New Invertebrate Vectors of Okadaic Acid from the North Atlantic Waters—Portugal (Azores and Madeira) and Morocco
Abstract
:1. Introduction
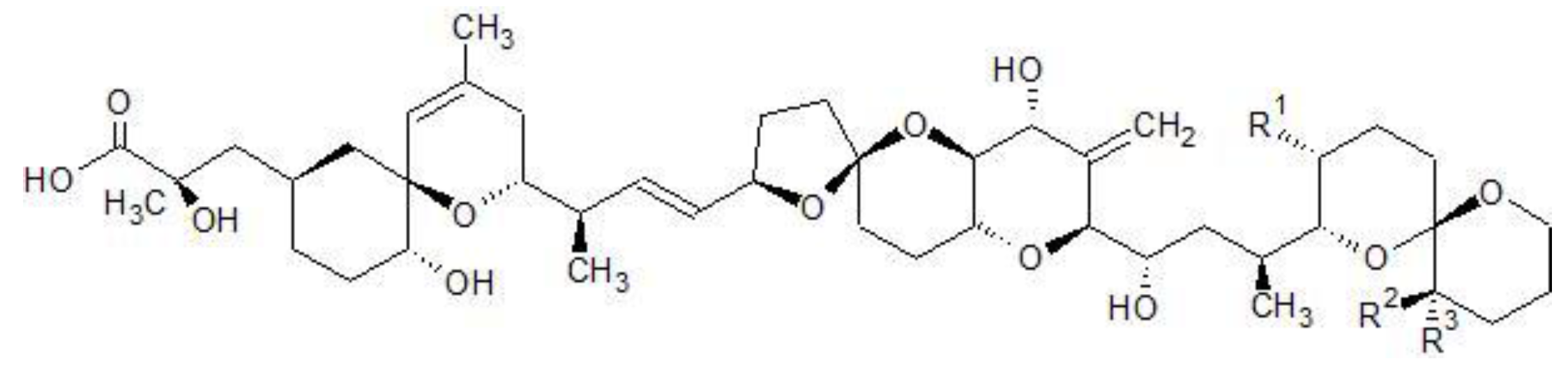
| Toxin | R1 | R2 | R3 |
|---|---|---|---|
| OA | CH3 | H | H |
| DTX1 | CH3 | CH3 | H |
| DTX2 | H | H | CH3 |
| Toxin | TEF | Reference |
|---|---|---|
| OA | 1 | [18] |
| DTX1 | 1 | [18] |
| DTX2 | 0.6 | [18] |
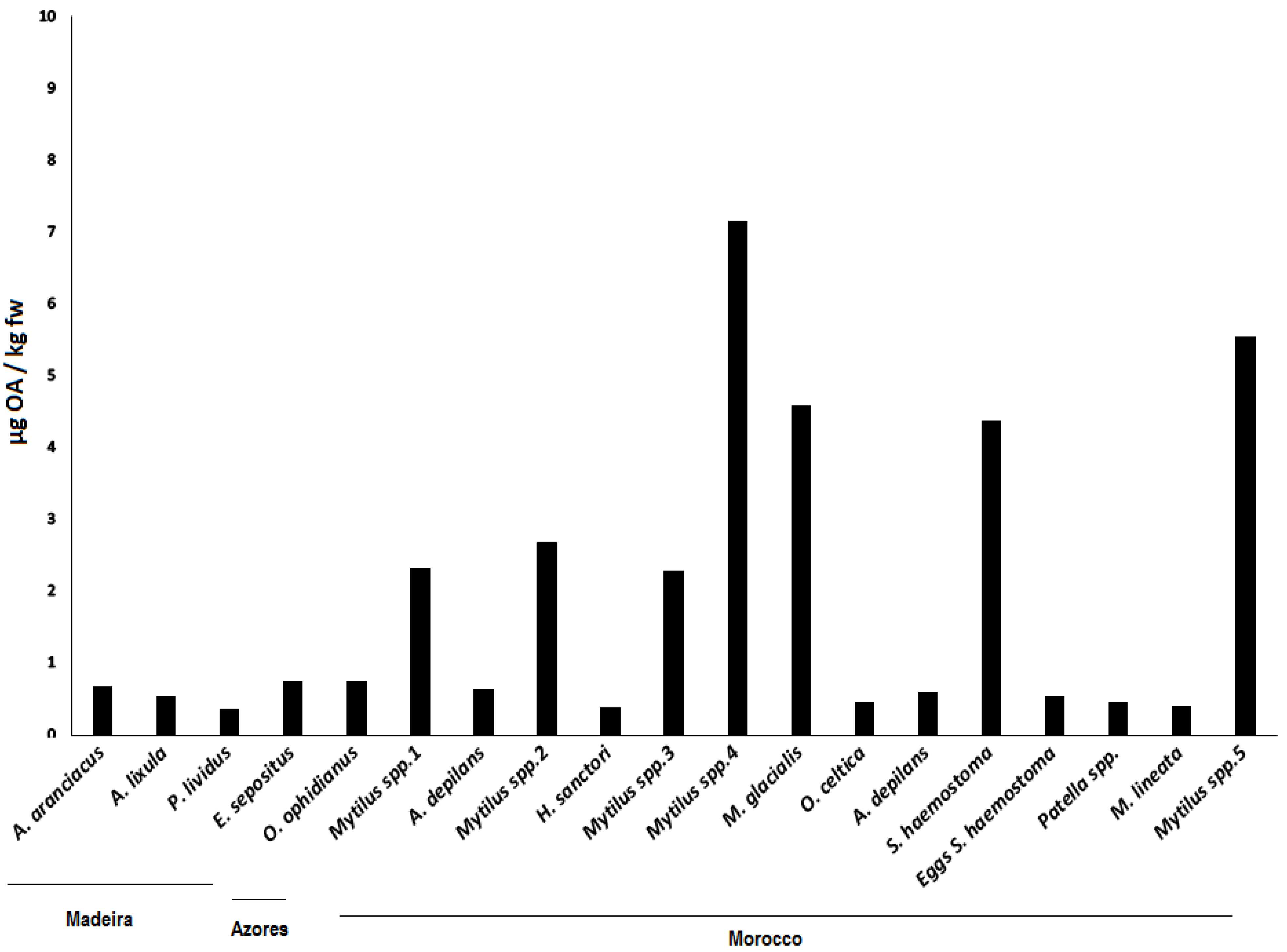
| Species | Trophic Level | Sampling Site(s) | Nr P Samples | AvNr | Edible | M. Status | Ref. |
|---|---|---|---|---|---|---|---|
| Astropecten aranciacus | 2nd level predator | Madeira | 1 | 2 | No | No | [22] |
| Echinaster sepositus | 2nd level predator | Madeira | 1 | 3 | No | No | [23] |
| Marthasterias glacialis | 2nd level predator | Madeira/Azores/Morocco | 8 | 1 | No | No | [24] |
| Ophidiaster ophidianus | Detritivorous | Madeira/Azores | 5 | 1 | No | No | [23] |
| Paracentrotus lividus | Grazer | Madeira/Azores/Morocco | 7 | 1 | Yes | No | [25] |
| Diadema africanum | Grazer | Madeira | 2 | 1 | No | No | [26] |
| Sphaerechinus granularis | Grazer | Azores | 4 | 1 | Yes | No | [27] |
| Arbacia lixula | Grazer | Madeira/Azores/Morocco | 9 | 4 | No | No | [28] |
| Holothuria(Platyperona)sanctori | Deposit feeder | Morocco | 4 | 1 | Yes | No | [29,30] |
| Pollicipes pollicipes | Filter feeder | Morocco | 3 | 35 | Yes | No | [24] |
| Monodonta lineata | Grazer | Morocco | 5 | 86 | Yes | No | [31] |
| Onchidella celtica | Grazer | Morocco | 1 | 50 | No | No | [32] |
| Pattela aspera | Grazer | Madeira | 2 | 15 | Yes | No | [24] |
| Patella spp. | Grazer | Morocco | 4 | 12 | Yes | No | [24] |
| Pattela candei | Grazer | Azores | 3 | 10 | Yes | No | [24] |
| Umbraculum umbraculum | Grazer | Madeira | 1 | 1 | No | No | [33] |
| Stramonita haemostoma | 2nd level predator | Madeira/Azores/Morocco | 5 | 15 | No | No | [34] |
| Charonia lampas | 3rd level predator | Madeira/Morocco | 3 | 1 | Yes | No | [35] |
| Cerithium vulgatum | Grazer | Morocco | 1 | 40 | Yes | No | [36] |
| Gibbula umbilicalis | Grazer | Morocco | 3 | 100 | Yes | No | [31] |
| Mytilus spp. | Filter feeder | Morocco | 4 | 30 | Yes | Yes | [37] |
2. Results and Discussion
| Analysis of Deviance | ||||
|---|---|---|---|---|
| Location | Factor | χ2 | df | p |
| Madeira | Organism | 3.9 | 3 | 0.28 |
| Rescaled model coefficients: sea urchin = 0.16; star fish = 0.71; gastropod = 1.6 × 10−8; limpet = 1.6 × 10−8 | ||||
| Azores | Organism | 2.1 | 3 | 0.56 |
| Rescaled model coefficients: sea urchin = 4.3 × 10−10; star fish = 1; gastropod = 0.5; limpet = 0.5 | ||||
| Morocco | Organism | 19.8 | 7 | <0.01 |
| Rescaled model coefficients: bivalve = 1; crustacean = 1 × 10−17; sea urchin = 1 × 10−17; star fish = 3.2 × 10−9; gastropod = 1.1 × 10−9; limpet = 1.1 × 10−9; sea snail = 6.4 × 10−9; sea cucumber = 1.1 × 10−9 | ||||
| Analysis of Deviance | ||||
|---|---|---|---|---|
| Location | Factor | χ2 | df | p |
| Madeira | Organism | 5.4 | 1 | <0.05 |
| Rescaled model coefficients: sea urchin = 0.45; star fish = 1.58 | ||||
| Morocco | Organism | 12.8 | 5 | <0.05 |
| Rescaled model coefficients: bivalve = 4; star fish = 1.15; gastropod = 0.44; limpet = 0.12; sea snail = 0.15; sea cucumber = 0.1 | ||||
3. Experimental Section
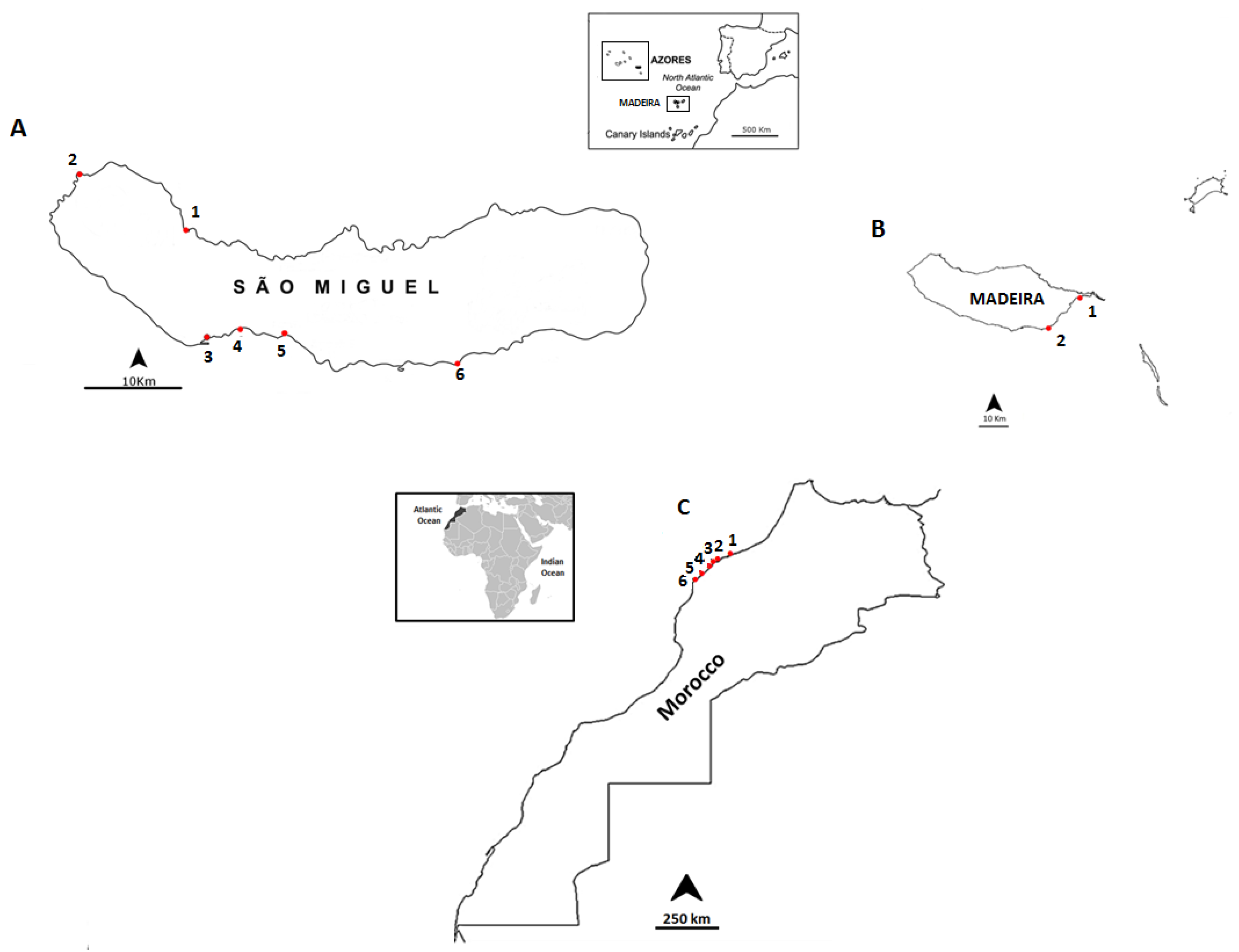
3.1. Selected Species and Sampling Sites
| Date | Location | Sampling Site | Geographic Coordinates |
|---|---|---|---|
| September 2012 | Madeira Island | Reis Magos | 32°39ʹ16.21ʹʹ N; 16°49ʹ05.29ʹʹ W |
| Caniçal | 32°44ʹ20.08ʹʹ N; 16°44ʹ17.55ʹʹ W | ||
| June 2013 | São Miguel Island | Cruzeiro | 37° 50ʹ31.19ʹʹ N; 25° 41ʹ33.61ʹʹ W |
| Étar | 37°44ʹ19.31ʹʹ N; 25°39ʹ38.84ʹʹ W | ||
| São Roque | 37°45ʹ15.35ʹʹ N; 25°38ʹ31.60ʹʹ W | ||
| Mosteiros | 37°53ʹ25.57ʹʹ N; 25°49ʹ14.72ʹʹ W | ||
| Lagoa | 37°44ʹ42.38ʹʹ N; 25°19ʹ.47ʹʹ W | ||
| Caloura | 37°42ʹ49.34ʹʹ N; 25°29ʹ54.54ʹʹ W | ||
| July 2013 | Morocco Coast | Casablanca corniche | 33°36ʹ01.2ʹʹ N; 7°39ʹ57.5ʹʹ W |
| El Jadida Haras | 33°14ʹ42.0ʹʹ N; 8°28ʹ37.5ʹʹ W | ||
| El Jadida Sâada | 33°14ʹ42.4ʹʹ N; 8°32ʹ26.9ʹʹ W | ||
| Sidi Bouzid | 33°13ʹ57.1ʹʹ N; 8°33ʹ20.9ʹʹ W | ||
| Mrizika | 32°57ʹ21.8ʹʹ N; 8°46ʹ53.2ʹʹ W | ||
| Oualidia | 32°43ʹ55.8ʹʹ N; 9°02ʹ57.6ʹʹ W |
3.2. Sample Extraction and Hydrolysis Procedure
3.3. Sample Analysis
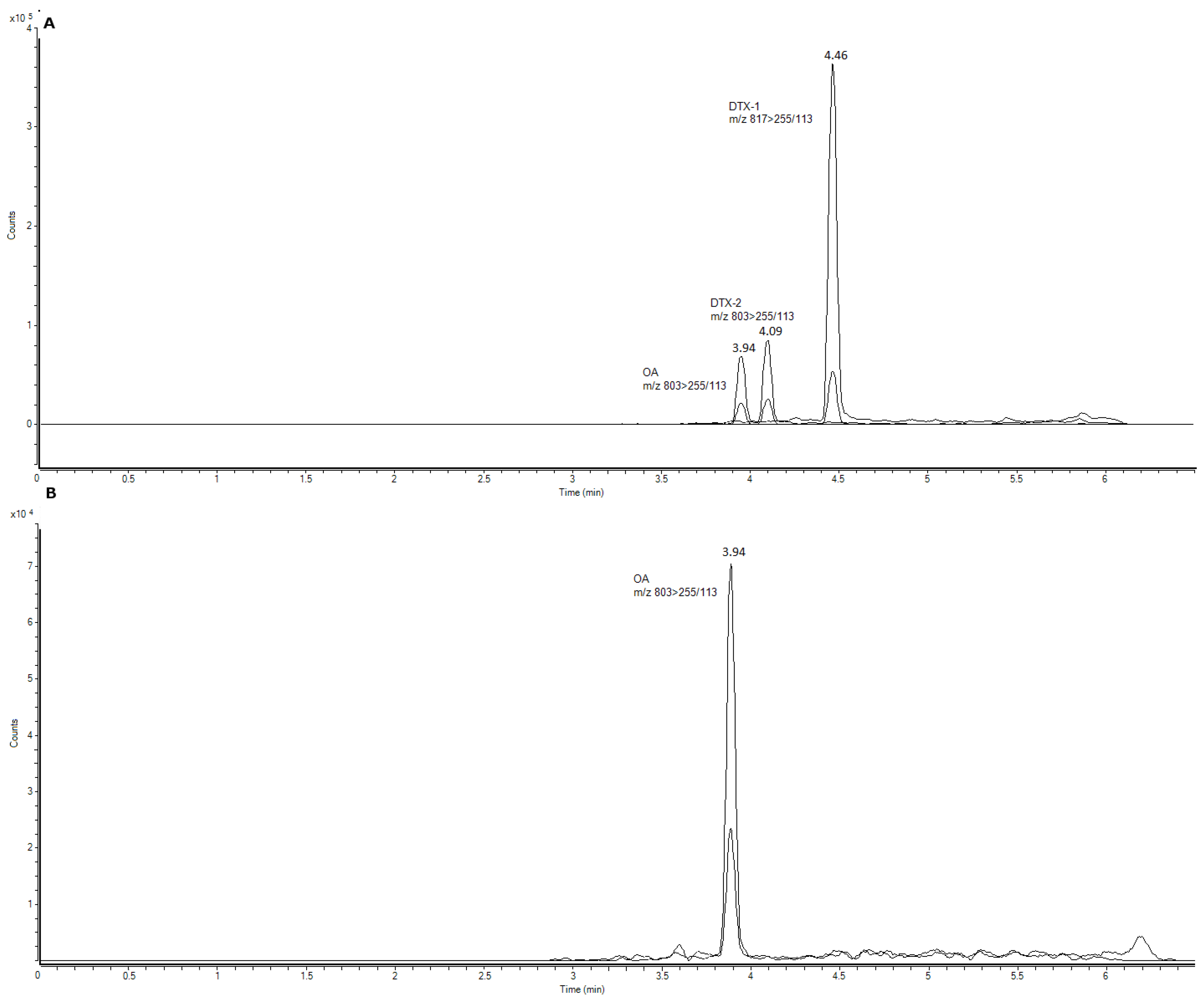
3.4. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References
- Schmitz, F.J.; Prasad, R.S.; Gopichand, Y.; Hossain, H.B.; van der Helm, D.; Schmidt, P. Acanthifolicin, a new episufide-containing polyether carboxylic acid from extracts of the marine sponge pandaros acanthifolium. J. Am. Chem. Soc. 1981, 103, 2467–2469. [Google Scholar] [CrossRef]
- Tachibana, K.; Scheurrer, P.J.; Tsukitani, Y.; Kikuchi, H.; Engen, D.V.; Clardy, J.; Gopichand, Y.; Schimitz, F.J. Okadaic acid, a cytotoxicity polyether from two marine sponges of the genus halichondria. J. Am. Chem. Soc. 1981, 103, 2469–2471. [Google Scholar] [CrossRef]
- MacKenzie, L.; Beuzenberg, V.; Holland, P.; McNabb, P.; Suzuki, T.; Selwood, A. Pectenotoxin and okadaic acid-based toxin profiles in dinophysis acuta and dinophysis acuminata from new zealand. Harmful Algae 2005, 4, 75–85. [Google Scholar] [CrossRef]
- Hu, T.M.; Curtis, J.M.; Walter, J.A.; Wright, J.L.C. Identification of DTX-4, a new water-soluble phosphatase inhibitor from the toxic dinoflagellate prorocentrum lima. J. Chem. Soc. Chem. Commun. 1995, 5, 597–599. [Google Scholar] [CrossRef]
- Caroppo, C.; Congestri, R.; Bruno, M. On the presence of phalacroma rotundatum in the southern adriatic sea (Italy). Aquat. Microb. Ecol. 1999, 17, 301–310. [Google Scholar] [CrossRef]
- Bialojan, C.; Takai, A. Inhibitory effect of a marine sponge toxin, okadaic acid, on protein phosphatases. Biochem. J. 1988, 256, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Takai, A.; Murata, M.; Torigoe, K.; Isobe, M.; Mieskes, G.; Yasumoto, T. Inhibitory effect of okadaic acid derivatives on protein phosphatases. A study on structure-affinity relationship. Biochem. J. 1992, 284, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Maneiro, E.; Rodas, V.L.; Costas, E.; Hernandez, J.M. Shellfish consumption: A major risk factor for colorectal cancer. Med. Hypotheses 2008, 70, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Prego-Faraldo, M.V.; Valdiglesias, V.; Laffon, B.; Eirin-Lopez, J.M.; Mendez, J. In vitro analysis of early genotoxic and cytotoxic effects of okadaic acid in different cell types of the mussel mytilus galloprovincialis. J. Toxicol. Environ. Health A 2015, 78, 814–824. [Google Scholar] [CrossRef] [PubMed]
- Yasumoto, T.; Oshima, Y.; Yamaguchi, M. Occurrence of a new type of shellfish poisoning in tohoku district. Bull. Jpn. Soc. Sci. Fish. 1978, 44, 1249–1255. [Google Scholar] [CrossRef]
- Elgarch, A.; Vale, P.; Rifai, S.; Fassouane, A. Detection of diarrheic shellfish poisoning and azaspiracid toxins in moroccan mussels: Comparison of the LC-MS method with the commercial immunoassay kit. Mar. Drugs 2008, 6, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Truan, D.; Lagos, M.; Santelices, J.P.; Diaz, J.C.; Lagos, N. Metabolic transformation of dinophysistoxin-3 into dinophysistoxin-1 causes human intoxication by consumption of o-acyl-derivatives dinophysistoxins contaminated shellfish. J. Toxicol. Sci. 2005, 30, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Scoging, A.; Bahl, M. Diarrhetic shellfish poisoning in the UK. Lancet 1998, 352, 117. [Google Scholar] [CrossRef]
- Reboreda, A.; Lago, J.; Chapela, M.J.; Vieites, J.M.; Botana, L.M.; Alfonso, A.; Cabado, A.G. Decrease of marine toxin content in bivalves by industrial processes. Toxicon 2010, 55, 235–243. [Google Scholar] [CrossRef] [PubMed]
- McCarron, P.; Kilcoyne, J.; Hess, P. Effects of cooking and heat treatment on concentration and tissue distribution of okadaic acid and dinophysistoxin-2 in mussels (mytilus edulis). Toxicon 2008, 51, 1081–1089. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.; Loreal, H.; Toyofuku, H.; Hess, P.; Karunasagar, I.; Ababouch, L. Assessment and Management of Biotoxin Risks in Bivalve Mollusks; FAO: Rome, Italy, 2011; p. 337. [Google Scholar]
- Hamano, Y.; Kinoshita, Y.; Yasumoto, T. Enteropathogenicity of diarrheic shellfish toxins in intestinal models. J. Food Hyg. Soc. Jpn. 1985, 27, 375–379. [Google Scholar] [CrossRef]
- Alexander, J.; Auðunsson, G.A.; Benford, D.; Cockburn, A.; Cravedi, J.P.; Dogliotti, E.; di Domenico, A.; Fernández-Cruz, M.L.; Fink-Gremmels, J.; Fürst, P.; et al. Opinion of the scientific panel on contaminants in the food chain on a request from the european commission on marine biotoxins in shellfish—Okadaic acid and analogues. EFSA J. 2008, 589, 1–62. [Google Scholar]
- Aune, T.; Larsen, S.; Aasen, J.A.; Rehmann, N.; Satake, M.; Hess, P. Relative toxicity of dinophysistoxin-2 (DTX-2) compared with okadaic acid, based on acute intraperitoneal toxicity in mice. Toxicon 2007, 49, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Regulation, C. COMMISSION REGULATION (EU) No 15/2011 of 10 January 2011 amending Regulation (EC) No 2074/2005 as regards recognised testing methods for detecting marine biotoxins in live bivalve molluscs. Off. J. Eur. Commun. 2011, L, 3–4. [Google Scholar]
- Silva, M.; Barreiro, A.; Rodriguez, P.; Otero, P.; Azevedo, J.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New invertebrate vectors for pst, spirolides and okadaic acid in the north atlantic. Mar. Drugs 2013, 11, 1936–1960. [Google Scholar] [CrossRef] [PubMed]
- Burla, H.; Ferlin, V.; Pabst, B.; Ribi, G. Notes on the ecology of astropecten aranciacus. Mar. Biol. 1972, 14, 235–241. [Google Scholar]
- Ferguson, J.C. Feeding activity in echinaster and its induction with dissolved nutrients. Biol. Bull. 1969, 136, 374–384. [Google Scholar] [CrossRef]
- Knox, G.A. Hard shores. In The Ecology of Seashores; Kennish, M.J., Ed.; CRC Press: Boca Raton, FL, USA, 2001; pp. 20–86. [Google Scholar]
- Lemée, R.; Boudouresque, C.F.; Gobert, J.; Malestroit, P.; Mari, X.; Meinesz, A.; Menager, V.; Ruitton, S. Feeding behaviour of paracentrotus lividus in the presence of caulerpa taxifolia introduced in the mediterranean sea. Oceanol. Acta 1995, 19, 245–253. [Google Scholar]
- Rodríguez, A.; Hernández, J.C.; Clemente, S.; Coppard, S.E. A new species of diadema (echinodermata: Echinoidea: Diadematidae) from the eastern atlantic ocean and a neotype designation of diadema antillarum (philippi, 1845). Zootaxa 2013, 3636, 144–170. [Google Scholar] [CrossRef]
- Martinez-Pita, I.; Sanches-Espana, A.; Garcia, F.J. Gonadal growth and reproduction in the sea urchin sphaerechinus granularis (lamarck 1816) in southern spain. Sci. Mar. 2008, 72, 603–611. [Google Scholar]
- Bulleri, F.; Benedetti-Cecchi, L.; Cinelli, F. Grazing by the sea urchins Arbacia lixula L. and Paracentrotus lividus Lam. in the Northwest Mediterranean. J. Exp. Mar. Biol. Ecol. 1999, 241, 81–85. [Google Scholar] [CrossRef]
- Navarro, P.G.; García-Sanz, S.; Barrio, J.M.; Tuya, F. Feeding and movement patterns of the sea cucumber holothuria sanctori. Mar. Biol. 2013, 160, 2957–2966. [Google Scholar] [CrossRef]
- Toral-Granda, V.; Lovatelli, A.; Vasconcellos, M. Scientific Committee Composed of Conand C., Hamel J.F., Mercier A., Purcell S. and Uthicke S. In Procedings of the International Workshop on the Sustainable Use and Management of Sea Cucumber Fisheries, SPC Beche-deMer Information Bulletin, Puerto Ayora, Galapagos Islands, Ecuador, 19–23 November 2008.
- Crothers, J.H. Common topshells: An introduction to the biology of osilinus lineatus with notes on other species in the genus. Field Stud. 2001, 10, 115–160. [Google Scholar]
- Dayrat, B. Review of the current knowledge of the systematics of onchidiidae (mollusca: Gastropoda: Pulmonata) with a checklist of nominal species. Zootaxa 2009, 2068, 1–26. [Google Scholar]
- Valdes, A. On the publication data, authorship, and type species of umbraculum and tylodina gastropoda; opisthobranchia; tylodinoidea. Nautilus 2001, 115, 29–34. [Google Scholar]
- Ramírez, R.; Tuya, F.; Haroun, R. Spatial patterns in the population structure of the whelk stramonita haemastoma (linnaeus, 1766) (gastropoda: Muricidae) in the canarian archipelago (eastern atlantic). Sci. Mar. 2009, 73, 431–437. [Google Scholar] [CrossRef]
- Lin, S.J.; Hwang, D.F. Possible source of tetrodotoxin in the starfish astropecten scoparius. Toxicon 2001, 39, 573–579. [Google Scholar] [CrossRef]
- Nicolaidou, A.; Nott, J.A. The role of the marine gastropod cerithium vulgatum in the biogeochemical cycling of metals. Biogeochem. Cycl. Sediment Ecol. 1999, 59, 137–146. [Google Scholar]
- Buschbaum, C.; Dittmann, S.; Hong, J.-S.; Hwang, I.; Strasser, M.; Thiel, M.; Valdivia, N.; Yoon, S.; Reise, K. Mytilid mussels: Global habitat engineers in coastal sediments. Helgol. Mar. Res. 2008, 63, 47–58. [Google Scholar] [CrossRef]
- Itoi, S.; Yoshikawa, S.; Asahina, K.; Suzuki, M.; Ishizuka, K.; Takimoto, N.; Mitsuoka, R.; Yokoyama, N.; Detake, A.; Takayanagi, C.; et al. Larval pufferfish protected by maternal tetrodotoxin. Toxicon 2014, 78, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Otero, P.; Alfonso, A.; Alfonso, C.; Vieytes, M.R.; Louzao, M.C.; Botana, A.M.; Botana, L.M. New protocol to obtain spirolides from alexandrium ostenfeldii cultures with high recovery and purity. Biomed. Chromatogr. 2010, 24, 878–886. [Google Scholar] [PubMed]
- European Union Reference Laboratory for Marine Biotoxins. Eu-Harmonised Standard Operating Procedure for Determination of Lipophilic Marine Biotoxins in Molluscs by LC-MS/MS. European Union Reference Laboratory for Marine Biotoxins. Available online: http://aesan.Msssi.Gob.Es/en/crlmb/web/procedimientos_crlmb/crlmb_standard_operating_procedures.Shtml (accessed on 3 February 2015).
- Zuur, A.F.; Ieno, E.N.; Walker, N.J.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer Science & Business Media: Berlin, Germany, 2009. [Google Scholar]
- Frees, E.W. Regression Modeling with Actuarial and Financial Applications; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Team, R.C. A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 30 June 2014).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, M.; Rodriguez, I.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. New Invertebrate Vectors of Okadaic Acid from the North Atlantic Waters—Portugal (Azores and Madeira) and Morocco. Toxins 2015, 7, 5337-5347. https://doi.org/10.3390/toxins7124885
Silva M, Rodriguez I, Barreiro A, Kaufmann M, Neto AI, Hassouani M, Sabour B, Alfonso A, Botana LM, Vasconcelos V. New Invertebrate Vectors of Okadaic Acid from the North Atlantic Waters—Portugal (Azores and Madeira) and Morocco. Toxins. 2015; 7(12):5337-5347. https://doi.org/10.3390/toxins7124885
Chicago/Turabian StyleSilva, Marisa, Inés Rodriguez, Aldo Barreiro, Manfred Kaufmann, Ana Isabel Neto, Meryem Hassouani, Brahim Sabour, Amparo Alfonso, Luis M. Botana, and Vitor Vasconcelos. 2015. "New Invertebrate Vectors of Okadaic Acid from the North Atlantic Waters—Portugal (Azores and Madeira) and Morocco" Toxins 7, no. 12: 5337-5347. https://doi.org/10.3390/toxins7124885
APA StyleSilva, M., Rodriguez, I., Barreiro, A., Kaufmann, M., Neto, A. I., Hassouani, M., Sabour, B., Alfonso, A., Botana, L. M., & Vasconcelos, V. (2015). New Invertebrate Vectors of Okadaic Acid from the North Atlantic Waters—Portugal (Azores and Madeira) and Morocco. Toxins, 7(12), 5337-5347. https://doi.org/10.3390/toxins7124885








