1. Introduction
Diarrheic Shellfish Poisoning (DSP) is a syndrome caused by the ingestion of organisms contaminated with the phosphatase inhibitors group of okadaic acid (OA) and its analogs; dynophysistoxin 1 and 2 (DTX1, DTX2) (
Figure 1). These diarrheic shellfish toxins (DST) were first isolated from two sponge species:
Halichondria okadai and
H. melanodocia [
1,
2], and are mainly produced by dynoflagellates of the genera
Dynophysis,
Phalacroma, and
Prorocentrum [
3,
4,
5]. Regarding the mechanism of action, these toxins are strong inhibitors of serine/threonine phosphatases, especially types 1 and 2A with a particularly high affinity to 2A [
6,
7]. This inhibition results in the increase of the phosphorylation of a number of proteins leading to significant cell alterations, being OA and DTX1,which were also reported as tumor promotors and inducers of genotoxicity and cytotoxicity at low concentrations in marine invertebrates [
8,
9].
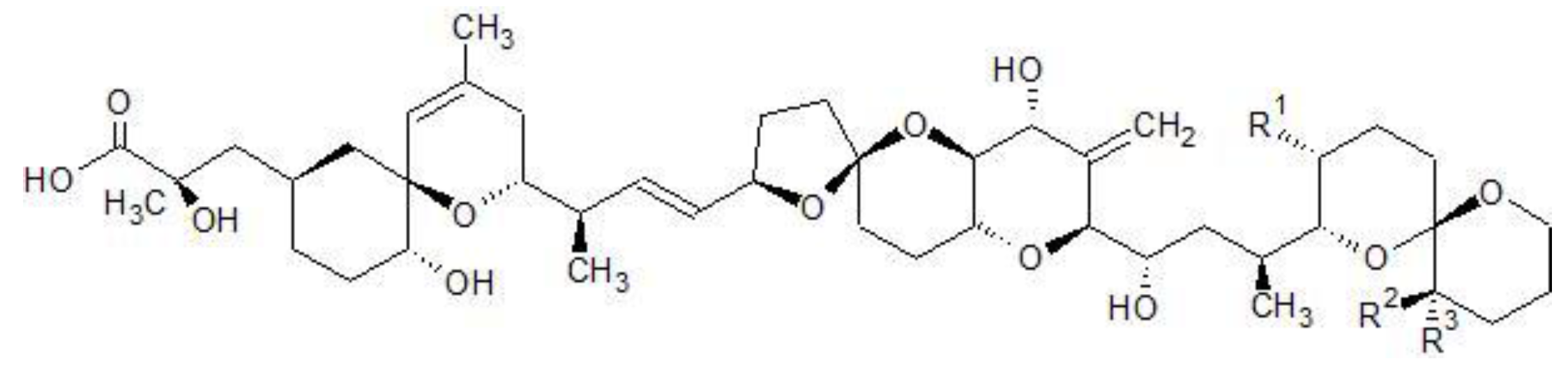
Figure 1.
Chemical structures of OA, DTX1&2.
Figure 1.
Chemical structures of OA, DTX1&2.
| Toxin | R1 | R2 | R3 |
|---|
| OA | CH3 | H | H |
| DTX1 | CH3 | CH3 | H |
| DTX2 | H | H | CH3 |
Although first reported in Japan, poisoning incidents occur all over the world and the most common route of intoxication is via ingestion of contaminated fish and shellfish [
10,
11,
12,
13]. These phycotoxins are heat and frost resistant and the taste and odor of the contaminated organisms remain unchanged [
14,
15,
16]. DSP is known for its serious gastrointestinal symptoms, from chills to diarrhea, and the severity of the intoxication depends on the amount of toxin that the patient was exposed to [
17].
Given their global incidence, DSTs are regulated worldwide. Studies on mice, together with epidemiologic studies, led to the establishment of the Lowest Observed Effect Level (LOEL) of 50 μg OA equivalents/person [
18]. Currently, the toxic equivalent factors (TEF) have been established, (
Table 1), and the limit value for the European Union is 160 µg OA equivalents/ kg shellfish meat (SM) [
18,
19]. Regarding detection methods, the European Food Safety Agency (EFSA) recommends the use of analytical techniques such as LC-MS/MS [
18]. This recommendation was later reinforced in 2011 by the European Commission (EC) suggesting that this technique should be established as a reference method for DST detection by 31 December 2014 [
20]. Analytical procedures are more reliable, do not have the ethical issues that come with the use of mice bioassay (MBA) technique, are able to identify several toxins in a mixture with high degree of sensitivity.
Table 1.
Toxic equivalent factors for OA and its analogs.
Table 1.
Toxic equivalent factors for OA and its analogs.
| Toxin | TEF | Reference |
|---|
| OA | 1 | [18] |
| DTX1 | 1 | [18] |
| DTX2 | 0.6 | [18] |
Prior research demonstrated that DSTs can be found in unusual vectors along the food chain, although monitoring for this group of toxins, exclusively in bivalves, is simplistic and underestimates the risk to public health [
21]. This argument is reinforced by the evidence that OA is bioaccumulated through the food web [
21]. In this study, we surveyed the Portuguese islands of Madeira (Madeira archipelago) and São Miguel (Azores archipelago), and the northwestern coast of Morocco for new vectors through intertidal and SCUBA diving harvesting. We collected 22 species of benthic organisms including gastropods (sea-snails, sea-slugs, and limpets), bivalves (mussels), crustaceans (barnacles), and echinoderms (starfishes, sea-urchins, and sea-cucumbers). The fact that most of the above-mentioned species are edible and commercially important species was a determining factor in the selection of these species (
Table 2). Inedible species were also sampled for their importance in the food chain. We believe that our data contribute to the development and updating of legislation regarding the monitoring procedures of these toxins in order to better protect public health.
Table 2.
Species sampled and their trophic level, average number of specimens comprising a pooled sample (AvNr), and number of samples collected (NrP Samples)—from Madeira in September 2012, São Miguel Island, Azores, in June 2013, and Morocco in July 2013— edibility and monitoring status (M. status). Availability of animals is dependent on their geographical distribution and ecology.
Table 2.
Species sampled and their trophic level, average number of specimens comprising a pooled sample (AvNr), and number of samples collected (NrP Samples)—from Madeira in September 2012, São Miguel Island, Azores, in June 2013, and Morocco in July 2013— edibility and monitoring status (M. status). Availability of animals is dependent on their geographical distribution and ecology.
| Species | Trophic Level | Sampling Site(s) | Nr P Samples | AvNr | Edible | M. Status | Ref. |
|---|
| Astropecten aranciacus | 2nd level predator | Madeira | 1 | 2 | No | No | [22] |
| Echinaster sepositus | 2nd level predator | Madeira | 1 | 3 | No | No | [23] |
| Marthasterias glacialis | 2nd level predator | Madeira/Azores/Morocco | 8 | 1 | No | No | [24] |
| Ophidiaster ophidianus | Detritivorous | Madeira/Azores | 5 | 1 | No | No | [23] |
| Paracentrotus lividus | Grazer | Madeira/Azores/Morocco | 7 | 1 | Yes | No | [25] |
| Diadema africanum | Grazer | Madeira | 2 | 1 | No | No | [26] |
| Sphaerechinus granularis | Grazer | Azores | 4 | 1 | Yes | No | [27] |
| Arbacia lixula | Grazer | Madeira/Azores/Morocco | 9 | 4 | No | No | [28] |
| Holothuria(Platyperona)sanctori | Deposit feeder | Morocco | 4 | 1 | Yes | No | [29,30] |
| Pollicipes pollicipes | Filter feeder | Morocco | 3 | 35 | Yes | No | [24] |
| Monodonta lineata | Grazer | Morocco | 5 | 86 | Yes | No | [31] |
| Onchidella celtica | Grazer | Morocco | 1 | 50 | No | No | [32] |
| Pattela aspera | Grazer | Madeira | 2 | 15 | Yes | No | [24] |
| Patella spp. | Grazer | Morocco | 4 | 12 | Yes | No | [24] |
| Pattela candei | Grazer | Azores | 3 | 10 | Yes | No | [24] |
| Umbraculum umbraculum | Grazer | Madeira | 1 | 1 | No | No | [33] |
| Stramonita haemostoma | 2nd level predator | Madeira/Azores/Morocco | 5 | 15 | No | No | [34] |
| Charonia lampas | 3rd level predator | Madeira/Morocco | 3 | 1 | Yes | No | [35] |
| Cerithium vulgatum | Grazer | Morocco | 1 | 40 | Yes | No | [36] |
| Gibbula umbilicalis | Grazer | Morocco | 3 | 100 | Yes | No | [31] |
| Mytilus spp. | Filter feeder | Morocco | 4 | 30 | Yes | Yes | [37] |
2. Results and Discussion
In this study, a total of 101 samples were collected from three different sampling sites:
Madeira (25 samples) in September 2012, São Miguel Island, Azores (37 samples), in June 2013 and the northwestern coast of Morocco (39 samples) in July 2013 (
Figure 2).
Several species belonging to distinct taxa were collected, comprising starfish (A. aranciacus, E. sepositus, M. glacialis, O. ophidianus), sea-urchins (A. lixula, D. africanum, P. lividus, S. granularis), sea-cucumber (H. sanctori), crustaceans (P. pollicipes), bivalves (Mytillus spp.) and gastropods (A. depilans, C. vulgatum, C. lampas, G. umbilicalis, M. lineata, O. celtica, Patella spp., P. candei, P. tenuis tenuis, P.aspera, S. haemostoma, and U. umbraculum).
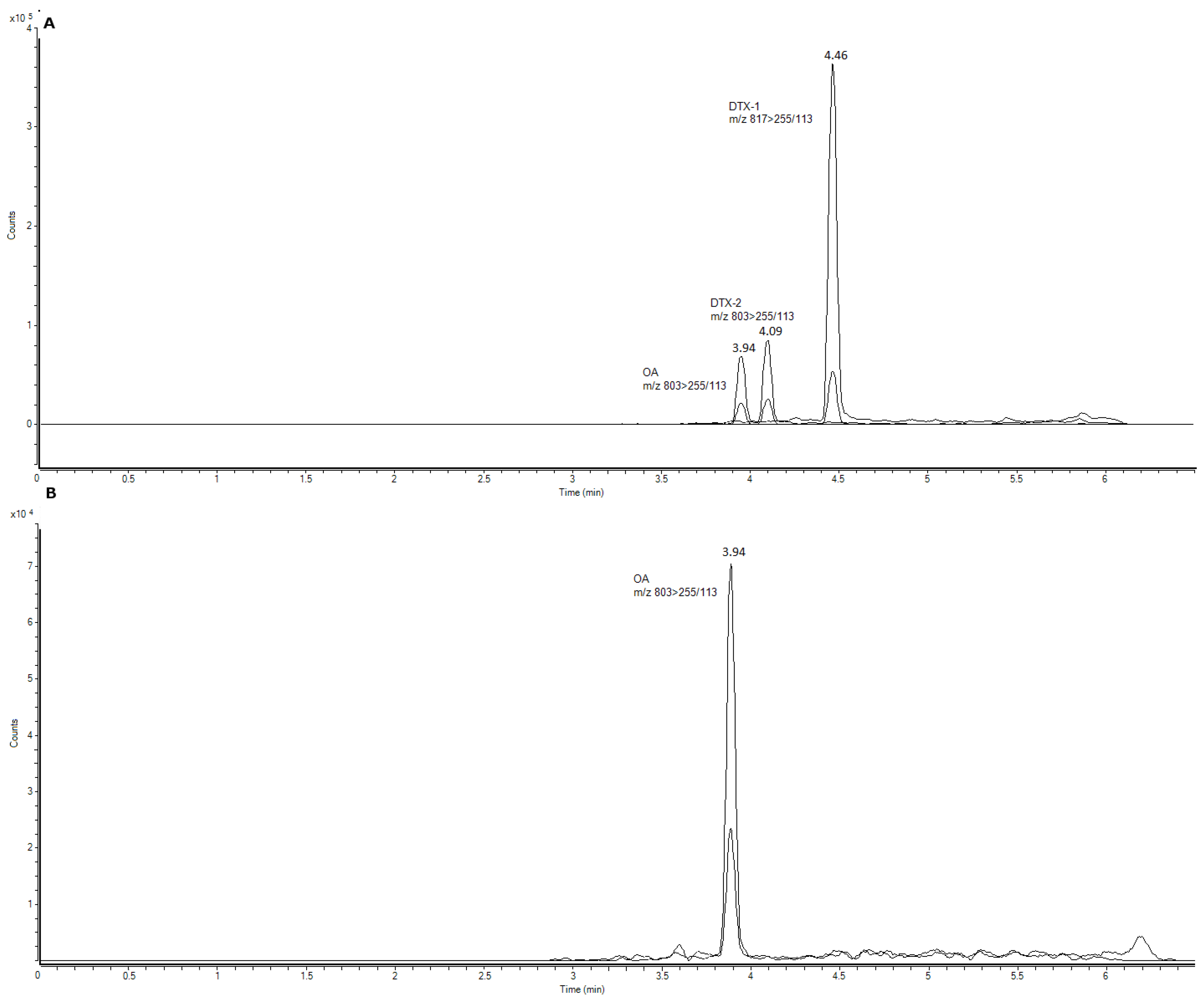
OA and its analogues were screened in the three sampling sites. A total of 19% of the samples contained quantifiable OA contents for OA (above the limit of quantification (LOQ)), but neither DTX1 nor DTX2 were detected (
Figure 3). DTX3 was also screened in all of the samples but was below the limit of detection (LOD) of the equipment. The LOD and LOQ for AO/DTX1/DTX2 in our equipment were 0.468 ng/mL and 1.56 ng/mL, respectively. In order to analyze and quantify OA/DTX1/DTX2 in the samples, a 50× step was added whereby the amounts of 1.85–35 ng/mL (0.37–7 µg/Kg) were detected. Following the official control method of the European Union, alkaline hydrolysis was performed in order to detect the esoteric forms of OA and DTX toxins. Within this method, a ratio of NaOH/HCl and a sample extract is required to maintain pH conditions and cannot be modified. For this reason, the samples cannot be concentrated. In order to maintain this ratio, the samples were diluted (50× dilution) and hydrolysis was done following the procedure. While the hydrolyzed samples were being analyzed, the diluted control without hydrolysis was also checked. In both samples, hydrolyzed and unhydrolyzed, OA could be detected in the samples with a higher concentration, but could not be quantified. This small amount was above the LOD but below the LOQ. No changes were observed in these peaks before and after hydrolysis. If the samples had been concentrated, the pH of the solution would have changed and the toxins could have degraded.
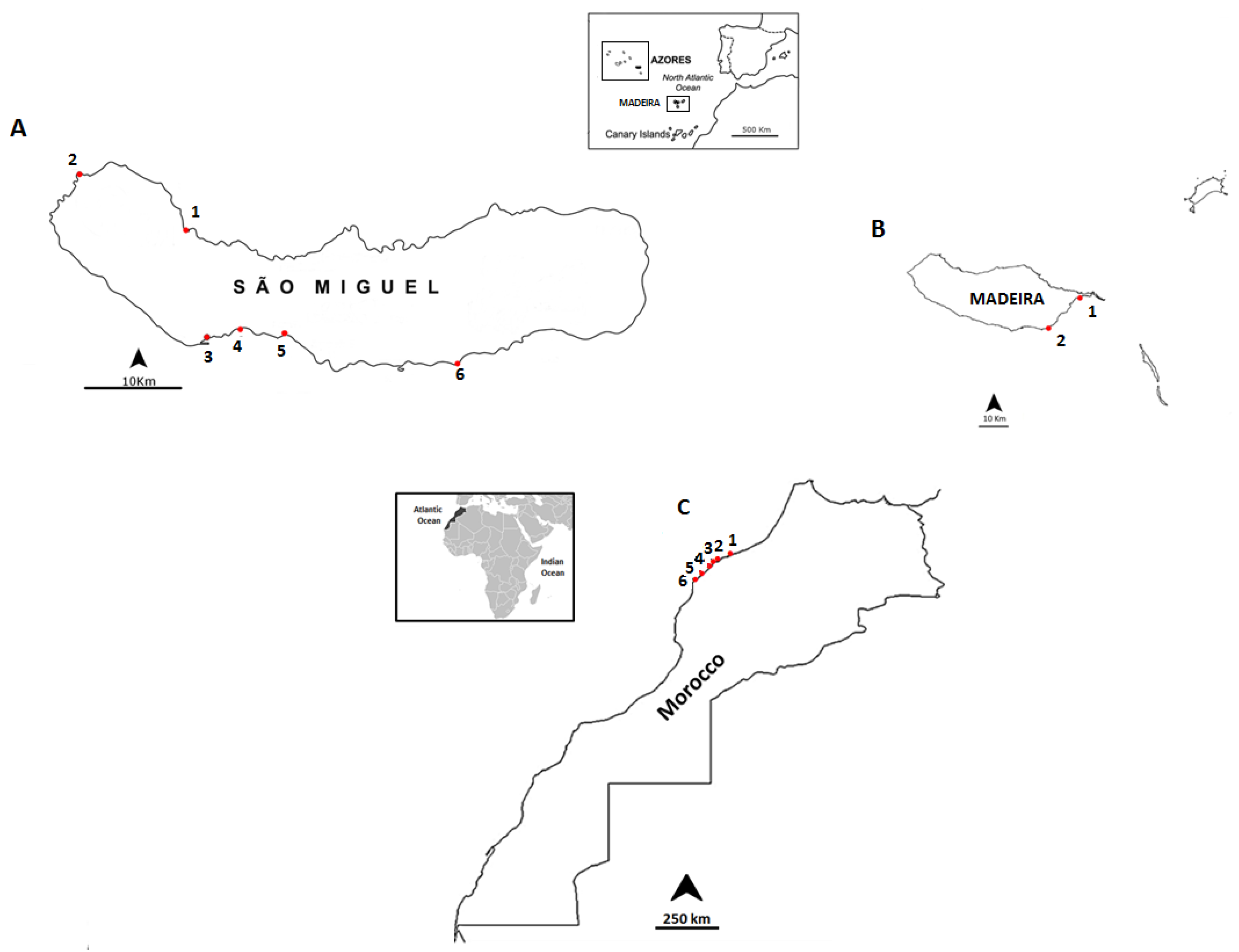
Figure 2.
Location of the sampling points: (A) the coast of São Miguel Island, Azores archipelago: 1-Cruzeiro; 2-Mosteiros; 3-Étar; 4-São Roque; 5-Lagoa; and 6-Caloura; (B) the coast of Madeira: 1-Reis Magos; 2-Caniçal; and (C) the northwestern coast of Morocco: 1-Casablanca Corniche; 2-El Jadida Haras; 3-El Jadida Sâada; 4-Sidi Bouzid; 5-Mrizika; and 6-Oualidia.
Figure 2.
Location of the sampling points: (A) the coast of São Miguel Island, Azores archipelago: 1-Cruzeiro; 2-Mosteiros; 3-Étar; 4-São Roque; 5-Lagoa; and 6-Caloura; (B) the coast of Madeira: 1-Reis Magos; 2-Caniçal; and (C) the northwestern coast of Morocco: 1-Casablanca Corniche; 2-El Jadida Haras; 3-El Jadida Sâada; 4-Sidi Bouzid; 5-Mrizika; and 6-Oualidia.
The majority of the samples with OA (73.7%), as well as the samples with the highest concentrations, were detected on the coast of Morocco, followed by Madeira (21.1%). This might be due to the fact that both sampling sites are at the same latitude. For São Miguel Island (Azores), only one measurable sample was detected in the starfish O. ophidianus.
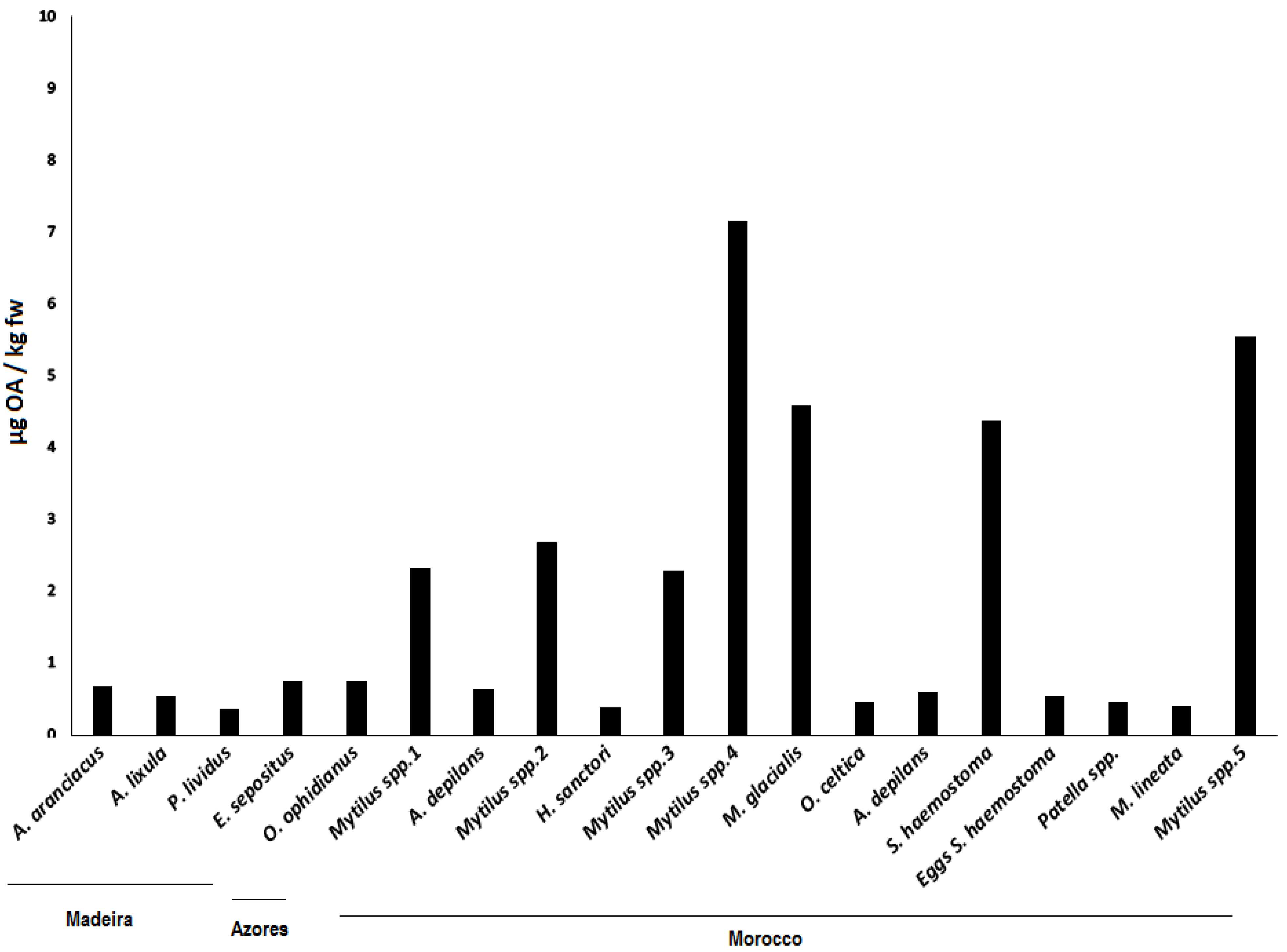
Figure 3.
OA (µg/Kg fresh weight (fw)) for all sampled groups of organisms in Madeira (Starfish—A. aranciacus, E. sepositus; Sea-urchins—A. lixula, P. lividus) Azores (Starfish—O. ophidianus) and Morocco (Starfish—M. glacialis; Sea-cucumbers—H. sanctori; Gastropods—A. depilans, O. celtica, S. haemostoma, Patella spp., M. lineata; Bivalves- Mytilus spp.).Numbers correspond to different sampling locations in Morocco: 1-Casablanca Corniche; 2-Sidi Bouzid; 3-El Jadida Saâda; 4-Mrizika; and 5-Oualidia.
Figure 3.
OA (µg/Kg fresh weight (fw)) for all sampled groups of organisms in Madeira (Starfish—A. aranciacus, E. sepositus; Sea-urchins—A. lixula, P. lividus) Azores (Starfish—O. ophidianus) and Morocco (Starfish—M. glacialis; Sea-cucumbers—H. sanctori; Gastropods—A. depilans, O. celtica, S. haemostoma, Patella spp., M. lineata; Bivalves- Mytilus spp.).Numbers correspond to different sampling locations in Morocco: 1-Casablanca Corniche; 2-Sidi Bouzid; 3-El Jadida Saâda; 4-Mrizika; and 5-Oualidia.
Average concentrations detected were all below the current limit implemented in the European legislation—160 µg OA equivalents/ kg shellfish meat [
18]—and ranged from 0.368 µg/kg fresh weigh (fw), in
P. lividus, to 7.157 µg/kg fw in
Mytilus spp.
Regarding statistics, the first step of the gamma hurdle model was a Generalized Linear Model (GLZ) performed with the data of presence/absence of OA, using binomial distribution error.
This model was applied separately to each geographical location. The results of the model’s analysis of deviance as well as the coefficients rescaled to a logistic probability [0,1] are shown in
Table 3. The “organism” did not turn out to be a significant factor, most likely due to the low number of samples with quantifiable OA contents, except in Morocco. In Morocco, the highest probability corresponded to the bivalve, whereas all other organisms had very low probabilities of containing OA. The second part of the model with the gamma error distribution analyzes the variation in OA concentration, showing quantifiable results among those samples. It was not possible to perform this analysis in Azores because there was only one single sample containing OA. The factor “organism” was significant both in Madeira and Morocco (
Table 4).In Madeira, OA only appeared in sea urchin and star fish, with star fish containing three times more OA, on average (1.58
versus 0.45,
Table 4). In Morocco, the bivalves contained much more OA than all the other organisms, the closest one being the sea star, with an average of approximately ¼ of bivalve OA content (4
versus 1.15,
Table 4).
Table 3.
Results of the binomial regression model for OA occurrence with “organism” as a factor.
Table 3.
Results of the binomial regression model for OA occurrence with “organism” as a factor.
| Analysis of Deviance |
|---|
| Location | Factor | χ2 | df | p |
|---|
| Madeira | Organism | 3.9 | 3 | 0.28 |
| Rescaled model coefficients: sea urchin = 0.16; star fish = 0.71; gastropod = 1.6 × 10−8; limpet = 1.6 × 10−8 |
| Azores | Organism | 2.1 | 3 | 0.56 |
| Rescaled model coefficients: sea urchin = 4.3 × 10−10; star fish = 1; gastropod = 0.5; limpet = 0.5 |
| Morocco | Organism | 19.8 | 7 | <0.01 |
| Rescaled model coefficients: bivalve = 1; crustacean = 1 × 10−17; sea urchin = 1 × 10−17; star fish = 3.2 × 10−9; gastropod = 1.1 × 10−9; limpet = 1.1 × 10−9; sea snail = 6.4 × 10−9; sea cucumber = 1.1 × 10−9 |
Table 4.
Results of the gamma regression model for OA occurrence with organism as factor.
Table 4.
Results of the gamma regression model for OA occurrence with organism as factor.
| Analysis of Deviance |
|---|
| Location | Factor | χ2 | df | p |
|---|
| Madeira | Organism | 5.4 | 1 | <0.05 |
| Rescaled model coefficients: sea urchin = 0.45; star fish = 1.58 |
| Morocco | Organism | 12.8 | 5 | <0.05 |
| Rescaled model coefficients: bivalve = 4; star fish = 1.15; gastropod = 0.44; limpet = 0.12; sea snail = 0.15; sea cucumber = 0.1 |
The high number of positive detections in Morocco could be explained by an increased eutrophication effect, due to larger population density, continental runoff, and industrial pollution. In comparison to the previous study, the amounts of OA detected are quite different, ranging from 0.58 µg/Kg fw to 429.41 µg/Kg fw [
21], even though the number of screened species is higher in the present study. Here, the higher concentrations that were detected could equally be due to higher anthropogenic inputs, although they were detected in different taxa, namely gastropods [
21]. From a statistical point of view, it is not possible to compare the bioaccumulation results between the studies due to the lower positive hits obtained in the present study.
Owing to the oligotrophic waters of Madeira and Azores archipelagos, bivalves are not common, which makes gastropods and echinoderms a good alternative for the monitoring of OA and its derivatives in both archipelagos.
We report nine new vectors for OA in the species
A. aranciacus,
A. lixula,
A. depilans,
E. sepositus,
H. sanctori,
O. celtica,
O. ophidianus,
Patella spp., and
S. haemostoma. We also detected OA in the eggs of
S. haemostoma indicating a potential parental transfer of the toxin to the offspring. These kind of cases have already been reported, as an example in
Takifugo rubripes larvae that are protected by maternal tetrodotoxin [
38].