Prevalence, Characterization, and Mycotoxin Production Ability of Fusarium Species on Korean Adlay (Coix lacrymal-jobi L.) Seeds
Abstract
:1. Introduction
2. Results
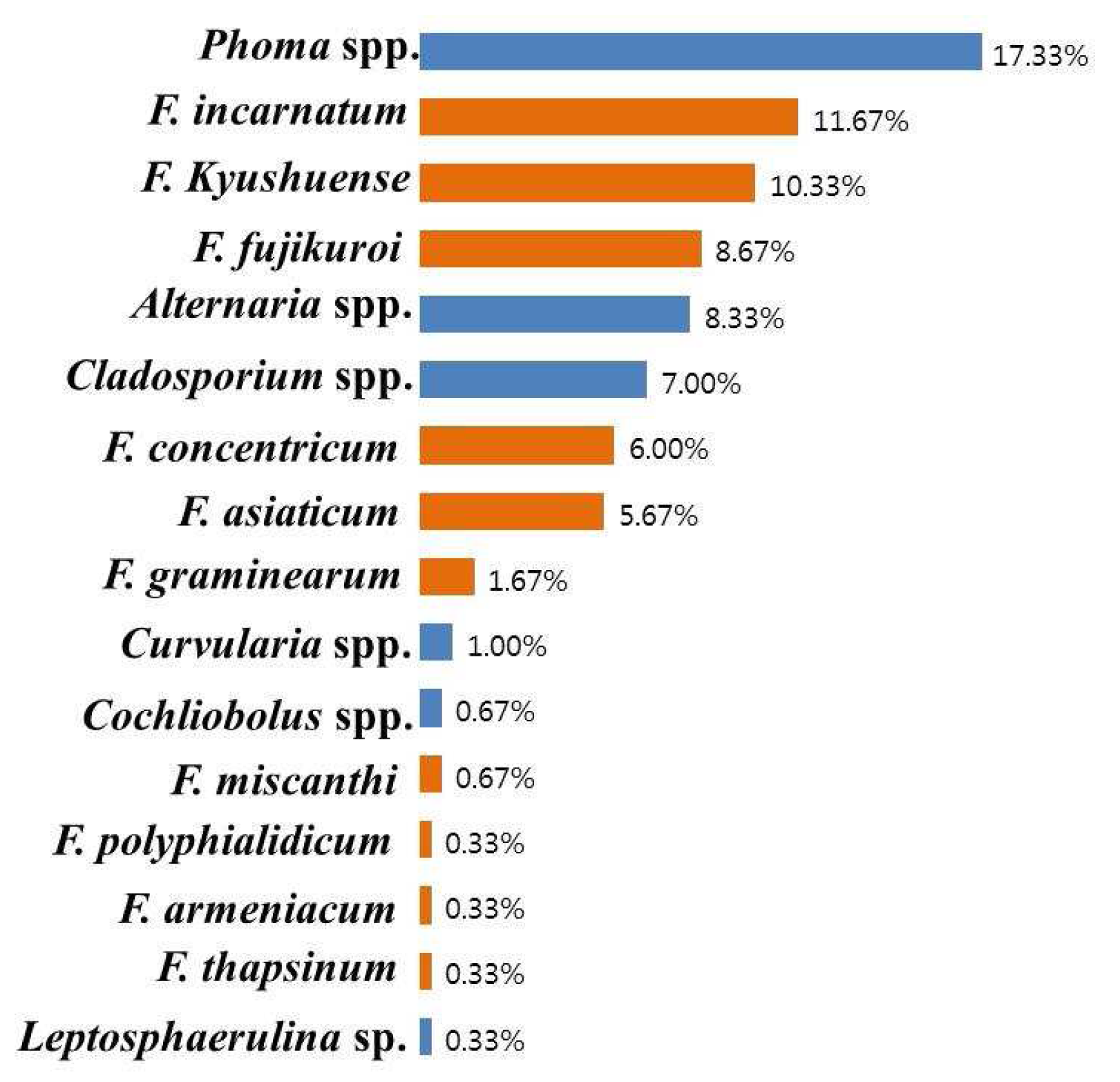
2.1. Mycofloral Incidence
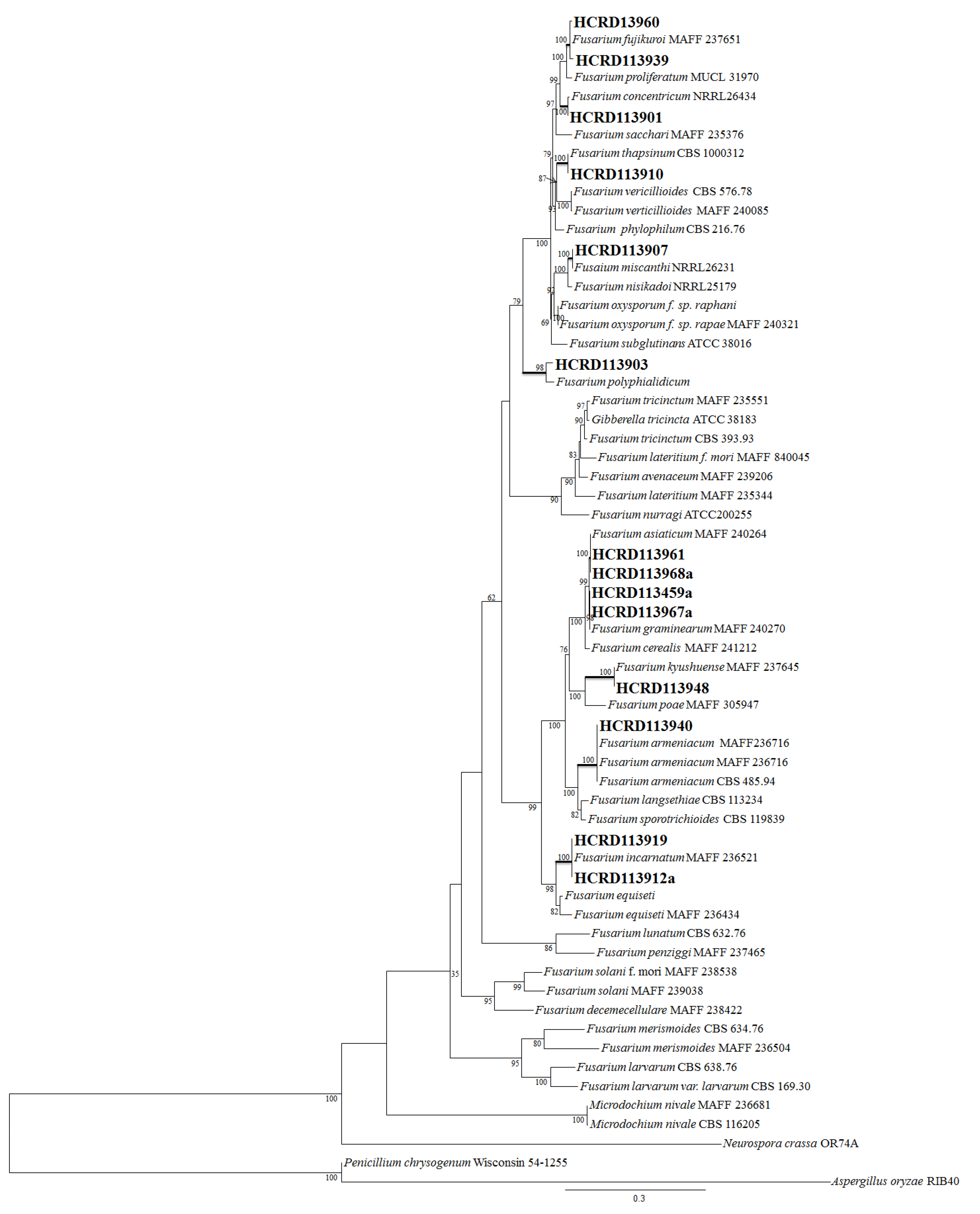
2.2. Incidence of Fusarium
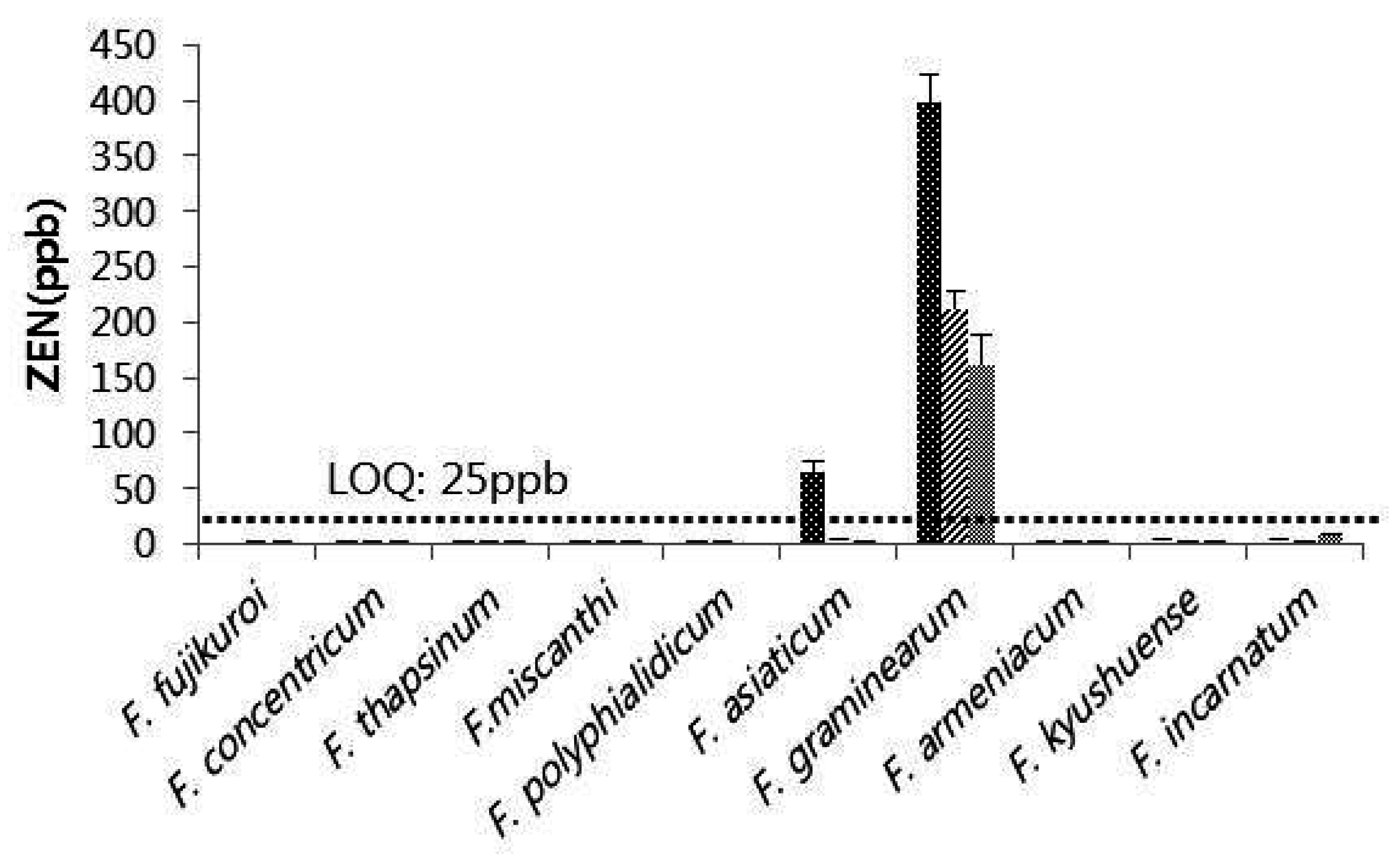
2.3. Occurance of Mycotoxin in Different Fusarium Species
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Plant Species and Sample Collection
5.2. Mycofloral Isolation and Analysis
5.3. Molecular Identification of the Isolates
5.3.1. DNA Extraction, PCR, and Purification
5.3.2. Sequencing and Phylogenetic Analysis
5.4. Analysis of Mycotoxins
5.4.1. Fusarium Inoculation for ELISA
5.4.2. Sample Preparation and Toxin Analysis
5.5. Morphological Characterization of Toxigenic Fusarium
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Akerele, O. Medicinal plants in traditional medicine. In Economic and Medicinal Plant Research; Wagner, H., Farnsworth, N., Eds.; Academic Press: London, UK, 1990; Volume 4, pp. 1–174. [Google Scholar]
- Barnes, P.M.; Powell-Griner, E.; McFann, K.; Nahin, R.L. Complementary and alternative medicine use among adults: United States, 2002. Semin. Integr. Med. 2004, 2, 54–71. [Google Scholar] [CrossRef]
- Lee, J.E.; Suh, M.H.; Yang, C.B. Characteristics of Job’s tear gruel by various mixing ratio, particle size and soaking time of Job’s tear and rice flour. Korean J. Food Cook. Sci. 2002, 18, 193–199. [Google Scholar]
- Ministry of Agriculture, Food and Rural Affair. Production Record of Cash Crops; Ministry of Agriculture, Food and Rural Affair: Sejong, Korea, 2015; p. 51.
- Jung, Y.J.; Kim, J.K.; Youn, K.S. Effect of roasting temperature on phycochemical pproperties of Job’s tears (Coix lachrymal-jobi L. var. ma-yeun) powder and extracts. Korean J. Food Preserv. 2006, 13, 477–482. [Google Scholar]
- Kim, H.K.; Cho, D.W.; Hahm, Y.T. The effects of Coix bran on lipid metabolism and glucose challenge in hyperlipidemic and diabetic rats. J. Korean Soc. Food Sci. Nutr. 2000, 29, 140–146. [Google Scholar]
- Park, G.S.; Lee, S.J. Effect of Job’s tears powder and green tea powder on the characteristics of quality of bread. J. Korean Soc. Food Sci. Nutr. 1999, 28, 1244–1250. [Google Scholar]
- Numata, M.; Yamamoto, A.; Moribayashi, A.; Yamada, H. Antitumor components isolated from the Chinese herbal medicine Coix lachrymal-jobi. Planta Med. 1994, 60, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, M.; Konno, C.; Hikino, H. Isolation and hypoglycemic activity of coixans A, B and C, glycans of Coix lachrymal-jobi L. var. ma-yuen seeds. Planta Med. 1986, 52, 64–65. [Google Scholar] [CrossRef] [PubMed]
- Ukita, T.; Tanimura, A. Studies on the anti-tumor component in the seeds of Coix lachrymal-jobi L. var. ma-yuen (Roman) STAPF. II: The structure of coixenolide. Chem. Pharm. Bull. 1961, 9, 43–46. [Google Scholar] [CrossRef]
- Choi, H.W.; Hong, S.K.; Kim, W.G.; Lee, Y.K. Diversity and pathogenicity of Fusarium species associated with head blight of Job’s tears. Korean J. Mycol. 2011, 39, 217–222. [Google Scholar] [CrossRef]
- Anonymous. List of Plant Disease in Korea, 5th ed.; The Korean Society of Plant Pathology: Seoul, Korea, 2009; p. 110. [Google Scholar]
- Bottalico, A.; Perrone, G. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant Pathol. 2002, 108, 611–624. [Google Scholar] [CrossRef]
- Steyn, P.S. Mycotoxins, general view, chemistry and structure. Toxicol. Lett. 2011, 82–83, 843–851. [Google Scholar] [CrossRef]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual; Blackwell Professional: Ames, IA, USA, 2006. [Google Scholar]
- Herron, D.A.; Wingfield, M.J.; Wingfield, B.D.; Rodas, C.A.; Manincowitz, S.; Steenkamp, E.T. Novel taxa in the Fusarium fujikuroi species complex from Pinus spp. Stud. Mycol. 2014, 80, 131–150. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.J.; Jiao, X.; Hu, Y.; Ku, X.; Gao, W. Mycobiota and mycotoxins in traditional medicinal seeds in China. Toxins 2015, 7, 3858–3875. [Google Scholar] [CrossRef] [PubMed]
- Langseth, W.; Bernhoft, A.; Rundberget, T.; Kosiak, B.; Gareis, M. Mycotoxin production and cytotoxicity of Fusarium strains isolated from Norwegian cereals. Mycopathologia 1999, 144, 103–113. [Google Scholar] [CrossRef]
- Aziz, N.H.; Youssef, Y.A.; El-Fouly, M.Z.; Moussa, L.A. Contamination of some medicinal plant samples and spices by fungi and their mycotoxins. Bot. Bull. Acad. Sin. 1998, 39, 279–285. [Google Scholar]
- Hashem, M.; Alamri, S. Contamination of common spices in Saudi Arabia markets with potential mycotoxin-producing fungi. Saudi J. Biol. Sci. 2010, 17, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Marin, S.; Sanchis, V.; Arnau, F.; Ramos, A.J.; Magan, N. Colonization and competitiveness of Aspergillus and Penicillium species on maize grain in the presence of Fusarium moniliforme and Fusarium proliferatum. Int. J. Food Microbiol. 1998, 45, 107–117. [Google Scholar] [CrossRef]
- Barros, G.; Torres, A.; Chulze, S. Aspergillus flavus population isolated from soil of Argentina’s peanut-growing region. Sclerotia production and toxigenic proflle. J. Sci. Food Agric. 2005, 85, 2349–2353. [Google Scholar]
- Kim, H.K.; Jang, H.S.; Choi, G.I.; Kim, H.J.; Kim, H.J.; Kim, H.L.; Cho, H.J.; Chan, L. Occurrence of mycotoxins in Korean grains and their simultaneous analysis. Korean J. Food Sci. Technol. 2013, 45, 111–119. [Google Scholar] [CrossRef]
- Mubatanhema, W.; Moss, M.O.; Frank, M.J.; Wilson, D.M. Prevalence of Fusarium species of the Liseola section on Zimbabwean corn and their ability to produce the mycotoxins zearalenone, moniliformin and fumonisin B1. Mycopathologia 1999, 48, 157–163. [Google Scholar] [CrossRef]
- Srobarova, A.; Pavlova, A. The pathogen localiuzation and ZEN concentration in wheat infected by Fusarium graminearum in relation to nutrition. Cereal Res. Commun. 1997, 25, 449–450. [Google Scholar]
- Adler, A.; Lew, H.; Edinger, W.; Oberforster, M. Occurance of moniliformin, deoxynivalenol and zearalenone in durum wheat (Triticum durum Desf.). Mycotoxin Res. 1995, 11, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Paul, N.C.; Deng, J.X.; Sang, H.K.; Choi, Y.P.; Yu, S.H. Distribution and antifungal activity of endophytic fungi in diferrent growth stages of chili pepper (Capsicum annuum L.) in Korea. Plant Pathol. J. 2012, 28, 10–19. [Google Scholar] [CrossRef]
- Paul, N.C.; Kim, W.K.; Woo, S.K.; Park, M.S.; Yu, S.H. Diversity of endophytic fungi associated with Taraxanum coreanum and their antifungal activity. Mycobiology 2006, 34, 185–190. [Google Scholar] [CrossRef] [PubMed]
- White, T.J.; Bruns, T.D.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press, Inc.: San Diego, CA, USA, 1990; pp. 315–322. [Google Scholar]
- Deng, J.X.; Paul, N.C.; Park, M.S.; Yu, S.H. Molecular characterization, morphology, and pathogenicity of Alternaria panax from aralicious plants in Korea. Mycol. Prog. 2013, 12, 383–396. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. ClustalX: Windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucl. Acids Res. 1997, 25, 487–488. [Google Scholar] [CrossRef]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]



| Structure | Characteristics * | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ff | Fc | Ft | Fm | Fp | Fa | Fg | Far | Fk | Fi | ||
| Macroconidia | Septa | 3 | 3–5 | 3 | 3–5 | 3 | 3–5 | 5–6 | 2–3 | 3–5 | 3–5 |
| Size | 38–41.1 × 3.26–3.24 | 33.08–45 × 2.5–2.75 | 21–29 × 2.5–2.7 | 40–57.2 × 3.8–4.1 | 27.87–30.1 × 2.8–3 | 39.63–60.5 × 4.69–6.22 | 54.7–65.2 × 4–5 | 25.5–27 × 3.5–4 | 31–41 × 4–5.5 | 23–36 × 3–4 | |
| Shape | Relatively slender | Relatively slender with no significant curvature | Relatively slender, slightly falcate or straight | Relatively slender | Relatively wide, straight, stout and robust | Curved to straight | Curved to straight with ventral surface | Prominently curved | Falcate to fusiform | Relatively slender with a curved dorsal surface | |
| Microconidia | Septa | 0–1 | 0–1 | 0 | 0 | 0–2 | - | - | - | 0–3 | 0–1 |
| Size | 6.48–16.19 × 1.14–2.27 | 6.14–9.36 × 2.29–2.6 | 6.2–7 × 2–2.5 | 7.5–11 × 5–7 | 8.76–16.71 × 2.46–4.51 | - | - | - | 18.7–20 × 2–4 | 4.5–9.6 × 2–3 | |
| Shape | Oval, club shaped with a flattened base | Oval, obovoid to allantoid | Club shaped with a flattened base | Pyriform | Fusiform or subclavate | - | - | - | Fusiform to falcate | Fusiform | |
| Primers | Primer Sequences | References |
|---|---|---|
| ITS5 | GGAAGTAAAAGTCGTAACAAGG | White et al. 1990 |
| ITS4 | TCCTCCGCTTATTGATATGC | |
| EF1 | ATGGGTAAGGAAGACAAGAC | O’Donnell et al. 2000 |
| EF2 | GGAAGTACCAGTGATCATGTT | |
| EF3 | GTAAGGAGGASAAGACTCACC | |
| EF2T | GGAAGTACCAGTGATCATGTT | |
| Btu-F-F01 | CAGACCGGTCAGTGCGTAA | Watanabe et al. 2011 |
| Btu-F-R01 | TTGGGGTCGAACATCTGCT |
| Gene | Initial Denaturing | Denaturing | Annealing | Extension | Final Extension | Cycle |
|---|---|---|---|---|---|---|
| ITS (ITS5, ITS4) | 94 °C, 10 m | 94 °C, 30 s | 55 °C, 30 s | 72 °C, 1 m | 72 °C, 10 m | 30 |
| EF (EF1, EF2) | 94 °C, 5 m | 94 °C, 30 s | 52 °C, 40 s | 72 °C, 1 m | 72 °C, 3 m | 35 |
| EF (EF3, EF2T) | 94 °C, 5 m | 94 °C, 30 s | 53 °C, 30 s | 72 °C, 1 m | 72 °C, 5 m | 40 |
| β-tubulin (BT2a, BT2b) | 94 °C, 5 m | 94 °C, 30 s | 60 °C, 30 s | 72 °C, 1 m | 72 °C, 3 m | 35 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
An, T.J.; Shin, K.S.; Paul, N.C.; Kim, Y.G.; Cha, S.W.; Moon, Y.; Yu, S.H.; Oh, S.-K. Prevalence, Characterization, and Mycotoxin Production Ability of Fusarium Species on Korean Adlay (Coix lacrymal-jobi L.) Seeds. Toxins 2016, 8, 310. https://doi.org/10.3390/toxins8110310
An TJ, Shin KS, Paul NC, Kim YG, Cha SW, Moon Y, Yu SH, Oh S-K. Prevalence, Characterization, and Mycotoxin Production Ability of Fusarium Species on Korean Adlay (Coix lacrymal-jobi L.) Seeds. Toxins. 2016; 8(11):310. https://doi.org/10.3390/toxins8110310
Chicago/Turabian StyleAn, Tae Jin, Kyu Seop Shin, Narayan Chandra Paul, Young Guk Kim, Seon Woo Cha, Yuseok Moon, Seung Hun Yu, and Sang-Keun Oh. 2016. "Prevalence, Characterization, and Mycotoxin Production Ability of Fusarium Species on Korean Adlay (Coix lacrymal-jobi L.) Seeds" Toxins 8, no. 11: 310. https://doi.org/10.3390/toxins8110310
APA StyleAn, T. J., Shin, K. S., Paul, N. C., Kim, Y. G., Cha, S. W., Moon, Y., Yu, S. H., & Oh, S.-K. (2016). Prevalence, Characterization, and Mycotoxin Production Ability of Fusarium Species on Korean Adlay (Coix lacrymal-jobi L.) Seeds. Toxins, 8(11), 310. https://doi.org/10.3390/toxins8110310






