Monoclonal Antibodies that Inhibit the Proteolytic Activity of Botulinum Neurotoxin Serotype/B
Abstract
:1. Introduction
2. Results and Discussion
2.1. Libraries Used for Monoclonal Antibody Generation
| Library | Immunizing Antigen | Vector | Library Size | mAbs |
|---|---|---|---|---|
| Human donor libraries | BoNT/A-E Toxoid | pYD2 | >107 | 1B10.1, 1B22, 2B23, 2B25.1, 4B19, B6.1 |
| Mouse B1 * | BoNT/B1 Okra | pYD4 | 4.0 × 107 | 16B3, 18A6, 18D10, 18E5, 18F2, 19A9, 19D22, 19G6 |
| Mouse B LC | BoNT/B1 LC | pYD4 | 5.0 × 107 | 31A5, 31E2, 31G2, 31H3, 34E8 |
2.2. Characterization of Monoclonal Antibodies
| scFv | mAb Origin | Epitope-Group | Affinity KD × 10−9 M (± SD) | Subserotype cross Reactivity | |||
|---|---|---|---|---|---|---|---|
| B LC | B1 | B2 | B3 | B4 | |||
| B6.1 | Human | I-3 | 0.24 (±0.14) | ++++ | +++ | +++ | +++ |
| 1B10.1 | Human | I-1 | 14.33 (±3.73) | ++ | ++ | + | +++ |
| 1B22 | Human | I-1 | 1.05 (±0.38) | ++++ | ++++ | ++++ | ++++ |
| 2B23 | Human | II | 0.60 (±0.22) | ++++ | ++++ | ++++ | ++++ |
| 2B25.1 | Human | I-1 | 0.24 (±0.13) | ++++ | ++++ | ++++ | ++++ |
| 4B19 | Human | II | 0.54 (±0.23) | ++++ | +++ | +++ | +++ |
| 16B3 | Mouse | I-1 | 0.84 (±0.22) | ++++ | ++++ | ++++ | ++++ |
| 18A6 | Mouse | I-3 | 4.21 (±0.36) | +++ | ++ | ++ | ++ |
| 18A7 | Mouse | I-2 | 13.5 (±3.02) | + | + | ++ | + |
| 18D10 | Mouse | I-2 | 7.53 (±1.72) | +++ | +++ | +++ | +++ |
| 18E5 | Mouse | I-1 | 1.00 (±0.17) | +++ | +++ | +++ | +++ |
| 18F2 | Mouse | III | 1.19 (±0.16) | + | +++ | ++ | - |
| 19A9 | Mouse | II | 3.16 (±0.17) | +++ | ++++ | +++ | ++++ |
| 19D22 | Mouse | I-1 | 9.31 (±1.98) | +++ | +++ | +++ | +++ |
| 31A5 | Mouse | I-1 | 1.75 (±0.13) | - | - | - | - |
| 31E2 | Mouse | III | 17.0 (±2.46) | - | - | - | - |
| 34E8 | Mouse | I-2 | 1.08 (±0.17) | - | - | - | - |
| 31G2 | Mouse | III | 3.34 (±0.37) | - | - | - | - |
| 31H3 | Mouse | I-2 | 1.39 (±0.34) | +++ | +++ | +++ | +++ |
2.3. Inhibition of BoNT LC Endopeptidase Activity
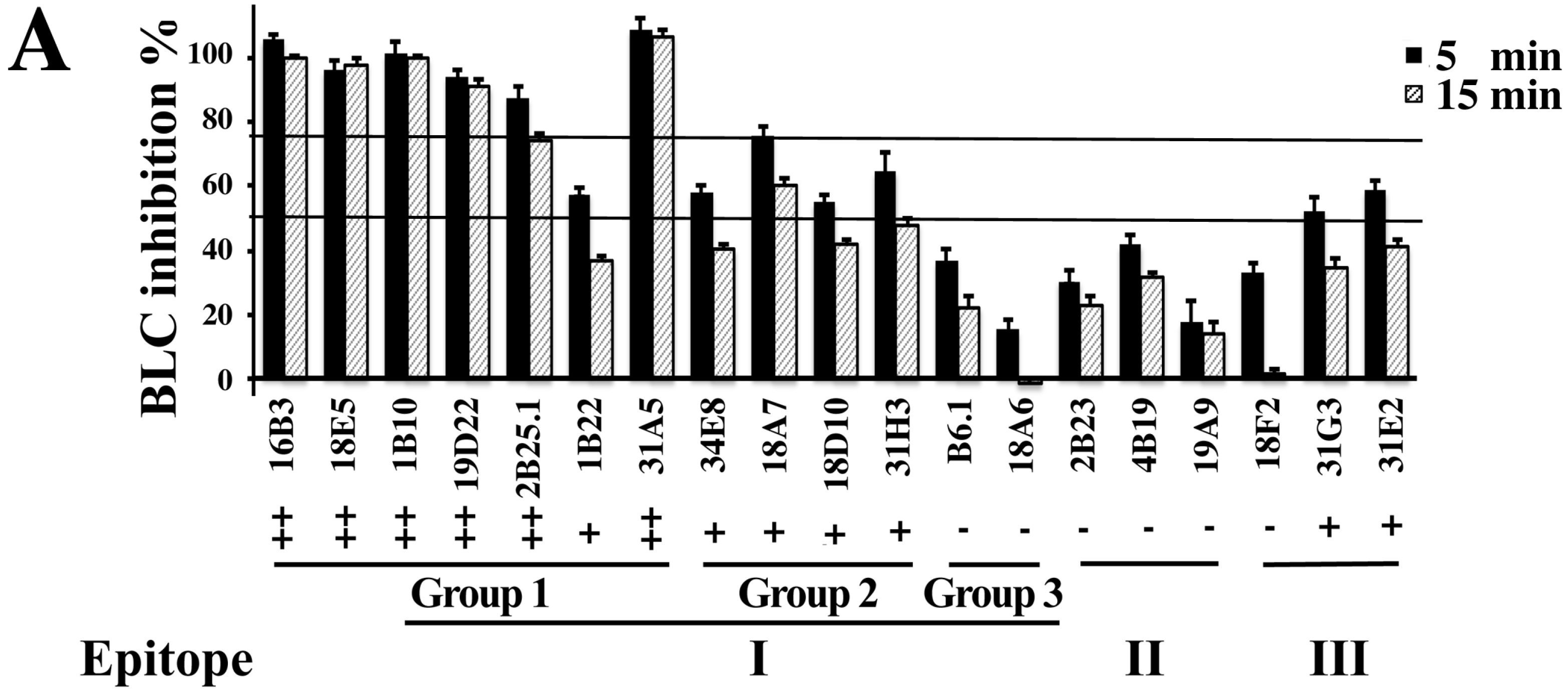

| mAb | Format | IC50 (×10−9 M) a | KD for BoNT/B1LC on Yeast (×10−9M) b | Epitope Group |
|---|---|---|---|---|
| 1B10.1 | IgG | 2.09 (2.04–2.14) | 118 b (69.0–197) | 1 |
| 16B3 | scFv | 5.21 (3.04–7.39) | 0.84 (0.40–1.28) | 1 |
| 18E5 | scFv | 7.01 (6.09–7.93) | 1.00 (0.66–1.34) | 1 |
| 19D22 | scFv | 5.47 (2.48–8.46) | 9.31 (5.35–13.3) | 1 |
| 18D10 | scFv | 59.0 (54.8–63.3) | 7.53 (4.09–11.0) | 2 |
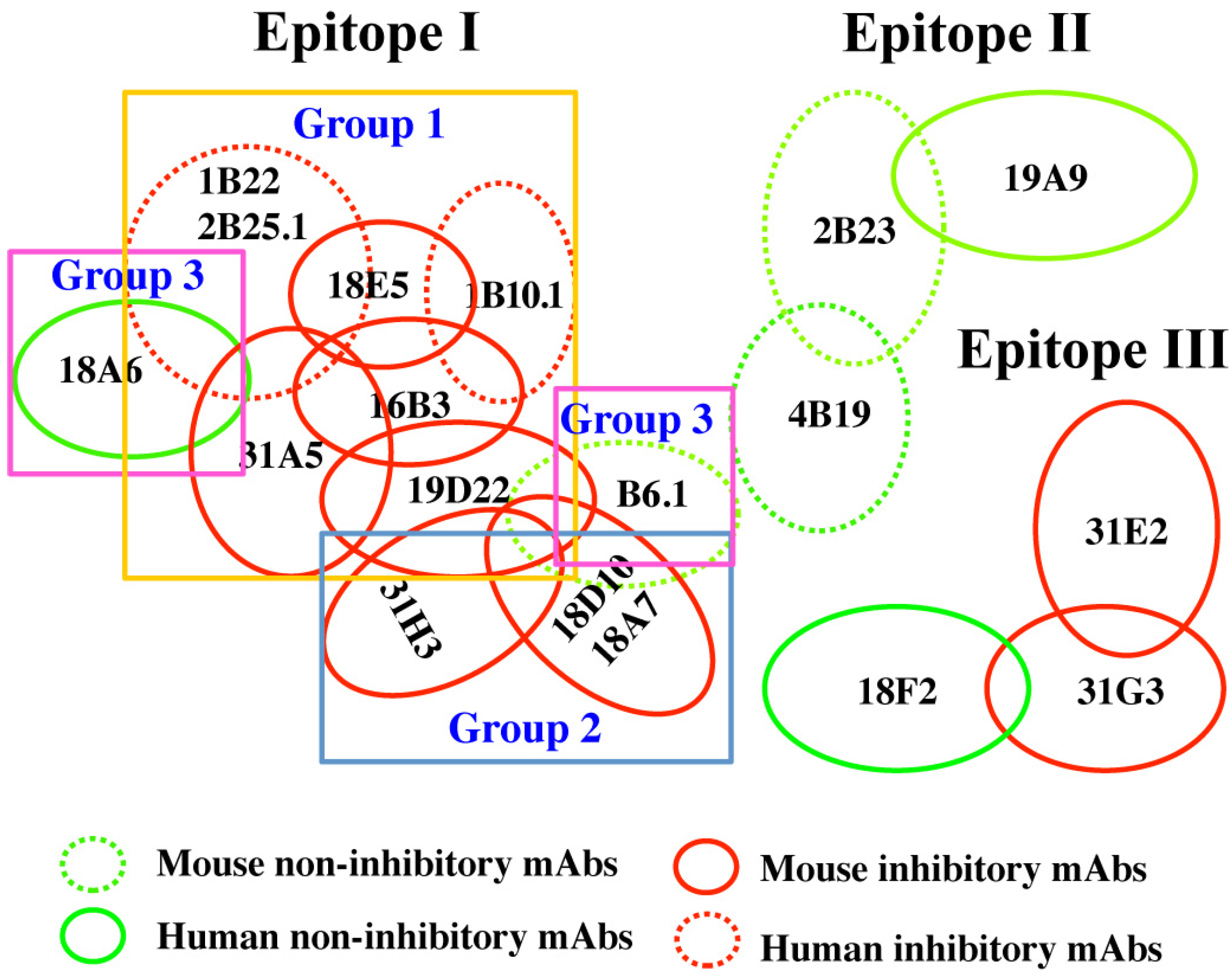
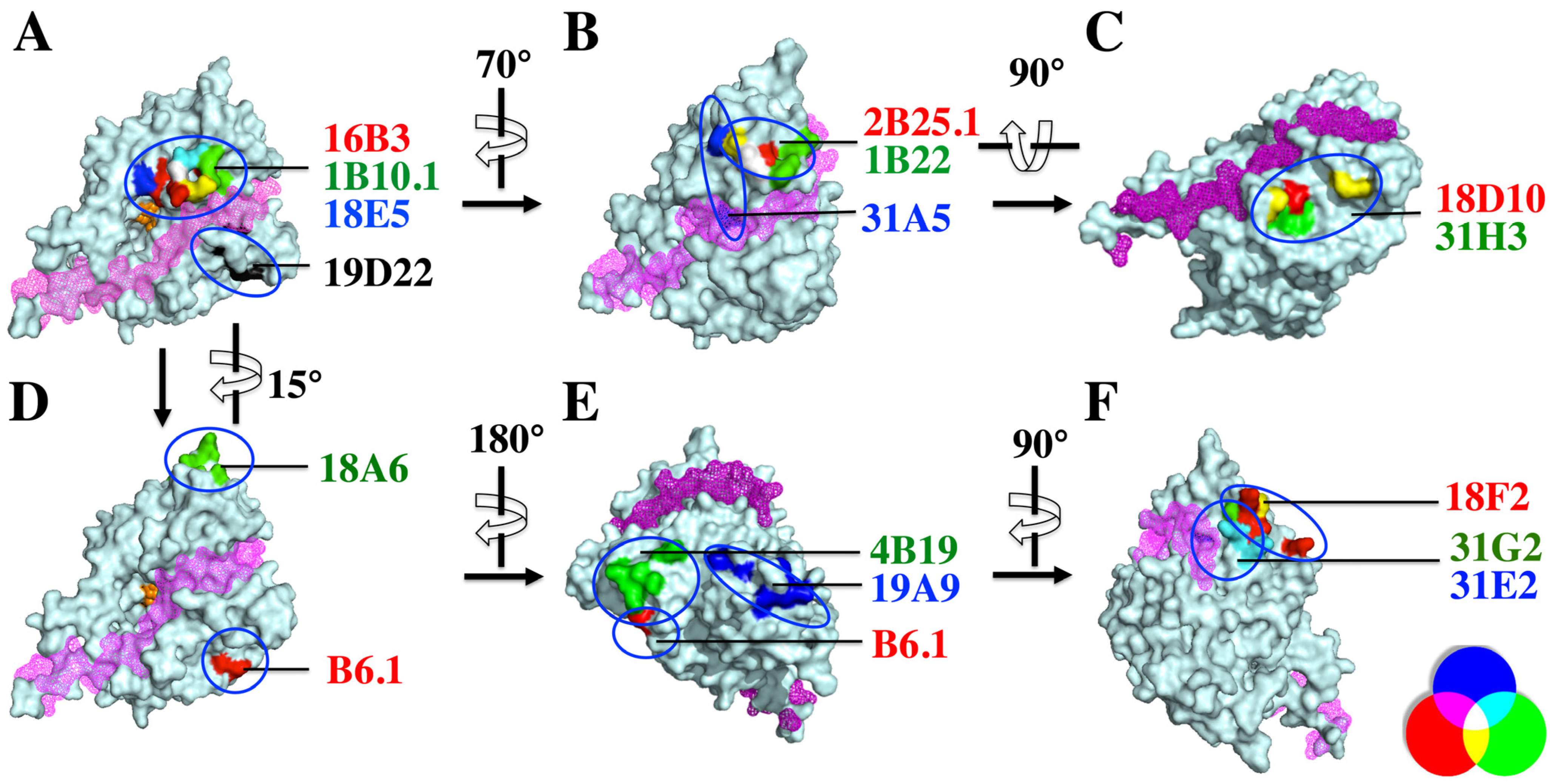
2.4. Defining the mAb Epitopes
| Epitope | mAb | Amino Acids Affecting mAb Binding |
|---|---|---|
| Epitope cluster I group 1 | 16B3 | G119, D120, Q177, N178, H179, R183, G185, I189 |
| 18E5 | Y117, G119, R121, R122, I132, A133, Q177, H179 | |
| 1B10.1 | R121, H179, D244 | |
| 2B25.1 | Y117, G119, E127, C308, I309 | |
| 1B22 | E127, P312, I309, I314, N315 | |
| 31A5 | G188, G185, P124, V135, H236, A133, I309 | |
| 19D22 | E22, A26, R31, P55 | |
| Epitope cluster I group 2 | 18A7 | R31, P55, T501 |
| 18D10 | R31, P55, T501 | |
| 31H3 | R31, R48, P55, F58 | |
| Epitope cluster I group3 | B6.1 | N81, K84 |
| 18A6 | N303, L306, V307, K320, D332, E334, K336, S338 | |
| Epitope cluster II | 4B19 | N6, N8, N10, D11, P12 |
| 19A9 | T392, I393, N396, S400, K402, D403, R409, Q411, V416 | |
| Epitope cluster III | 18F2 | K285, V341, E342K, D345, L347 |
| 31G2 | V341, S343, K346, K349 | |
| 31E2 | L238, F295, K326, G237, K346, K349 |
2.5. The Epitope of the Inhibitory mAb 1B10.1 Includes Active Site Residues
| Mutation of BoNT/B1 LC | Mean KD a (±SD) | Ratio Mutant/Wild Type | ΔΔG | |
|---|---|---|---|---|
| R121A | No binding - ≥5.0 | - | ≥5.0 | |
| R122A | 89.57 (±2.31) | 0.54 | −0.36 | |
| V123A | 106.25 (±4.45) | 0.64 | −0.26 | |
| P124A | 105.90 (±8.06) | 0.64 | −0.26 | |
| L125A | 105.95 (±1.20) | 0.64 | −0.26 | |
| I132A | 99.38 (±2.86) | 0.60 | −0.30 | |
| Q177A | 127.10 (±6.51) | 0.76 | −0.16 | |
| N178A | 110.55 (±9.12) | 0.66 | −0.24 | |
| H179A | 14621.50 (±3178) | 87.92 | 2.61 | |
| F180A | 90.55 (±15.63) | 0.54 | −0.35 | |
| R183A | 100.25 (±23.8) | 0.60 | −0.29 | |
| E184A | 89.29 (±52.3) | 0.54 | −0.36 | |
| F186A | 97.24 (±25.4) | 0.58 | −0.31 | |
| V243A | 99.78 (±18.3) | 0.60 | −0.30 | |
| D244A | 1349.37 (±523) | 8.11 | 1.22 | |
| D245A | 123.65 (±20.7) | 0.74 | −0.17 | |
| Q264A | 127.75 (±21.6) | 0.77 | −0.15 | |
| K290A | 154.20 (±74.8) | 0.93 | −0.04 | |
| Q293A | 114.88 (±22.1) | 0.69 | −0.22 | |
| N294A | 126.05 (±26.8) | 0.76 | −0.16 | |
| G297A | 127.70 (± 34.2) | 0.77 | −0.15 | |
| I298A | 97.71 (±11.7) | 0.59 | −0.31 | |
| R301A | 93.56 (±20.4) | 0.56 | −0.33 | |
| D512A | 119.30 (±50.9) | 0.72 | −0.19 | |
| N514A | 145.10 (±26.6) | 0.87 | −0.08 | |
| V515A | 115.80 (±20.1) | 0.70 | −0.21 | |
| D516A | 274.90 (±42.8) | 1.65 | 0.29 | |
| Wild Type | 166.30 (±84.7) | - | - |
3. Experimental Section
3.1. Ethics Section
3.2. Oligonucleotides for Library Construction
3.3. Strains, Media, Antibodies, and Toxin
3.4. Protein Expression and Purification
3.5. Mouse Immunization and Spleen Harvest
3.6. Yeast Displayed scFv Library Construction and Library Sorting
3.7. Measurement of Yeast Displayed scFv KD
3.8. Classification of mAbs Based on Overlap of Epitopes
3.9. MAb Inhibition of Synaptobrevin-2 Cleavage by BoNT/B LC
3.10. Determination of IC50 Values
3.11. Random Mutation Library Construction and Sorting
3.12. Site-Directed Mutagenesis
3.13. Fine Epitope Mapping of mAb 1B10.1
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AAALAC/I | Assessment and Accreditation of Laboratory Animal Care, International |
| BoNTs | Botulinum neurotoxins |
| mAb | Monoclonal antibody |
| HC | Heavy chain |
| LC | Light chain |
| HC | BoNT binding domain |
| HN | BoNT translocation domain |
| KD | Equilibrium binding constant |
| CHO | Chinese hamster ovary |
| IgG | Immunoglobulin G |
| Fab | Antigen binding fragment of immunoglobulin with variable domain and first constant domain |
| scFv | Single-chain variable fragment |
| FACS | Fluorescent activated cell sorting |
| sb2 | Synaptobrevin-2 |
| SNAP 25 | Synaptosomal-associated protein, 25kDa |
| Vh | Heavy chain variable region |
| Vk | Kappa chain variable region |
| Wt | wild type |
| IMAC | Immobilized metal affinity chromatography |
| PCR | polymerase chain reaction |
| PBS | Phosphate buffered saline |
| SD-CAA | Selective growth dextrose casamino acids media |
| SG-CAA | Selective growth galactose casamino acids media |
| SDS-PAGE | SDS-polyacrylamide gel electrophoresis |
| MFI | Mean fluorescence intensity |
| FRET | Fluorescence Resonance Energy Transfer |
| YsCsY | Yellow fluorescent protein (YFP)-synaptobevin-2-cyan fluorescent protein (CFP)-synaptobevin-2-YFP |
| PE | Phycoerythrin |
| IC50 | the half maximal inhibitory concentration |
| ΔΔG | Change of Gibbs free energy |
References
- Gill, M.D. Bacterial toxins: A table of lethal amounts. Microbiol. Rev. 1982, 46, 86–94. [Google Scholar] [PubMed]
- Arnon, S.S.; Schechter, R.; Inglesby, T.V.; Henderson, D.A.; Bartlett, J.G.; Ascher, M.S.; Eitzen, E.; Fine, A.D.; Hauer, J.; Layton, M.; et al. Botulinum toxin as a biological weapon: medical and public health management. JAMA 2001, 285, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Lacy, D.B.; Tepp, W.; Cohen, A.C.; DasGupta, B.R.; Stevens, R.C. Crystal structure of botulinum neurotoxin type A and implications for toxicity. Nat. Struct. Biol. 1998, 5, 898–902. [Google Scholar] [CrossRef] [PubMed]
- Chai, Q.; Arndt, J.W.; Dong, M.; Tepp, W.H.; Johnson, E.A.; Chapman, E.R.; Stevens, R.C. Structural basis of cell surface receptor recognition by botulinum neurotoxin B. Nature 2006, 444, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Dolly, J.O.; Black, J.; Williams, R.S.; Melling, J. Acceptors for botulinum neurotoxin reside on motor nerve terminals and mediate its internalization. Nature 1984, 307, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.; Yeh, F.; Tepp, W.H.; Dean, C.; Johnson, E.A.; Janz, R.; Chapman, E.R. SV2 is the protein receptor for botulinum neurotoxin A. Science 2006, 312, 592–596. [Google Scholar] [CrossRef] [PubMed]
- Montecucco, C. How do tetanus and botulinum toxins bind to neuronal membranes? Trends Biochem. Sci. 1986, 11, 315–317. [Google Scholar] [CrossRef]
- Rummel, A.; Mahrhold, S.; Bigalke, H.; Binz, T. The HCC-domain of botulinum neurotoxins A and B exhibits a singular ganglioside binding site displaying serotype specific carbohydrate interaction. Mol. Microbiol. 2004, 51, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Simpson, L.L. Kinetic studies on the interaction between botulinum toxin type A and the chloinergic neuromuscular junction. J. Pharmacol. Expt. Ther. 1980, 212, 16–21. [Google Scholar]
- Blaustein, R.O.; Germann, W.J.; Finkelstein, A.; DasGupta, B.R. The N-terminal half of the heavy chain of botulinum type A neurotoxin forms channels in planar phospholipid bilayers. FEBS Lett. 1987, 226, 115–120. [Google Scholar] [CrossRef]
- Fischer, A.; Montal, M. Single molecule detection of intermediates during botulinum neurotoxin translocation across membranes. Proc. Natl. Acad. Sci. USA 2007, 104, 10447–10452. [Google Scholar] [CrossRef] [PubMed]
- Schiavo, G.; Benfenati, F.; Poulain, B.; Rossetto, O.; de Laureto, P.P.; DasGupta, B.R.; Montecucco, C. Tetanus and botulinum-B neurotoxins block neurotransmitter release by proteolytic cleavage of synaptobrevin. Nature 1992, 359, 832–835. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Karalewitz, A.P.; Barbieri, J.T. Insights into the different catalytic activities of Clostridium neurotoxins. Biochemistry 2012, 51, 3941–3947. [Google Scholar] [CrossRef] [PubMed]
- Breidenbach, M.A.; Brunger, A.T. Substrate recognition strategy for botulinum neurotoxin serotype A. Nature 2004, 432, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Arnon, S.S.; Schechter, R.; Maslanka, S.E.; Jewell, N.P.; Hatheway, C.L. Human botulism immune globulin for the treatment of infant botulism. N. Engl. J. Med. 2006, 354, 462–471. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Investigational heptavalent botulinum antitoxin (HBAT) to replace licensed botulinum antitoxin AB and investigational botulinum antitoxin E. MMWR Morb. Mortal Wkly. Rep. 2010, 59, 299. [Google Scholar]
- Nowakowski, A.; Wang, C.; Powers, D.B.; Amersdorfer, P.; Smith, T.J.; Montgomery, V.A.; Sheridan, R.; Blake, R.; Smith, L.A.; Marks, J.D. Potent neutralization of botulinum neurotoxin by recombinant oligoclonal antibody. Proc. Natl. Acad. Sci. USA 2002, 99, 11346–11350. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Rodriguez, C.; Levy, R.; Arndt, J.W.; Forsyth, C.M.; Razai, A.; Lou, J.; Geren, I.; Stevens, R.C.; Marks, J.D. Molecular evolution of antibody cross-reactivity for two subtypes of type A botulinum neurotoxin. Nat. Biotechnol. 2007, 25, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Geren, I.; Garcia-Rodriguez, C.; Forsyth, C.M.; Wen, W.; Knopp, K.; Brown, J.; Smith, T.; Smith, L.A.; Marks, J.D. Affinity maturation of human botulinum neurotoxin antibodies by light chain shuffling via yeast mating. Protein Eng. Des. Sel. 2010, 23, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Garcia-Rodriguez, C.; Manzanarez, G.; Silberg, M.A.; Conrad, F.; Bettencourt, J.; Pan, X.; Breece, T.; To, R.; Li, M.; et al. Engineered domain-based assays to identify individual antibodies in oligoclonal combinations targeting the same protein. Anal. Biochem. 2012, 430, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Meng, Q.; Li, M.; Silberg, M.A.; Conrad, F.; Bettencourt, J.; To, R.; Huang, C.; Ma, J.; Meyer, K.; Shimizu, R.; et al. Domain-based assays of individual antibody concentrations in an oligoclonal combination targeting a single protein. Anal. Biochem. 2012, 421, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Eubanks, L.M.; Hixon, M.S.; Jin, W.; Hong, S.; Clancy, C.M.; Tepp, W.H.; Baldwin, M.R.; Malizio, C.J.; Goodnough, M.C.; Barbieri, J.T. An in vitro and in vivo disconnect uncovered through high-throughput identification of botulinum neurotoxin A antagonists. Proc. Natl. Acad. Sci. USA 2007, 104, 2602–2607. [Google Scholar] [CrossRef] [PubMed]
- Lebeda, F.J.; Cer, R.Z.; Mudunuri, U.; Stephens, R.; Singh, B.R.; Adler, M. The zinc-dependent protease activity of the botulinum neurotoxins. Toxins 2010, 2, 978–997. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.-P.; Davis, J.; Wang, S.; Park, J.G.; Nambiar, M.P.; Schmidt, J.J.; Millard, C.B. Small molecules showing significant protection of mice against botulinum neurotoxin serotype A. PLoS ONE 2010, 5, e10129. [Google Scholar] [CrossRef] [PubMed]
- Kiris, E.; Burnett, J.C.; Kane, C.D.; Bavari, S. Recent advances in botulinum neurotoxin inhibitor development. Curr. Top Med. Chem. 2014, 14, 2044–2061. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.A.; Ho, M. Cargo-delivery platforms for targeted delivery of inhibitor cargos against botulism. Curr. Top Med. Chem. 2014, 14, 2081–2093. [Google Scholar] [CrossRef] [PubMed]
- Edupuganti, O.P.; Ovsepian, S.V.; Wang, J.; Zurawski, T.H.; Schmidt, J.J.; Smith, L.; Lawrence, G.W.; Dolly, J.O. Targeted delivery into motor nerve terminals of inhibitors for SNARE-cleaving proteases via liposomes coupled to an atoxic botulinum neurotoxin. FEBS J. 2012, 279, 2555–2567. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Thompson, A.A.; Fan, Y.; Lou, J.; Conrad, F.; Ho, M.; Pires-Alves, M.; Wilson, B.A.; Stevens, R.C.; Marks, J.D. A single-domain llama antibody potently inhibits the enzymatic activity of botulinum neurotoxin by binding to the non-catalytic alpha-exosite binding region. J. Mol. Biol. 2010, 397, 1106–1118. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Geren, I.; Dong, J.; Lou, J.; Wen, W.; Conrad, F.; Smith, T.J.; Smith, L.A.; Ho, M.; Pires-Alves, M.; Wilson, B.A.; Marks, J.D. Monoclonal antibodies targeting the alpha-exosite of botulinum neurotoxin serotype/A inhibit catalytic activity. PLoS ONE 2015, 10, e0135306. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.; Forsyth, C.M.; LaPorte, S.L.; Geren, I.N.; Smith, L.A.; Marks, J.D. Fine and domain-level epitope mapping of botulinum neurotoxin type A neutralizing antibodies by yeast surface display. J. Mol. Biol. 2007, 365, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, S.; Eswaramoorthy, S. Structural analysis of the catalytic and binding sites of Clostridium botulinum neurotoxin B. Nat. Struct. Mol. Biol. 2000, 7, 693–699. [Google Scholar] [CrossRef]
- Brunger, A.T.; Breidenbach, M.A.; Jin, R.; Fischer, A.; Santos, J.S.; Montal, M. Botulinum neurotoxin heavy chain belt as an intramolecular chaperone for the light chain. PLoS Pathog. 2007, 3, e113. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Sidhu, S.S.; Wells, J.A. Two-state selection of conformation-specific antibodies. Proc. Natl. Acad. Sci. USA 2009, 106, 3071–3076. [Google Scholar] [CrossRef] [PubMed]
- Eswaramoorthy, S.; Kumaran, D.; Keller, J.; Swaminathan, S. Role of metals in the biological activity of Clostridium botulinum neurotoxins. Biochemistry 2004, 43, 2209–2216. [Google Scholar] [CrossRef] [PubMed]
- Foran, P.; Shone, C.C.; Dolly, J.O. Differences in the protease activities of tetanus and botulinum B toxins revealed by the cleavage of vesicle-associated membrane protein and various sized fragments. Biochemistry 1994, 33, 15365–15374. [Google Scholar] [CrossRef] [PubMed]
- Olson, M.A.; Armendinger, T.L. Free-energy contributions to complex formation between botulinum neurotoxin type B and synaptobrevin fragment. Protein Eng. 2002, 15, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Marks, J.D.; Hoogenboom, H.R.; Bonnert, T.P.; McCafferty, J.; Griffiths, A.D.; Winter, G. By-passing immunization. Human antibodies from V-gene libraries displayed on phage. J. Mol. Biol. 1991, 222, 581–597. [Google Scholar] [CrossRef]
- Schier, R.; Bye, J.M.; Apell, G.; McCall, A.; Adams, G.P.; Malmqvist, M.; Weiner, L.M.; Marks, J.D. Isolation of high affinity monomeric human anti-c-erbB-2 single chain Fv using affinity driven selection. J. Mol. Biol. 1996, 255, 28–43. [Google Scholar] [CrossRef] [PubMed]
- Pires-Alves, M.; Ho, M.; Aberle, K.K.; Janda, K.D.; Wilson, B.A. Tandem fluorescent proteins as enhanced FRET-based substrates for botulinum neurotoxin activity. Toxicon 2009, 53, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Goh, C.H.; Brothers, M.C.; Wang, S.; Young, R.L.; Ou, Y.; Lui, J.N.; Kalafatis, M.; Lan, X.; Wolf, A.E.; et al. Glycine insertion at protease cleavage site of SNAP25 resists cleavage but enhances affinity for botulinum neurotoxin serotype A. Protein Sci. 2012, 21, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Razai, A.; Garcia-Rodriguez, C.; Lou, J.; Geren, I.N.; Forsyth, C.M.; Robles, Y.; Tsai, R.; Smith, T.J.; Smith, L.A.; Siegel, R.W.; et al. Molecular evolution of antibody affinity for sensitive detection of botulinum neurotoxin type A. J. Mol. Biol. 2005, 351, 158–169. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, Y.; Dong, J.; Lou, J.; Wen, W.; Conrad, F.; Geren, I.N.; Garcia-Rodriguez, C.; Smith, T.J.; Smith, L.A.; Ho, M.; et al. Monoclonal Antibodies that Inhibit the Proteolytic Activity of Botulinum Neurotoxin Serotype/B. Toxins 2015, 7, 3405-3423. https://doi.org/10.3390/toxins7093405
Fan Y, Dong J, Lou J, Wen W, Conrad F, Geren IN, Garcia-Rodriguez C, Smith TJ, Smith LA, Ho M, et al. Monoclonal Antibodies that Inhibit the Proteolytic Activity of Botulinum Neurotoxin Serotype/B. Toxins. 2015; 7(9):3405-3423. https://doi.org/10.3390/toxins7093405
Chicago/Turabian StyleFan, Yongfeng, Jianbo Dong, Jianlong Lou, Weihua Wen, Fraser Conrad, Isin N. Geren, Consuelo Garcia-Rodriguez, Theresa J. Smith, Leonard A. Smith, Mengfei Ho, and et al. 2015. "Monoclonal Antibodies that Inhibit the Proteolytic Activity of Botulinum Neurotoxin Serotype/B" Toxins 7, no. 9: 3405-3423. https://doi.org/10.3390/toxins7093405
APA StyleFan, Y., Dong, J., Lou, J., Wen, W., Conrad, F., Geren, I. N., Garcia-Rodriguez, C., Smith, T. J., Smith, L. A., Ho, M., Pires-Alves, M., Wilson, B. A., & Marks, J. D. (2015). Monoclonal Antibodies that Inhibit the Proteolytic Activity of Botulinum Neurotoxin Serotype/B. Toxins, 7(9), 3405-3423. https://doi.org/10.3390/toxins7093405







