Ricin Trafficking in Cells
Abstract
:1. Introduction

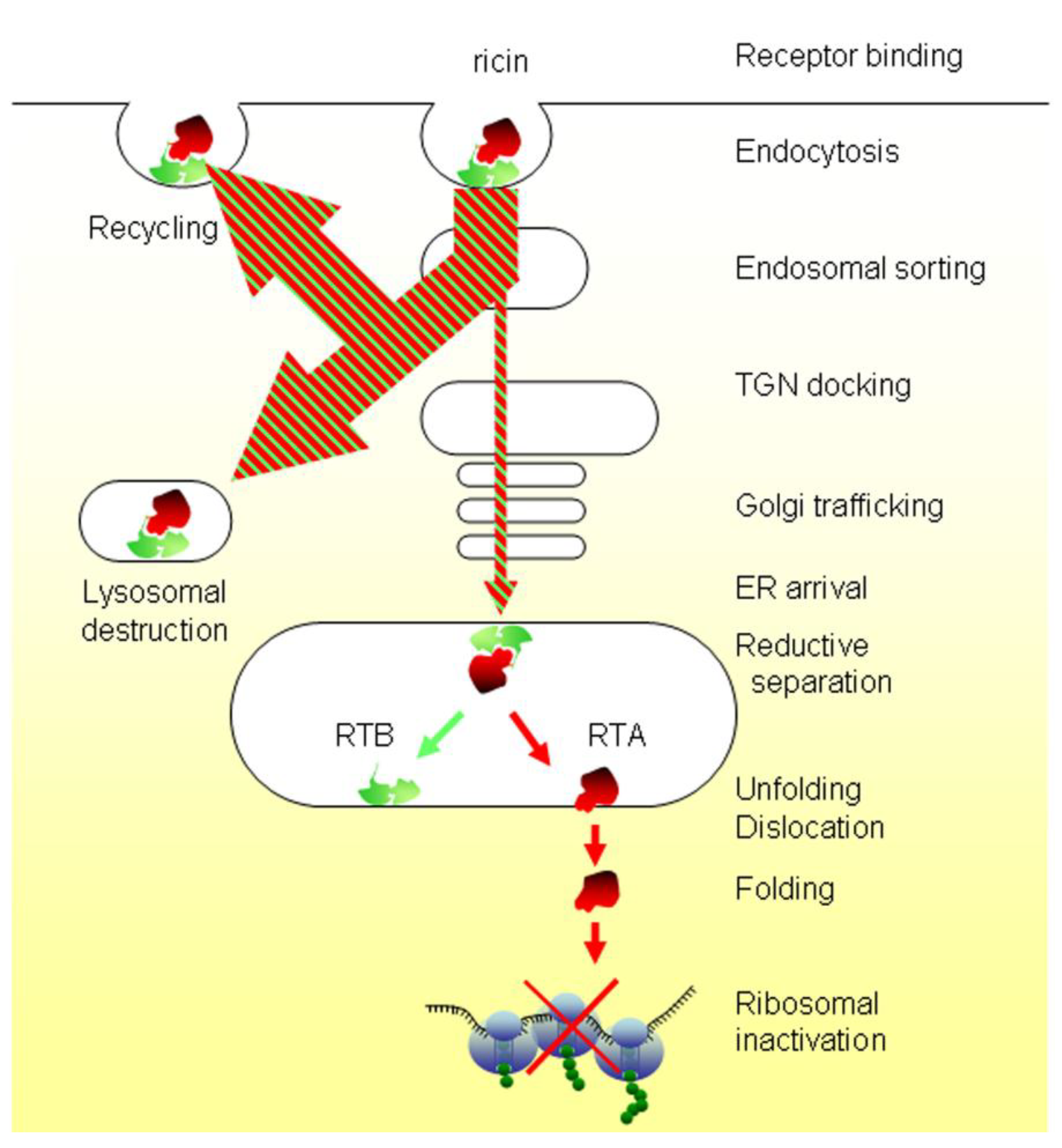
2. Er Luminal Events
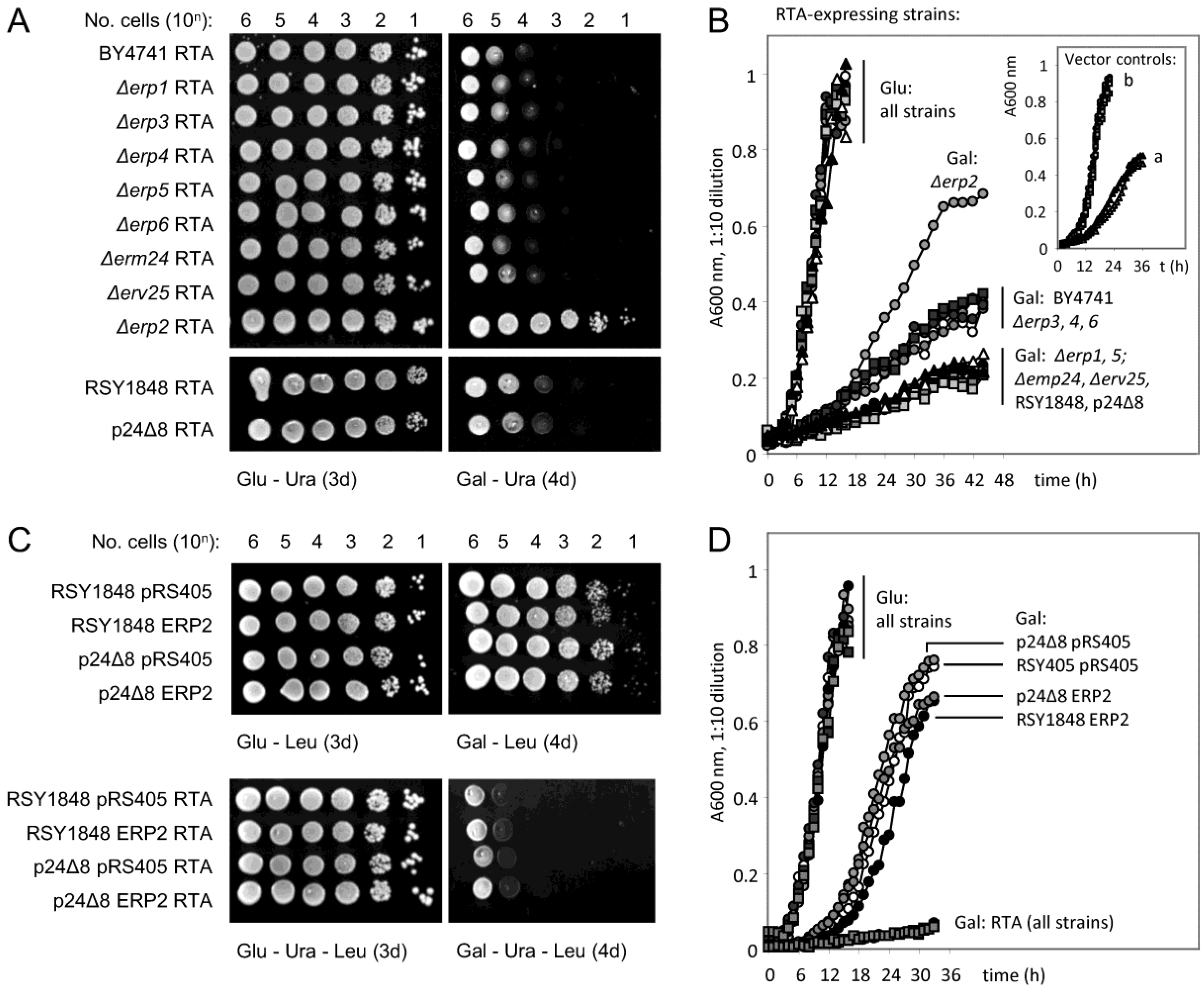
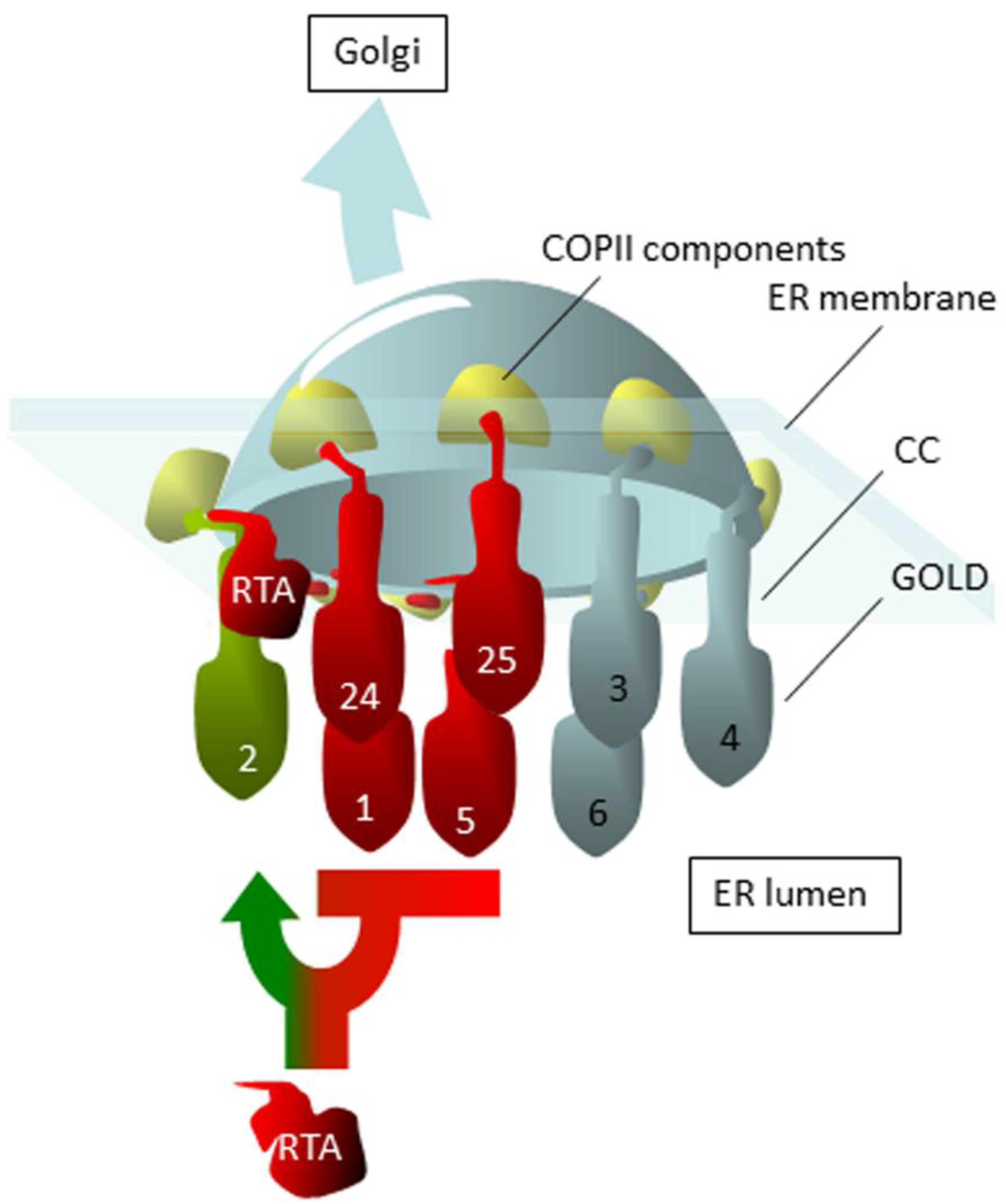
3. ER–Golgi Cycling Precedes Dislocation


4. ER-Cytosol Dislocation
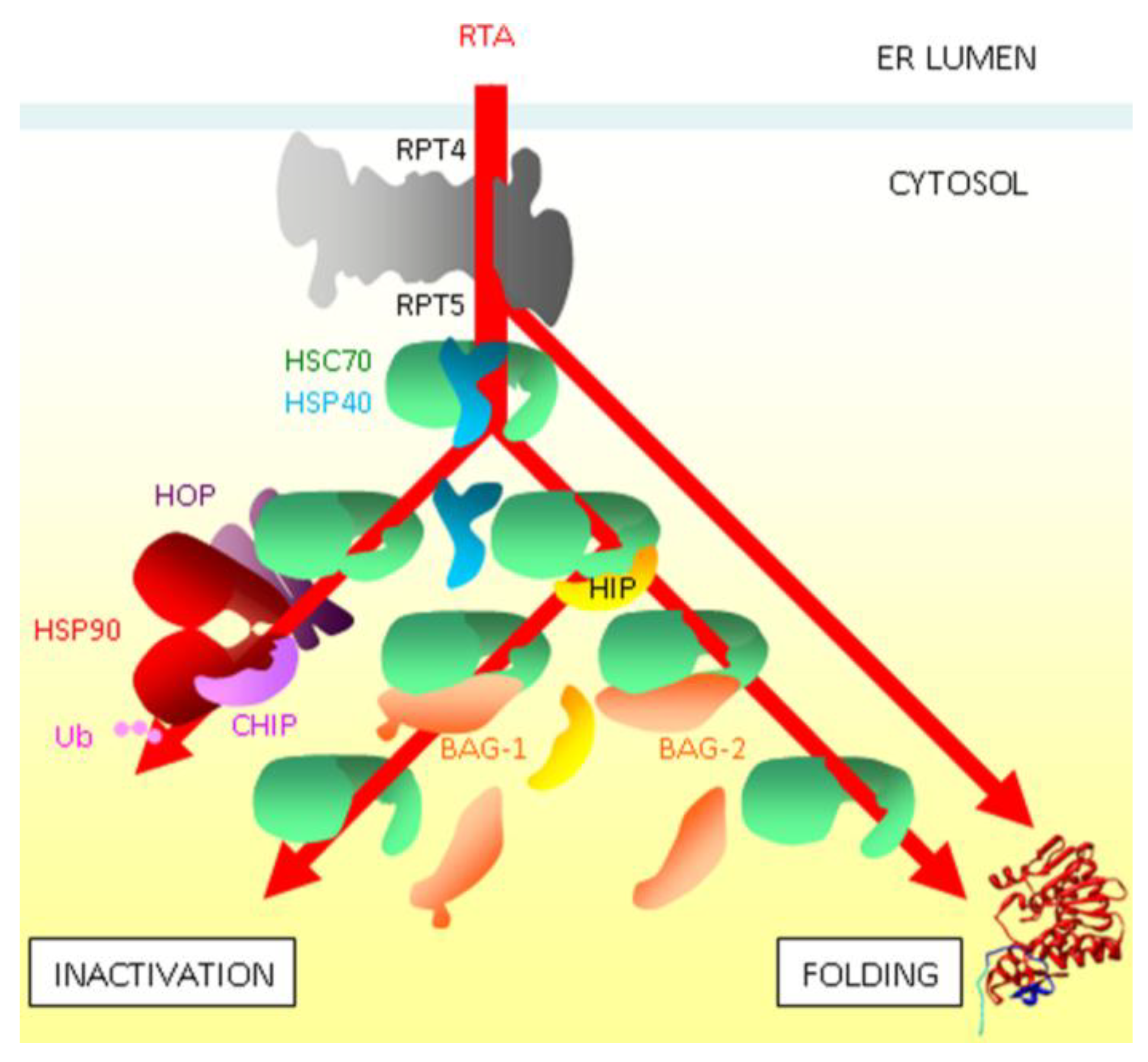
5. Cytosolic Triage
6. Concluding Remarks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Spilsberg, B.; van Meer, G.; Sandvig, K. Role of lipids in the retrograde pathway of ricin intoxication. Traffic 2003, 4, 544–552. [Google Scholar] [CrossRef]
- Spooner, R.A.; Watson, P.D.; Marsden, C.J.; Smith, D.C.; Moore, K.A.; Cook, J.P.; Lord, J.M.; Roberts, L.M. Protein disulphide-isomerase reduces ricin to its A and B chains in the endoplasmic reticulum. Biochem. J. 2004, 383, 285–293. [Google Scholar] [CrossRef]
- Sandvig, K.; Olsnes, S.; Pihl, A. Kinetics of binding of the toxic lectins abrin and ricin to surface receptors of human cells. J. Biol. Chem. 1976, 251, 3977–3984. [Google Scholar] [PubMed]
- Simpson, J.C.; Smith, D.C.; Roberts, L.M.; Lord, J.M. Expression of mutant dynamin protects cells against diphtheria toxin but not against ricin. Exp. Cell. Res. 1998, 239, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Moya, M.; Dautry-Varsat, A.; Goud, B.; Louvard, D.; Boquet, P. Inhibition of coated pit formation in Hep2 cells blocks the cytotoxicity of diphtheria toxin but not that of ricin toxin. J. Cell. Biol. 1985, 101, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Sandvig, K.; Olsnes, S.; Petersen, O.W.; van Deurs, B. Inhibition of endocytosis from coated pits by acidification of the cytosol. J. Cell. Biochem. 1988, 36, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.J.; Callewaert, N.; Contreras, R.; Khorana, H.G. Structure and function in rhodopsin: High-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl. Acad. Sci. USA 2002, 99, 13419–13424. [Google Scholar] [CrossRef] [PubMed]
- Crispin, M.; Chang, V.T.; Harvey, D.J.; Dwek, R.A.; Evans, E.J.; Stuart, D.I.; Jones, E.Y.; Lord, J.M.; Spooner, R.A.; Davis, S.J. A human embryonic kidney 293T cell line mutated at the Golgi alpha-mannosidase II locus. J. Biol. Chem. 2009, 284, 21684–21695. [Google Scholar] [CrossRef] [PubMed]
- Dang, H.; Klokk, T.I.; Schaheen, B.; McLaughlin, B.M.; Thomas, A.J.; Durns, T.A.; Bitler, B.G.; Sandvig, K.; Fares, H. Derlin-dependent retrograde transport from endosomes to the Golgi apparatus. Traffic 2011, 12, 1417–1431. [Google Scholar] [CrossRef] [PubMed]
- Elling, U.; Taubenschmid, J.; Wirnsberger, G.; O’Malley, R.; Demers, S.P.; Vanhaelen, Q.; Shukalyuk, A.I.; Schmauss, G.; Schramek, D.; Schnuetgen, F.; et al. Forward and reverse genetics through derivation of haploid mouse embryonic stem cells. Cell. Stem cell. 2011, 9, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Rutenber, E.; Katzin, B.J.; Ernst, S.; Collins, E.J.; Mlsna, D.; Ready, M.P.; Robertus, J.D. Crystallographic refinement of ricin to 2.5 A. Proteins 1991, 10, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Spooner, R.A.; Watson, P.; Smith, D.C.; Boal, F.; Amessou, M.; Johannes, L.; Clarkson, G.J.; Lord, J.M.; Stephens, D.J.; Roberts, L.M. The secretion inhibitor Exo2 perturbs trafficking of Shiga toxin between endosomes and the trans-Golgi network. Biochem. J. 2008, 414, 471–484. [Google Scholar] [CrossRef] [PubMed]
- Rapak, A.; Falnes, P.O.; Olsnes, S. Retrograde transporrt of mutant ricin to the endoplasmic reticulum with subsequent translocation to cytosol. Proc. Natl. Acad. Sci. USA 1997, 94, 3783–3788. [Google Scholar] [CrossRef] [PubMed]
- Wesche, J.; Rapak, A.; Olsnes, S. Dependence of ricin toxicity on translocation of the toxin A-chain from the endoplasmic reticulum to the cytosol. J. Biol. Chem. 1999, 274, 34443–34449. [Google Scholar] [CrossRef] [PubMed]
- Endo, Y.; Mitsui, K.; Motizuki, M.; Tsurugi, K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes. The site and the characteristics of the modification in 28 S ribosomal RNA caused by the toxins. J. Biol. Chem. 1987, 262, 5908–5912. [Google Scholar] [PubMed]
- Jandhyala, D.M.; Thorpe, C.M.; Magun, B. Ricin and Shiga toxins: effects on host cell signal transduction. Curr. Top. Microbiol. Immunol. 2012, 357, 41–65. [Google Scholar] [PubMed]
- Tesh, V.L. The induction of apoptosis by Shiga toxins and ricin. Curr. Top. Microbiol. Immunol. 2012, 357, 137–178. [Google Scholar]
- Llorente, A.; Rapak, A.; Schmid, S.L.; van Deurs, B.; Sandvig, K. Expression of mutant dynamin inhibits toxicity and transport of endocytosed ricin to the Golgi apparatus. J. Cell. Biol. 1998, 140, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Grimmer, S.; Iversen, T.G.; van Deurs, B.; Sandvig, K. Endosome to Golgi transport of ricin is regulated by cholesterol. Mol. Biol. Cell. 2000, 11, 4205–4216. [Google Scholar] [CrossRef] [PubMed]
- Day, P.J.; Owens, S.R.; Wesche, J.; Olsnes, S.; Roberts, L.M.; Lord, J.M. An interaction between ricin and calreticulin that may have implications for toxin trafficking. J. Biol. Chem. 2001, 276, 7202–7208. [Google Scholar] [CrossRef] [PubMed]
- Birkeli, K.A.; Llorente, A.; Torgersen, M.L.; Keryer, G.; Tasken, K.; Sandvig, K. Endosome-to-Golgi transport is regulated by protein kinase A type II alpha. J. Biol. Chem. 2003, 278, 1991–1997. [Google Scholar] [CrossRef] [PubMed]
- Utskarpen, A.; Slagsvold, H.H.; Iversen, T.G.; Walchli, S.; Sandvig, K. Transport of ricin from endosomes to the Golgi apparatus is regulated by Rab6A and Rab6A’. Traffic 2006, 7, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Grimmer, S.; Spilsberg, B.; Hanada, K.; Sandvig, K. Depletion of sphingolipids facilitates endosome to Golgi transport of ricin. Traffic 2006, 7, 1243–1253. [Google Scholar] [CrossRef] [PubMed]
- Pust, S.; Dyve, A.B.; Torgersen, M.L.; van Deurs, B.; Sandvig, K. Interplay between toxin transport and flotillin localization. PLoS One 2010, 5. [Google Scholar] [CrossRef] [PubMed]
- Iversen, T.G.; Skretting, G.; Llorente, A.; Nicoziani, P.; van Deurs, B.; Sandvig, K. Endosome to Golgi transport of ricin is independent of clathrin and of the Rab9- and Rab11-GTPases. Mol. Biol. Cell. 2001, 12, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Tcatchoff, L.; Andersson, S.; Utskarpen, A.; Klokk, T.I.; Skanland, S.S.; Pust, S.; Gerke, V.; Sandvig, K. Annexin A1 and A2: Roles in retrograde trafficking of Shiga toxin. PLoS One 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Van Deurs, B.; Petersen, O.W.; Olsnes, S.; Sandvig, K. Delivery of internalized ricin from endosomes to cisternal Golgi elements is a discontinuous, temperature-sensitive process. Exp. Cell. Res. 1987, 171, 137–152. [Google Scholar]
- Van Deurs, B.; Sandvig, K.; Petersen, O.W.; Olsnes, S.; Simons, K.; Griffiths, G. Estimation of the amount of internalized ricin that reaches the trans-Golgi network. J. Cell. Biol. 1988, 106, 253–267. [Google Scholar]
- Emmanuel, F.; Turpin, E.; Alfsen, A.; Frenoy, J.P. Separation of ricin A- and B-chains after dithiothreitol reduction. Anal. Biochem. 1988, 173, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.C.; Roberts, L.M.; Lord, J.M. Catalytic and cytotoxic activities of recombinant ricin A chain mutants with charged residues added at the carboxyl terminus. Protein Expr. Purif. 1995, 6, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Spooner, R.A.; Lord, J.M. How ricin and Shiga toxin reach the cytosol of target cells: Retrotranslocation from the endoplasmic reticulum. Curr. Top. Microbiol. Immunol. 2012, 357, 19–40. [Google Scholar] [PubMed]
- Bellisola, G.; Fracasso, G.; Ippoliti, R.; Menestrina, G.; Rosen, A.; Solda, S.; Udali, S.; Tomazzolli, R.; Tridente, G.; Colombatti, M. Reductive activation of ricin and ricin A-chain immunotoxins by protein disulfide isomerase and thioredoxin reductase. Biochem. Pharmacol. 2004, 67, 1721–1731. [Google Scholar] [CrossRef] [PubMed]
- Pasetto, M.; Barison, E.; Castagna, M.; della Cristina, P.; Anselmi, C.; Colombatti, M. Reductive activation of type 2 ribosome-inactivating proteins is promoted by transmembrane thioredoxin-related protein. J. Biol. Chem. 2012, 287, 7367–7373. [Google Scholar] [CrossRef] [PubMed]
- Irvine, A.G.; Wallis, A.K.; Sanghera, N.; Rowe, M.L.; Ruddock, L.W.; Howard, M.J.; Williamson, R.A.; Blindauer, C.A.; Freedman, R.B. Protein disulfide-isomerase interacts with a substrate protein at all stages along its folding pathway. PLoS One 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.S.; Tolleson, W.H.; Chirtel, S.J. Thermal inactivation of ricin using infant formula as a food matrix. J. Agric. Food Chem. 2006, 54, 7300–7304. [Google Scholar] [CrossRef] [PubMed]
- Fiani, M.L.; Blum, J.S.; Stahl, P.D. Endosomal proteolysis precedes ricin A-chain toxicity in macrophages. Arch. Biochem. Biophys. 1993, 307, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Blum, J.S.; Fiani, M.L.; Stahl, P.D. Proteolytic cleavage of ricin A chain in endosomal vesicles. Evidence for the action of endosomal proteases at both neutral and acidic pH. J. Biol. Chem. 1991, 266, 22091–22095. [Google Scholar] [PubMed]
- Spooner, R.A.; Hart, P.J.; Cook, J.P.; Pietroni, P.; Rogon, C.; Hohfeld, J.; Roberts, L.M.; Lord, J.M. Cytosolic chaperones influence the fate of a toxin dislocated from the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 2008, 105, 17408–17413. [Google Scholar] [CrossRef] [PubMed]
- Gregers, T.F.; Skanland, S.S.; Walchli, S.; Bakke, O.; Sandvig, K. BiP negatively affects ricin transport. Toxins 2013, 5, 969–982. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Spooner, R.A.; Allen, S.C.; Guise, C.P.; Ladds, G.; Schnöder, T.; Schmitt, M.J.; Lord, J.M.; Roberts, L.M. Folding-competent and folding-defective forms of ricin A chain have different fates after retrotranslocation from the endoplasmic reticulum. Mol. Biol. Cell. 2010, 21, 2543–2554. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.; Navarro-Garcia, F.; Huerta, J.; Burress, H.; Massey, S.; Ireton, K.; Teter, K. Hsp90 is required for transfer of the cholera toxin A1 subunit from the endoplasmic reticulum to the cytosol. J. Biol. Chem. 2010, 285, 31261–31267. [Google Scholar] [CrossRef] [PubMed]
- Redmann, V.; Gardner, T.; Lau, Z.; Morohashi, K.; Felsenfeld, D.; Tortorella, D. Novel class of potential therapeutics that target ricin retrograde translocation. Toxins 2014, 6, 33–53. [Google Scholar] [CrossRef]
- Slominska-Wojewodzka, M.; Gregers, T.F.; Walchli, S.; Sandvig, K. EDEM is involved in retrotranslocation of ricin from the endoplasmic reticulum to the cytosol. Mol. Biol. Cell. 2006, 17, 1664–1675. [Google Scholar] [CrossRef] [PubMed]
- Slominska-Wojewodzka, M.; Pawlik, A.; Sokolowska, I.; Antoniewicz, J.; Wegrzyn, G.; Sandvig, K. The role of EDEM2 compared with EDEM1 in ricin transport from the endoplasmic reticulum to the cytosol. Biochem. J. 2014, 457, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Sokolowska, I.; Walchli, S.; Wegrzyn, G.; Sandvig, K.; Slominska-Wojewodzka, M. A single point mutation in ricin A-chain increases toxin degradation and inhibits EDEM1-dependent ER retrotranslocation. Biochem. J. 2011, 436, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Mayerhofer, P.U.; Cook, J.P.; Wahlman, J.; Pinheiro, T.T.; Moore, K.A.; Lord, J.M.; Johnson, A.E.; Roberts, L.M. Ricin A chain insertion into endoplasmic reticulum membranes is triggered by a temperature increase to 37 °C. J. Biol. Chem. 2009, 284, 10232–10242. [Google Scholar] [CrossRef] [PubMed]
- Bernsel, A.; Viklund, H.; Hennerdal, A.; Elofsson, A. TOPCONS: Consensus prediction of membrane protein topology. Nucleic Acids Res. 2009, 37, W465–W468. [Google Scholar] [CrossRef] [PubMed]
- Hennerdal, A.; Elofsson, A. Rapid membrane protein topology prediction. Bioinformatics 2011, 27, 1322–1323. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.H.; Ploegh, H.L.; Weissman, J.S. Road to ruin: Targeting proteins for degradation in the endoplasmic reticulum. Science 2011, 334, 1086–1090. [Google Scholar] [CrossRef] [PubMed]
- Allen, S.C.; Moore, K.A.; Marsden, C.J.; Fulop, V.; Moffat, K.G.; Lord, J.M.; Ladds, G.; Roberts, L.M. The isolation and characterization of temperature-dependent ricin A chain molecules in Saccharomyces cerevisiae. FEBS J. 2007, 274, 5586–5599. [Google Scholar] [CrossRef] [PubMed]
- Parikh, B.A.; Tortora, A.; Li, X.P.; Tumer, N.E. Ricin inhibits activation of the unfolded protein response by preventing splicing of the HAC1 mRNA. J. Biol. Chem. 2008, 283, 6145–6153. [Google Scholar] [CrossRef] [PubMed]
- Budnik, A.; Stephens, D.J. ER exit sites—Localization and control of COPII vesicle formation. FEBS Lett. 2009, 583, 3796–3803. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.; Kim, W.; Belden, W.J.; Spear, E.D.; Barlowe, C.; Ng, D.T. Distinct retrieval and retention mechanisms are required for the quality control of endoplasmic reticulum protein folding. J. Cell. Biol. 2001, 155, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Wakana, Y.; Takai, S.; Nakajima, K.; Tani, K.; Yamamoto, A.; Watson, P.; Stephens, D.J.; Hauri, H.P.; Tagaya, M. Bap31 is an itinerant protein that moves between the peripheral endoplasmic reticulum (ER) and a juxtanuclear compartment related to ER-associated Degradation. Mol. Biol. Cell. 2008, 19, 1825–1836. [Google Scholar] [CrossRef] [PubMed]
- Strating, J.R.; Bouw, G.; Hafmans, T.G.; Martens, G.J. p24 Proteins from the same subfamily are functionally nonredundant. Biochimie 2011, 93, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Strating, J.R.; Martens, G.J. The p24 family and selective transport processes at the ER-Golgi interface. Biol. Cell 2009, 101, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Marzioch, M.; Henthorn, D.C.; Herrmann, J.M.; Wilson, R.; Thomas, D.Y.; Bergeron, J.J.; Solari, R.C.; Rowley, A. Erp1p and Erp2p, partners for Emp24p and Erv25p in a yeast p24 complex. Mol. Biol. Cell. 1999, 10, 1923–1938. [Google Scholar] [CrossRef] [PubMed]
- Elrod-Erickson, M.J.; Kaiser, C.A. Genes that control the fidelity of endoplasmic reticulum to Golgi transport identified as suppressors of vesicle budding mutations. Mol. Biol. Cell. 1996, 7, 1043–1058. [Google Scholar] [CrossRef] [PubMed]
- Springer, S.; Chen, E.; Duden, R.; Marzioch, M.; Rowley, A.; Hamamoto, S.; Merchant, S.; Schekman, R. The p24 proteins are not essential for vesicular transport in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 2000, 97, 4034–4039. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed]
- Page, R.D. TreeView: An application to display phylogenetic trees on personal computers. Compt. Appl. Biosci. 1996, 12, 357–358. [Google Scholar]
- Anantharaman, V.; Aravind, L. The GOLD domain, a novel protein module involved in Golgi function and secretion. Genome Biol. 2002, 3. [Google Scholar] [CrossRef]
- Ciufo, L.F.; Boyd, A. Identification of a lumenal sequence specifying the assembly of Emp24p into p24 complexes in the yeast secretory pathway. J. Biol. Chem. 2000, 275, 8382–8388. [Google Scholar] [CrossRef] [PubMed]
- Contreras, I.; Yang, Y.; Robinson, D.G.; Aniento, F. Sorting signals in the cytosolic tail of plant p24 proteins involved in the interaction with the COPII coat. Plant. Cell Physiol. 2004, 45, 1779–1786. [Google Scholar] [CrossRef] [PubMed]
- Stamnes, M.A.; Craighead, M.W.; Hoe, M.H.; Lampen, N.; Geromanos, S.; Tempst, P.; Rothman, J.E. An integral membrane component of coatomer-coated transport vesicles defines a family of proteins involved in budding. Proc. Natl. Acad. Sci. USA 1995, 92, 8011–8015. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Li, X.P.; Tumer, N.E. N-glycosylation does not affect the catalytic activity of ricin a chain but stimulates cytotoxicity by promoting its transport out of the endoplasmic reticulum. Traffic 2012, 13, 1508–1521. [Google Scholar] [CrossRef] [PubMed]
- Simpson, J.C.; Dascher, C.; Roberts, L.M.; Lord, J.M.; Balch, W.E. Ricin cytotoxicity is sensitive to recycling between the endoplasmic reticulum and the Golgi complex. J. Biol. Chem. 1995, 270, 20078–20083. [Google Scholar] [CrossRef] [PubMed]
- Pawar, V.; De, A.; Briggs, L.; Omar, M.M.; Sweeney, S.T.; Lord, J.M.; Roberts, L.M.; Spooner, R.A.; Moffat, K.G. RNAi screening of Drosophila (Sophophora) melanogaster S2 cells for ricin sensitivity and resistance. J. Biomol. Screen 2011, 16, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Kanehara, K.; Xie, W.; Ng, D.T. Modularity of the Hrd1 ERAD complex underlies its diverse client range. J. Cell. Biol. 2010, 188, 707–716. [Google Scholar] [CrossRef] [PubMed]
- Redmann, V.; Oresic, K.; Tortorella, L.L.; Cook, J.P.; Lord, M.; Tortorella, D. Dislocation of ricin toxin A chains in human cells utilizes selective cellular factors. J. Biol. Chem. 2011, 286, 21231–21238. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Spooner, R.A.; Hampton, R.Y.; Lord, J.M.; Roberts, L.M. Cytosolic entry of Shiga-like toxin a chain from the yeast endoplasmic reticulum requires catalytically active Hrd1p. PLoS One 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Sato, B.K.; Schulz, D.; Do, P.H.; Hampton, R.Y. Misfolded membrane proteins are specifically recognized by the transmembrane domain of the Hrd1p ubiquitin ligase. Mol. Cell. 2009, 34, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Hazes, B.; Read, R.J. Accumulating evidence suggests that several AB-toxins subvert the endoplasmic reticulum-associated protein degradation pathway to enter target cells. Biochemistry 1997, 36, 11051–11054. [Google Scholar] [CrossRef] [PubMed]
- Bazirgan, O.A.; Garza, R.M.; Hampton, R.Y. Determinants of RING-E2 fidelity for Hrd1p, a membrane-anchored ubiquitin ligase. J. Biol. Chem. 2006, 281, 38989–39001. [Google Scholar] [CrossRef] [PubMed]
- Kikkert, M.; Doolman, R.; Dai, M.; Avner, R.; Hassink, G.; van Voorden, S.; Thanedar, S.; Roitelman, J.; Chau, V.; Wiertz, E. Human HRD1 is an E3 ubiquitin ligase involved in degradation of proteins from the endoplasmic reticulum. J. Biol. Chem. 2004, 279, 3525–3534. [Google Scholar] [CrossRef] [PubMed]
- Bar-Nun, S. The role of p97/Cdc48p in endoplasmic reticulum-associated degradation: From the immune system to yeast. Curr. Top. Microbiol. Immunol. 2005, 300, 95–125. [Google Scholar] [PubMed]
- Tsai, Y.C.; Weissman, A.M. Ubiquitylation in ERAD: Reversing to go forward? PLoS Biol. 2011, 9. [Google Scholar] [CrossRef]
- Ernst, R.; Claessen, J.H.; Mueller, B.; Sanyal, S.; Spooner, E.; van der Veen, A.G.; Kirak, O.; Schlieker, C.D.; Weihofen, W.A.; Ploegh, H.L. Enzymatic blockade of the ubiquitin-proteasome pathway. PLoS Biol. 2011, 8. [Google Scholar] [CrossRef]
- Lipson, C.; Alalouf, G.; Bajorek, M.; Rabinovich, E.; Atir-Lande, A.; Glickman, M.; Bar-Nun, S. A proteasomal ATPase contributes to dislocation of endoplasmic reticulum-associated degradation (ERAD) substrates. J. Biol. Chem. 2008, 283, 7166–7175. [Google Scholar] [CrossRef] [PubMed]
- Deeks, E.D.; Cook, J.P.; Day, P.J.; Smith, D.C.; Roberts, L.M.; Lord, J.M. The low lysine content of ricin A chain reduces the risk of proteolytic degradation after translocation from the endoplasmic reticulum to the cytosol. Biochemistry 2002, 41, 3405–3413. [Google Scholar] [CrossRef] [PubMed]
- Pietroni, P.; Vasisht, N.; Cook, J.P.; Roberts, D.M.; Lord, J.M.; Hartmann-Petersen, R.; Roberts, L.M.; Spooner, R.A. The proteasome cap RPT5/Rpt5p subunit prevents aggregation of unfolded ricin A chain. Biochem. J. 2013, 453, 435–445. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spooner, R.A.; Lord, J.M. Ricin Trafficking in Cells. Toxins 2015, 7, 49-65. https://doi.org/10.3390/toxins7010049
Spooner RA, Lord JM. Ricin Trafficking in Cells. Toxins. 2015; 7(1):49-65. https://doi.org/10.3390/toxins7010049
Chicago/Turabian StyleSpooner, Robert A., and J. Michael Lord. 2015. "Ricin Trafficking in Cells" Toxins 7, no. 1: 49-65. https://doi.org/10.3390/toxins7010049
APA StyleSpooner, R. A., & Lord, J. M. (2015). Ricin Trafficking in Cells. Toxins, 7(1), 49-65. https://doi.org/10.3390/toxins7010049




