The Effects of Bee Venom Acupuncture on the Central Nervous System and Muscle in an Animal hSOD1G93A Mutant
Abstract
:1. Introduction
2. Results
2.1. BV Treatment Improves Motor Functions in Symptomatic hSOD1G93A Mice
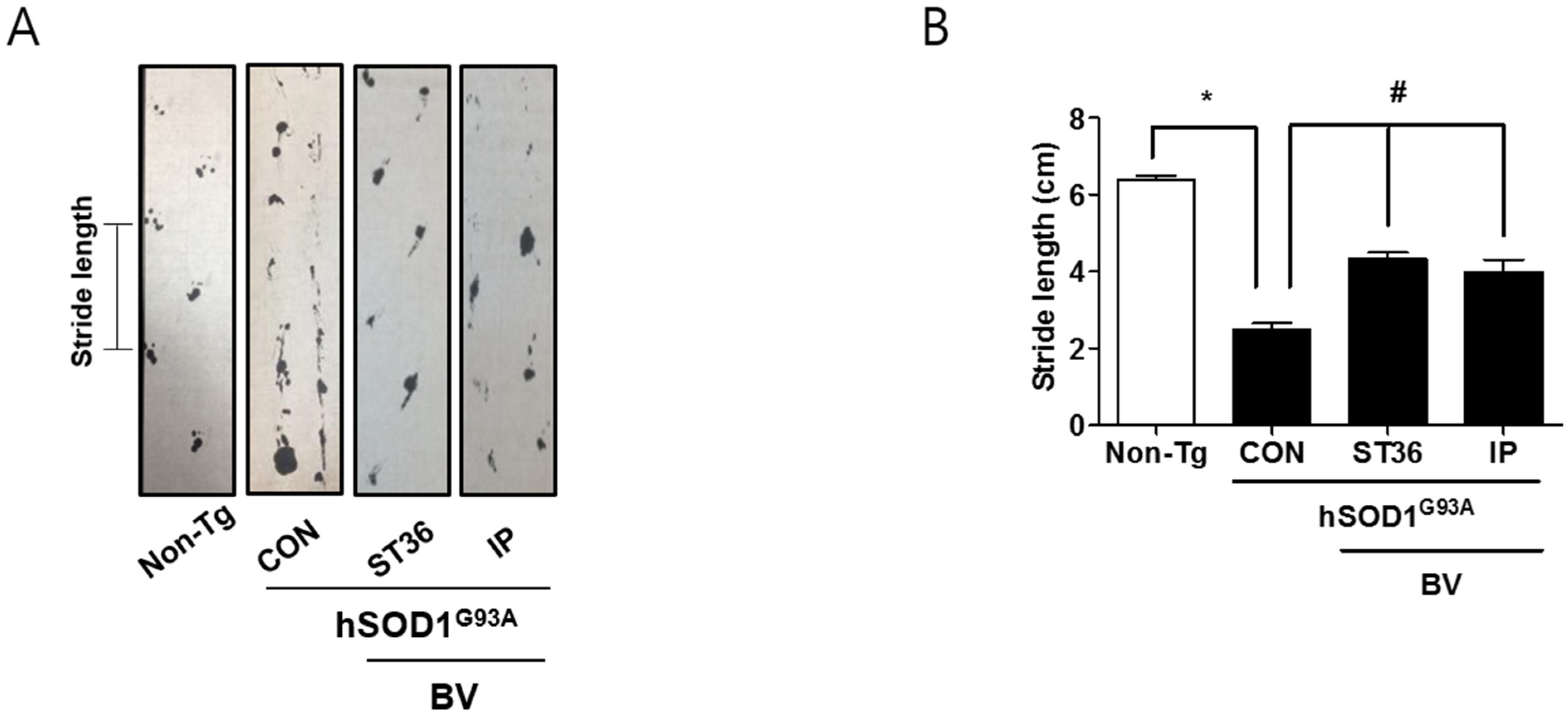
2.2. BV Treatment Reduces Motor Neuron Cell Death in Symptomatic hSOD1G93A Mice
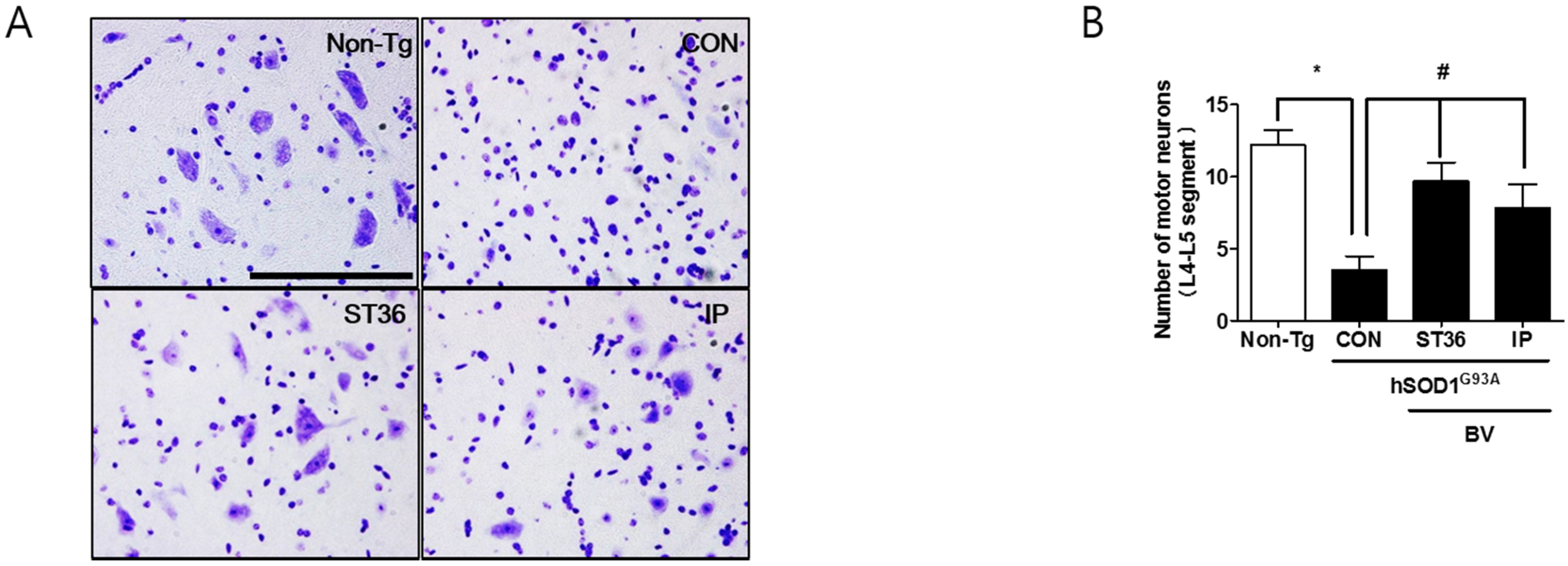
2.3. BVA at ST36 Augments Anti-Neuroinflammation
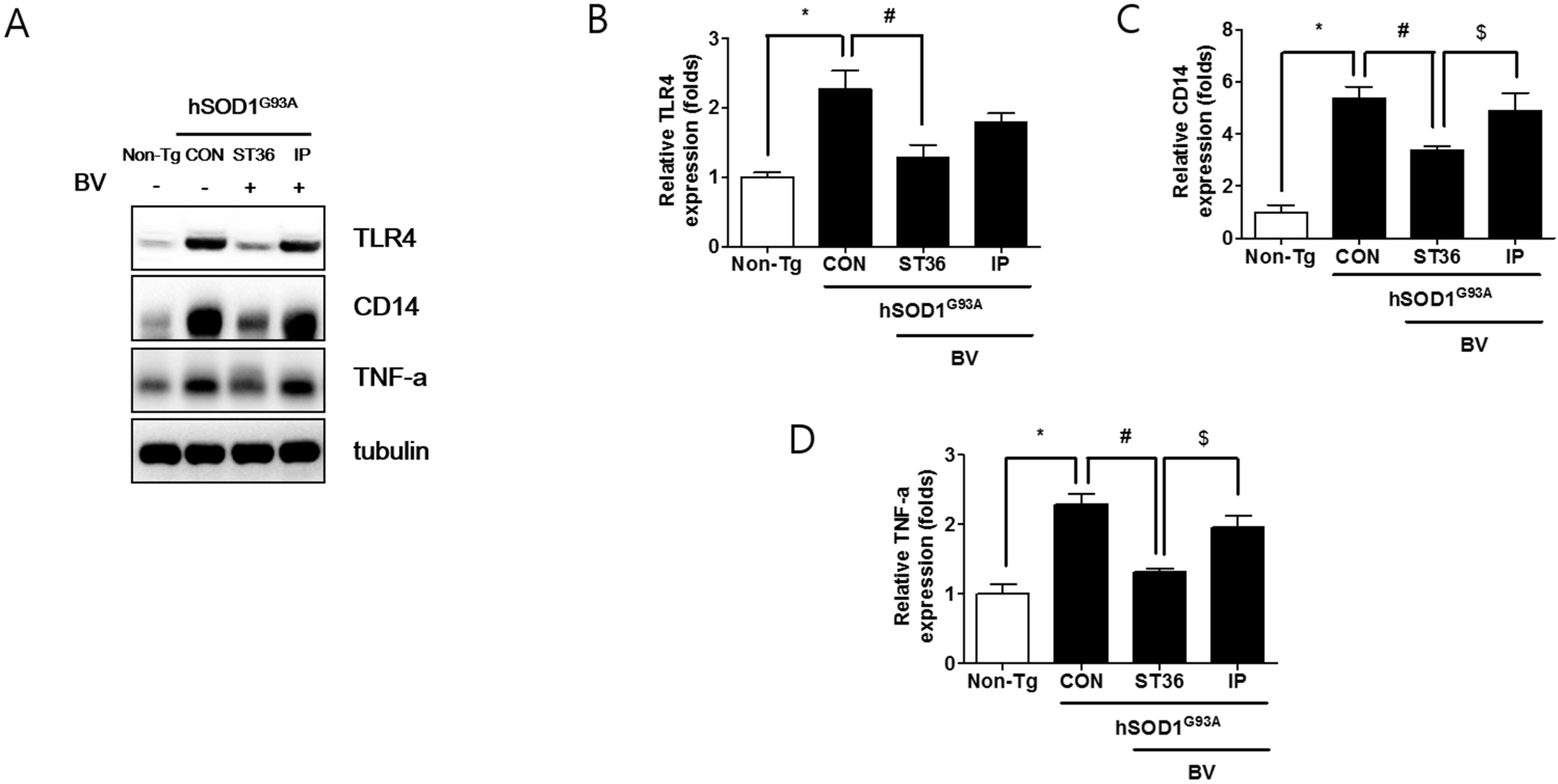
2.4. BVA at ST36 Prevents Muscle Atrophy in Symptomatic hSOD1G93A Transgenic Mice

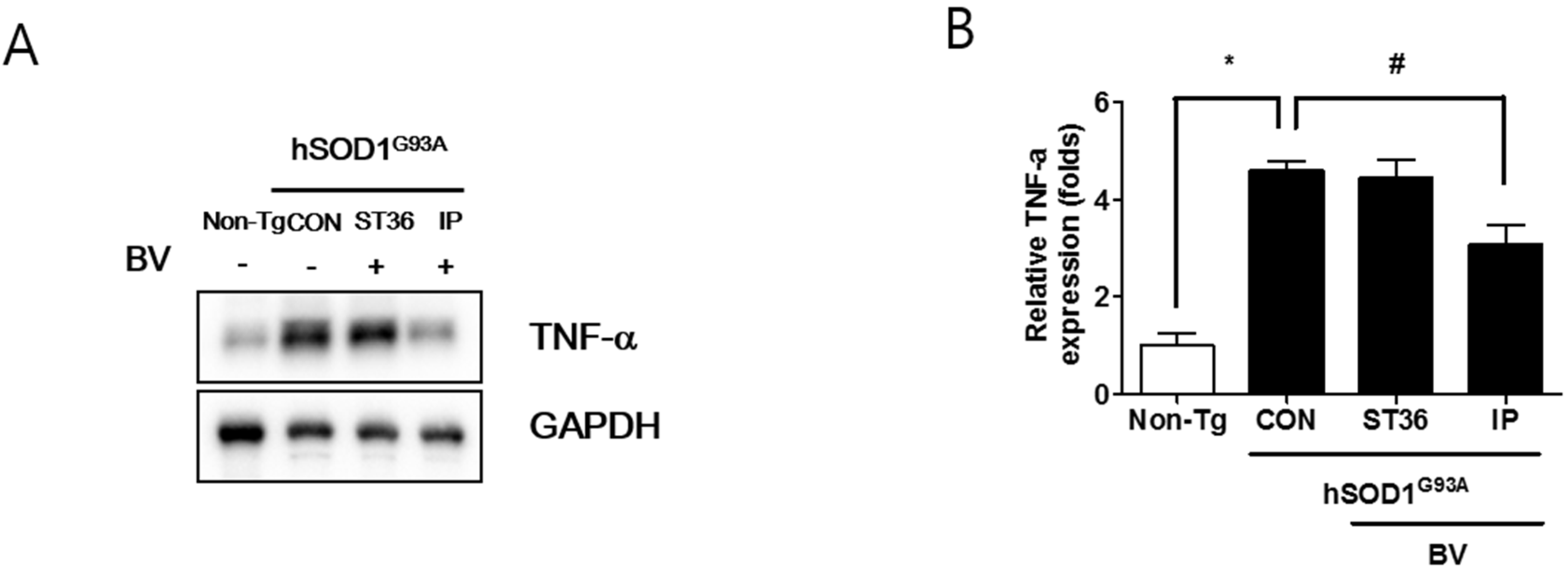
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Bee Venom Treatment
4.3. Footprint Test
4.4. Western Blot Analysis
4.5. Tissue Preparation
4.6. Nissl Staining
4.7. H&E Staining
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rosen, D.R.; Siddique, T.; Patterson, D.; Figlewicz, D.A.; Sapp, P.; Hentati, A.; Donaldson, D.; Goto, J.; O’Regan, J.P.; Deng, H.X.; et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 1993, 362, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Synofzik, M.; Fernandez-Santiago, R.; Maetzler, W.; Schols, L.; Andersen, P.M. The human G93A SOD1 phenotype closely resembles sporadic amyotrophic lateral sclerosis. J. Neurol. Neurosurg. Psychiatr. 2010, 81, 764–767. [Google Scholar] [CrossRef] [PubMed]
- Moller, T. Neuroinflammation in Huntington’s disease. J. Neural Transm. 2010, 117, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Weydt, P.; Moller, T. Neuroinflammation in the pathogenesis of amyotrophic lateral sclerosis. Neuroreport 2005, 16, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Rojas, F.; Cortes, N.; Abarzua, S.; Dyrda, A.; van Zundert, B. Astrocytes expressing mutant SOD1 and TDP43 trigger motoneuron death that is mediated via sodium channels and nitroxidative stress. Front. Cell. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Bowerman, M.; Vincent, T.; Scamps, F.; Perrin, F.E.; Camu, W.; Raoul, C. Neuroimmunity dynamics and the development of therapeutic strategies for amyotrophic lateral sclerosis. Front. Cell Neurosci. 2013, 7, 214. [Google Scholar] [CrossRef] [PubMed]
- McGeer, P.L.; McGeer, E.G. Inflammatory processes in amyotrophic lateral sclerosis. Muscle Nerve 2002, 26, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Meissner, F.; Molawi, K.; Zychlinsky, A. Mutant superoxide dismutase 1-induced IL-1beta accelerates ALS pathogenesis. Proc. Natl. Acad. Sci. USA 2010, 107, 13046–13050. [Google Scholar] [CrossRef] [PubMed]
- Sekizawa, T.; Openshaw, H.; Ohbo, K.; Sugamura, K.; Itoyama, Y.; Niland, J.C. Cerebrospinal fluid interleukin 6 in amyotrophic lateral sclerosis: Immunological parameter and comparison with inflammatory and non-inflammatory central nervous system diseases. J. Neurol. Sci. 1998, 154, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Fotinos, A.; Mao, L.L.; Atassi, N.; Zhou, E.W.; Ahmad, S.; Guan, Y.; Berry, J.D.; Cudkowicz, M.E.; Wang, X. Neuroprotective agents target molecular mechanisms of disease in ALS. Drug Discov. Today 2015, 20, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.B.; Lee, H.J.; Han, H.J.; Mar, W.C.; Kang, S.K.; Yoon, O.B.; Beitz, A.J.; Lee, J.H. The water-soluble fraction of bee venom produces antinociceptive and anti-inflammatory effects on rheumatoid arthritis in rats. Life Sci. 2002, 71, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Somerfield, S.D.; Brandwein, S. Bee venom and adjuvant arthritis. J. Rheumatol. 1988, 15, 1878. [Google Scholar] [PubMed]
- Kim, J.I.; Yang, E.J.; Lee, M.S.; Kim, Y.S.; Huh, Y.; Cho, I.H.; Kang, S.; Koh, H.K. Bee venom reduces neuroinflammation in the MPTP-induced model of Parkinson’s disease. Int. J. Neurosci. 2011, 121, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.J.; Jiang, J.H.; Lee, S.M.; Yang, S.C.; Hwang, H.S.; Lee, M.S.; Choi, S.M. Bee venom attenuates neuroinflammatory events and extends survival in amyotrophic lateral sclerosis models. J. Neuroinflamm. 2010, 7. [Google Scholar] [CrossRef]
- Korean Pharmacopuncture Institute. Pharmacopuncturology, 2nd ed.; Elsevier Korea: Seoul, Korea, 2011. [Google Scholar]
- Strudwick, M.W.; Hinks, R.C.; Choy, S.T. Point injection as an alternative acupuncture technique—An exploratory study of responses in healthy subjects. Acupunct. Med. 2007, 25, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Wilms, H.; Zecca, L.; Rosenstiel, P.; Sievers, J.; Deuschl, G.; Lucius, R. Inflammation in Parkinson’s diseases and other neurodegenerative diseases: Cause and therapeutic implications. Curr. Pharm Des. 2007, 13, 1925–1928. [Google Scholar] [CrossRef] [PubMed]
- Yang, E.J.; Jiang, J.H.; Lee, S.M.; Hwang, H.S.; Lee, M.S.; Choi, S.M. Electroacupuncture reduces neuroinflammatory responses in symptomatic amyotrophic lateral sclerosis model. J. Neuroimmunol. 2010, 223, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.Y.; Dai, Y.; Wang, S.L.; Yamanaka, H.; Kobayashi, K.; Obata, K.; Chen, J.; Noguchi, K. Differential activation of p38 and extracellular signal-regulated kinase in spinal cord in a model of bee venom-induced inflammation and hyperalgesia. Mol. Pain. 2008, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, S. The effects of sa-am acupuncture treatment on respiratory physiology parameters in amyotrophic lateral sclerosis patients: A pilot study. Evid. Based Complement. Alternat. Med. 2013, 2013. [Google Scholar] [CrossRef]
- Liang, S.; Christner, D.; Du Laux, S.; Laurent, D. Significant neurological improvement in two patients with amyotrophic lateral sclerosis after 4 weeks of treatment with acupuncture injection point therapy using enercel. J. Acupunct. Meridian Stud. 2011, 4, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Chen, X.; Bao, J.; Bai, Y.; Lu, H.; Wang, Q.; Liu, Y.; Yuan, C.; Li, W.; Liu, Z.; et al. The use of integrative therapies in patients with amyotrophic lateral sclerosis in Shanghai, China. Evid. Based Complement. Alternat. Med. 2013, 2013, 613596. [Google Scholar] [PubMed]
- Wasner, M.; Klier, H.; Borasio, G.D. The use of alternative medicine by patients with amyotrophic lateral sclerosis. J. Neurol. Sci. 2001, 191, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Lariviere, W.R.; Melzack, R. The bee venom test: A new tonic-pain test. Pain 1996, 66, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Kwon, Y.B.; Lee, J.D.; Lee, H.J.; Han, H.J.; Mar, W.C.; Kang, S.K.; Beitz, A.J.; Lee, J.H. Bee venom injection into an acupuncture point reduces arthritis associated edema and nociceptive responses. Pain 2001, 90, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Kim, H.W.; Li, J.; Kwon, Y.B. Effect of bee venom acupuncture on methamphetamine-induced hyperactivity, hyperthermia and Fos expression in mice. Brain Res. Bull. 2011, 84, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Krasnianski, A.; Deschauer, M.; Neudecker, S.; Gellerich, F.N.; Muller, T.; Schoser, B.G.; Krasnianski, M.; Zierz, S. Mitochondrial changes in skeletal muscle in amyotrophic lateral sclerosis and other neurogenic atrophies. Brain 2005, 128, 1870–1876. [Google Scholar] [CrossRef] [PubMed]
- Echaniz-Laguna, A.; Zoll, J.; Ponsot, E.; N’Guessan, B.; Tranchant, C.; Loeffler, J.P.; Lampert, E. Muscular mitochondrial function in amyotrophic lateral sclerosis is progressively altered as the disease develops: A temporal study in man. Exp. Neurol. 2006, 198, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Manzano, R.; Toivonen, J.M.; Olivan, S.; Calvo, A.C.; Moreno-Igoa, M.; Munoz, M.J.; Zaragoza, P.; Garcia-Redondo, A.; Osta, R. Altered expression of myogenic regulatory factors in the mouse model of amyotrophic lateral sclerosis. Neurodegener. Dis. 2011, 8, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Kaspar, B.K.; Llado, J.; Sherkat, N.; Rothstein, J.D.; Gage, F.H. Retrograde viral delivery of IGF-1 prolongs survival in a mouse ALS model. Science 2003, 301, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.S.; Jeong, H.S.; Park, H.J.; Baik, Y.; Yoon, M.H.; Choi, C.B.; Koh, H.G. A proposed transpositional acupoint system in a mouse and rat model. Res. Vet. Sci. 2008, 84, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Filali, M.; Lalonde, R.; Rivest, S. Sensorimotor and cognitive functions in a SOD1(G37R) transgenic mouse model of amyotrophic lateral sclerosis. Behav. Brain Res. 2011, 225, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, R.; Olivan, S.; Osta, R.; Navarro, X. Evolution of gait abnormalities in SOD1(G93A) transgenic mice. Brain Res. 2011, 1406, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Cai, M.; Choi, S.M.; Song, B.K.; Son, I.; Kim, S.; Yang, E.J. Scolopendra subspinipes mutilans attenuates neuroinflammation in symptomatic hSOD1(G93A) mice. J. Neuroinflamm. 2013, 10. [Google Scholar] [CrossRef]
- Irugalbandara, Z.E. Simplified differentiation of Nissl granules stained by toluidine blue in paraffin sections. Stain Technol. 1960, 35, 47–48. [Google Scholar] [PubMed]
- Cai, M.; Shin, B.Y.; Kim, D.H.; Kim, J.M.; Park, S.J.; Park, C.S.; Won, H.; Hong, N.D.; Kang, D.H.; Yutaka, Y.; et al. Neuroprotective effects of a traditional herbal prescription on transient cerebral global ischemia in gerbils. J. Ethnopharmacol. 2011, 138, 723–730. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, M.; Choi, S.-M.; Yang, E.J. The Effects of Bee Venom Acupuncture on the Central Nervous System and Muscle in an Animal hSOD1G93A Mutant. Toxins 2015, 7, 846-858. https://doi.org/10.3390/toxins7030846
Cai M, Choi S-M, Yang EJ. The Effects of Bee Venom Acupuncture on the Central Nervous System and Muscle in an Animal hSOD1G93A Mutant. Toxins. 2015; 7(3):846-858. https://doi.org/10.3390/toxins7030846
Chicago/Turabian StyleCai, MuDan, Sun-Mi Choi, and Eun Jin Yang. 2015. "The Effects of Bee Venom Acupuncture on the Central Nervous System and Muscle in an Animal hSOD1G93A Mutant" Toxins 7, no. 3: 846-858. https://doi.org/10.3390/toxins7030846
APA StyleCai, M., Choi, S.-M., & Yang, E. J. (2015). The Effects of Bee Venom Acupuncture on the Central Nervous System and Muscle in an Animal hSOD1G93A Mutant. Toxins, 7(3), 846-858. https://doi.org/10.3390/toxins7030846




