Development of an Innovative in Vitro Potency Assay for Anti-Botulinum Antitoxins
Abstract
:1. Introduction
2. Results
2.1. Binding of BoNT/B to Syt-II40-60 Peptide
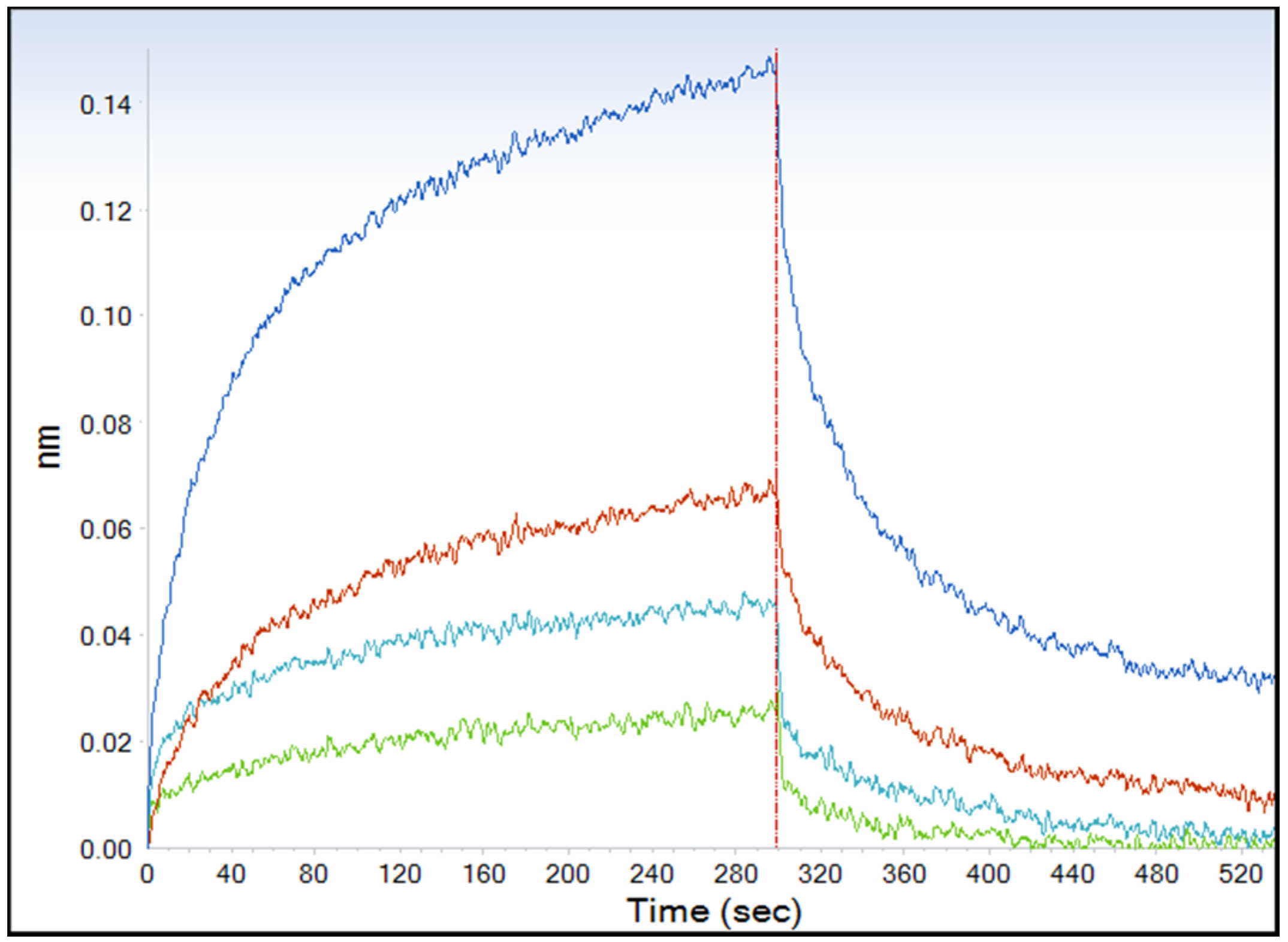
2.1.1. Application of Bio-Layer Interferometry (BLI) for the Analysis of BoNT/B-Syt-II40-60 Interaction
2.2. Establishment of an in Vitro Anti-BoNT/B Potency Assay
2.3. Evaluating the Performance of the in Vitro Assay in Quantitating Neutralizing Antibody Concentration in Unknown Antitoxin Preparations
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Materials
4.2.1. Peptides
4.2.2. Toxins and Antitoxins
4.3. Peptide Conjugation to Magnetic Beads
4.4. In Vivo Mouse Neutralization Antibody Test
4.5. In Vitro Neutralization Assay
4.6. Endo-Peptidase Activity Assay
4.7. UPLC Analysis
4.8. Bead ELISA
4.9. Biolayer Interferometry
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hatheway, C. Botulism. In Laboratory Diagnosis of Infectious Diseases; Balows, A., Hausler, W.J., Jr., Ohashi, M., Turano, A., Lennete, E.H., Eds.; Springer: New York, NY, USA, 1988; pp. 111–133. [Google Scholar]
- Barash, J.R.; Arnon, S.S. A novel strain of clostridium botulinum that produces type B and type H botulinum toxins. J. Infect. Dis. 2014, 209, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Turton, K.; Chaddock, J.A.; Acharya, K.R. Botulinum and tetanus neurotoxins: Structure, function and therapeutic utility. Trends Biochem. Sci. 2002, 27, 552–558. [Google Scholar] [CrossRef]
- Schiavo, G.; Matteoli, M.; Montecucco, C. Neurotoxins affecting neuroexocytosis. Physiol. Rev. 2000, 80, 717–766. [Google Scholar] [PubMed]
- Sobel, J. Botulism. Clin. Infect. Dis. 2005, 41, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). CSTE botulism surveillance summary 2014. Available online: https://www.cdc.gov/nationalsurveillance/pdfs/botulism_cste_2014.pdf (accessed on 19 July 2016).
- Dong, M.; Yeh, F.; Tepp, W.H.; Dean, C.; Johnson, E.A.; Janz, R.; Chapman, E.R. SV2 is the protein receptor for botulinum neurotoxin A. Science 2006, 312, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Arnon, S.S.; Schechter, R.; Inglesby, T.V.; Henderson, D.A.; Bartlett, J.G.; Ascher, M.S.; Eitzen, E.; Fine, A.D.; Hauer, J.; Layton, M.; et al. Botulinum toxin as a biological weapon: Medical and public health managment. J. Am. Med. Assoc. 2001, 285, 1059–1070. [Google Scholar] [CrossRef]
- Tacket, C.O.; Shandera, W.X.; Mann, J.M.; Hargrett, N.T.; Blake, P.A. Equine antitoxin use and other factors that predict outcome in type a foodborne botulism. Am. J. Med. 1984, 76, 794–798. [Google Scholar] [CrossRef]
- European Directorate for the Quality of Medicines and Healthcare. Botulinum antitoxin. In European Pharmacopoeia, 8th ed.; EDQM Council of Europe: Strasbourg, France, 2014; p. 1029. [Google Scholar]
- Dressler, D.; Dirnberger, G. Botulinum toxin antibody testing: Comparison between the immunoprecipitation assay and the mouse diaphragm assay. Eur Neurol. 2001, 45, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Hanna, P.A.; Jankovic, J. Mouse bioassay versus western blot assay for botulinum toxin antibodies: Correlation with clinical response. Neurology 1998, 50, 1624–1629. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, C.Y.; Smith, L.A.; West, M.W.; Boles, J.W.; Brown, J.E. Evaluation of a botulinum fragment C-based elisa for measuring the humoral immune response in primates. Biologicals 2003, 31, 17–24. [Google Scholar] [CrossRef]
- Palace, J.; Nairne, A.; Hyman, N.; Doherty, T.V.; Vincent, A. A radioimmuno-precipitation assay for antibodies to botulinum A. Neurology 1998, 50, 1463–1466. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Tepp, W.H.; Clancy, C.M.; Borodic, G.E.; Johnson, E.A. A neuronal cell-based botulinum neurotoxin assay for highly sensitive and specific detection of neutralizing serum antibodies. FEBS Lett. 2007, 581, 4803–4808. [Google Scholar] [CrossRef] [PubMed]
- Frisk, M.L.; Lin, G.; Johnson, E.A.; Beebe, D.J. Synaptotagmin II peptide-bead conjugate for botulinum toxin enrichment and detection in microchannels. Biosens. Bioelectron. 2010, 26, 1929–1935. [Google Scholar] [CrossRef] [PubMed]
- Wictome, M.; Newton, K.; Jameson, K.; Hallis, B.; Dunnigan, P.; Mackay, E.; Clarke, S.; Taylor, R.; Gaze, J.; Foster, K.; et al. Development of an in vitro bioassay for clostridium botulinum type B neurotoxin in foods that is more sensitive than the mouse bioassay. Appl. Environ. Microbiol. 1999, 65, 3787–3792. [Google Scholar] [PubMed]
- Kalb, S.R.; Moura, H.; Boyer, A.E.; McWilliams, L.G.; Pirkle, J.L.; Barr, J.R. The use of Endopep-MS for the detection of botulinum toxins A, B, E, and F in serum and stool samples. Anal. Biochem. 2006, 351, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.G.; Liu, Y.; Rigsby, P.; Sesardic, D. An improved method for development of toxoid vaccines and antitoxins. J. Immunol. Methods 2008, 337, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Hallis, B.; James, B.A.; Shone, C.C. Development of novel assays for botulinum type A and B neurotoxins based on their endopeptidase activities. J. Clin. Microbiol. 1996, 34, 1934–1938. [Google Scholar] [PubMed]
- Rosen, O.; Feldberg, L.; Gura, S.; Brosh-Nissimov, T.; Guri, A.; Zimhony, O.; Shapiro, E.; Beth-Din, A.; Stein, D.; Ozeri, E.; et al. Early, real-time medical diagnosis of botulism by endopeptidase-mass spectrometry. Clin. Infect. Dis. 2015, 61, e58–e61. [Google Scholar] [CrossRef] [PubMed]
- Boyer, A.E.; Moura, H.; Woolfitt, A.R.; Kalb, S.R.; McWilliams, L.G.; Pavlopoulos, A.; Schmidt, J.G.; Ashley, D.L.; Barr, J.R. From the mouse to the mass spectrometer: Detection and differentiation of the endoproteinase activities of botulinum neurotoxins A-G by mass spectrometry. Anal. Chem. 2005, 77, 3916–3924. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Goodnough, M.C.; Malizio, C.J.; Pirkle, J.L.; Barr, J.R. Detection of botulinum neurotoxin A in a spiked milk sample with subtype identification through toxin proteomics. Anal. Chem. 2005, 77, 6140–6146. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Baudys, J.; Kalb, S.R.; Barr, J.R. Improved detection of botulinum neurotoxin type A in stool by mass spectrometry. Anal. Biochem. 2011, 412, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Bjornstad, K.; Tevell Aberg, A.; Kalb, S.R.; Wang, D.; Barr, J.R.; Bondesson, U.; Hedeland, M. Validation of the Endopep–MS method for qualitative detection of active botulinum neurotoxins in human and chicken serum. Anal. Bioanal. Chem. 2014, 406, 7149–7161. [Google Scholar] [CrossRef] [PubMed]
- Kalb, S.R.; Krilich, J.C.; Dykes, J.K.; Luquez, C.; Maslanka, S.E.; Barr, J.R. Detection of botulinum toxins A, B, E, and F in foods by Endopep-MS. J. Agric. Food Chem. 2015, 63, 1133–1141. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Arndt, J.W.; Dong, M.; Tepp, W.H.; Johnson, E.A.; Chapman, E.R.; Stevens, R.C. Structural basis of cell surface receptor recognition by botulinum neurotoxin B. Nature 2006, 444, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Rummel, A.; Binz, T.; Brunger, A.T. Botulinum neurotoxin B recognizes its protein receptor with high affinity and specificity. Nature 2006, 444, 1092–1095. [Google Scholar] [CrossRef] [PubMed]
- Barr, J.R.; Moura, H.; Boyer, A.E.; Woolfitt, A.R.; Kalb, S.R.; Pavlopoulos, A.; McWilliams, L.G.; Schmidt, J.G.; Martinez, R.A.; Ashley, D.L. Botulinum neurotoxin detection and differentiation by mass spectrometry. Emerg. Infect. Dis. 2005, 11, 1578–1583. [Google Scholar] [CrossRef] [PubMed]
- Bowmer, E.J. Preparation and assay of the international standards for clostridium botulinum types A, B, C, D and E antitoxins. Bull. World Health Organ. 1963, 29, 701–709. [Google Scholar] [PubMed]
- Food and Drug Administration (FDA). Highlights of prescribing information. Available online: http://www.fda.gov/ucm/groups/fdagov-public/@fdagov-bio-gen/documents/document/ucm345147.pdf (accessed on 19 July 2016).
- Hatheway, C.H.; Snyder, J.D.; Seals, J.E.; Edell, T.A.; Lewis, G.E., Jr. Antitoxin levels in botulism patients treated with trivalent equine botulism antitoxin to toxin types A, B, and E. J. Infect. Dis 1984, 150, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Du, Z.W.; Pier, C.L.; Tepp, W.H.; Zhang, S.C.; Johnson, E.A. Sensitive and quantitative detection of botulinum neurotoxin in neurons derived from mouse embryonic stem cells. Biochem. Biophys. Res. Commun. 2011, 404, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Pellett, S.; Tepp, W.H.; Toth, S.I.; Johnson, E.A. Comparison of the primary rat spinal cord cell (RSC) assay and the mouse bioassay for botulinum neurotoxin type A potency determination. J. Pharmacol. Toxicol. Methods 2010, 61, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Stahl, A.M.; Ruthel, G.; Torres-Melendez, E.; Kenny, T.A.; Panchal, R.G.; Bavari, S. Primary cultures of embryonic chicken neurons for sensitive cell-based assay of botulinum neurotoxin: Implications for therapeutic discovery. J. Biomol. Screen. 2007, 12, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Bao, S.; Yin, J.; Cai, K.; Hou, X.; Xiao, L.; Tu, W.; Wang, Q.; Wang, H. Dominant antigenic peptides located at the heavy chain terminal of botulinum neurotoxin B contain receptor-binding sites for synaptotagmin II. Biochem. Biophys. Res. Commun. 2008, 374, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Strotmeier, J.; Willjes, G.; Binz, T.; Rummel, A. Human synaptotagmin-ii is not a high affinity receptor for botulinum neurotoxin B and G: Increased therapeutic dosage and immunogenicity. FEBS Lett. 2012, 586, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Whelan, S.M.; Elmore, M.J.; Bodsworth, N.J.; Brehm, J.K.; Atkinson, T.; Minton, N.P. Molecular cloning of the clostridium botulinum structural gene encoding the type B neurotoxin and determination of its entire nucleotide sequence. Appl. Environ. Microbiol. 1992, 58, 2345–2354. [Google Scholar] [PubMed]
- Zichel, R.; Mimran, A.; Keren, A.; Barnea, A.; Steinberger-Levy, I.; Marcus, D.; Turgeman, A.; Reuveny, S. Efficacy of a potential trivalent vaccine based on hc fragments of botulinum toxins A, B, and E produced in a cell-free expression system. Clin. Vaccine Immunol. 2010, 17, 784–792. [Google Scholar] [CrossRef] [PubMed]
- European Directorate for the Quality of Medicines and Healthcare. Botulinum antitoxin. In European Pharmacopoeia, 7th ed.; EDQM Council of Europe: Strasbourg, France, 2011; Volume 1, p. 949. [Google Scholar]
- Rosen, O.; Feldberg, L.; Gura, S.; Zichel, R. Improved detection of botulinum type E by rational design of a new peptide substrate for endopeptidase-mass spectrometry assay. Anal. Biochem. 2014, 456, 50–52. [Google Scholar] [CrossRef] [PubMed]
- Rosen, O.; Feldberg, L.; Gura, S.; Zichel, R. A new peptide substrate for enhanced botulinum neurotoxin type B detection by endopeptidase-liquid chromatography-tandem mass spectrometry/multiple reaction monitoring assay. Anal. Biochem. 2015, 473, 7–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Baudys, J.; Krilich, J.; Smith, T.J.; Barr, J.R.; Kalb, S.R. A two-stage multiplex method for quantitative analysis of botulinum neurotoxins type A, B, E, and F by MALDI-TOF mass spectrometry. Anal. Chem. 2014, 86, 10847–10854. [Google Scholar] [CrossRef] [PubMed]

| Preparation | Anti-BoNT/B Antibody Potencies | |||||
| Mouse Assay (IU/mL) | Endopep Assay (IU/mL) | |||||
| Average (n) | SD | CV (%) | Average (n) | SD | CV (%) | |
| F-1 | 4875 (3) | 650 | 13 | 4178 (4) | 171 | 4.1 |
| F-2 | 3798 (3) | 605 | 16 | 3843 (3) | 469 | 12.2 |
| F-3 | 3800 (3) | 0 | 0 | 3722 (3) | 294 | 7.9 |
| F-1/2 * | 2438 ** | NA | NA | 2860 (3) | 250 | 8.7 |
| F-3/1.5 * | 2533 ** | NA | NA | 2576 (3) | 290 | 11.3 |
| I-1 | 1330 (1) | NA | NA | 1403 (3) | 54 | 3.8 |
| I-2 | 1014 (1) | NA | NA | 1330 (3) | 143 | 10.8 |
| I-3 | 874 (1) | NA | NA | 1105 (3) | 96 | 8.7 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosen, O.; Ozeri, E.; Barnea, A.; David, A.B.; Zichel, R. Development of an Innovative in Vitro Potency Assay for Anti-Botulinum Antitoxins. Toxins 2016, 8, 276. https://doi.org/10.3390/toxins8100276
Rosen O, Ozeri E, Barnea A, David AB, Zichel R. Development of an Innovative in Vitro Potency Assay for Anti-Botulinum Antitoxins. Toxins. 2016; 8(10):276. https://doi.org/10.3390/toxins8100276
Chicago/Turabian StyleRosen, Osnat, Eyal Ozeri, Ada Barnea, Alon Ben David, and Ran Zichel. 2016. "Development of an Innovative in Vitro Potency Assay for Anti-Botulinum Antitoxins" Toxins 8, no. 10: 276. https://doi.org/10.3390/toxins8100276
APA StyleRosen, O., Ozeri, E., Barnea, A., David, A. B., & Zichel, R. (2016). Development of an Innovative in Vitro Potency Assay for Anti-Botulinum Antitoxins. Toxins, 8(10), 276. https://doi.org/10.3390/toxins8100276