Canine Cyanotoxin Poisonings in the United States (1920s–2012): Review of Suspected and Confirmed Cases from Three Data Sources
Abstract
:1. Introduction
2. Results and Discussion
2.1. Harmful Algal Bloom-Related Illness Surveillance System (HABISS)
| Website address | Owner | Date of access |
|---|---|---|
| State Resources | ||
| http://myfwc.com/research/redtide/task-force/reports-presentations/resource-guide/ | Florida Fish and Wildlife Conservation Commission | 3 May 13 |
| http://www.kdheks.gov/algae-illness/index.htm | Kansas Department of Health and Environment | 3 May 13 |
| http://www.deq.virginia.gov/Programs/Water/WaterQualityInformationTMDLs/WaterQualityMonitoring/VirginiaHarmfulAlgalBloomTaskForce.aspx | Virginia Department of Environmental Quality | 3 May 13 |
| Federal Agency Resources | ||
| http://www.cdc.gov/nceh/hsb/hab/default.htm | Centers for Disease Control and Prevention | 3 May 13 |
| http://www.epa.gov/gmpo/habpage.html | U.S. Environmental Protection Agency | 3 May 13 |
| http://pubs.usgs.gov/fs/2006/3147/pdf/FS2006_3147.pdf | U.S. Geological Survey | 3 May 13 |
| Private Entity Resources | ||
| http://www.mote.org/index.php?src=faq&refno=142&category=Florida%20red%20tide | Mote Marine Laboratory | 3 May 13 |
| http://www.whoi.edu/redtide/ | Woods Hole Oceanographic Institute | 3 May 13 |
2.2. Suspected and Confirmed Canine CyanoHAB Intoxication Cases from the Veterinary Medical Teaching Hospital (VMTH) Necropsy and Biopsy Case Records, University of California, Davis
2.3. Historical Reports from a Review of Scientific Publications, Media, and Other Electronically Available Sources
| # | Year | State | Reported Exposure | Cyanotoxin | # Dead | Breed 1 | #Sick | Breed 1 | Ref |
|---|---|---|---|---|---|---|---|---|---|
| 1 | late 1920s | CA | 0 | 1 | [25] | ||||
| 2 | 1944 | IA | Anabaena flos-aquae | 1 | [26,27,28,29] | ||||
| 3 | 1948 | MN | A. lemmermannii | 2 | [26] | ||||
| 4 | 1948 | MN | Microcystis aeruginosa | 2 | [26,29] | ||||
| 5 | 1948 | IA | Anabaena flosaquae | 2 | [26,28,29] | ||||
| 6 | 1952 | IA | Anabaena flos-aquae | 15 | [26,28,30,31] | ||||
| 7 | 1969 | FL | Microcystis aeruginosa2 | 1 | German Shepherd | [32] | |||
| 8 | 1976 | WA | Anabaena flos-aquae | 4 | 7 | [33,34,35,36] | |||
| 9 | 1977 | MT | Anabaena flos-aquae | Anatoxin-a | 8 | [37,38,39] | |||
| 10 | 1977 | WA | Anabaena flos-aquae | 1 | [40] | ||||
| 11 | 1978 | WA | Anabaena flos-aquae | 0 | 1 | German Shepherd | [34] | ||
| 12 | 1979 | PA | Anabaena | 0 | 1 | [41] | |||
| 13 | 1980 | MT | Cyanobacteria | 2 | [42] | ||||
| 14 | 1981 | ID | Anabaena flos-aquae | Anatoxin-a | 2 | [43,44,45] | |||
| 15 | 1982 | WA | Anabaena flos-aquae | 2 | Black Labrador (2) | 1 | [36,44] | ||
| 16 | 1985 | WI | 2 | Collie, Labrador | 1 | [46,47] | |||
| 17 | 1985 | SD | Anabaena flos-aquae | Anatoxin-a(s) | 9 | German Shepherd | [48] | ||
| 18 | 1985 | SD | Anabaena flos-aquae | Anatoxin-a(s) | 5 | [48] | |||
| 19 | 1986 | WI | Cyanobacteria | 3 | [49] | ||||
| 20 | 1986 | NJ | Cyanobacteria | 1 | [50] | ||||
| 21 | 1989 | WI | Cyanobacteria | 0 | 2 | [51] | |||
| 22 | 1989–1990 | WA | Anabaena flos-aquae | 1 | 5 | [52,53,54] | |||
| 23 | 1990s | UT | Cyanobacteria | 2 | [55] | ||||
| 24 | 1990 | MS | Blue-green algae | 14 | [56,57] | ||||
| 25 | 1990 | IN | Anabaena flos-aquae | Anatoxin-a | 2 | [58] | |||
| 26 | 1991 | SD | Blue-green algae | 1 | [59] | ||||
| 27 | 1991 | OR | Anabaena | 5 | English Springer Spaniels (2), Cocker Spaniel | [60,61,62] | |||
| 28 | 1991 | CA | Microcystis aeruginosa | 1 | Golden Retriever | [24] | |||
| 29 | 1997 | WA | Cyanobacteria | 1 | Brittany Spaniel | [63] | |||
| 30 | 1997 | CA | Blue-green algae | 1 | Field Spaniel | [64] | |||
| 31 | 1997 | WA | Microcystis aeruginosa | Microcystins | 1 | Golden Retriever | [65,66] | ||
| 32 | 1998 | MN | Blue-green algae | 1 | [67] | ||||
| 33 | 1998 | MD | Microcystis aeruginosa2 | 2 | [68] | ||||
| 34 | 1998 | MA | Anabaena sp. | 2 | 2 | Black Labrador mix | [69,70] | ||
| 35 | 1999 | NY | Anatoxin-a 2 | 2 | Black Labrador-Golden Retriever mix, Chocolate Labrador | [71,72,73,74] | |||
| 36 | 1999 | VT | Cyanobacteria | 2 | Labrador Retriever | [72,75] | |||
| 37 | 1999 | ID | Cyanobacteria | 6 | [76,77] | ||||
| 38 | 1999 | ID | Cyanotoxin | 1 | [78,79] | ||||
| 39 | 2000 | NY | Blue-green algae | Anatoxin-a | 2 | [73] | |||
| 40 | 2000 | ID | Blue-green algae | 3 | Chocolate Labrador Retriever | [80,81,82] | |||
| 41 | 2000–2001 | OR | Cyanobacteria | 2 | [36] | ||||
| 42 | 2001 | CA | 5 | Australian Shepherd-English Setter mix, Mutt | [36,83,84] | ||||
| 43 | 2002 | NM | Lyngbya (“Mermaid’s hair”) | Neosaxitoxin | 0 | 1 | Golden Retriever | [85,86] | |
| 44 | 2002 | NM | Lyngbya (“Mermaid’s hair”) | 1 | [85] | ||||
| 45 | 2002 | CA | Anabaena, Lyngbya, Planktothrix | Anatoxin-a | 1 | [83,87] | |||
| 46 | 2002 | CA | Anabaena, Lyngbya, Planktothrix | Anatoxin-a | 2 | [83,87] | |||
| 47 | 2002 | VT | Microcystins, Anatoxin-a | 1 | [73,88,89] | ||||
| 48 | 2003 | WI | Blue-green algae 2 | 1 | [90] | ||||
| 49 | 2003 | SD | 2 | Chocolate Labrador | [91] | ||||
| 50 | 2004 | NE | Anabaena | Microcystins | 3 | [92,93,94,95] | |||
| 51 | 2004 | NE | Cyanobacteria | 3 | Yellow Labrador, Sheep-dog, Australian Shepherd | [95,96,97] | |||
| 52 | 2004 | NY | Anabaena, Microcystis | Microcystins, Anatoxin-a | 1 | Labrador Retriever | 1 | [98,99,100] | |
| 53 | 2004 | WI | Anabaena, Microcystis, Aphanizomenon | 1 | Labrador Retriever | 1 | [101,102,103] | ||
| 54 | 2004 | MN | Cyanobacteria | 1 | [104] | ||||
| 55 | 2004 | CA | Cyanotoxin 2 | 1 | [83] | ||||
| 56 | 2004 | ID | Cyanobacteria | 1 | Labrador | 1 | [105] | ||
| 57 | 2004 | MN | Microcystis2 | 1 | [106] | ||||
| 58 | 2004 | WA | 1 | Chocolate Labrador-Doberman mix | [107,108] | ||||
| 59 | 2005 | WA | Anabaena | 3 | [36,108] | ||||
| 60 | 2005 | WI | 1 | [109] | |||||
| 61 | 2005 | IA | Cyanobacteria | 0 | 1 | [110] | |||
| 62 | 2006 | NE | Anabaena sp. | Anatoxin-a | 2 | German Shorthair Pointer | [111,112] | ||
| 63 | 2006 | WA | Anabaena, Microcystis, Aphanizomenon | Anatoxin-a | 2 | 1 | [36,113,114] | ||
| 64 | 2006 | ID | Cyanobacteria | 0 | 1 | [115] | |||
| 65 | 2007 | MN | Cyanobacteria | 1 | Yellow Labrador | [116,117,118] | |||
| 66 | 2007 | MI | Microcystin 2 | 1 | Border collie | [119,120] | |||
| 67 | 2007 | MN | Cyanobacteria | 1 | [121] | ||||
| 68 | 2007 | KS | 3 | 1 | [122] | ||||
| 69 | 2007 | MT | Toxic blue-green algae | 1 | [123] | ||||
| 70 | 2007 | MT | Toxic blue-green algae | 0 | 1 | [123] | |||
| 71 | 2007 | WA | Cyanobacteria | 2 | Hunting dogs (2) | [124] | |||
| 72 | 2007 | MN | Cyanobacteria | 3 | Golden Retriever, Cocker Spaniel, Bernese Mountain Dog | [121,125] | |||
| 73 | 2007 | NM | Blue-green algae | 1 | Jack Russell Terrier | [126,127,128] | |||
| 74 | 2007 | WI | Blue-green algae | 1 | Brittany Spaniel | [129] | |||
| 75 | 2007 | WA | Anatoxin-a 2 | 1 | [114] | ||||
| 76 | 2008 | KS | Blue-green algae | 1 | Australian Shepherd | 2 | Australian Shepherd, Labradoodle | [130] | |
| 77 | 2008 | MT | Cyanobacteria | 1 | [131] | ||||
| 78 | 2008 | WA | 2 | Labrador Retriever (2) | [114] | ||||
| 79 | 2008 | WA | 2 | 1 | [114] | ||||
| 80 | 2008 | MN | Cyanobacteria | 3 | [132] | ||||
| 81 | 2009 | WI | Cyanobacteria | 2 | [133] | ||||
| 82 | 2009 | WI | Cyanobacteria | 1 | Australian Terrier | [134] | |||
| 83 | 2009 | WA | Blue-green algae | 0 | 2 | [135] | |||
| 84 | 2009 | NM | 2 | Pit Bull (2) | [136,137] | ||||
| 85 | 2009 | ND | Microcystis | 1 | [138] | ||||
| 86 | 2009 | CA | Cyanobacteria | 1 | Blue Heeler cross | [139] | |||
| 87 | 2009 | CA | Blue-green algae | 1 | [140] | ||||
| 88 | 2009 | OR | Anatoxin-a | 4 | Border Collie, Labrador mix, Husky, Blue Heeler | [141,142,143,144,145] | |||
| 89 | 2009 | MN | 1 | Rat Terrier | [146] | ||||
| 90 | 2009 | TX | 0 | 2 | [147] | ||||
| 91 | 2009 | NM | 1 | Labrador | [136] | ||||
| 92 | 2009 | MN | Blue-green algae | 1 | Black Labrador | [148,149,150,151] | |||
| 93 | 2009 | WA | Blue-green algae 2 | 1 | Labrador Retriever | [152] | |||
| 94 | 2009 | IN | Blue-green algae 2 | 1 | Golden Retriever | [153] | |||
| 95 | 2010 | WI | 1 | [154] | |||||
| 96 | 2010 | WI | Blue-green algae | 1 | 1 | [154] | |||
| 97 | 2010 | OH | Cyanobacteria | 3 | Black Labrador Retriever, Rat Terrier, Golden Retriever | 1 | Cairn Terrier | [155,156] | |
| 98 | 2010 | OH | 2 | [157] | |||||
| 99 | 2010 | ND | 3 types toxic algae | 1 | [158] | ||||
| 100 | 2010 | OR | Blue-green algae | Anatoxin-a | 1 | Labrador | [145,159] | ||
| 101 | 2010 | NY | Blue-green algae | 1 | [160] | ||||
| 102 | 2010 | MT | Blue-green algae | 1 | Australian Shepherd | [131,161] | |||
| 103 | 2011 | MA | Blue-green algae | 0 | 1 | [162] | |||
| 104 | 2011 | OR | Algal scum | 1 | Jack Russell Terrier | [163] | |||
| 105 | 2011 | KS | Cyanobacteria | 3 | German Shepherd? | 1 | [164] | ||
| 106 | 2011 | OR | Green scum | 1 | Springer Spaniel | [165] | |||
| 107 | 2011 | OH | Blue green algae | Negative for microcystin | 1 | Labrador Retriever mix | [166,167] | ||
| 108 | 2011 | KS | Microcystis | 1 | Briard | [168] | |||
| 109 | 2012 | OK | Blue green algae | 2 | [169] | ||||
| 110 | 2012 | WI | 1 | [170] | |||||
| 111 | 2012 | IN | Blue green algae | 2 | Short-Haired Pointer, Labrador mix | 2 | [171,172] | ||
| 112 | 2012 | NY | 2 | [173] | |||||
| 113 | 2012 | CA | 0 | 1 | [174] | ||||
| 114 | unknown | CA | 1 | Pit Bull | [175] | ||||
| 115 | unknown | CA | 0 | 1 | [176] | ||||
| Total | 215 | 45 |
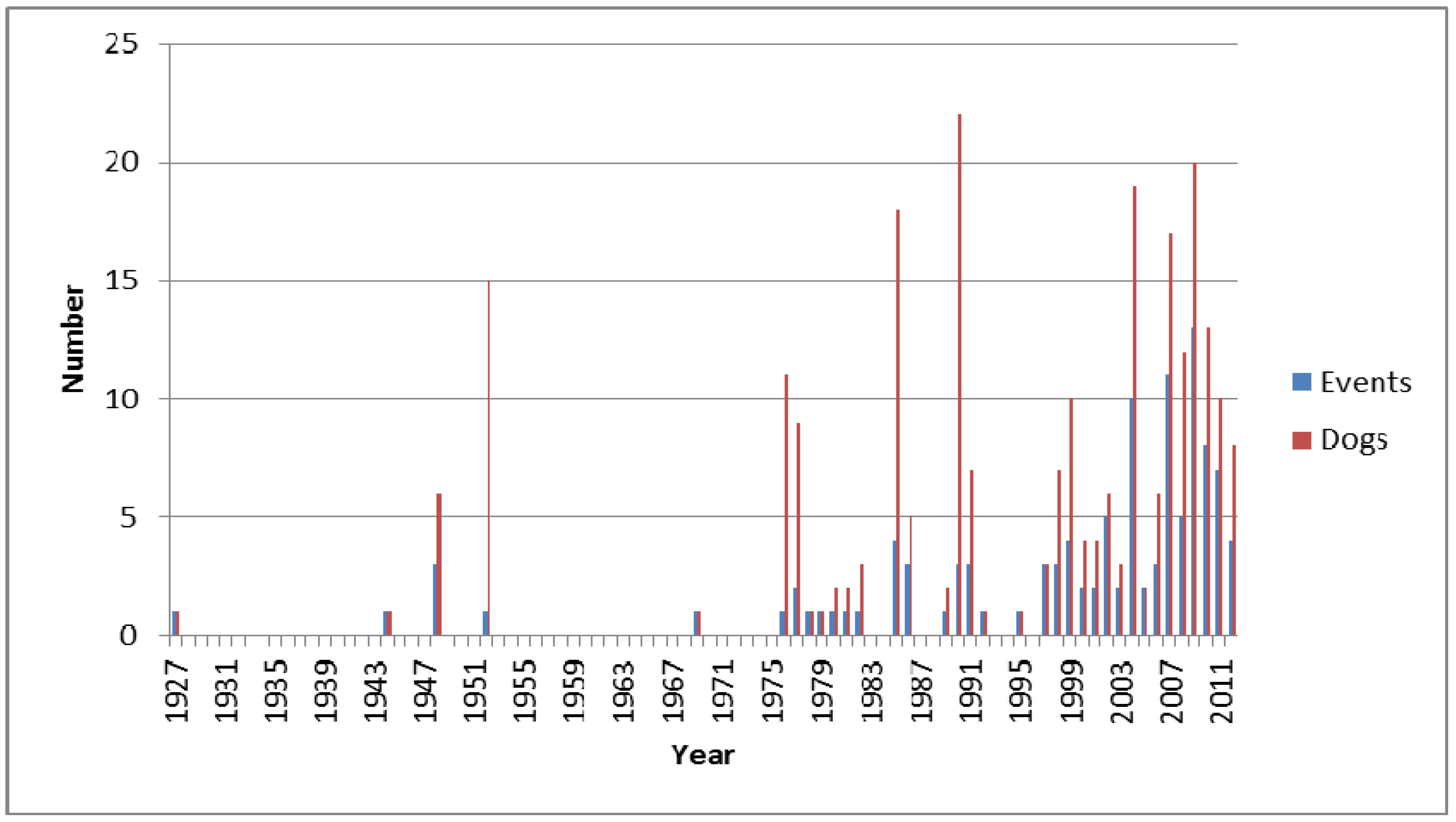
2.4. Dogs, CyanoHABs, and Public Health
| Number reported | Source of reports Dates of reports | ||
|---|---|---|---|
| HABISS 2007–2011 | Media Search Late 1920s–2012 | VMTH 1984–2011 | |
| Number of discrete cyanoHAB events associated with canine illness or death reported during the monitoring period | 55 | 115 | 44 |
| Number of sick or dead dogs attributed to cyanotoxin exposure across all reported cyanoHAB events during the monitoring period | 63 | 260 | 45 |
| Number of sick or dead dogs attributed to anatoxin-a or anatoxin-a(s) exposure | 12 | 44 | 2 |
| Number of sick or dead dogs attributed to microcystin or other hepatotoxic cyanotoxin exposure | 3 | 5 | 43 |
| Number (%) of cases where cyanoHAB intoxication was biochemically confirmed | 8 (13%) | 20 (8%) | 2 (4%) |
| Number (%) of cases that were published in peer-reviewed scientific literature | 0 | 62 (25%) | 1 (2%) |
3. Experimental Section
3.1. Harmful Algal Bloom-Related Illness Surveillance System (HABISS)
3.2. Potential and Confirmed Canine CyanoHAB Cases from the Veterinary Medical Teaching Hospital (VMTH), University of California, Davis
3.3. Historical Reports from Scientific Publications, Media and Electronic Sources
3.4. Overlap of Canine Cyanotoxin Intoxication Cases Identified Using the Three Sources
3.5. Limitations
4. Conclusions
Acknowledgments
Disclaimer
Conflicts of Interest
References
- Sivonen, K.; Jones, G. Cyanobacterial Toxins. In Toxic Cyanobacteria in Water. A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E. and F.N. Spon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Stewart, I.; Carmichael, W.; Backer, L. Cyanobacteria. In Water and Sanitation-Related Diseases and the Environment: Challenges, Interventions and Preventive Measures; Selendy, J., Ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2011; pp. 95–110. [Google Scholar]
- Backer, L.; McGillicuddy, D.J., Jr. Harmful algal blooms at the interface between coastal oceanography and human health. Oceanography 2006, 19, 94–106. [Google Scholar] [CrossRef]
- Backer, L.; Moore, S. Harmful Algal Blooms: Future Threats in a Warmer World. In Environmental Pollution and Its Relation to Climate Change; Nemr, A., Ed.; Nova Science Publishers, Inc: New York, NY, USA, 2010; pp. 485–512. [Google Scholar]
- Paerl, H.W.; Paul, V.J. Climate change: Links to global expansion of harmful cyanobacteria. Water Res. 2012, 46, 1349–1363. [Google Scholar] [CrossRef]
- Stewart, I.; Webb, P.M.; Schluter, P.J.; Fleming, L.E.; Burns, J.W., Jr.; Gantar, M.; Backer, L.C.; Shaw, G.R. Epidemiology of recreational exposure to freshwater cyanobacteria—An international prospective cohort study. BMC Public Health 2006, 6. [Google Scholar] [CrossRef]
- Backer, L.C.; Carmichael, W.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.B.; Nierenberg, K.; Hill, V.; Kieszak, S.M.; et al. Recreational exposure to low concentrations of microcystins during an algal bloom in a small lake. Mar. Drugs 2008, 6, 389–406. [Google Scholar] [CrossRef]
- Backer, L.C.; McNeel, S.V.; Barber, T.; Kirkpatrick, B.; Williams, C.; Irvin, M.; Zhou, Y.; Johnson, T.B.; Nierenberg, K.; Aubel, M.; et al. Recreational exposure to microcystins during algal blooms in two California lakes. Toxicon 2010, 55, 909–921. [Google Scholar] [CrossRef]
- Backer, L.C. Cyanobacterial harmful algal blooms (CyanoHABs): Developing a public health response. Lake Reserv. Manag. 2002, 18, 20–31. [Google Scholar] [CrossRef]
- Senior, V.E. Algal poisoning in Saskatchewan. Can. J. Comp. Med. Vet. Sci. 1960, 24, 26–31. [Google Scholar]
- Codd, G.A.; Edwards, C.; Beattle, K.A.; Barr, W.M.; Gunn, G.J. Fatal attraction to cyanobacteria? Nature 1992, 359, 110–111. [Google Scholar]
- Zaias, J.; Backer, L.; Fleming, L. Harmful Algal Blooms. In Human-Animal Medicine Clinical Approaches to Zoonoses, Toxicants, and Other Shared Health Risks; Rabinowitz, P., Conti, L., Eds.; Sanders: Maryland Heights, MO, USA, 2010; pp. 91–104. [Google Scholar]
- Landsberg, J.H. The effects of harmful algal blooms on aquatic organisms. Rev. Fish. Sci. 2002, 10, 113–390. [Google Scholar] [CrossRef]
- Beasley, V.R.; Cook, W.O.; Dahlem, A.M.; Hooser, S.B.; Lovell, R.A.; Valentine, W.M. Algae intoxication in livestock and waterfowl. Vet. Clin. North Am. Food Anim. Pract. 1989, 5, 345–361. [Google Scholar]
- Beasley, V.R.; Dahlem, A.M.; Cook, W.O.; Valentine, W.M.; Lovell, R.A.; Hooser, S.B.; Harada, K.; Suzuki, M.; Carmichael, W.W. Diagnostic and clinically important aspects of cyanobacterial (blue-green algae) toxicoses. J. Vet. Diagn. Investig. 1989, 1, 359–365. [Google Scholar] [CrossRef]
- Cheng, Y.S.; Zhou, Y.; Irvin, C.M.; Kirkpatrick, B.; Backer, L.C. Characterization of aerosols containing microcystin. Mar. Drugs 2007, 5, 136–150. [Google Scholar] [CrossRef]
- Codd, G.A.; Bell, S.G.; Kaya, K.; Ward, C.J.; Beattie, K.A.; Metcalf, J.S. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Rankin, K.; Alroy, K.; Kudela, R.; Oates, S.; Murray, M.; Miller, M.A. Treatment of cyanobacterial (microcystin) toxicosis using oral cholestyramine: Case report of a dog from Montana. Toxins 2013, 5, 1051–1063. [Google Scholar] [CrossRef]
- Mereish, K.A.; Solow, R. Effect of antihepatotoxic agents against microcystin-LR toxicity in cultured rat hepatocytes. Pharm. Res. 1990, 7, 256–259. [Google Scholar]
- Mereish, K.A.; Bunner, D.L.; Ragland, D.R.; Creasia, D.A. Protection against microcystin-LR-induced hepatotoxicity by Silymarin: Biochemistry, histopathology, and lethality. Pharm. Res. 1991, 8, 273–277. [Google Scholar] [CrossRef]
- Rao, P.V.L.; Jayaraj, R.; Bhaskar, A.S. Protective efficacy and the recovery profile of certain chemoprotectants against lethal poisoning by microcystin-LR in mice. Toxicon 2004, 44, 723–730. [Google Scholar] [CrossRef]
- Rao, P.V.L.; Gupta, N.; Jayaraj, R. Screening of certain chemoprotectants against cyclic peptide toxin microcystin-LR. Indian J. Pharmacol. 2004, 36, 87–92. [Google Scholar]
- Kurek, J. Managing the Acutely Convulsing Patient: Generalized Tremors, Cluster Seizures, and Status Epileptics. Pacific Tide. 2013. Available online: http://www.pacificveterinaryspecialists.com/newsletters/Pacific-Tide-June-2013.pdf (accessed on 7 June 2013).
- DeVries, S.E.; Galey, F.D.; Namikoshi, M.; Woo, J.C. Clinical and pathologic findings of blue-green algae (Microcystis aeruginosa) intoxication in a dog. J. Vet. Diagn. Investig. 1993, 5, 403–408. [Google Scholar] [CrossRef]
- California Department of Water Resources. Clear Lake Water Quality Investigation; The Resources Agency: Sacramento, CA, USA, 1966; p. 248. Available online: http://www.water.ca.gov/waterdatalibrary/docs/historic/Bulletins/Bulletin_143/Bulletin_143-2__1966.pdf (accessed on 7 June 2013).
- Schwimmer, D.; Schwimmer, M. Medical Aspects of Phycology. In Algae, Man and the Environment; Jackson, D., Ed.; Syracuse University Press: Syracuse, NY, USA, 1968; pp. 279–358. [Google Scholar]
- Anonymous. Pigs Die after Drinking Water from OKOBOJI. Vindicator and Republican, 19 October 1944. [Google Scholar]
- Rose, E.T. Toxic algae in Iowa lakes. Proc. Iowa Acad. Sci. 1953, 60, 738–745. [Google Scholar]
- Yoo, R.S.; Carmichael, W.W.; Hoehn, R.C.; Hrudey, S.E. Cyanobacterial (Blue-Green algal) Toxins: A Resource Guide; AWWA Research Foundation and American Water Works Association: Denver, CO, USA, 1995. [Google Scholar]
- Anonymous. Lake Water not Only Made Dog Sick—It Killed Animal! LeMars Globe Post, 11 December 1952. [Google Scholar]
- Firkins, G.S. Toxic algae poisoning. Iowa State Coll. Vet. 1953, 15, 151–153. [Google Scholar]
- Brown, L. Report Nearly Completed on Dying Lake. The Lakeland Ledger, 7 May 1973. [Google Scholar]
- Anonymous. Lake Kills Only Dogs. The Spokesman Review, 9 October 1976. [Google Scholar]
- Young, L. Toxic Algae Discovered in Spokane Arm of Lake Roosevelt. The Spokesman Review, 29 September 1978. [Google Scholar]
- Soltero, R.A.; Nichols, D.G. The Recent Blue-Green Algal Blooms of Long Lake, Washington. In The Water Environment: Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 143–159. [Google Scholar]
- Jacoby, J.M.; Kann, J. The occurrence and response to toxic cyanobacteria in the Pacific Northwest, North America. Lake Reserv. Manag. 2007, 23, 123–143. [Google Scholar] [CrossRef]
- Anonymous. Forest Service Officials Eye Hebgen Lake Closure. Idaho Falls Post Register, 29 June 1977; 14. [Google Scholar]
- Anonymous. Hebgen Lake: Swimming Barred After 14 Cows Die. The Spokesman Review, 2 July 1977; 1. [Google Scholar]
- Juday, R.E.; Keller, E.J.; Horpestad, A.; Bahls, L.L.; Glasser, S. A toxic bloom of Anabaena Flos-Aquae in Hebgen Reservoir Montana in 1977. In The Water Environment: Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 103–112. [Google Scholar]
- Anonymous. Warning Signs Planned. Spokane Daily Chronicle, 20 July 1977. [Google Scholar]
- Billings, W.H. Water-Associated Human Illness in North-East Pennsylvania and Its Suspected Association with Blue-Green Algal Blooms. In The Water Environment: Algal Toxins and Health; Carmichael, W.W., Ed.; Plenum Press: New York, NY, USA, 1981; pp. 243–255. [Google Scholar]
- Anonymous. Toxic Algae Blooming. Spokesman Review, 2 August 1980. [Google Scholar]
- Associated Press, Algae Fouls Lake, Kills Dogs, Cows. Spokane Chronicle, 9 November 1982.
- Associated Press, Dogs, Cows Die after Drinking Polluted Water. Lewiston Morning Tribune, 10 November 1982.
- Kann, J.; Falter, C.M. Blue-Green Algae Toxicity in Black Lake, Kootenai County, Idaho; Idaho Water Resources Research Institute: Moscow, ID, USA, 1985; p. 55. Available online: http://contentdm.lib.uidaho.edu/cdm4/item_viewer.php?CISOROOT=/idahowater&CISOPTR=91 (accessed on 7 June 2013).
- Anonymous. Access to 2 Lakes Restricted after the Deaths of 2 Dogs. The Milwaukeee Journal, 25 June 1985. [Google Scholar]
- Sonzogni, W.C.; Repavich, W.M.; Standridge, J.H.; Wedepohl, R.E.; Vennie, J.G. A note on algal toxins in Wisconsin waters experiencing blue-green algae blooms. Lake Reserv. Manag. 1988, 4, 281–285. [Google Scholar] [CrossRef]
- Mahmood, N.A.; Carmichael, W.W.; Pfahler, D. Anticholinesterase poisonings in dogs from a cyanobacterial (blue-green algae) bloom dominated by Anabaena flos-aquae. Am. J. Vet. Res. 1988, 49, 500–503. [Google Scholar]
- Dadisman, Q. Toxic Algae Linked to Death of Cows, Dogs. The Milwaukee Sentinel, 7 March 1987. [Google Scholar]
- Anonymous. Algae Prompting Clifton to Empty Main Memorial Pond. The Record, 15 June 1986. [Google Scholar]
- Anonymous. Demise of Herons Laid to Algae. Star Tribune, 25 June 1989. [Google Scholar]
- Associated Press, Health Officials Blame Cats’ Deaths on Algae Toxin in Lake. Anchorage Daily News, 3 December 1989.
- Associated Press, Algae Bloom in Lake Baffles Experts. Eugene Register Guard, 24 December 1989.
- Associated Press, Eight Pets Killed by Poisonous Algae Bloom. Spokane Chronicle, 25 December 1989.
- Winterton, D. Report: Toxic Algae in East Canyon Reservoir. Standard Examiner, 6 March 2011. [Google Scholar]
- Environmental Protection Agency (EPA). Lake Washington—No Longer Muddying Up the Waters. Section 319 Success Stories; EPA: Washington, DC, USA, 2010; p. 1. Available online: http://www.epa.gov/owow/NPS/Section319I/MS.html (accessed on 7 June 2013).
- Anonymous. Algae Threat Gone from Lake. The Commercial Appeal, 23 October 1990. [Google Scholar]
- Carmichael, W.W. Toxic freshwater blue-green algae (cyanobacteria): An overlooked health threat. Health Environ. Digest. 1991, 5, 14. [Google Scholar]
- Anonymous. Deadly Blue-Green Algae was Found in Mina Lake. USA Today, 30 August 1991. [Google Scholar]
- Anonymous. Algae Kills Dogs. Ellensburg Daily Record, 11 October 1991. [Google Scholar]
- Anonymous. Five Hunting Dogs Killed by Algae in Drinking Water. The Oregonian, 11 October 1991. [Google Scholar]
- McAllister, T. Hunters Share Sad Story of Dogs Poisoned by Algae to Alert Others to Danger. The Oregonian, 23 October 1991. [Google Scholar]
- Gorham, M.E. Blue-Green Algae can be Deadly to Dogs, Other Animals. The Columbian, 3 July 1997. [Google Scholar]
- Marks, J. Water in California’s Carmel River is Deemed Unsafe to Drink. Monterey County Herald, 25 September 1997. [Google Scholar]
- Calvan, B.C. Is Dog Toxic Lake’s First Victim?—Officials Concerned over Spreading Microbes. The Seattle Times, 26 September 1997. [Google Scholar]
- Johnston, B.R.; Jacoby, J.M. Cyanobacterial toxicity and migration in a mesotrophic lake in western Washington, USA. Hydrobiologia 2003, 495, 79–91. [Google Scholar] [CrossRef]
- Anonymous. Algae Prompts DNR to Issue Lake Warning. St. Paul Pioneer Press, 1 August 1998. [Google Scholar]
- Anonymous. Maryland Department of Agriculture: Protect Animals from Blue-Green Algae. PR Newswire. 25 September 1998, p. 8022. Available online: http://www.thefreelibrary.com/Maryland+Department+of+Agriculture%3A+Protect+Animals+From+Blue-Green...-a053028837 (accessed on 7 June 2013).
- Leaning, J. Toxic Algae Killed Dogs at Nickerson. Cape Cod Times, 12 December 1998. [Google Scholar]
- Leaning, J. Fourth Dog Sickens in Park. Cape Cod Times, 12 December 1998. [Google Scholar]
- Moore, S.; Lanphear, M. Dog Deaths Laid to Poison Lake Algea. Press Republican, 8 September 1999. [Google Scholar]
- Moore, S. Lake Algae Blooms Still Pose Threat. Press Republican, 9 September 1999. [Google Scholar]
- Boyer, G.; Watzin, M.C.; Shambaugh, A.D.; Satchwell, M.F.; Rosen, B.R.; Mihuc, T. The Occurrence of Cyanobacterial Toxins in Lake Champlain. In Lake Champlain: Partnerships and Research in the New Millennium; Manley, T.O., Manley, P.L., Mihuc, T.B., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2004; pp. 241–257. [Google Scholar]
- Boyer, G.L. Cyanobacterial toxins in New York and the lower Great Lakes ecosystems. Adv. Exp. Med. Biol. 2008, 619, 153–165. [Google Scholar] [CrossRef]
- Bazilchuk, N. Toxin in Lake Champlain Algae Kills Two N.Y. Dogs. The Burlington Free Press, 8 September 1999. [Google Scholar]
- Anonymous. Threat of Troubled Waters. Dog Deaths Lead Officials to Check River. Times News, 12 November 1999. [Google Scholar]
- Anonymous. Mystery of Dog Death Deepens. Times News, 18 November 1999. [Google Scholar]
- Associated Press, Scientists: Dog Died Because of Algae. Lewiston Morning Tribune, 7 January 2000.
- Anonymous. Scientists Confirm Algae Killed Dog Near Lake Lowell. Times News, 7 January 2000. [Google Scholar]
- Anonymous. Officials Wait on Final Report of Dog Deaths. USDA to Release Preliminary Findings on Algae Next Week. Times News, 22 November 2000. [Google Scholar]
- Anonymous. Second Dog Dies Along Snake. Times News, 15 November 2000. [Google Scholar]
- Anonymous. Toxicologists Confirm Snake River Algae Species. The Times-News, 9 December 2000. [Google Scholar]
- Hill, H. Dog Deaths in Humboldt and Mendocino County Water Bodies Possibly Related to Cyanobacterial Toxicity. 2005. Available online: http://www.waterboards.ca.gov/water_issues/programs/bluegreen_algae/docs/workgroup110805/harriethill.pdf (accessed on 7 June 2013).
- Holschuh, A. Danger on the Beach. Is Toxic Algae Killing Dogs at Big Lagoon? North Coast Journal Weekly. 29 November 2001. Available online: http://www.northcoastjournal.com/112901/cover1129.html (accessed on 7 June 2013).
- Soussan, T. Warning Issued for Dogs at Lake. Albuquerque Journal, 16 August 2002. [Google Scholar]
- Thilsted, J. New Mexico Department of Agriculture, Veterinary Diagnostic Services, Elephant Butte canine poisoning cyanobacteria. Personal communication, 22 July 2002. [Google Scholar]
- Puschner, B.; Hoff, B.; Tor, E.R. Diagnosis of anatoxin-a poisoning in dogs from North America. J. Vet. Diagn. Investig. 2008, 20, 89–92. [Google Scholar] [CrossRef]
- Associated Press, Algae Warning Issued by State. Times Argus, 11 September 2002.
- Silverman, A. Algae Suspected in Dog Deaths. The Burlington Free Press, 11 September 2002. [Google Scholar]
- Ansami, R. Deadly Algae Invades Lakes. Daily Globe, 8 August 2003. [Google Scholar]
- Smith, D. Algae Blamed for Dogs’ Death. Star Tribune, 16 November 2003. [Google Scholar]
- Gaarder, N. Toxin Closes Buccaneer Bay’s Lake. Omaha World Herald, 6 May 2004. [Google Scholar]
- Laukaitis, A.J. Toxic Algae Closes Buccaneer Bay Lake. Lincoln Journal Star, 7 May 2004. [Google Scholar]
- Brakhage, P.A. Cyanobacteria, the Nebraska Experience; Nebraska Department of Environmental Quality: Lincoln, NE, USA, 2009; p. 5. Available online: http://www.deq.state.ne.us/Publica.nsf/pages/WAT145 (accessed on 7 June 2013).
- Walker, S.R.; Lund, J.C.; Schumacher, D.G.; Brakhage, P.A.; McManus, B.C.; Miller, J.D.; Augustine, M.M.; Carney, J.J.; Holland, R.S.; Hoagland, K.D.; et al. Nebraska experience. Adv. Exp. Med. Biol. 2008, 619, 139–152. [Google Scholar] [CrossRef]
- Anonymous. Sarpy Lakes Closed. Channel 6 News. 17 May 2004. Available online: http://www.wowt.com/home/headlines/778382.html (accessed on 7 June 2013).
- Gaarder, N. Lake Algae Blamed for Dog Deaths. Water trouble. Omaha World Herald, 18 May 2004. [Google Scholar]
- Goldberg, D. Lake Algae may be Killing Animals, Birds. Syracuse Post Standard, 4 June 2004. [Google Scholar]
- Goldberg, D. Advisories Posted at Lake; Toxic Algae Prompts Officials to Close Lake Neatahwanta. Syracuse Post Standard, 5 June 2004. [Google Scholar]
- Spencer, K.M.; Boyer, G. Dog Deaths of Summer on Lake Neatahwanta. Upper Peninsula Student Research Symposium in Chemistry and Related Fields; Lake Superior State University, Michigan Tech: Sault Sainte Marie, MI, USA, 2005. Available online: http://www.chemistry.mtu.edu/~djchesne/SRS/SRS_05/Abstracts/Spencer.pdf (accessed on 7 June 2013).
- Sensenbrenner, L. Pond Algae is Blamed for Pet Dog’s Seizures. Capital Times, 10 June 2004. [Google Scholar]
- Novak, B.; Sensenbrenner, L. Toxin Scare Hits Area Lakes; Kegonsa Lake Closed after Dog Suffers Convulsions. The Capital Times, 9 June 2004. [Google Scholar]
- Williams, B. Dog Dies after Swim in Kegonsa. Algae Toxin in Water may be to Blame; Officials will Still Test. Wisconsin State Journal, 11 June 2004. [Google Scholar]
- Lindon, M.; Heiskary, S. Microcystin Levels in Eutrophic South Central Minnesota Lakes; Minnesota Pollution Control Agency: Saint Paul, MN, USA, 2007; p. 53. Available online: http://www.pca.state.mn.us/index.php/view-document.html?gid=15393 (accessed on 7 June 2013).
- Jacoby, J. Brownless Algae may be Deadly to Dogs. Baker City Herald, 1 October 2004. [Google Scholar]
- Fode, M. Blue-Green Algae Bloom Causes at Least One Dog Death at Lake Benton. Pipestone County Star, 2004. [Google Scholar]
- Leventis, A. Toxic Algae Claims Another Family Pet; Blue-Green Algae in Steilacoom Lake Likely Killed a Family Dog. Lake Homeowners are Running out of Options to Treat the Toxic Algae. The News Tribune, 4 October 2004. [Google Scholar]
- Hanowell, R. Toxic Blue-Green Algae Blooms. Tacoma Pierce County Health Department Veterinary Newsletter; Tacoma Pierce County Health Department: Tacoma, WA, USA, 2005; p. 1. Available online: http://www.tpchd.org/files/library/e15475924fa7b08d.pdf (accessed on 7 June 2013).
- Novak, B. Toxic Algae Warning Issued; Scum on Area Lakes, Ponds Threaten People, Pets. The Capital Times, 14 July 2005. [Google Scholar]
- Anonymous. Popular Pond may be Contaminated. The Hawk-Eye, 22 July 2005. [Google Scholar]
- Anonymous. Toxic Algae on Private Lakes. WOWT.com News. 28 May 2006. Available online: http://www.wowt.com/home/headlines/2886311.html?site=mobile (accessed on 7 June 2013).
- Anonymous. Toxic Algae Surfaces at Thomas Lakes. Suburban Newspapers, 8 June 2006. [Google Scholar]
- McCormick, J. Toxic Algae may Have Killed 2 Dogs at Lake. Kitsap Sun, 6 June 2006. [Google Scholar]
- Hardy, J. Dog Deaths from Cyanobacteria Spurs Lake Monitoring. Zoonotic Disease Newsletter. 2008. Available online: http://www.ifish.net/board/showthread.php?t=229467 (accessed on 7 June 2013).
- Zimowsky, P. Algae in Some Idaho Waters can Sicken or Kill Dogs. The Idaho Statesman, 8 September 2006. [Google Scholar]
- Debner, M. Toxic Algae Found in Lakes Linked to dog’s Death. Angler Insider. 20 June 2007. Available online: http://anglerinsider.com/forum/showthread.php?8639-Toxic-Algae-Found-In-Lakes-Linked-To-Dog-s-Death (accessed on 7 June 2013).
- Marohn, K. Dog’s Death Spurs Algae Warning. St. Cloud Times, 20 June 2007. [Google Scholar]
- Smith, D. Outdoors Almanac: Four Dogs Have Died from Blue-Green Algae Poisoning. Star Tribune, 12 August 2007. [Google Scholar]
- Machniak, C. ‘The Water in This Lake Killed My Dog’; Dog Owner, Neighbors at Odds over Canine’s Cause of Death. Flint Journal, 3 July 2007. [Google Scholar]
- Anonymous. Lake Algae is Focus in Probe of Dog’s Death. Detroit Free Press, 7 July 2007. [Google Scholar]
- Smith, D. Blue-Green Algae Poisoning Has Killed Four Dogs. Star Tribune, 11 August 2007. [Google Scholar]
- Ratzlaff, A. Dog Deaths at Local Reservoir may be Linked to Algae Toxins. Hillsboro Free Press, 11 July 2007. [Google Scholar]
- Anonymous. Algae Blooms Forming on State Waters Again. KULR8.com. 19 July 2007. Available online: http://www.kulr8.com/news/state/8614757.html (accessed on 7 June 2013).
- Hamel, K. Watch for Cyanobacteria “Blue-Green Algae” on Your Lake. Waterline. September 2008, p. 1, 3. Available online: http://www.walpa.org/documents/Waterline/Waterline_Sept08.pdf (accessed on 7 June 2013).
- Kiava, L. 2 More Minn. Dogs Dead from Blue-Green Algae. WCCO TV. 9 August 2007. Available online: http://www.duckhuntingchat.com/forum/viewtopic.php?f=11&t=40464 (accessed on 7 June 2013).
- Anonymous. Caution Urged after Dog Dies at Elephant Butte. Las Cruces Sun-News, 17 September 2007. [Google Scholar]
- Anonymous. Elephant Butte Algae may Have Killed Dog. El Paso Times, 17 September 2007. [Google Scholar]
- Anonymous. Possible Algae Poisoning at Elephant Butte. Ruidoso News, 18 September 2007. [Google Scholar]
- Cardin, F. We must Address the Problem of Blue-Green Algae. The Post-Crescent, 26 October 2007. [Google Scholar]
- McCartney, D. Danger to Dogs can Lurk in Pond Water, Woman Finds. The Wichita Eagle, 1 September 2008. [Google Scholar]
- Chase, C. Toxic Algae Bloom Poisons Dog. Daily Interlake.com. 16 September 2010. Available online: http://www.dailyinterlake.com/news/local_montana/article_9b5e46a0-c12c-11df-8429-001cc4c03286.html (accessed on 7 June 2013).
- Anonymous. DNR Cautions Dog Owners about Fatal Algae Toxin. The Fulton County News, 6 November 2008. [Google Scholar]
- Anonymous. Stinky Blue-Green Algae Blamed for Dog Deaths.Algae is Blooming in Response to Drought and Fertilizer Runoffs from Farms. NBCnews.com. 27 September 2009. Available online: http://www.nbcnews.com/id/33045773/ns/us_news-environment/t/stinky-blue-green-algae-blamed-dog-deaths/ (accessed on 7 June 2013).
- Goiffon, T. Dog Deaths and Blue Green Algae. Aussie Talk. 2009. Available online: http://dunhamlakeaustralianterriers.com/aussie-talk/page/5 (accessed on 7 June 2013).
- Hamel, K. Freshwater Algae Control Program: Report to the Washington State Legislature (2008–2009); Department of Ecology: Washington, DC, USA, 2009; pp. 1–32. Available online: https://fortress.wa.gov/ecy/publications/summarypages/0910082.html (accessed on 7 June 2013).
- Meeks, A. Algae in Lake Water Suspected in Dogs’ Deaths. Alamogordo Daily News, 19 July 2009. [Google Scholar]
- Meeks, A. Algae in Elephant Butte Water Suspected in Dogs’ Deaths. Las Cruces Sun-News, 18 July 2009. [Google Scholar]
- North Dakota State University Extension Service. It’s not Too Early to Watch for Blue-Green Algae. Agweek. 13 July 2009. Available online: http://www.agweek.com/event/article/id/246600/publisher_ID/1/ (accessed on 7 June 2013).
- Burns, R. Killer Algae on the Van Duzen. The Journal, 31 July 2009. [Google Scholar]
- California Regional Water Quality Control Board, Agencies Warn to Avoid Exposure to Toxic Blue Green Algae in Three Northern CA Rivers. News Release. 20 August 2009. Available online: http://cdm16658.contentdm.oclc.org/cdm/singleitem/collection/p267501ccp2/id/2384/rec/20 (accessed on 7 June 2013).
- Bolt, G. Tests Find Dog Died of Toxin in Algae. All Four Dogs that were at Elk Creek Near Elkton Now are Presumed to Have Died from the Water. The Register Guard, 11 September 2009. [Google Scholar]
- Terry, L. OSU Confirms Algae Killed Dogs. The Oregonian, 10 September 2009. [Google Scholar]
- Rillos, L. Four Dogs Die: ‘Something Seriously Toxic is on the Banks’. KVAL.COM. 24 August 2009. Available online: http://www.kpic.com/news/local/54754657.html (accessed on 7 June 2013).
- Freeman, M. Dog Dies from Toxic Algae, Lab Tests Reveal. Health Officials Hope This will be a Wake-Up Call for People to Stay Away from Water When Warned. Mail Tribune, 11 september 2009. [Google Scholar]
- Lyon, C. DWP Develops resources for cyanobacteria and drinking water. Pipeline. 2011, pp. 4–6. Available online: http://public.health.oregon.gov/HealthyEnvironments/DrinkingWater/Operations/Treatment/Documents/algae/Pipeline_Algae.pdf (accessed on 7 June 2013).
- Anonymous. Dogs are Endangered by Lake Algae. Antigo Daily Journal, 1 September 2009. [Google Scholar]
- Anonymous. River: Slow Flow Merits Caution. San Marcos Daily Record, 5 September 2009. [Google Scholar]
- Lien, D. Algae Exposure Suspected in Death of Dog in Southern Minnesota. Pioneer Press, 19 September 2009. [Google Scholar]
- Anonymous. Dog Dead from Fox Lake Algae. Jackson County Pilot, 18 September 2009. [Google Scholar]
- Associated Press. Dog dies after exposure to toxic algae in Twin Cities. Post Bulletin.com. 18 September 2009. Available online: http://ww2.postbulletin.com/newsmanager/templates/localnews_story.asp?z=2&a=416864 (accessed on 7 June 2013).
- Imrie, R. What’s Ugly, Smells, Kills Dogs? Blue-Green Algae. Seattle Times, 27 September 2009. [Google Scholar]
- Robinson, E. Lacamas, Round Lakes Closed after Pet Dog Dies. Columbian, 18 September 2009. [Google Scholar]
- Lowe, S. Warning Dog Owners: Blue-Green Algae can be Possible Hazard. South Bend Tribune, 15 November 2009. [Google Scholar]
- Polk County Association of Lakes and Rivers. Meeting Minutes. 28 October 2010. Available online: http://pcalr.org/wppcalr/wp-content/uploads/2011/12/Minutes_102010.pdf (accessed on 7 June 2013).
- Anonymous. Dog Dies, Owner Ill after Animal Swims in Algae-Choked Grand Lake St. Marys. Sandusky Register, 31 July 2010. [Google Scholar]
- Hunt, S.; McGlade, C. Algae may be Killing Pets; At Least Three Dogs Dead, Nine Humans Ill. The Columbus Dispatch, 2 August 2010. [Google Scholar]
- Anonymous. Low Toxin Levels Found in Burr Oak State Park Lake. The Athens News, 5 August 2010. [Google Scholar]
- North Dakota Department of Health. State Health Department Issues Blue-Green Algae Bloom Advisory. 3 August 2010. Available online: http://www.ndhan.gov/data/mrNews/Harmful%20Algae%20Lake%20Josephine.pdf (accessed on 7 June 2013).
- Freeman, M. Algae Toxin Levels Prompt Alarm. Blue-Green Algae Registers 400 Times Higher than Mark That Killed Dog. Mail Tribune, 2 September 2010. [Google Scholar]
- Anonymous. Algae Forces Closing of Honeoye Beach. Rochester Democrat and Chronicle, 29 August 2010. [Google Scholar]
- Lutz, A. Flathead Dog Sick with Blue, Green Algae. KPAX.COM. 14 September 2010. Available online: http://www.kpax.com/news/dog-sick-with-blue-green-algae/ (accessed on 7 June 2013).
- Simpson, N. Pembroke Pond Closed after Girls Get Sick. The Patriot Ledger, 8 June 2011. [Google Scholar]
- Freeman, M. Health Warnings Issued after Algae Discoveries. Mail Tribune, 30 July 2011. [Google Scholar]
- Kansas State University. Lethal Ingestion: Diagnostic Lab Analyzes Blue-Green Algae in Recent Dog Deaths. Lifelines. 2011. Available online: http://www.vet.k-state.edu/depts/development/lifelines/1109.htm#algae (accessed on 7 June 2013).
- Creighton, A. Yoncalla Dog Dies after River Outing, Blue-Green Algae Likely the Cause. The News Review Today, 4 August 2011. [Google Scholar]
- Ferenchik, M. Toxic Algae in Pond could be Cause of Dog’s Death. The Columbus Dispatch, 9 September 2011. [Google Scholar]
- Ferenchik, M. Tests Find no Algae Liver Toxin in Dog. The Columbus Dispatch, 17 September 2011. [Google Scholar]
- Van der Merwe, D.; Sebbag, L.; Nietfeld, J.C.; Aubel, M.T.; Foss, A.; Carney, E. Investigation of a Microcystis aeruginosa cyanobacterial freshwater harmful algal bloom associated with acute microcystin toxicosis in a dog. J. Vet. Diagn. Invest. 2012, 24, 679–687. [Google Scholar] [CrossRef]
- Anonymous. Blue-Green Algae is Believed to Have Killed to Dogs at Lake Ellsworth. 7 News KSWO. 19 May 2012. Available online: http://www.kswo.com/story/18564203/blue-green-algae-is-believed-to-have-killed-to-dogs-at-lake-ellsworth (accessed on 7 June 2013).
- Buelow, J. Blue-Green Algae Bloom Blamed for Dog’s Death. Tomahawk Leader, 10 July 2012. [Google Scholar]
- Widener, A. Dogs Die after Swimming in Salamonie Reservoir. WANE.COM. 18 July 2012. Available online: http://www.wane.com/dpp/news/dogs-die-after-swimming-in-reservoir (accessed on 7 June 2013).
- Wiehe, J. State Warns of Toxic Algae after Lake Trip Dooms Dogs. Fort Wayne Journal Gazette, 21 July 2012. [Google Scholar]
- Miller, R. Toxic Blue-Green Algae Blooms in Area Waters. Olean Times Herald, 8 September 2012. [Google Scholar]
- Anonymous. Blue Green Algae. NeighborHound Watch. 15 August 2012. Available online: http://www.dogheirs.com/events/528 (accessed on 7 June 2013).
- Cassoli, D.; Downes, S. Cool cases from the E. R. Part 2. 2013. Available online: http://www.vcaspecialtyvets.com/ckfinder/userfiles/files/Northwest/Winterfest%202012/Cassoli% 20and%20Downes%20-%20Cool%20Cases%20-%20Part%202.pdf (accessed on 7 June 2013).
- U.S. Environmental Protection Agency. Reconsideration of California’s 2006 Section 303(d) List Omission of Microcystin Toxin Listings for Three Klamath River Segments and Determination to Add Microcystin Toxins Listing for Klamath River Hydrologic Unit (HU), Middle HA Hydrologic Area (HA), Oregon to Iron Gate; USA EPA Water Division: Francisco, CA, USA, 13 March 2008; pp. 1–12. Available online: http://www.epa.gov/region9/water/tmdl/303d-pdf/Klamath-SWRCB303d-final.pdf (accessed on 7 June 2013).
- U.S. Environmental Protection Agency. Climate Change Indicators in the United States. 1 May 2013. Available online: http://www.epa.gov/climatechange/science/indicators/weather-climate/temperature.html (accessed on 7 June 2013).
- Carmichael, W.W.; Evans, W.R.; Yin, Q.Q.; Bell, P.; Moczydlowski, E. Evidence for paralytic shellfish poisons in the freshwater cyanobacterium Lyngbya wollei (Farlow ex. Gomont) comb. nov. Appl. Environ. Microbiol. 1997, 63, 3104–3110. [Google Scholar]
- Carmichael, W.W.; Biggs, D.F.; Gorham, P.R. Toxicology and pharmacological action of Anabaena flos-aquae toxin. Science 1975, 187, 542–544. [Google Scholar]
- Devlin, J.P.; Edwards, O.E.; Gorham, P.R.; Hunter, N.R.; Pike, R.K.; Stavric, B. Anatoxin-a, a toxic alkaloid from Anabaena flos-aquae NRC-44h. Can. J. Chem. 1977, 55, 1367–1371. [Google Scholar] [CrossRef]
- Negri, A.P.; Jones, G.J. Bioaccumulation of paralytic shellfish poisoning (PSP) toxins from the cyanobacterium Anabaena circinalis by the freshwater mussel Alathyria condola. Toxicon 1995, 33, 667–678. [Google Scholar] [CrossRef]
- Kankaanpää, H.T.; Holliday, J.; Schröder, H.; Goddard, T.J.; von Fister, R.; Carmichael, W.W. Cyanobacteria and prawn farming in northern New South Wales, Australia—A case study on cyanobacteria diversity and hepatotoxin bioaccumulation. Toxicol. Appl. Pharmacol. 2005, 203, 243–256. [Google Scholar] [CrossRef]
- Smith, J.L.; Haney, J.F. Foodweb transfer, accumulation, and depuration of microcystins, a cyanobacterial toxin, in pumpkinseed sunfish (Lepomis gibbos). Toxicon 2006, 48, 580–589. [Google Scholar] [CrossRef]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Staedler, M.; Miller, W.A.; Toy-Choutka, S.; Dominik, C.; et al. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS One 2010, 5, e12576. [Google Scholar]
- Quiblier, C.; Wood, S.; Echenique-Subiabre, I.; Heath, M.; Villeneuve, A.; Humbert, J.-F. A review of current knowledge on toxic benthic freshwater cyanobacteria—ecology, toxin production and risk management. Water Res 2013. [Google Scholar] [CrossRef]
- Kudela, R.M. Characterization and deployment of Solid Phase Adsorption Toxin Tracking (SPATT) resin for monitoring of microcystins in fresh and saltwater. Harmful Algae 2011, 11, 117–125. [Google Scholar] [CrossRef]
- Backer, L.C.; Grindem, C.B.; Corbett, W.T.; Cullins, L.; Hunter, J.L. Pet dogs as sentinels for environmental contamination. Sci. Total Environ. 2001, 274, 161–169. [Google Scholar]
- Paerl, H.W.; Huisman, J. Blooms like it hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef]
- Paerl, H.W.; Huisman, J. Climate change: A catalyst for global expansion of harmful cyanobacterial blooms. Environ. Microbiol. Rep. 2009, 1, 27–37. [Google Scholar] [CrossRef]
- DogHeirsTeam. Toxic Blue-Green Algae in Luna Pier. NeighborHound Watch. 21 November 2011. Available online: http://www.dogheirs.com/events/179 (accessed on 7 June 2013).
- NCCOS Phytoplankton Monitoring Network homepage. Available online: http://products.coastalscience.noaa.gov/pmn/ (accessed on 3 May 2013).
- Glynn, M.; Backer, L. Collecting Public Health Surveillance Data: Creating a Surveillance System. In Principles and Practice of Public Health Surveillance; Lee, L., Teutsch, S., Thacker, S., St. Louis, M., Eds.; Oxford University Press: New York, NY, USA, 2010; pp. 44–64. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Backer, L.C.; Landsberg, J.H.; Miller, M.; Keel, K.; Taylor, T.K. Canine Cyanotoxin Poisonings in the United States (1920s–2012): Review of Suspected and Confirmed Cases from Three Data Sources. Toxins 2013, 5, 1597-1628. https://doi.org/10.3390/toxins5091597
Backer LC, Landsberg JH, Miller M, Keel K, Taylor TK. Canine Cyanotoxin Poisonings in the United States (1920s–2012): Review of Suspected and Confirmed Cases from Three Data Sources. Toxins. 2013; 5(9):1597-1628. https://doi.org/10.3390/toxins5091597
Chicago/Turabian StyleBacker, Lorraine C., Jan H. Landsberg, Melissa Miller, Kevin Keel, and Tegwin K. Taylor. 2013. "Canine Cyanotoxin Poisonings in the United States (1920s–2012): Review of Suspected and Confirmed Cases from Three Data Sources" Toxins 5, no. 9: 1597-1628. https://doi.org/10.3390/toxins5091597
APA StyleBacker, L. C., Landsberg, J. H., Miller, M., Keel, K., & Taylor, T. K. (2013). Canine Cyanotoxin Poisonings in the United States (1920s–2012): Review of Suspected and Confirmed Cases from Three Data Sources. Toxins, 5(9), 1597-1628. https://doi.org/10.3390/toxins5091597




