; Pelhate, M.; Yasuhara, T. Threonine6-bradykinin in the venom of the wasp Colpa interrupta (F.) presynaptically blocks nicotinic synaptic transmission in the insect CNS. Comp. Biochem. Physiol. 1990, 96C, 157–162. [Google Scholar] [CrossRef]
- Piek, T.; Hue, B.; Mony, L.; Nakajima, T.; Pelhate, M.; Yasuhara, T. Block of synaptic transmission in insect CNS by toxins from the venom of the wasp Megascolia flavifrons (FAB.). Comp. Biochem. Physiol. 1987, 87C, 287–295. [Google Scholar] [CrossRef]
- Eldefrawi, A.T.; Eldefrawi, M.E.; Konno, K.; Mansour, N.A.; Nakanishi, K.; Oltz, E.; Usherwood, P.N.R. Structure and synthesis of a potent glutamate receptor antagonist in wasp venom. Proc. Natl. Acad. Sci. USA 1988, 85, 4910–4913. [Google Scholar] [CrossRef] [PubMed]
- Piek, T.; Fokkens, R.H.; Karst, H.; Kruk, C.; Lind, A.; van Marle, J.; Nakajima, T.; Nibbering, N.M.M.; Shinozaki, H.; Spanjer, W.; et al. Polyamine like toxins—A new class of pesticides? In Neurotox ′88: Molecular Basis of Drug and Pesticide Action; Lunt, G.G., Ed.; Elsevier: Amsterdam, The Netherlands, 1988; pp. 61–76. [Google Scholar]
- Nakanishi, K.; Goodnow, R.; Konno, K.; Niwa, M.; Bukownik, R.; Kalimopoulos, T.A.; Usherwood, P.N.R.; Eldefrawi, A.T.; Eldefrawi, M.E. Philanthotoxin-433 (PhTX-433), a non-competitive glutamate receptor inhibitor. Pure Appl. Chem. 1990, 62, 1223–1230. [Google Scholar] [CrossRef]
- Piek, T.; Hue, B. Philanthotoxins, a new class of neuroactive polyamines, block nicotinic transmission in the insect CNS. Comp. Biochem. Physiol. 1989, 93c, 403–406. [Google Scholar] [CrossRef]
- Konno, K.; Hisada, M.; Itagaki, Y.; Naoki, H.; Kawai, N.; Miwa, A.; Yasuhara, T.; Takayama, H. Isolation and structure of pompilidotoxins (PMTXs), novel neurotoxins in solitary wasp venoms. Biochem. Biophys. Res. Commun. 1998, 250, 612–616. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Kawai, N. Pompilidotoxins: Novel peptide neurotoxins blocking sodium channel inactivation from solitary wasp venom. Curr. Med. Chem. Central Nerv. Syst. Agents 2004, 4, 139–146. [Google Scholar] [CrossRef]
- Konno, K.; Miwa, A.; Takayama, H.; Hisada, M.; Itagaki, Y.; Naoki, H.; Yasuhara, T.; Kawai, N. α-Pompilidotoxin (α-PMTX), a novel neurotoxin from the venom of a solitary wasp, facilitates transmission in the crustacean neuromuscular synapse. Neurosci. Lett. 1997, 238, 99–102. [Google Scholar] [CrossRef]
- Konno, K.; Hisada, M.; Naoki, H.; Itagaki, Y.; Yasuhara, T.; Nakata, Y.; Miwa, A.; Kawai, N. Molecular determinants of binding of a wasp toxin (PMTXs) and its analogs in the Na+ channels proteins. Neurosci. Lett. 2000, 285, 29–32. [Google Scholar] [CrossRef]
- Harsch, A.; Konno, K.; Takayama, H.; Kawai, N.; Robinson, H. Effects of α-pompilidotoxin on synchronized firing in networks of rat cortical neurons. Neurosci. Lett. 1998, 252, 49–52. [Google Scholar] [CrossRef]
- Miyawaki, T.; Tsubokawa, H.; Yokota, H.; Ogura, K.; Konno, K.; Masuzawa, T.; Kawai, N. Differential effects of novel wasp toxin on rat hippocampal interneurons. Neurosci. Lett. 2002, 328, 25–28. [Google Scholar] [CrossRef]
- Yokota, H.; Tsubokawa, H.; Miyawaki, T.; Konno, K.; Nakayama, H.; Masuzawa, T.; Kawai, N. Modulation of synaptic transmissions in hippocampal CA1 neurons by a novel neurotoxin (β-PMTX) derived from wasp venom. Neurosci. Res. 2001, 41, 365–371. [Google Scholar] [CrossRef]
- Sahara, Y.; Gotoh, M.; Konno, K.; Miwa, A.; Tsubokawa, H.; Robinson, H.P.C.; Kawai, N. A new class of neurotoxin from wasp venom slows inactivation of sodium current. Eur. J. Neurosci. 2000, 12, 1961–1970. [Google Scholar] [CrossRef] [PubMed]
- Kawai, N.; Konno, K. Molecular determinants of two neurotoxins that regulate sodium current inactivation in rat hippocampus. Neurosci. Lett. 2004, 361, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, E.; Maejima, H.; Yamaoka, K.; Konno, K.; Kawai, N.; Shimizu, E.; Yokote, S.; Nakayama, H.; Seyama, I. Novel wasp toxin discriminates between neuronal and cardiac sodium channels. Mol. Pharmacol. 2001, 59, 1457–1463. [Google Scholar]
- Schiavon, E.; Stevens, M.; Zaharenko, A.J.; Konno, K.; Tytgat, J.; Wanke, E. Voltage-gated sodium channels isoform-specific effects of pompilidotoxins. FEBS J. 2010, 277, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, J.; Olivera, B.M.; Bosmans, F. Animal toxins influence voltage-gated sodium channel function. Handb. Exp. Pharmacol. 2014, 221, 203–229. [Google Scholar] [PubMed]
- Oliveira, J.S.; Redaelli, E.R.; Zaharenko, A.; Cassulini, R.R.; Konno, K.; Pimenta, D.C.; de Freitas, J.C.; Clare, J.J.; Wanke, E. Nav 1.1–1.6 sodium channels binding specificity to sea anemone toxins: Unexpected contributions from changes in the IV/S3-S4 outer loop. J. Biol. Chem. 2004, 279, 33323–33335. [Google Scholar] [CrossRef] [PubMed]
- Honma, T.; Shiomi, K. Peptide toxins in sea anemones: Structural and functional aspects. Marine Biotech. 2006, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pucca, M.B.; Cerni, F.A.; Pinheiro, E.L., Jr.; Bordon, K.C.F.; Amorim, F.G.; Cordeiro, F.A.; Longhim, H.T.; Cremonez, C.M.; Oliveira, G.H.; Arantes, E.C. Tityus serrulatus venom—A lethal cocktail. Toxicon 2015, 108, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Bosmans, F.; Swartz, K.J. Targeting voltage sensors in sodium channels with spider toxins. Trends Pharmacol. Sci. 2010, 31, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Grieco, T.M.; Raman, I.M. Production of resurgent current in NaV1.6-null Purkinje neurons by slowing sodium channel inactivation with β-pompilidotoxin. J. Neurosci. 2004, 24, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Webster, R.G.; Brain, K.L.; Wilson, R.H.; Grem, L.L.; Vincent, A. Oxaliplatin induces hyperexcitability at motor and autonomic neuromuscular junctions through effects on voltage-gated sodium channels. Br. J. Pharmacol. 2005, 146, 1027–1039. [Google Scholar] [CrossRef] [PubMed]
- Khaliq, Z.M.; Raman, I.M. Relative contributions of axonal and somatic Na channels to action potential initiation in cerebellar Purkinje neurons. J. Neurosci. 2006, 26, 1935–1944. [Google Scholar] [CrossRef] [PubMed]
- Yokote, S.; Setoguchi, R.; Shimizu, E.; Mishima, N.; Kawahara, K.; Kuniyasu, A.; Shirasaki, T.; Takahama, K.; Konno, K.; Kawai, N.; et al. A synthetic approach to develop peptide inhibitors selective for brain-type sodium channels on the basis of pompilidotoxin structure. Heterocycles 2009, 79, 925–933. [Google Scholar]
- Magloire, V.; Czrnecki, A.; Anwander, H.; Streit, J. β-Pompilidotoxin modulated spontaneous activity and persistent sodium currents in spinal networks. Neuroscience 2011, 172, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Teramoto, N.; Zhu, H.-L.; Yotsu-Yamashita, M.; Inai, T.; Cunnane, T.C. Resurgent-like currents in mouse vas deferens myocytes are mediated by NaV1.6 voltage-gated sodium channels. Pflugers Arch. 2012, 464, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Hisada, M.; Naoki, H.; Itagaki, Y.; Kawai, N.; Miwa, A.; Yasuhara, T.; Morimoto, Y.; Nakata, Y. Structure and biological activities of eumenine mastoparan-AF (EMP-AF), a novel mast cell degranulating peptide in the venom of the solitary wasp (Anterhynchium flavomarginatum micado). Toxicon 2000, 38, 1505–1515. [Google Scholar] [CrossRef]
- Cabrera, M.P.S.; Souza, B.M.; Fontana, R.; Konno, K.; Palma, M.S.; de Azevedo, W.F., Jr.; Ruggiero Neto, J. Conformation and lytic activity of eumenine mastoparan: A new antimicrobial peptide from wasp venom. J. Peptide Res. 2004, 64, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Sforça, M.L.; Oyama, S., Jr.; Canduri, F.; Lorenzi, C.C.B.; Pertinez, T.A.; Konno, K.; Souza, B.M.; Palma, M.S.; Ruggiero Neto, J.; de Azevedo, W.F., Jr.; et al. How C-terminal carboxyamidation alters the mast cell degranulating activity of peptides from the venom of the eumenine solitary wasp. Biochemistry 2004, 43, 5608–5617. [Google Scholar] [CrossRef] [PubMed]
- Murata, K.; Shinada, T.; Ohfune, Y.; Hisada, M.; Yasuda, A.; Naoki, H.; Nakajima, T. Novel mastoparan and protonectin analogs isolated from a solitary wasp, Orancistrocerus drewseni drewseni. Amino Acids 2009, 37, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Lee, S.H. Isolation and molecular cloning of venom peptides from Orancistrocerus drewseni (Hymenoptera: Eumenidae). Toxicon 2010, 55, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Hisada, M.; Naoki, H.; Itagaki, Y.; Fontana, R.; Rangel, M.; Oliveira, J.S.; Cabrera, M.P.; Ruggiero Neto, J.; Hide, I.; et al. Eumenitin, a novel antimicrobial peptide from the venom of the solitary eumenine wasp Eumenes rubronotatus. Peptides 2006, 27, 2624–2631. [Google Scholar] [CrossRef] [PubMed]
- Arcisio-Miranda, M.; Cabrera, M.P.S.; Konno, K.; Rangel, M.; Procopio, J. Effects of the cationic antimicrobial peptide Eumenitin from the venom of solitary wasp Eumenes rubronotatus in planar lipid bilayers: Surface charge and pore formation activity. Toxicon 2008, 51, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Rangel, M.; Cabrera, M.P.S.; Kazuma, K.; Ando, K.; Wang, X.; Kato, M.; Nihei, K.; Hirata, I.Y.; Cross, T.; Garcia, A.N.; et al. Chemical and biological characterization of four new antimicrobial and α-helical peptides from the venoms of two solitary eumenine wasps. Toxicon 2011, 57, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Lee, S.H. Differential gene expression profiles in the venom gland/sac of Eumenes pomiformis (Hymenoptera: Eumenidae). Toxicon 2010, 55, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Rangel, M.; Oliveira, J.S.; Cabrera, M.P.S.; Fontana, R.; Hirata, I.Y.; Nakata, Y.; Mori, K.; Kawano, M.; Fuchino, H.; et al. Decoralin, a novel linear cationic α-helical peptide from the venom of the solitary eumenine wasp Oreumenes decoratus. Peptides 2007, 28, 2320–2327. [Google Scholar] [CrossRef] [PubMed]
- Konno, K.; Hisada, M.; Fontana, R.; Lorenzi, C.C.B.; Naoki, H.; Itagaki, Y.; Miwa, A.; Kawai, N.; Nakata, Y.; Yasuhara, T.; et al. Anoplin, a novel antimicrobial peptide from the venom of the solitary wasp Anoplius samariensis. Biochim. Biophys. Acta 2001, 1550, 70–80. [Google Scholar] [CrossRef]
- Cabrera, M.P.S.; Arcisio-Miranda, M.; Costa, S.T.B.; Konno, K.; Ruggiero, J.R.; Procopio, J.; Ruggiero Neto, J. Study of the mechanism of action of Anoplin, a helical antimicrobial decapeptide with ion channel-like activity, and the role of the amidated C-terminus. J. Peptide Sci. 2008, 14, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Munk, J.K.; Uggerhøj, L.E.; Poulsen, T.J.; Frimodt-Møller, N.; Wimmer, R.; Nyberg, N.T.; Hansen, P.R. Synthetic analogs of anoplin show improved antimicrobial activities. J. Pept. Sci. 2013, 19, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Jindrichova, B.; Burketova, L.; Novotna, Z. Novel properties of antimicrobial peptide anoplin. Biochem. Biophys. Res. Commun. 2014, 444, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, J.; Zheng, X.; Yang, X.; Ma, P.; Cai, Y.; Zhang, B.; Chen, Y. Design of novel analogues of short antimicrobial peptide anoplin with improved antimicrobial activity. J. Pept. Sci. 2014, 20, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Daben, M.; Libardo, J.; Nagella, S.; Lugo, A.; Pierce, S.; Angeles-Boza, A.M. Copper-binding tripeptide motif increases potency of the antimicrobial peptide Anoplin via Reactive Oxygen Species generation. Biochem. Biophys. Res. Commun. 2015, 456, 446–451. [Google Scholar]
- Kuhn-Nentwig, L. Antimicrobial and cytolytic peptides of venomous arthropods. CMLS Cell. Mol. Life Sci. 2003, 60, 2651–2668. [Google Scholar] [CrossRef] [PubMed]
- Moreau, S.J.M. “It stings a bit but it cleans well”: Venoms of Hymenoptera and their antimicrobial potential. J. Insect Physiol. 2013, 59, 186–204. [Google Scholar] [CrossRef] [PubMed]
- Piek, T. Neurotoxic kinins from wasp and ant venoms. Toxicon 1991, 29, 139–149. [Google Scholar] [CrossRef]
- Konno, K.; Palma, M.S.; Hirata, I.Y.; Juliano, M.A.; Juliano, L.; Yasuhara, T. Identification of bradykinins in solitary wasp venoms. Toxicon 2002, 40, 309–312. [Google Scholar] [CrossRef]
- Konno, K.; Hisada, M.; Naoki, H.; Itagaki, Y.; Yasuhara, T.; Juliano, M.A.; Juliano, L.; Palma, M.S.; Nakajima, T. Isolation and sequence determination of peptides in the venom of the spider wasp Cyphononyx dorsalis guided by matrix-assisted laser desorption/ionization time of flight (MALDI-TOF) mass spectrometry. Toxicon 2001, 39, 1257–1260. [Google Scholar] [CrossRef]
- Picolo, G.; Hisada, M.; Sciani, J.M.; Conceição, I.M.; Machado, M.; Oliveira, V.; de Melo, R.L.; Cury, Y.; Konno, K.; Hayashi, M.A.F. Bradykinin-related peptides from the venom of the solitary wasp Cyphononyx fulvognathus. Biochem. Pharmacol. 2010, 79, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Arimoto, H.; Kinumi, T.; Oba, Y.; Uemura, D. Identification of proteins from venom of the paralytic spider wasp, Cyphononyx dorsalis. Insect Biochem. Mol. Biol. 2007, 37, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Lee, S.H. Identification and characterization of venom proteins of two solitary wasps, Eumenes pomiformis and Orancistrocerus drewseni. Toxicon 2010, 56, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Frølund, S.; Bella, A.; Kristensen, A.S.; Ziegler, H.L.; Witt, M.; Olsen, C.A.; Strømgaard, K.; Henrik Franzyk, H.; Jaroszewski, J.W. Assessment of structurally diverse philanthotoxin analogues for inhibitory activity on ionotropic Glutamate receptor subtypes: Discovery of nanomolar, nonselective, and use-dependent antagonists. J. Med. Chem. 2010, 53, 7441–7451. [Google Scholar] [CrossRef] [PubMed]
- Nihei, K.; Kazuma, K.; Ando, K.; Konno, K. Chemical and biological characterization of a novel neuropeptide in the venom of solitary digger wasp. Toxicon 2012, 60, 144. [Google Scholar] [CrossRef]
- Orchard, I.; Lange, A.B.; Bendena, W.G. FMRFamide-related peptides: A multifunctional family of structurally related neuropeptides in insects. Adv. Insect Physiol. 2001, 28, 267–329. [Google Scholar]
- Peeff, N.M.; Orchard, I.; Lange, A.B. Isolation, sequence, and bioactivity of PDVDHVFLRFamide and ADVGHVFLRFamide peptides from the locust central nervous system. Peptides 1994, 15, 387–392. [Google Scholar] [CrossRef]
- Holman, G.M.; Cook, B.J.; Nachman, R.J. Isolation, primary structure and synthesis of Leucomyosuppressin, an insect neuropeptide that inhibits spontaneous contractions of the cockroach hindgut. Comp. Biochem. Physiol. 1986, 85C, 329–333. [Google Scholar]
- Hisada, M.; Konno, K.; Itagaki, Y.; Naoki, H.; Nakajima, T. Sequencing wasp venom peptides by endopeptidase digestion and nested collision-induced dissociation/post-source decay method. Rapid Commun. Mass Spectr. 2002, 16, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Hisada, M.; Satake, H.; Masuda, K.; Aoyama, M.; Murata, K.; Shinada, T.; Iwashita, T.; Ohfune, Y.; Nakajima, T. Molecular components and toxicity of the venom of the solitary wasp, Anoplius samariensis. Biochem. Biophys. Res. Commun. 2005, 330, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Baek, J.H.; Yoon, K.A. Differential properties of venom peptides and proteins in solitary vs. social hunting wasps. Toxins 2016, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Hisada, M.; Konno, K.; Itagaki, Y.; Naoki, H.; Nakajima, T. Advantages of using nested collision induced dissociation/post-source decay with matrix-assisted laser desorption/ionization time of flight mass spectrometry: Sequencing of novel peptides from wasp venom. Rapid Commun. Mass Spectr. 2000, 14, 1828–1834. [Google Scholar] [CrossRef]
- Konno, K. Bioactive peptides in solitary wasp venoms. In Program & Abstracts, Proceedings of the 10th IST Asia Pacific Conference on Animal, Plants and Microbial Toxins, Changsha, China, June 2014; p. 18.


| Name | Sequence | Name | Sequence |
|---|---|---|---|
| Mastoparan | INLKALAALAKKIL-NH2 | EMP-OD | GRILSFIKGLAEHL-NH2 |
| EMP-AF | INLLKIAKGIIKSL-NH2 | Eumenitin | LNLKGIFKKVKSLLT |
| EMP-ER | FDIMGLIKKVAGAL-NH2 | Eumenitin-R | LNLKGLIKKVASLLN |
| EMP-EF | FDVMGIIKKIASAL-NH2 | Eumenitin-F | LNLKGLFKKVASLLT |
| EpVP2a | FDLLGLVKKVASAL-NH2 | EpVP1 | INLKGLIKKVASLLT |
| Anoplin | GLLKRIKTLL-NH2 | Decoralin | SLLSLIRKLIT |
| Name | Sequence |
|---|---|
| Cd-125 | DTARLKWH |
| As-126 | pQDPPVVKMK-NH2 |
| Bm-10 | pQTAPVPKAISK-NH2 |
| Orancis-Protonectin | ILGIITSLLKSL-NH2 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konno, K.; Kazuma, K.; Nihei, K.-i. Peptide Toxins in Solitary Wasp Venoms. Toxins 2016, 8, 114. https://doi.org/10.3390/toxins8040114
Konno K, Kazuma K, Nihei K-i. Peptide Toxins in Solitary Wasp Venoms. Toxins. 2016; 8(4):114. https://doi.org/10.3390/toxins8040114
Chicago/Turabian StyleKonno, Katsuhiro, Kohei Kazuma, and Ken-ichi Nihei. 2016. "Peptide Toxins in Solitary Wasp Venoms" Toxins 8, no. 4: 114. https://doi.org/10.3390/toxins8040114
APA StyleKonno, K., Kazuma, K., & Nihei, K.-i. (2016). Peptide Toxins in Solitary Wasp Venoms. Toxins, 8(4), 114. https://doi.org/10.3390/toxins8040114





