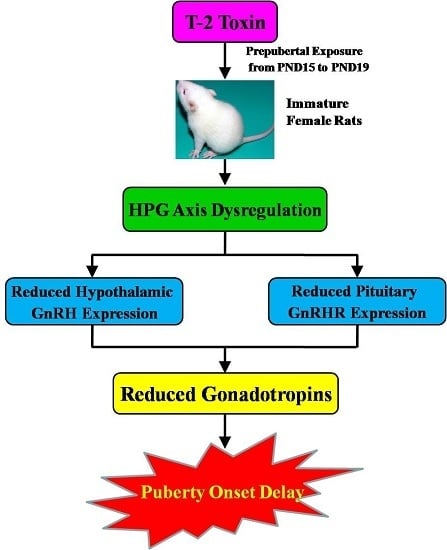
Delay of the Onset of Puberty in Female Rats by Prepubertal Exposure to T-2 Toxin
Abstract
:1. Introduction
2. Results
2.1. General Observations of Female Rats Following Prepubertal Exposure to T-2 Toxin
| Groups | Body Weight (g) | ||
|---|---|---|---|
| PND15 | PND19 | PND35 | |
| Vehicle control | 37.13 ± 1.18 | 47.18 ± 2.20 | 143.56 ± 5.80 |
| T-2 toxin treatment | 37.42 ± 1.22 | 47.73 ± 1.11 | 142.98 ± 5.26 |
2.2. Effects of Prepubertal Exposure to T-2 Toxin on the Days of Vaginal Opening (VO), First Dioestrus (D1), and First Estrus (E1) in Regular Estrous Cycles

2.3. Effect of Prepubertal Exposure to T-2 Toxin on the Vaginal Estrus
2.4. Effect of Prepubertal Exposure to T-2 Toxin on the Development of Reproductive Organs
| Groups | Relative Organic Weight (mg/g) | Corpora Lutea | ||
|---|---|---|---|---|
| Uterus | Ovary | Vagina | ||
| Vehicle control | 1.16 ± 0.28 | 0.49 ± 0.10 | 1.05 ± 0.27 | 9.6 ± 4.7 |
| T-2 toxin treatment | 0.83 ± 0.40 * | 0.33 ± 0.08 * | 0.91 ± 0.31 | 1.6 ± 0.6 ** |
2.5. Histopathological Alterations of Reproductive Organs
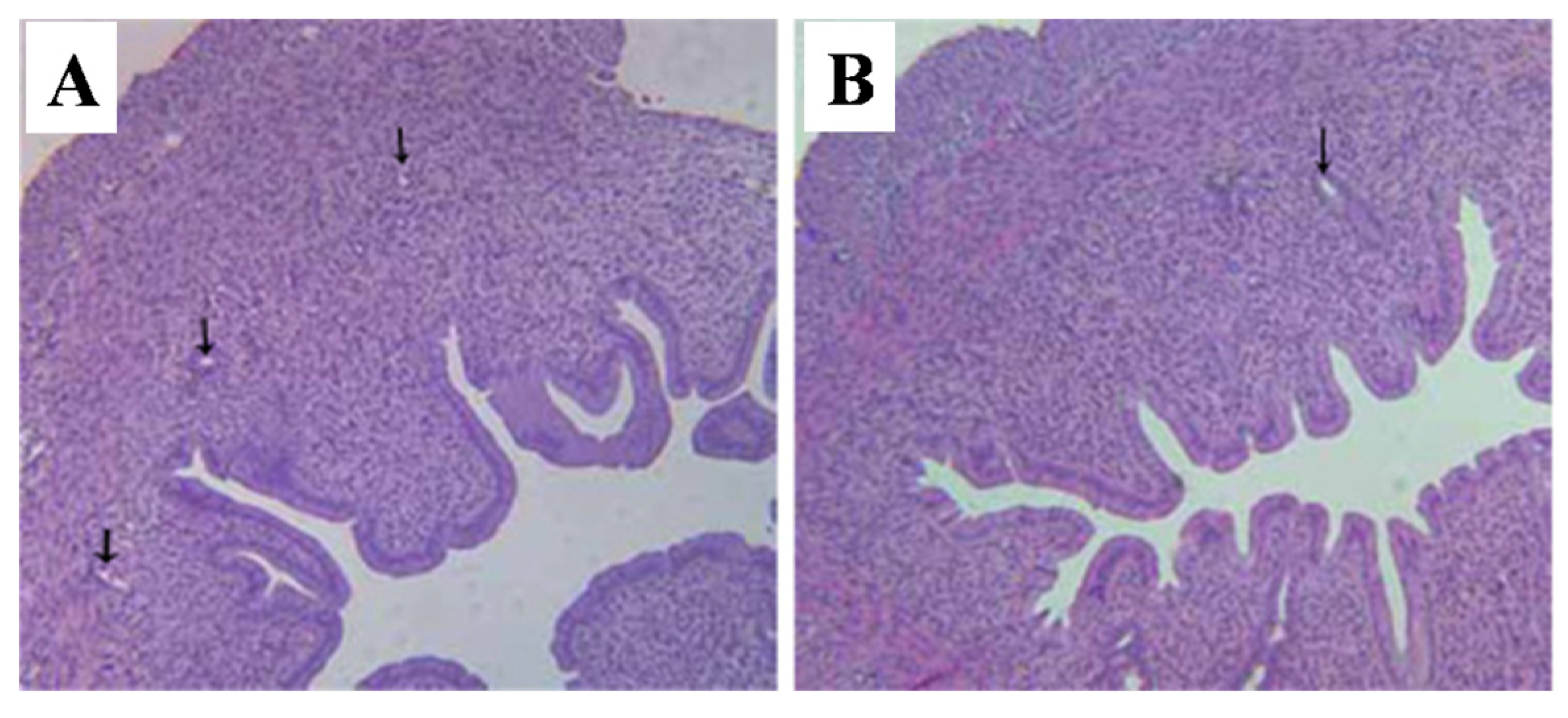
2.5.1. Uterus
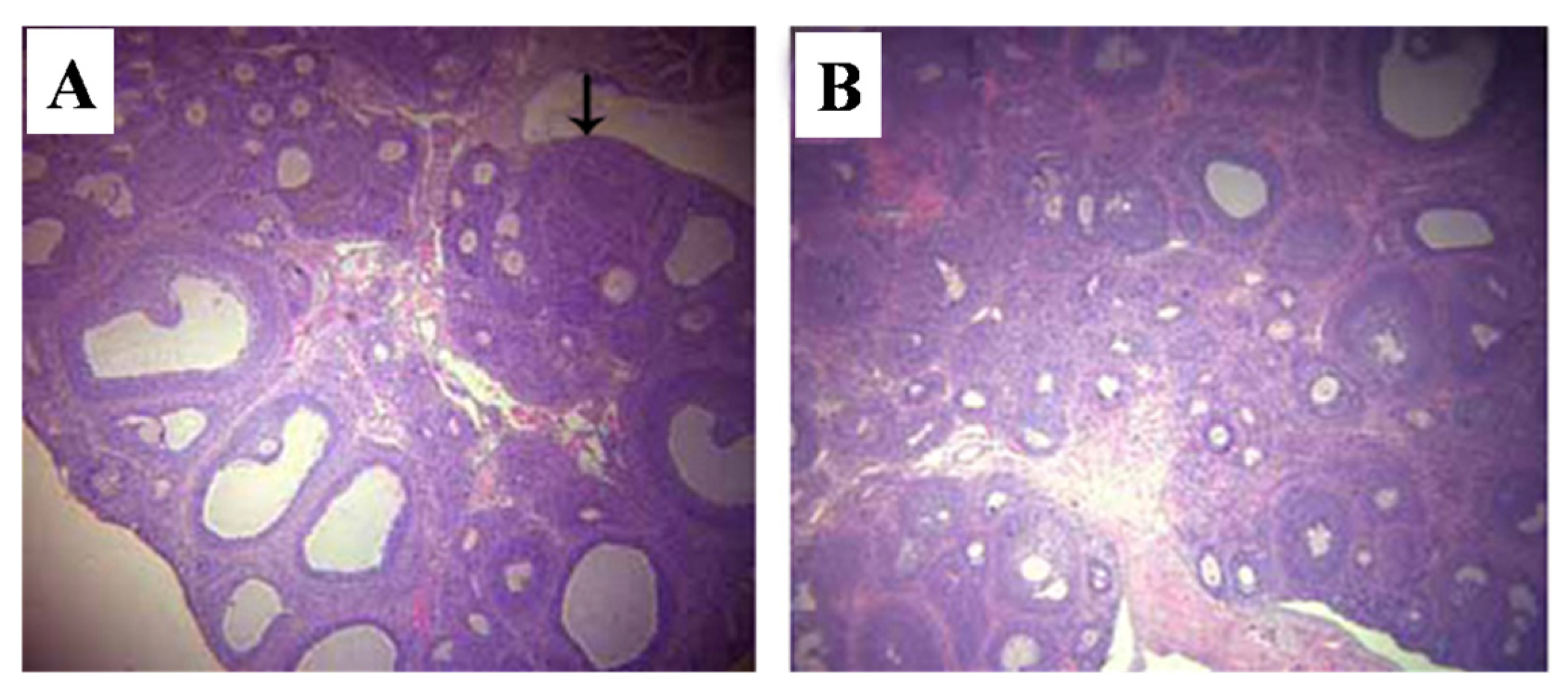
2.5.2. Ovaries
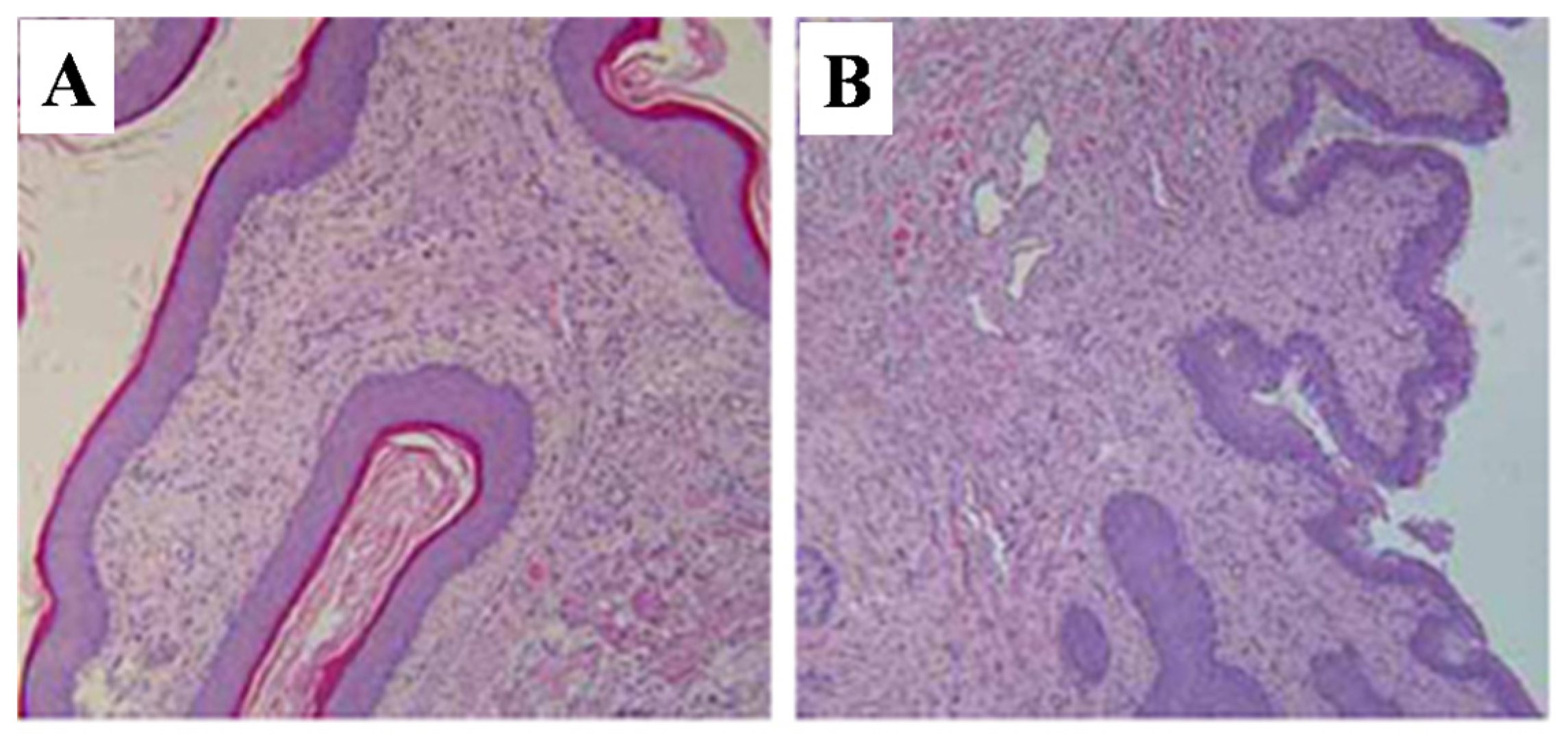
2.5.3. Vagina
2.6. Effects of Prepubertal Exposure to T-2 Toxin on the Levels of Serum Gonadotropins
| Groups | Serum Gonadotropins | ||
|---|---|---|---|
| LH (ng/mL) | FSH (ng/mL) | Estradiol (pg/mL) | |
| Vehicle control | 8.84 ± 0.39 | 9.65 ± 0.95 | 43.42 ± 4.58 |
| T-2 toxin treatment | 7.03 ± 0.17 ** | 8.19 ± 0.78 | 33.06 ± 3.04 * |
2.7. Effects of Prepubertal Exposure to T-2 Toxin on the mRNA Expressions of GnRH and GnRHR

3. Discussion
4. Experimental Section
4.1. Animals (Study Number 2011-005)
4.2. Chemicals and Reagents
4.3. Experimental Procedures
4.3.1. Mating and Experimental Design
4.3.2. Experiment I
4.3.3. Experiment II
4.4. Examination and Biochemical Determinations
4.4.1. Vaginal Opening Examination
4.4.2. Hormone Assays
4.4.3. RNA Extraction and RT-PCR
| Gene Name | Sequence | Product Size (bp) | Annealing Temperature (°C) | |
|---|---|---|---|---|
| GnRH | Sense | 5′-AGCACTGGTCCTATGGGT TG-3′ | 105 | 56 |
| Antisence | 5′-GGGGTTCTGCCATTTGATCCA-3′ | |||
| GnRHR | Sense | 5′-GTATGCTGGAGAGTACTCTGCA-3′ | 380 | 56 |
| Antisence | 5′-GGATGATGAAGAGGCAGCTGAAG-3′ | |||
| β-actin | Sense | 5′-TCGGTCATCACTATCGGCAAT-3′ | 550 | 56 |
| Antisence | 5′-GTATGCTGGAGAGTTACTCTGCA-3′ | |||
4.5. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kakarla, N.; Bradshaw, K.D. Disorders of pubertal development: Precocious puberty. Semin. Reprod. Med. 2003, 21, 339–352. [Google Scholar] [PubMed]
- Chulani, V.L.; Gordon, L.P. Adolescent growth and development. Prim. Care 2014, 41, 465–487. [Google Scholar] [CrossRef] [PubMed]
- Lam, T.; Williams, P.L.; Lee, M.M.; Korrick, S.A.; Birnbaum, L.S.; Burns, J.S.; Sergeyev, O.; Revich, B.; Altshul, L.M.; Patterson, D.G., Jr.; et al. Prepubertal organochlorine pesticide concentrations and age of pubertal onset among Russian boys. Environ. Int. 2014, 73, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Naicker, N.; Norris, S.A.; Mathee, A.; Becker, P.; Richter, L. Lead exposure is associated with a delay in the onset of puberty in South African adolescent females: Findings from the birth to twenty cohort. Sci. Total Environ. 2010, 408, 4949–4954. [Google Scholar] [CrossRef] [PubMed]
- Massart, F.; Meucci, V.; Saggese, G.; Soldani, G. High growth rate of girls with precocious puberty exposed to estrogenic mycotoxins. J. Pediatr. 2008, 152, 690–695. [Google Scholar] [CrossRef] [PubMed]
- Reilly, M.P.; Saca, J.C.; Hamilton, A.; Solano, R.F.; Rivera, J.R.; Whitehouse-Innis, W.; Parsons, J.G.; Dearth, R.K. Prepubertal exposure to arsenic(III) suppresses circulating insulin-like growth factor-1 (IGF-1) delaying sexual maturation in female rats. Reprod. Toxicol. 2014, 44, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Si, J.; Han, X.; Zhang, F.; Xin, Q.; An, L.; Li, G.; Li, C. Perinatal exposure to low doses of tributyltin chloride advances puberty and affects patterns of estrous cyclicity in female mice. Environ. Toxicol. 2012, 27, 662–670. [Google Scholar] [CrossRef] [PubMed]
- Nah, W.H.; Park, M.J.; Gye, M.C. Effects of early prepubertal exposure to bisphenol A on the onset of puberty, ovarian weights, and estrous cycle in female mice. Clin. Exp. Reprod. Med. 2011, 38, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Zorrilla, L.M.; Gibson, E.K.; Stoker, T.E. The effects of simazine, a chlorotriazine herbicide, on pubertal development in the female Wistar rat. Reprod. Toxicol. 2010, 29, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Nesic, K.; Ivanovic, S.; Nesic, V. Fusarial toxins: Secondary metabolites of Fusarium fungi. Rev. Environ. Contam. Toxicol. 2014, 228, 101–120. [Google Scholar] [PubMed]
- Cortinovis, C.; Pizzo, F.; Spicer, L.J.; Caloni, F. Fusarium mycotoxins: Effects on reproductive function in domestic animals—A review. Theriogenology 2013, 80, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Li, R.; Xiao, S.; Diao, H.; Viveiros, M.M.; Song, X.; Ye, X. Postweaning exposure to dietary zearalenone, a mycotoxin, promotes premature onset of puberty and disrupts early pregnancy events in female mice. Toxicol. Sci. 2013, 132, 431–442. [Google Scholar] [CrossRef] [PubMed]
- Ewuola, E.O.; Egbunike, G.N. Effects of dietary fumonisin B1 on the onset of puberty, semen quality, fertility rates and testicular morphology in male rabbits. Reproduction 2010, 139, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Gbore, F.A. Growth performance and puberty attainment in growing pigs fed dietary fumonisin B(1). J. Anim. Physiol. Anim. Nutr. 2009, 93, 761–767. [Google Scholar] [CrossRef] [PubMed]
- Doi, K.; Ishigami, N.; Sehata, S. T-2 toxin-induced toxicity in pregnant mice and rats. Int. J. Mol. Sci. 2008, 9, 2146–2158. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, W.; Cheng, G.; Huang, L.; Chen, D.; Tao, Y.; Pan, Y.; Hao, H.; Wu, Q.; Wan, D.; et al. High risk of embryo-fetal toxicity: Placental transfer of T-2 toxin and its major metabolite HT-2 toxin in BeWo cells. Toxicol. Sci. 2014, 137, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Sehata, S.; Kiyosawa, N.; Makino, T.; Atsumi, F.; Ito, K.; Yamoto, T.; Teranishi, M.; Baba, Y.; Uetsuka, K.; Nakayama, H.; et al. Morphological and microarray analysis of T-2 toxin-induced rat fetal brain lesion. Food Chem. Toxicol. 2004, 42, 1727–1736. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.Y.; Zhang, Y.F.; Liang, A.M.; Kong, X.F.; Li, Y.X.; Ma, K.W.; Jing, A.H.; Feng, S.Y.; Qiao, X.L. Toxic effects of T-2 toxin on reproductive system in male mice. Toxicol. Ind. Health 2010, 26, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Ndossi, D.G.; Frizzell, C.; Tremoen, N.H.; Fæste, C.K.; Verhaegen, S.; Dahl, E.; Eriksen, G.S.; Sørlie, M.; Connolly, L.; Ropstad, E. An in vitro investigation of endocrine disrupting effects of trichothecenes deoxynivalenol (DON), T-2 and HT-2 toxins. Toxicol. Lett. 2012, 214, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Tu, D.; Yuan, L.Y.; Yi, J.E.; Tian, Y. T-2 toxin regulates steroid hormone secretion of rat ovarian granulosa cells through cAMP-PKA pathway. Toxicol. Lett. 2015, 232, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, L.; Wang, Y.; Wu, R.; Hua, C.; Zhao, J.; Liu, Y.; Peng, S. Effects of low dose T-2 toxin on secretion of gonadotropin-releasing hormone in the immortalized hypothalamic GT1-7 cell line. Toxicon 2015, 100, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Smyth, C.E.; Knee, R.; Wilkinson, M.; Murphy, P.R. Decline in basic fibroblast growth factor (FGF-2) mRNA expression in female rat hypothalamus at puberty. J. Neuroendocrinol. 1997, 9, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Ojeda, S.R.; Skinner, M.K. Puberty in the rat. In The Physiology of Reproduction; Neill, J.D., Ed.; Academic Press/Elsevier: San Diego, CA, USA, 2006; pp. 2061–2126. [Google Scholar]
- Davis, L.K.; Murr, A.S.; Best, D.S.; Fraites, M.J.; Zorrilla, L.M.; Narotsky, M.G.; Stoker, T.E.; Goldman, J.M.; Cooper, R.L. The effects of prenatal exposure to atrazine on pubertal and postnatal reproductive indices in the female rat. Reprod. Toxicol. 2011, 32, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Kinouchi, R.; Matsuzaki, T.; Iwasa, T.; Gereltsetseg, G.; Nakazawa, H.; Kunimi, K.; Kuwahara, A.; Yasui, T.; Irahara, M. Prepubertal exposure to glucocorticoid delays puberty independent of the hypothalamic Kiss1-GnRH system in female rats. Int. J. Dev. Neurosci. 2012, 30, 596–601. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.Z.; Zhao, H.; Chen, B.Y. Decreased hypothalamic aromatization in female rats of true precocious puberty. Neurosci. Lett. 2004, 366, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Buck Louis, G.M.; Gray, L.E., Jr.; Marcus, M.; Ojeda, S.R.; Pescovitz, O.H.; Witchel, S.F.; Sippell, W.; Abbott, D.H.; Soto, A.; Tyl, R.W.; et al. Environmental factors and puberty timing: Expert panel research needs. Pediatrics 2008, 121, S192–S207. [Google Scholar] [CrossRef] [PubMed]
- Ponzo, O.J.; Szwarcfarb, B.; Rondina, D.; Carbone, S.; Reynoso, R.; Scacchi, P.; Moguilevsky, J.A. Changes in the sensitivity of gonadotrophin axis to leptin during sexual maturation in female rats. Neuro. Endocrinol. Lett. 2001, 22, 427–431. [Google Scholar] [PubMed]
- Dutlow, C.M.; Rachman, J.; Jacobs, T.W.; Millar, R.P. Prepubertal increase in gonadotropin-releasing hormone mRNA, gonadotropin-releasing hormone precursor, and subsequent maturation of precursor processing in male rats. J. Clin. InvestIG. 1992, 90, 2496–2501. [Google Scholar] [CrossRef] [PubMed]
- Urbanski, H.F.; Ojeda, S.R. Activation of luteinizing hormone release advances the onset of female puberty. Neuroendocrinology 1987, 46, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Thuvander, A.; Wikman, C.; Gadhasson, I. In vitro exposure of human lymphocytes to trichothecenes: Individual variation in sensitivity and effects of combined exposure on lymphocyte function. Food Chem. Toxicol. 1999, 37, 639–648. [Google Scholar] [CrossRef]
- Maruniakova, N.; Kadasi, A.; Sirotkin, A.V.; Bulla, J.; Kolesarova, A. T-2 toxin and its metabolite HT-2 toxin combined with insulin-like growth factor-I modify progesterone secretion by porcine ovarian granulosa cells. J. Environ. Sci. Health 2014, 49, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Weidner, M.; Hüwel, S.; Ebert, F.; Schwerdtle, T.; Galla, H.J.; Humpf, H.U. Influence of T-2 and HT-2 toxin on the blood-brain barrier in vitro: New experimental hints for neurotoxic effects. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Ravindran, J.; Agrawal, M.; Gupta, N.; Rao, P.V. Alteration of blood brain barrier permeability by T-2 toxin: Role of MMP-9 and inflammatory cytokines. Toxicology 2011, 280, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Genazzani, A.R.; Bernardi, F.; Monteleone, P.; Luisi, S.; Luisi, M. Neuropeptides, neurotransmitters, neurosteroids, and the onset of puberty. Ann. N. Y. Acad. Sci. 2000, 900, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Weekley, L.B.; O’Rear, C.E.; Kimbrough, T.D.; Llewellyn, G.C. Acute and chronic effects of the trichothecene mycotoxin T-2 on rat brain regional concentrations of serotonin, tryptophan, and tyrosine. Vet. Hum. Toxicol. 1989, 31, 221–224. [Google Scholar] [PubMed]
- Wang, J.; Fitzpatrick, D.W.; Wilson, J.R. Effect of dietary T-2 toxin on biogenic monoamines in discrete areas of the rat brain. Food Chem. Toxicol. 1993, 31, 191–197. [Google Scholar] [CrossRef]
- Taylor, M.J.; Smart, R.A.; Sharma, R.P. Relationship of the hypothalamic-pituitary-adrenal axis with chemically induced immunomodulation. I. Stress-like response after exposure to T-2 toxin. Toxicology 1989, 56, 179–195. [Google Scholar] [CrossRef]
- Dedes, I. Kisspeptins and the control of gonadotrophin secretion. Syst. Biol. Reprod. Med. 2012, 58, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Rhie, Y.J. Kisspeptin/G protein-coupled receptor-54 system as an essential gatekeeper of pubertal development. Ann. Pediatr. Endocrinol. Metab. 2013, 18, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Skorupskaite, K.; George, J.T.; Anderson, R.A. The kisspeptin-GnRH pathway in human reproductive health and disease. Hum. Reprod. Update 2014, 20, 485–500. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.; Chou, Y.Y.; Wu, Y.M.; Lin, C.C.; Lin, S.J.; Lee, C.C. Phthalates may promote female puberty by increasing kisspeptin activity. Hum. Reprod. 2013, 28, 2765–2773. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Du, G.; Zhang, W.; Huang, H.; Chen, D.; Wu, D.; Wang, X. Short-term neonatal/prepubertal exposure of dibutyl phthalate (DBP) advanced pubertal timing and affected hypothalamic kisspeptin/GPR54 expression differently in female rats. Toxicology 2013, 314, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Tena-Sempere, M. Kisspeptin/GPR54 system as potential target for endocrine disruption of reproductive development and function. Int. J. Androl. 2010, 33, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chang, F.; Bai, Y.; Chen, F.; Zhang, J.; Chen, L. Bisphenol A enhances kisspeptin neurons in anteroventral periventricular nucleus of female mice. J. Endocrinol. 2014, 221, 201–213. [Google Scholar] [CrossRef] [PubMed]
- OECD/OCDE 440. OECD Guideline for the Testing of Chemicals Uterotrophic Bioassay in Rodents: A Short-term Screening Test for Oestrogenic Properties. Adopted: 16 October 2007. Available online: http://www.oecd-ilibrary.org/docserver/download/9744001e.pdf?expires=1442589960&id=id&accname=guest&checksum=A3D94275E94C1228C4735E35D33226BD (accessed on 26 October 2015).
- Joint FAO/WHO Expert Committee on Food Additives (JECFA). Safety Evaluation of Certain Mycotoxins in Food—Prepared by the Fifty-Sixth Meeting of the Joint FAO/WHO Expert Committee on Food Additives; WHO Food Additives Series; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001; Volume 47. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion on the risks for animal and public health related to the presence of T-2 and HT-2 toxin in food and feed. EFSA J. 2011, 9, 2481–2665. [Google Scholar]
- Speijers, G.J.A. Precision of estimates of an ADI (or TDI or PTWI). Regul. Toxicol. Pharmacol. 1999, 30, S87–S93. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, N.; Arias, P.; Szwarcfarb, B.; Ponzo, O.; Carbone, S.; Moguilevsky, J.; Scacchi, P.; Reynoso, R.M. Reproductive axis response to repeated lipopolysaccharide administration in peripubertal female rats. J. Physiol. Biochem. 2010, 66, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jing, L.; Yuan, H.; Peng, S.Q. T-2 toxin induces apoptosis in ovarian granulosa cells of rats through reactive oxygen species-mediated mitochondrial pathway. Toxicol. Lett. 2011, 202, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Cano-Sancho, G.; Marín, S.; Ramos, A.J.; Sanchis, V. Exposure assessment of T2 and HT2 toxins in Catalonia (Spain). Food Chem. Toxicol. 2012, 50, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Sirot, V.; Fremy, J.M.; Leblanc, J.C. Dietary exposure to mycotoxins and health risk assessment in the second French total diet study. Food Chem. Toxicol. 2013, 52, 1–11. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, R.; Wang, Y.-M.; Zhang, L.-S.; Zhang, L.; Zhao, Z.-M.; Zhao, J.; Peng, S.-Q. Delay of the Onset of Puberty in Female Rats by Prepubertal Exposure to T-2 Toxin. Toxins 2015, 7, 4668-4683. https://doi.org/10.3390/toxins7114668
Yang R, Wang Y-M, Zhang L-S, Zhang L, Zhao Z-M, Zhao J, Peng S-Q. Delay of the Onset of Puberty in Female Rats by Prepubertal Exposure to T-2 Toxin. Toxins. 2015; 7(11):4668-4683. https://doi.org/10.3390/toxins7114668
Chicago/Turabian StyleYang, Rong, Yi-Mei Wang, Li-Shi Zhang, Li Zhang, Zeng-Ming Zhao, Jun Zhao, and Shuang-Qing Peng. 2015. "Delay of the Onset of Puberty in Female Rats by Prepubertal Exposure to T-2 Toxin" Toxins 7, no. 11: 4668-4683. https://doi.org/10.3390/toxins7114668
APA StyleYang, R., Wang, Y.-M., Zhang, L.-S., Zhang, L., Zhao, Z.-M., Zhao, J., & Peng, S.-Q. (2015). Delay of the Onset of Puberty in Female Rats by Prepubertal Exposure to T-2 Toxin. Toxins, 7(11), 4668-4683. https://doi.org/10.3390/toxins7114668