Staphylococcus epidermidis and Staphylococcus haemolyticus: Molecular Detection of Cytotoxin and Enterotoxin Genes
Abstract
:1. Background
2. Results
2.1. Detection of Enterotoxin Genes
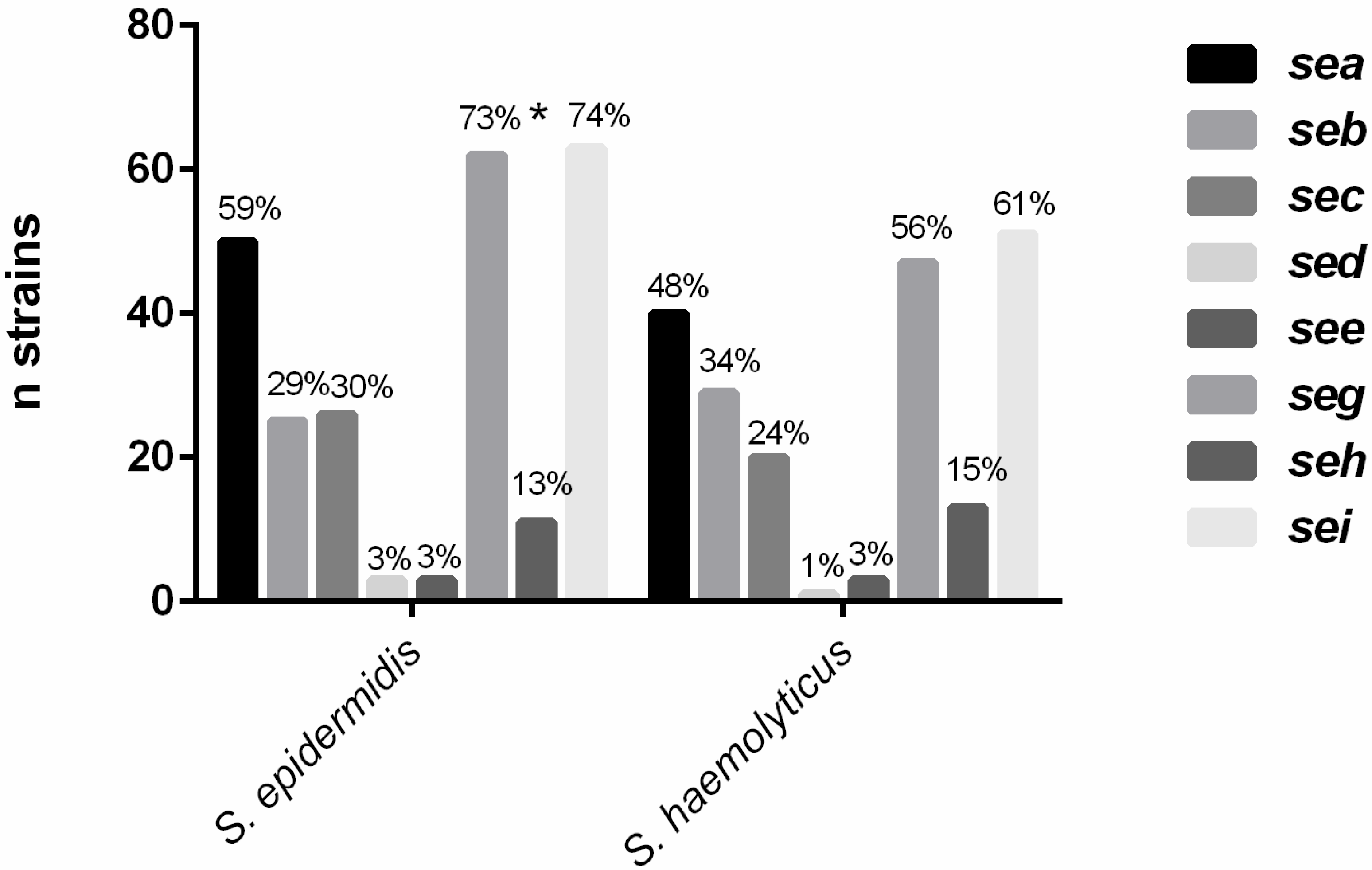
2.2. Molecular and Phenotypic Detection of Cytotoxins
| Organisms (n) | hla * | hlb | hld | α-toxin | β-toxin | δ-toxin | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | n | % | n | % | n | % | |
| S. epidermidis (85) | 79 | 92.9 | 79 | 92.9 | 81 | 95.3 | 24 | 28.2 | 25 | 29.4 | - | - |
| S. haemolyticus (84) | 77 | 91.7 | - | - | - | - | 70 | 83.3 | 68 | 81 | 34 | 40.5 |
| Total (169) | 156 | 92.3 | 79 | 92.9 | 81 | 95.3 | 94 | 55.6 | 93 | 55 | 34 | 40.5 |
| Staphylococcus haemolyticus | Staphylococcus epidermidis | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Genes | Genes | Genes | ||||||||
| Toxin | hla+ | hla− | Total | hla+ * | hla− * | Total | Toxin | hlb+ | hlb− | Total |
| n(%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | n (%) | ||
| α-Toxin + | 65 (78) | 5 (6) | 70 (84) | 24 (28) | 0 | 24 (28) | β-Toxin + | 24(28) | 1 (1) | 25 (29) |
| α-Toxin − | 12 (14) | 2 (2) | 14 (16) | 55 (65) | 6 (7) | 61 (72) | β-Toxin − | 55 (65) | 5 (6) | 60 (71) |
| Total | 77 (92) | 7 (8) | 84 (100) | 79 (93) | 6 (7) | 85 (100) | Total | 79 (93) | 6 (7) | 85 (100) |
3. Discussion
4. Material and Methods
4.1. Isolates
4.2. Species Identification
4.3. Detection of Enterotoxin Genes
| Name | Product | Sequence | Reference | Amplicon Size (bp) |
|---|---|---|---|---|
| sea-1 | Enterotoxin A | TTGGAAACGGTTAAAACGAA | [29] | 120 |
| sea-2 | GAACCTTCCCATCAAAAACA | |||
| seb-1 | Enterotoxin B | TCGCATCAAACTGACAAACG | [29] | 478 |
| seb-2 | GCAGGTACTCTATAAGTGCC | |||
| sec-1 | Enterotoxin C | GACATAAAAGCTAGGAATTT | [29] | 257 |
| sec-2 | AAATCGGATTAACATTATCC | |||
| sed-1 | Enterotoxin D | CTAGTTTGGTAATATCTCCT | [29] | 317 |
| sed-2 | TAATGCTATATCTTATAGGG | |||
| see-1 | Enterotoxin E | CAAAGAAATGCTTTAAGCAATCTTAGGCCAC | [29] | 170 |
| see-2 | CTTACCGCCAAAGCTG | |||
| seg-1 | Enterotoxin G | AATTATGTGAATGCTCAACCCGATC | [36] | 642 |
| seg-2 | AAACTTATATGGAACAAAAGGTACTAGTTC | |||
| sei-1 | Enterotoxin H | CAATCACATCATATGCGAAAGCAG | [36] | 376 |
| seh-2 | CATCTACCCAAACATTAGCACC | |||
| sei-1 | Enterotoxin I | CTCAAGGTGATATTGGTGTAGG | [36] | 576 |
| sei-2 | AAAAAACTTACAGGCAGTCCATCTC | |||
| hla/yidD_epid-1 | α-hemolysin/yidD | TTTCKCCACTTACACCMCC | This study | 160 |
| hla/yidD_epid-2 | GGAACAGGATCAAAGCCACCT | |||
| hlb_epid-1 | β-hemolysin | TGGTGGCGTTGGTATTGTGA | This study | 541 |
| hlb_epid-2 | ACCCCAAGATTTCACGGACC | |||
| hla_haem-1 | α-hemolysin | TGGGCCATAAACTTCAATCGC | This study | 72 |
| hla-haem-2 | ACGCCACCTACATGCAGATTT | |||
| hld-epid-1 | δ-hemolysin | ATGGCAGCAGATATCATTTC | [30] | 444 |
| hld-epid-2 | CGTGAGCTTGGGAGAGAC |
4.4. Detection of Hemolysin Genes
4.5. Phenotypic Production of β-and δ-Cytotoxins
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of interest
References
- Vuong, C.; Otto, M. Staphylococcus epidermidisinfections. Microbes Infect. 2002, 4, 481–489. [Google Scholar] [CrossRef]
- Irlinger, F. Safety assessment of dairy microorganisms: Coagulase-negative staphylococci. Int. J. Food Microbiol. 2008, 126, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Podkowik, M.; Bystroń, J.; Bania, J. Genotypes, antibiotic resistance, and virulence factors of staphylococci from ready-to-eat food. Foodborne Pathog. Dis. 2012, 9, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Cunha, M.L.R.S.; Calsolari, R.A.O.; Araújo-Júnior, J.P. Detection of enterotoxin and toxic shock syndrome toxin 1 genes in Staphylococcus, with emphasis on coagulase-negative staphylococci. Microbiol. Immunol. 2007, 51, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Barretti, P.; Montelli, A.C.; Batalha, J.E.; Caramori, J.C.; Cunha, M.L.R.S. The role of virulence factors in the outcome of staphylococcal peritonitis in CAPD patients. BMC Infect. Dis. 2009, 9, 212. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.L.; Llewelyn, M.J. Superantigen-induced proliferation of human CD4+CD25− T cells is followed by a switch to a functional regulatory phenotype. J. Immunol. 2010, 185, 6591–6598. [Google Scholar] [CrossRef] [PubMed]
- Frase, J.D.; Proft, T. The bacterial superantigen and superantigen-like proteins. Immunol. Rev. 2008, 225, 226–243. [Google Scholar] [CrossRef] [PubMed]
- Dinges, M.M.; Orwin, P.M.; Schlievert, P.M. Exotoxins of Staphylococcus aureus. Clin. Microbiol. Rev. 2000, 13, 16–34. [Google Scholar] [CrossRef] [PubMed]
- Coelho, S.M.O.; Reinoso, E.; Pereira, I.A.; Soares, L.C.; Demo, M.; Bogni, C.; Souza, M.M.S. Virulence factors and antimicrobial resistence of Staphylococcus aureus isolated from bovine mastitis in Rio de Janeiro. Pesq. Vet. Bras. 2009, 29, 369–374. [Google Scholar] [CrossRef]
- Peng, H.L.; Novick, R.P.; Kreiswirth, B.; Kornblum, J.; Schlievert, P. Cloning, characterization, and sequencing of an accessory gene regulator (agr) in Staphylococcus aureus. J. Bacteriol. 1988, 170, 4365–4372. [Google Scholar] [PubMed]
- McKevitt, A.I.; Bjornson, G.L.; Mauracher, C.A.; Scheifele, D.W. Amino acid sequence of a deltalike toxin from Staphylococcus epidermidis. Infect. Immun. 1990, 58, 1473–1475. [Google Scholar] [PubMed]
- Even, S.; Leroy, S.; Charlier, C.; Zakour, N.B.; Chacornac, J.P.; Lebert, I.; Jamet, E.; Desmonts, M.H.; Coton, E.; Pochet, S.; et al. Low occurrence of safety hazards in coagulase negative staphylococci isolated from fermented food stuffs. Int. J. Food Microbiol. 2010, 139, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Okee, M.S.; Joloba, M.L.; Okello, M.; Najjuka, F.C.; Katabazi, F.A.; Bwanga, F.; Nanteza, A.; Kateete, D.P. Prevalence of virulence determinants in Staphylococcus epidermidis from ICU patients in Kampala, Uganda. J. Infect. Dev. Ctries. 2012, 6, 242–250. [Google Scholar] [PubMed]
- Schleifer, K.H.; Kloos, W.E. Isolation and characterization of staphylococci from human skin. Int. J. Syst. Bacteriol. 1975, 25, 50–61. [Google Scholar] [CrossRef]
- Rudd, K.E.; Humphery-Smith, I.; Wasinger, V.C.; Bairoch, A. Low molecular weight proteins: A challenge for post-genomic research. Electrophoresis 1998, 19, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Lavèn, M.; Klepsch, M.; de Gier, J.W.; Bitter, W.; van Ulsen, P.; Luirink, J. Role for Escherichia coli YidD in Membrane Protein Insertion. J. Bacteriol. 2011, 193, 5242–5251. [Google Scholar] [CrossRef] [PubMed]
- Davenport, K.W.; Daligault, H.E.; Minogue, T.D.; Bishop-Lilly, K.A.; Broomall, S.M.; Bruce, D.C.; Chain, P.S.; Coyne, S.R.; Frey, K.G.; Gibbons, H.S.; et al. Complete Genome Assembly of Staphylococcus epidermidis. Genome Announc. 2014, 2, e01059-14. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.F.; Miceli, M.H.; Anaissie, E.J. Mucosa or skin as source of coagulase-negative staphylococcal bacteraemia? Lancet Infect. Dis. 2004, 4, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Falcone, M.; Giannella, M.; Raponi, G.; Mancini, C.; Venditti, M. Teicoplanin use and emergence of Staphylococcus haemolyticus: Is there a link? Clin. Microbiol. Infect. 2006, 12, 96–97. [Google Scholar] [CrossRef] [PubMed]
- Mack, D.; Rohde, H.; Harris, L.G.; Davies, A.P.; Horstkotte, M.A.; Knobloch, J.K. Biofilm formation in medical device-related infection. Int. J. Artif. Organs 2006, 29, 343–359. [Google Scholar] [PubMed]
- Michelin, A.F.; Carlos, I.Z. Interaction of staphylococcal exterotoxins with the immmune system of the host. Rev. Ciênc. Farm 2003, 24, 83–95. [Google Scholar]
- De Freitas Guimarães, F.; Nóbrega, D.B.; Richini-Pereira, V.B.; Marson, P.M.; de Figueiredo Pantoja, J.C.; Langoni, H. Enterotoxin genes in coagulase-negative and coagulase-positive staphylococci isolated from bovine milk. J. Dairy Sci. 2013, 96, 2866–2872. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.B.; Antonelli, C.M.; Pereira Franchi, E.P.L.; Cunha, M.L.R.S. Detection of enterotoxin a in coagulase-negative staphylococci isolated from nutrition students. Arch. Clin. Microbiol. 2012, 3. Available online: http://imedpub.com/ojs/index.php/acmicrob/article/view/414 (accessed on 10 September 2014). [Google Scholar] [CrossRef]
- Crass, B.; Bergdoll, M.S. Involvement of coagulase-negative staphylococci in toxic shock syndrome. J. Clin. Microbiol. 1986, 23, 43–45. [Google Scholar] [PubMed]
- Cunha, M.L.R.S.; Rugolo, L.M.S.S.; Lopes, C.A.M. Study of virulence factors in coagulase-negative staphylococci isolated from newborns. Mem. Inst. Oswaldo Cruz 2006, 101, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, N.G.; Pereira, V.C.; Araújo-Júnior, J.P.; Cunha, M.L.R.S. Molecular detection of enterotoxins E, G, H and I in Staphylococcus aureus and coagulase-negative staphylococci isolated from clinical samples of newborns in Brazil. J. App. Microbiol. 2011, 111, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Jett, M.; Ionin, B.; Das, R.; Neill, R. The staphylococcal enterotoxins. In Molecular Medical Microbiology; Sussman, M., Ed.; Academic Press: San Diego, CA, USA, 2001; pp. 1089–1116. [Google Scholar]
- Lammler, C.; Akineden, O.; Annemuller, C.; Wolter, W.; Zschock, M. Molecular analysis of virulence factors of Staphylococcus aureus isolated from bovine sub-clinical mastitis. In Proceedings of the Symposium on Immunology of Ruminant Mammary Gland, Stresa, Italy, 11–14 June 2002; pp. 226–330.
- Akineden, O.; Annemuller, C.; Hassan, A.A.; Lammler, C.; Wolter, W.; Zschock, M. Toxin genes and other characteristics of Staphylococcus aureus isolates from milk of cows with mastitis. Clin. Diagn. Lab. Immunol. 2001, 8, 959–964. [Google Scholar] [CrossRef] [PubMed]
- Rosec, J.P.; Gigaud, O. Staphylococcal enterotoxin genes of classical and new types detected by PCR in France. J. Food Microbiol. 2002, 77, 61–70. [Google Scholar] [CrossRef]
- Madhusoodanan, J.; Seo, K.S.; Remortel, B.; Park, J.Y.; Hwang, S.Y.; Fox, L.K.; Park, Y.H.; Deobald, C.F.; Wang, D.; Liu, S.; et al. An enterotoxin-bearing pathogenicity island in Staphylococcus epidermidis. J. Bacteriol. 2011, 193, 1854–1862. [Google Scholar] [CrossRef] [PubMed]
- Jarraud, S.; Mougel, C.; Thioulouse, J.; Lina, G.; Meugnier, H.; Forey, F.; Nesme, X.; Etienne, J.; Vandenesch, F. Relationships between Staphylococcus aureus genetic background, virulence factors, agr groups (alleles), and human disease. Infect. Immun. 2002, 70, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Huseby, M.; Shi, K.; Brown, C.K.; Digre, J.; Mengistu, F.; Seo, K.S.; Bohach, G.A.; Schlievert, P.M.; Ohlendorf, D.H.; Earhart, C.A. Structure and biological activities of beta toxin from Staphylococcus aureus. J. Bacteriol. 2007, 189, 8719–8726. [Google Scholar] [CrossRef] [PubMed]
- Dziewanowska, K.; Edwards, V.M.; Deringer, J.R.; Bohach, G.A.; Guerra, D.J. Comparison of the β-toxins from Staphylococcus aureus and Staphylococcus intermedius. Arch. Biochem. Biophys. 1996, 335, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Bedidi-Madani, N.; Greenland, T.; Richard, Y. Exoprotein and slime production by coagulase-negative staphylococci isolated from goats’ milk. Vet. Microbiol. 1998, 59, 139–145. [Google Scholar] [CrossRef]
- Nataro, J.P.; Corcoran, L.; Zirin, S.; Swink, S.; Taichman, N.; Goin, J.; Harris, M.C. Prospective analysis of coagulase-negative staphylococcal infection in hospitalized infants. J. Pediatr. 1994, 125, 798–804. [Google Scholar] [CrossRef]
- Moraveji, Z.; Tabatabaei, M.; Aski, S.H.; Khoshbakht, R. Characterization of hemolysins of Staphylococcus strains isolated from human and bovine, southern Iran. Iran. J. Vet. Res. 2014, 15, 326–330. [Google Scholar]
- Novick, R.P. Autoinduction and signal transduction in the regulation of staphylococcal virulence. Mol. Microbiol. 2003, 48, 1429–1449. [Google Scholar] [CrossRef] [PubMed]
- Dordet-Frisoni, E.; Dorchies, G.; De, A.C.; Talon, R.; Leroy, S. Genomic diversity in Staphylococcus xylosus. Appl. Environ. Microbiol. 2007, 73, 7199–7209. [Google Scholar] [CrossRef] [PubMed]
- Wuster, A.; Babu, M.M. Conservation and evolutionary dynamics of the agr cell-to-cell communication system across firmicutes. J. Bacteriol. 2008, 190, 743–746. [Google Scholar] [CrossRef] [PubMed]
- Gemmel, C.G. Coagulase-negative staphylococci. J. Med. Microbial. 1986, 22, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Koneman, E.W.; Allen, S.D.; Janda, W.M.; Schreckenberger, P.C.; Winn, W.C., Jr. Color Atlas and Textbook of Diagnostic Microbiology, 5th ed.; Lippincott: Philadelphia, PA, USA, 1997. [Google Scholar]
- Cunha, M.L.R.S.; Sinzato, Y.K.; Silveira, L.V.A. Comparision of methods for identification of Coagulase-negative Staphylococci. Mem. Inst. Oswaldo Cruz 2004, 99, 855–860. [Google Scholar] [CrossRef]
- Couto, I.; Pereira, S.; Miragaia, M.; Sanches, I.S.; Lencastre, H. Identification of clinical staphylococcal isolates from humans by Internal Transcribed Spacer PCR. J. Clin. Microbiol. 2001, 39, 3099–3103. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.M.; Tyler, S.D.; Ewan, E.P.; Ashton, F.E.; Pollard, D.R.; Rozee, K.R. Detection of genes for enterotoxins, exfoliative toxins, and toxic shock syndrome toxin 1 in Staphylococcus aureus by the polymerase chain reaction. J. Clin. Microbiol. 1991, 29, 426–430. [Google Scholar] [PubMed]
- Marconi, C.; Cunha, M.L.R.S.; Araujo, J.P., Jr.; Rugolo, L.M.S.S. Standardization of the PCR technique for the detection of delta toxin in Staphylococcus spp. J. Venom. Anim. Toxins Incl. Trop. Dis. 2005, 11, 117–128. [Google Scholar] [CrossRef]
- Hébert, G.A.; Hancock, G.A. Synergistic hemolysis exhibited by species of Staphylococci. J. Clin. Microbiol. 1985, 22, 409–415. [Google Scholar] [PubMed]
- Freer, J.H.; Arbuthnott, J.P. Toxins of Staphylococcus aureus. Pharmacol. Ther. 1983, 19, 55–106. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinheiro, L.; Brito, C.I.; De Oliveira, A.; Martins, P.Y.F.; Pereira, V.C.; Da Cunha, M.D.L.R.d.S. Staphylococcus epidermidis and Staphylococcus haemolyticus: Molecular Detection of Cytotoxin and Enterotoxin Genes. Toxins 2015, 7, 3688-3699. https://doi.org/10.3390/toxins7093688
Pinheiro L, Brito CI, De Oliveira A, Martins PYF, Pereira VC, Da Cunha MDLRdS. Staphylococcus epidermidis and Staphylococcus haemolyticus: Molecular Detection of Cytotoxin and Enterotoxin Genes. Toxins. 2015; 7(9):3688-3699. https://doi.org/10.3390/toxins7093688
Chicago/Turabian StylePinheiro, Luiza, Carla Ivo Brito, Adilson De Oliveira, Patrícia Yoshida Faccioli Martins, Valéria Cataneli Pereira, and Maria De Lourdes Ribeiro de Souza Da Cunha. 2015. "Staphylococcus epidermidis and Staphylococcus haemolyticus: Molecular Detection of Cytotoxin and Enterotoxin Genes" Toxins 7, no. 9: 3688-3699. https://doi.org/10.3390/toxins7093688
APA StylePinheiro, L., Brito, C. I., De Oliveira, A., Martins, P. Y. F., Pereira, V. C., & Da Cunha, M. D. L. R. d. S. (2015). Staphylococcus epidermidis and Staphylococcus haemolyticus: Molecular Detection of Cytotoxin and Enterotoxin Genes. Toxins, 7(9), 3688-3699. https://doi.org/10.3390/toxins7093688








