Ultrasound-Guided Nerve Block with Botulinum Toxin Type A for Intractable Neuropathic Pain
Abstract
:1. Introduction
2. Case Reports
2.1. Case 1
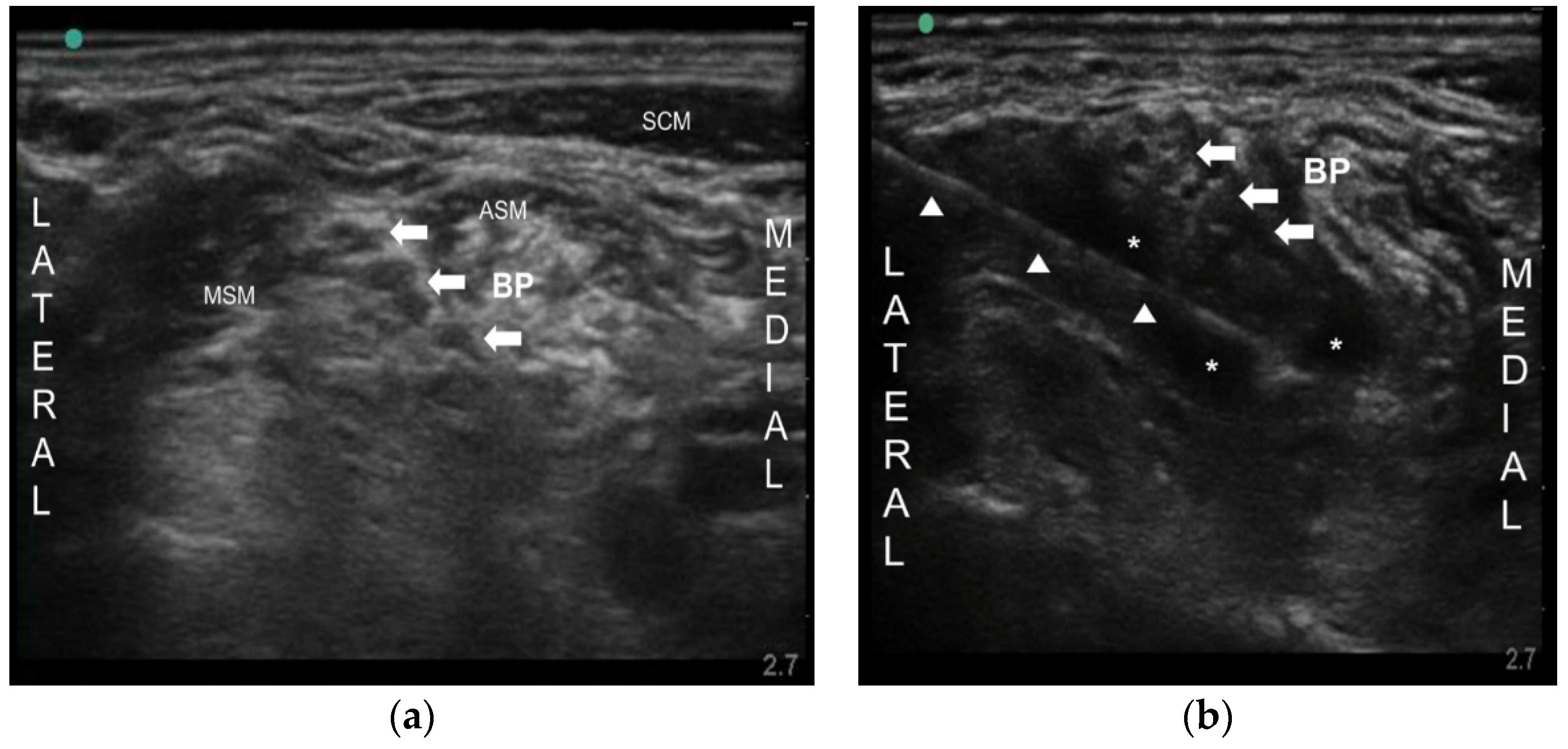
2.2. Case 2
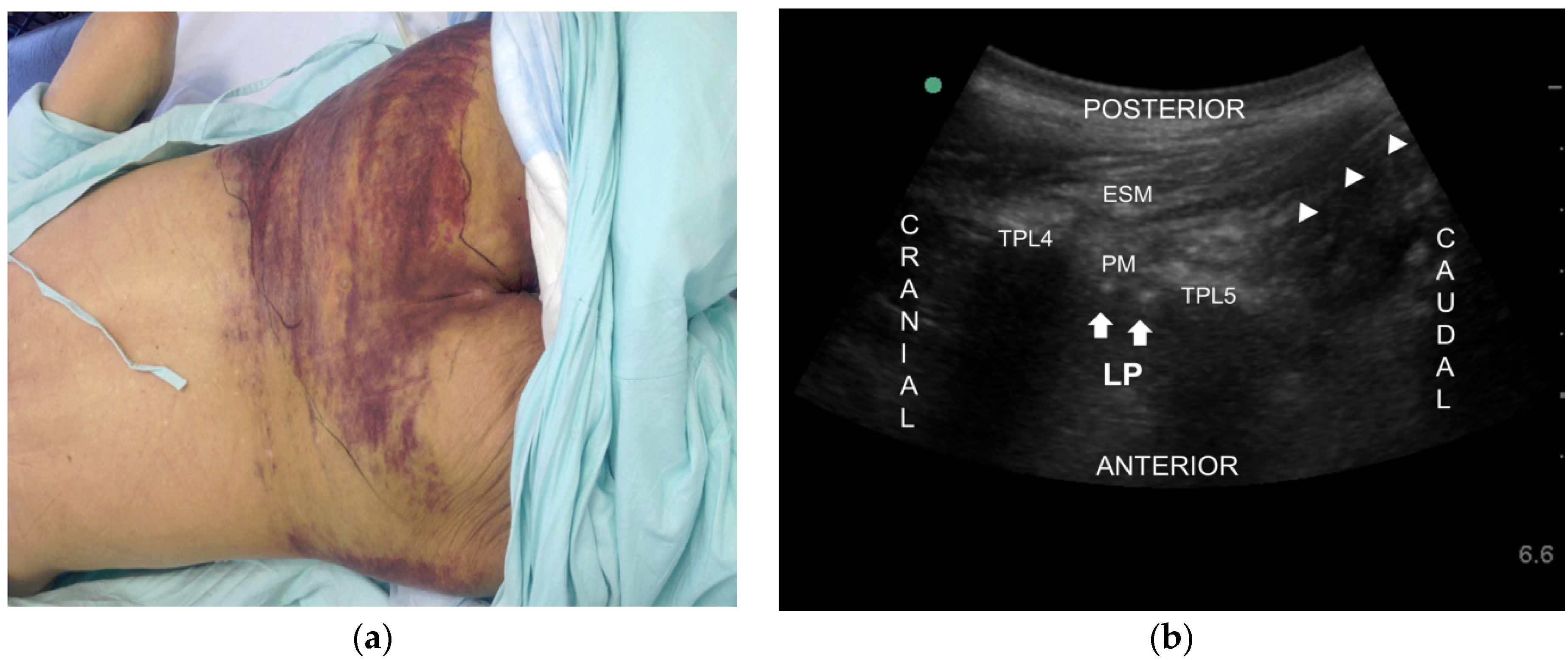
3. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mersky, H.; Bogduk, N. Classification of Chronic Pain; IASP Press: Seattle, WA, USA, 1994. [Google Scholar]
- Park, H.J.; Moon, D.E. Pharmacologic management of chronic pain. Korean J. Pain 2010, 23, 99–108. [Google Scholar] [CrossRef]
- Jensen, M.P.; Chodroff, M.J.; Dworkin, R.H. The impact of neuropathic pain on health-related quality of life: Review and implications. Neurology 2007, 68, 1178–1182. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Park, H.J.; Lee, S.H.; Moon, D.E. Ganglion impar block with botulinum toxin type A for chronic perineal pain -a case report. Korean J. Pain 2010, 23, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.H. Herpes Zoster and Postherpetic Neuralgia: Practical Consideration for Prevention and Treatment. Korean J. Pain 2015, 28, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Tesfaye, S.; Vileikyte, L.; Rayman, G.; Sindrup, S.; Perkins, B.; Baconja, M.; Vinik, A.I.; Boulton, A.J. Painful diabetic peripheral neuropathy: Consensus recommendations on diagnosis, assessment and management. Diabetes Metab. Res. Rev. 2011, 27, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.A.; Schütz, S.G.; Simpson, D.M. Botulinum toxin for neuropathic pain and spasticity: An overview. Pain Manag. 2014, 4, 129–151. [Google Scholar] [CrossRef] [PubMed]
- Kapural, L.; Stillman, M.; Kapural, M.; McIntyre, P.; Guirgius, M.; Mekhail, N. Botulinum toxin occipital nerve block for the treatment of severe occipital neuralgia: A case series. Pain Pract. 2007, 7, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.; Silva, S.; Cottrell, C. Botulinum toxin type-A (BOTOX) in the treatment of occipital neuralgia: A pilot study. Headache 2008, 48, 1476–1481. [Google Scholar] [CrossRef] [PubMed]
- Yuan, R.Y.; Sheu, J.J.; Yu, J.M.; Chen, W.T.; Tseng, I.J.; Chang, H.H.; Hu, C.J. Botulinum toxin for diabetic neuropathic pain: A randomized double-blind crossover trial. Neurology 2009, 72, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.T.; Yuan, R.Y.; Chiang, S.C.; Sheu, J.J.; Yu, J.M.; Tseng, I.J.; Yang, S.K.; Chang, H.H.; Hu, C.J. OnabotulinumtoxinA improves tactile and mechanical pain perception in painful diabetic polyneuropathy. Clin. J. Pain 2013, 29, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Mackey, S.; Hui, H.; Xong, D.; Zhang, Q.; Zhang, D. Subcutaneous injection of botulinum toxin A is beneficial in postherpetic neuralgia. Pain Med. 2010, 11, 1827–1833. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Nam, T.S.; Paik, K.S.; Leem, J.W. Involvement of peripherally released substance P and calcitonin gene-related peptide in mediating mechanical hyperalgesia in a traumatic neuropathy model of the rat. Neurosci. Lett. 2004, 360, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.H.; Kim, D.W.; Nam, T.S.; Paik, K.S.; Leem, J.W. Peripheral glutamate receptors contribute to mechanical hyperalgesia in a neuropathic pain model of the rat. Neuroscience 2004, 128, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Burstein, R.; Zhang, X.; Levy, D.; Aoki, K.R.; Brin, M.F. Selective inhibition of meningeal nociceptors by botulinum neurotoxin type A: Therapeutic implications for migraine and other pains. Cephalalgia 2014, 34, 853–869. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moon, Y.E.; Choi, J.H.; Park, H.J.; Park, J.H.; Kim, J.H. Ultrasound-Guided Nerve Block with Botulinum Toxin Type A for Intractable Neuropathic Pain. Toxins 2016, 8, 18. https://doi.org/10.3390/toxins8010018
Moon YE, Choi JH, Park HJ, Park JH, Kim JH. Ultrasound-Guided Nerve Block with Botulinum Toxin Type A for Intractable Neuropathic Pain. Toxins. 2016; 8(1):18. https://doi.org/10.3390/toxins8010018
Chicago/Turabian StyleMoon, Young Eun, Jung Hyun Choi, Hue Jung Park, Ji Hye Park, and Ji Hyun Kim. 2016. "Ultrasound-Guided Nerve Block with Botulinum Toxin Type A for Intractable Neuropathic Pain" Toxins 8, no. 1: 18. https://doi.org/10.3390/toxins8010018
APA StyleMoon, Y. E., Choi, J. H., Park, H. J., Park, J. H., & Kim, J. H. (2016). Ultrasound-Guided Nerve Block with Botulinum Toxin Type A for Intractable Neuropathic Pain. Toxins, 8(1), 18. https://doi.org/10.3390/toxins8010018




