Trichothecenes: From Simple to Complex Mycotoxins
Abstract
:1. Introduction
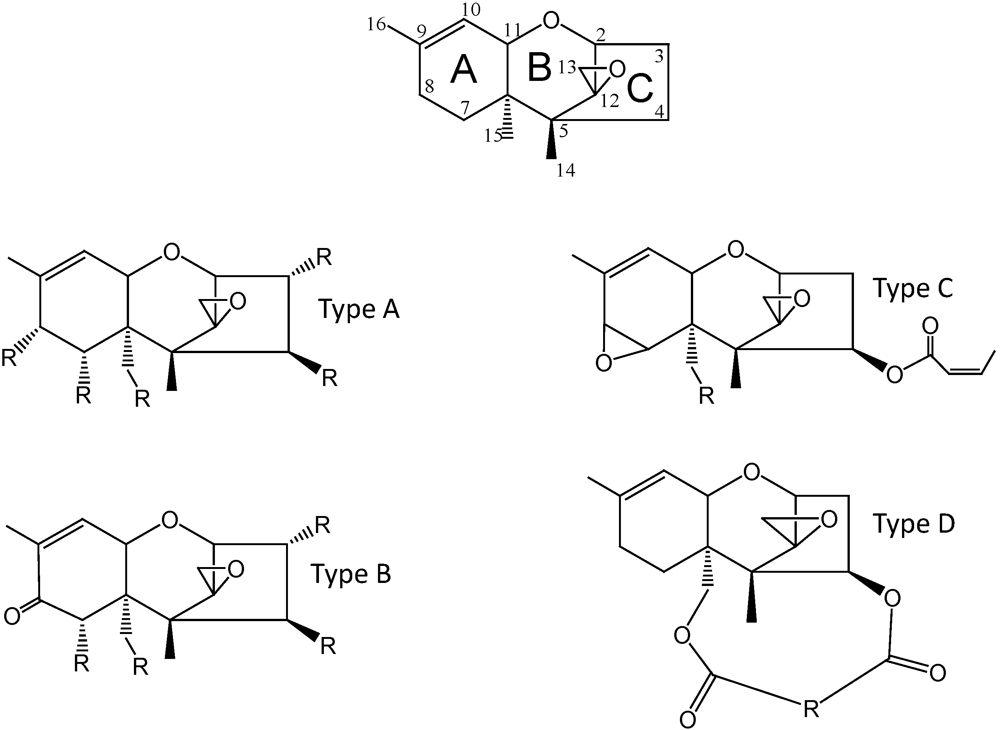
2. Trichothecene Biosynthesis in Fusarium
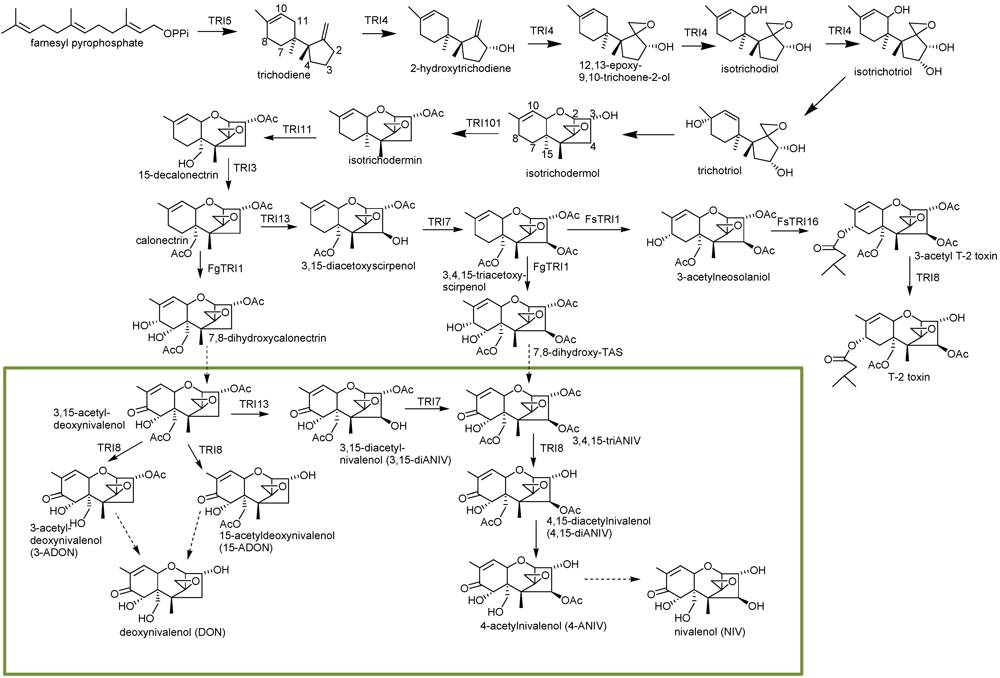
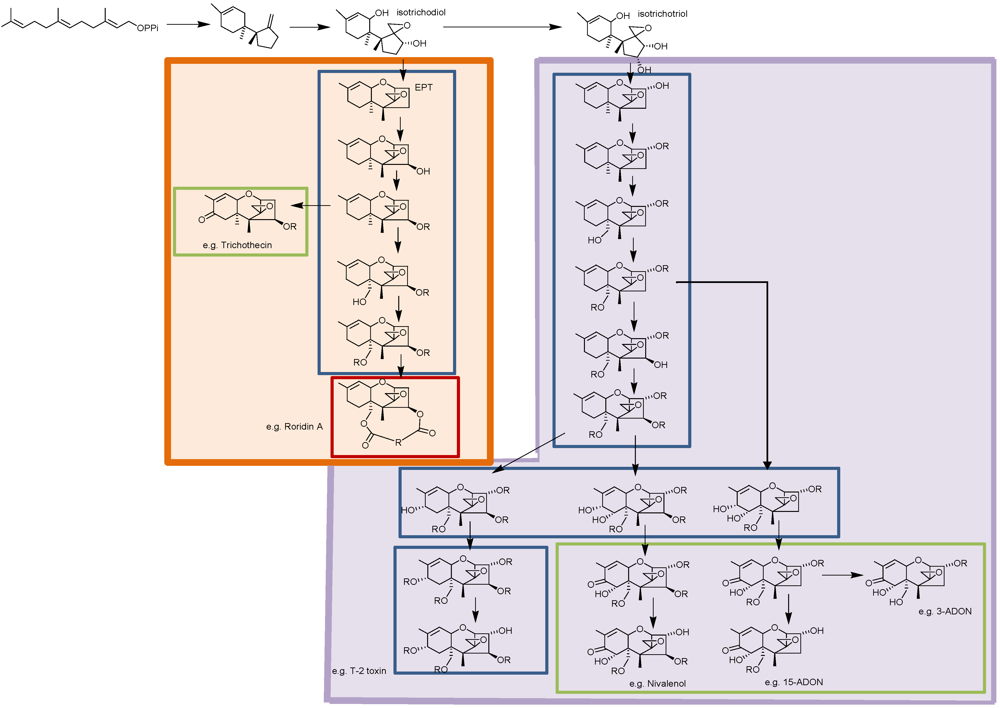
3. Trichothecene Biosynthesis in Other Genera
4. Genetic Approach to Trichothecene Classification
| Trichothecene | Simple or Macrocyclic (S or M) | Type 1 | Group 2 | Type 3 |
|---|---|---|---|---|
| 1 trichodermol | S | A | I | d |
| 2 trichodermin | S | A | I | d |
| 3 4,15-Diacetoxyscirpenol (DAS) | S | A | t | |
| 4 neosolaniol | S | A | II | t |
| 5 T-2 toxin | S | A | II | t |
| 6 isotrichodermol | S | A | I | t |
| 7 calonectrin | S | A | t | |
| 8 7,8-dihydroxy calonectrin | S | A | II | t |
| 9 harzianum A | S | A | d | |
| 10 nivalenol (NIV) | S | B | III | t |
| 11 deoxynivalenol (DON) | S | B | III | t |
| 12 fusarenon-X | S | B | III | t |
| 13 trichothecin | S | B | III | d |
| 14 trichothecinol A | S | B | III | t |
| 15 crotocin | S | C | d | |
| 16 satratoxin H | M | D | d | |
| 17 roridin A | M | D | d | |
| 18 baccharin | M | D | d | |
| 19 verrucarin A | M | D | d |
5. Conclusions
Acknowledgments
References
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism-from biochemistry to genomics. Nat. Rev. Microbiol. 2005, 3, 937–947. [Google Scholar]
- Ueno, Y. Mode of action of trichothecenes. Pure Appl. Chem. 1977, 49, 1737–1745. [Google Scholar]
- Parry, D.W.; Jenkinson, P.; McLeod, L. Fusarium ear blight (scab) in small grain cereals-a review. Plant Pathol. 1995, 44, 207–238. [Google Scholar]
- McMullen, M.; Jones, R.; Gallenberg, D. Scab of wheat and barley: A re-emerging disease of devastating impact. Plant Dis. 1997, 81, 1340–1348. [Google Scholar]
- Cole, R.A.; Jarvis, B.B.; Schweikert, M.A. Handbook of Secondary Metabolites; Academic Press: New York, NY, USA, 2003; pp. 199–560. [Google Scholar]
- Desjardins, A.E. Fusarium Mycotoxins Chemistry, Genetics and Biology; APS Press: Eagan, MN, USA, 2006; pp. 1–260. [Google Scholar]
- Bean, G.A.; Fernando, T.; Jarvis, B.B.; Burton, B. The isolation and identification of trichothecene metabolites from a plant pathogenic strain of Myrothecium roridum. J. Nat. Prod. 1984, 47, 727–729. [Google Scholar] [CrossRef] [PubMed]
- Kuti, J.O.; Ng, T.J.; Bean, G.A. Possible involvement of a pathogen-produced trichothecene metabolite in Myrothecium leaf spot of muskmelon. Physiol. Mol. Plant Pathol. 1989, 34, 41–54. [Google Scholar] [CrossRef]
- Pestka, J.J.; Forstell, J.H. Inhibition of human lymphocyte transformation by the macrocyclic trichothecenes roridin A and verrucarin A. Toxicol. Lett. 1988, 41, 215–222. [Google Scholar]
- Degenkolb, T.; Dieckmann, R.; Nielsen, K.F.; Gräfenhan, T.; Theis, C.; Zafari, D.; Chaverri, P.; Ismaiel, A.; Brückner, H.; von Döhren, H.; et al. The Trichoderma brevicompactum clade: A separate lineage with new species, new peptaibiotics, and mycotoxins. Mycol. Prog. 2008, 7, 177–219. [Google Scholar] [CrossRef]
- Farr, D.F.; Bills, G.F.; Chamuris, G.P.; Rossman, A.Y. Fungi on Plants and Plant Products in the United States; APS Press: Eagan, MN, USA, 1989. [Google Scholar]
- Carrasco, L.; Barbacid, M.; Vazquez, D. The trichodermin group of antibiotics, inhibitors of peptide bond formation by eukaryotic ribosomes. Biochim. Biophys. Acta 1973, 312, 368–376. [Google Scholar]
- Cundliffe, E.; Cannon, M.; Davies, J. Mechanism of inhibition of eukaryotic protein synthesis by trichothecene fungal toxins. Proc. Natl. Acad. Sci. USA 1974, 71, 30–34. [Google Scholar] [CrossRef]
- Cundliffe, E.; Davies, J.E. Inhibition of initiation, elongation, and termination of eukaryotic protein synthesis by trichothecene fungal toxins. Antimicrob. Agents Chemother. 1977, 11, 491–499. [Google Scholar] [PubMed]
- Ueno, Y. The toxicology of mycotoxins. Crit. Rev. Toxicol. 1985, 14, 99–132. [Google Scholar]
- Pace, J.G.; Watts, M.R.; Canterbury, W.J. T-2 mycotoxin inhibits mitochondrial protein synthesis. Toxicon 1988, 26, 77–85. [Google Scholar]
- McLaughlin, J.E.; Bin-Umer, M.A.; Tortora, A.; Mendeze, N.; McCormick, S.; Tumer, N.E. A genome-wide screen in Saccharomyces cerevisiae reveals a critical role for the mitochondria in the toxicity of a trichothecene mycotoxin. Proc. Natl. Acad. Sci. USA 2009, 106, 21883–21888. [Google Scholar]
- Ueno, Y.; Matsumoto, H. Inactivation of some thiol-enzymes by trichothecene mycotoxins from Fusarium species. Chem. Pharm. Bull. 1975, 23, 2439–2442. [Google Scholar] [PubMed]
- Suneja, S.K.; Wagle, D.S.; Ram, G.C. Effect of oral administration of T-2 toxin on glutathione shuttle enzymes, microsomal reductases and lipid peroxidation in rat liver. Toxicon 1989, 27, 995–1001. [Google Scholar]
- Riley, R.T.; Norred, W.P. Mechanisms of Mycotoxicity. In The Mycota VI: Human and Animal Relationships; Howard, D.H., Miller, J.D., Eds.; Springer-Verlag: New York, NY, USA, 1996; pp. 193–211. [Google Scholar]
- Middlebrook, J.L.; Leatherman, D.L. Specific associations of T-2 toxin with mammalian cells. Biochem. Pharmacol. 1989, 38, 3093–3102. [Google Scholar]
- Wannemacher, R.W.; Winer, S.L. Trichothecene Mycotoxins. In Medical Aspects of Chemical and Biological Warfare; Sidell, R.R., Takafuji, E.T., Franz, D.R., Eds.; Office of the Surgeon General at TMM Publications: Washington, DC, USA, 1977; pp. 655–676. [Google Scholar]
- Pestka, J.J. Mechanisms of deoxynivalenol-induced gene expression and apoptosis. Food Addit. Contam. 2008, 22, 1128–1140. [Google Scholar]
- McLean, M. The phytotoxicity of Fusarium metabolites: An update since 1989. Mycopathologia 1996, 133, 163–179. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.H.; Hohn, T.M.; McCormick, S.P. Reduced virulence of Gibberella zeae caused by disruption of a trichothecene toxin biosynthetic gene. Mol. Plant-Microbe Interact. 1995, 61, 1923–1930. [Google Scholar]
- Proctor, R.H.; Hohn, T.M.; McCormick, S.P. Restoration of wild-type virulence to Tri5 disruption mutants of Gibberella zeae via gene reversion and mutant complementation. Microbiology 1997, 143, 2583–2591. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E.; Proctor, R.H.; Bai, G.; McCormick, S.P.; Shaner, G.; Beuchley, G.; Hohn, T.M. Reduced virulence of trichothecene-non-producing mutants of Gibberella zeae in wheat field tests. Mol. Plant-Microbe Interact. 1996, 9, 1996–1023. [Google Scholar]
- Grove, J.F. The trichothecenes and their biosynthesis. Prog. Chem. Org. Nat. Prod. 2007, 88, 63–130. [Google Scholar]
- Ueno, Y. Toxicological features of T-2 toxin and related trichothecenes. Fundam. Appl. Toxicol. 1984, 4, S124–S132. [Google Scholar]
- Ueno, Y. Mode of action of trichothecenes. Ann. Nutr. Aliment. 1977, 31, 885–900. [Google Scholar]
- Altomare, C.; Logrieco, A.; Bottalico, A.; Mulé, G.; Moretti, A.; Evidente, A. Production of type S trichothecenes and enniatin B by Fusarium sambucinum Fuckel sensu lato. Mycopathologia 1995, 129, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, C.; Barthel, J.; Engelhardt, G.; Bauer, J.; Meyer, K. Simultaneous determination of type A, B and D trichothecenes and their occurrence in cereals and cereal products. Food Addit. Contam. A 2009, 26, 1273–1289. [Google Scholar]
- Garvey, G.S.; McCormick, S.P.; Rayment, I. Structural and functional characterization of the TRI101 trichothecene 3-0-acetyltransferase from Fusarium sporotrichioides and Fusarium graminearum: Kinetic insights to combating Fusarium head blight. J. Biol. Chem. 2008, 283, 1660–1669. [Google Scholar] [PubMed]
- Hohn, T.M.; VanMiddlesworth, F. Purification and characterization of the sesquiterpene cyclase trichodiene synthetase from Fusarium sporotrichioides. Arch. Biochem. Biophys. 1986, 251, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Hohn, T.M.; Beremand, P.D. Isolation and nucleotide sequence of a sesquiterpene cyclase gene from the trichothecene-producing fungus Fusarium sporotrichioides. Gene 1989, 79, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Hohn, T.M.; Desjardins, A.E.; McCormick, S.P. The Tri4 gene of Fusarium sporotrichioides encodes a cytochrome P450 monooxygenase involved in trichothecene biosynthesis. Mol. Plant-Microbe Interact. 1995, 248, 95–102. [Google Scholar]
- McCormick, S.P.; Alexander, N.J.; Proctor, R.H. Fusarium Tri4 encodes a multifunctional oxygenase required for trichothecene biosynthesis. Can. J. Microbiol. 2006, 52, 636–642. [Google Scholar] [PubMed]
- McCormick, S.P.; Taylor, S.L.; Plattner, R.D.; Beremand, M.N. Bioconversion of possible T-2 toxin precursors by a mutant strain of Fusarium sporotrichioides. Appl. Environ. Microbiol. 1990, 56, 702–706. [Google Scholar] [PubMed]
- McCormick, S.P.; Alexander, N.J.; Trapp, S.E.; Hohn, T.M. Disruption of TRI101, the gene encoding trichothecene 3-O-acetyltransferase, from Fusarium sporotrichioides. Appl. Environ. Microbiol. 1999, 65, 5252–5256. [Google Scholar] [PubMed]
- Kimura, M.; Kaneko, I.; Komiyama, M.; Takatsuki, A.; Koshino, H.; Yoneyama, K.; Yamaguchi, I. Trichothecene 3-O-acetyltransferase protects both the producing organism and transformed yeast from related mycotoxins. J. Biol. Chem. 1998, 273, 1654–1661. [Google Scholar] [PubMed]
- Alexander, N.J.; Hohn, T.M.; McCormick, S.P. The TRI11 gene of Fusarium sporotrichioides encodes a cytochrome P450 monooxygenase required for C-15 hydroxylation in trichothecene biosynthesis. Appl. Environ. Microbiol. 1998, 64, 221–225. [Google Scholar] [PubMed]
- McCormick, S.P.; Hohn, T.M.; Desjardins, A.E. Isolation and characterization of Tri3, a gene encoding 15-O-acetyltransferase from Fusarium sporotrichioides. Appl. Environ. Microbiol. 1996, 62, 353–359. [Google Scholar] [PubMed]
- Garvey, G.S.; McCormick, S.P.; Alexander, N.J.; Rayment, I. Structural and functional characterization of TRI3 trichothecene 15-O-acetyltransferase from Fusarium sporotrichioides. Protein Sci. 2009, 18, 747–761. [Google Scholar] [PubMed]
- Lee, T.; Han, Y.-K.; Kim, K.-H.; Yun, S.-H.; Lee, Y.-W. Tri13 and Tri7 determine deoxynivalenol- and nivalenol-producing chemotypes of Gibberella zeae. Appl. Environ. Microbiol. 2002, 68, 2148–2154. [Google Scholar] [PubMed]
- Brown, D.W.; McCormick, S.P.; Alexander, N.J.; Proctor, R.H.; Desjardins, A.E. Inactivation of a cytochrome P-450 is a determinant of trichothecene diversity in Fusarium species. Fungal Genet. Biol. 2002, 36, 224–233. [Google Scholar] [CrossRef] [PubMed]
- McCormick, S.P.; Alexander, N.J. Fusarium Tri8 encodes a trichothecene C-3 esterase. Appl. Environ. Microbiol. 2002, 68, 2959–2964. [Google Scholar] [CrossRef] [PubMed]
- McCormick, S.P.; Harris, L.J.; Alexander, N.J.; Ouellet, T.; Saparno, A.; Allard, S.; Desjardins, A.E. Tri1 in Fusarium graminearum encodes a P450 oxygenase. Appl. Environ. Microbiol. 2004, 70, 2044–2051. [Google Scholar] [PubMed]
- McCormick, S.P.; Alexander, N.J.; Proctor, R.H. Heterologous expression of two trichothecene P450 genes in Fusarium verticillioides. Can. J. Microbiol. 2006, 52, 220–226. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; McCormick, S.P.; Waalwijk, C.; van der Lee, T.; Proctor, R.H. The genetic basis for 3-ADON and 15-ADON trichothecene chemotypes in Fusarium graminearum. Fungal Genet. Biol. 2011, 48, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Trapp, S.E.; Hohn, T.M.; McCormick, S.P.; Jarvis, B.B. Characterization of the gene cluster for biosynthesis of macrocyclic trichothecenes in Myrothecium roridum. Mol. Gen. Genet. 1998, 257, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Tokai, T.; Koshino, H.; Takahashi-Ando, N.; Sato, M.; Fujimura, M.; Kimura, M. Fusarium Tri4 encodes a key multifunctional cytochrome P450 monooxygenase for four consecutive oxygenation steps in trichothecene biosynthesis. Biochim. Biophys. Acta 2007, 353, 412–417. [Google Scholar]
- Gallo, A.; Mulé, G.; Favilla, M.; Altomare, C. Isolation and characterisation of a trichodiene synthase homologous gene in Trichoderma harzianum. Physiol. Mol. Plant Pathol. 2004, 65, 11–20. [Google Scholar] [CrossRef]
- Tijerino, A.; Cardoza, R.E.; Moraga, J.; Malmierca, M.G.; Vicente, F.; Aleu, J.; Collado, I.G.; Gutiérrez, S.; Monte, E.; Hermosa, R. Overexpression of trichodiene synthase gene tri5 increases trichodermin production and antimicrobial activity in Trichoderma brevicompactum. Fungal Genet. Biol. 2011, 48, 285–296. [Google Scholar] [CrossRef] [PubMed]
- McCormick, S.P.; Alexander, N.J. Myrothecium roridum Tri4 encodes a multifunctional oxygenase required for three oxygenation steps. Can. J. Microbiol. 2007, 53, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Iida, A.; Knoisha, K.; Kubo, H.; Tomioka, K.; Tokuda, H.; Nishino, H. Trichothecinols A, B and C. Potent anti-tumor promoting sesquiterpenoids from the fungus Trichothecium roseum. Tetrahedron Lett. 1996, 51, 9219–9220. [Google Scholar]
- Hohn, T.M.; McCormick, S.P.; Desjardins, A.E. Evidence for a gene cluster involving trichothecene-pathway biosynthetic genes in Fusarium sporotrichioides. Curr. Genet. 1993, 24, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Keller, N.P.; Hohn, T.M. Metabolic pathway gene clusters in filamentous fungi. Fungal Genet. Biol. 1997, 21, 17–29. [Google Scholar]
- Alexander, N.J.; Proctor, R.H.; McCormick, S.P. Genes, gene clusters, and biosynthesis of trichothecenes and fumonisins in Fusarium. Toxin Rev. 2009, 28, 198–215. [Google Scholar] [CrossRef]
- Proctor, R.H.; McCormick, S.P.; Alexander, N.J.; Desjardins, A.E. Evidence that a secondary metabolic biosynthetic gene cluster has grown by gene relocation during evolution of the filamentous fungus Fusarium. Mol. Microbiol. 2009, 74, 1128–1142. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.W.; Dyer, R.B.; McCormick, S.P.; Kendra, D.F.; Plattner, R.D. Functional demarcation of the Fusarium core trichothecene gene cluster. Fungal Genet. Biol. 2004, 41, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Meek, I.B.; Peplow, A.W.; Ake, C., Jr.; Phillips, T.D.; Beremand, M.N. Tri1 encodes the cytochrome P450 monooxygenase for C-8 hydroxylation during trichothecene biosynthesis in Fusarium sporotrichioides. Appl. Environ. Microbiol. 2003, 69, 1607–1613. [Google Scholar] [PubMed]
- Peplow, A.W.; Meek, I.B.; Wiles, M.C.; Phillips, T.D.; Beremand, M.N. Tri16 is required for esterification of position C-8 during trichothecene mycotoxin production by Fusarium sporotrichioides. Appl. Environ. Microbiol. 2003, 69, 5935–5940. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; McAlees, A.J.; Shingu, Y.; Yoneyama, K.; Yamaguchi, I. The mystery of the trichothecene 3-O-acetyltransferase gene. FEBS Lett. 1998, 435, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; McCormick, S.P.; Larson, T.M.; Jurgenson, J.E. Expression of Tri15 in Fusarium sporotrichioides. Curr. Genet. 2004, 45, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Seong, K.-Y.; Pasquali, M.; Zhou, X.; Song, J.; Hilbrun, K.; McCormick, S.P.; Dong, Y.; Xu, J.R.; Kistler, H.C. Global gene regulation by Fusarium transcription factors Tri6 and Tri10 reveals adaptations for toxin biosynthesis. Mol. Microbiol. 2009, 72, 354–367. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Tokai, T.; Takahashi-Ando, N.; Ohsato, S.; Fujimura, M. Molecular and genetic studies of Fusarium trichothecene biosynthesis: Pathways, genes and evolution. Biosci. Biotechnol. Biochem. 2007, 2105–2123. [Google Scholar] [PubMed]
- Grove, J.F. Non-macrocyclic trichothecenes. Nat. Prod. Rep. 1988, 5, 187–209. [Google Scholar]
- Kimura, M.; Shingu, Y.; Yoneyama, K.; Yamaguchi, I. Features of Tri101, the trichothecene3-O-acetyltransferase gene, related to the self-defense mechanism in Fusarium graminearum. Biosci. Biotechnol. Biochem. 1998, 62, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Islam, Z.; Shinozuka, J.; Harkema, J.R.; Pestka, J.J. Purification and comparative neurotoxicity of the trichothecenes satratoxin G and roridin L2 from Stachybotrys chartarum. J. Toxicol. Environ. Health A 2009, 72, 1242–1251. [Google Scholar] [CrossRef] [PubMed]
- Bondy, G.S.; McCormick, S.P.; Beremand, M.N.; Pestka, J.J. Murine lymphocyte proliferation impaired by substituted neosolaniols and calonectrins-Fusarium metabolites associated with trichothecene biosynthesis. Toxicon 1991, 29, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; McCormick, S.P.; Ziegenhorn, S.L. Phytotoxicity of selected trichothecenes using Chlamydomonas reinhardtii as a model system. Nat. Toxins 1999, 7, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Desjardins, A.E.; McCormick, S.P.; Appell, M. Structure-activity relationships of trichothecene toxins in an Arabidopsis thaliana leaf assay. J. Agric. Food Chem. 2007, 55, 6487–6492. [Google Scholar] [PubMed]
- Starkey, D.E.; Ward, T.J.; Aoki, T.; Gale, L.R.; Kistler, H.C.; Geiser, D.M.; Suga, H.; Tóth, B.; Varga, J.; O'Donnell, K. Global molecular surveillance reveals novel Fusarium head blight species and trichothecene toxin diversity. Fungal Genet. Biol. 2007, 44, 1191–1204. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; van der Lee, T.; Yang, X.; Yu, D.; Waalwijk, C. Fusarium populations on Chinese barley show a dramatic gradient in mycotoxin profiles. Phytopathology 2008, 98, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Zhang, Z.; van der Lee, T.; Chen, W.Q.; Xu, J.; Xu, J.S.; Yang, L.; Yu, D.; Waalwijk, C.; Feng, J. Population genetic analyses of Fusarium asiaticum populations from barley suggest a recent shift favoring 3ADON producers in southern China. Phytopathology 2010, 100, 328–336. [Google Scholar] [PubMed]
- Druzhinina, I.S.; Kubicek, C.P.; Komoń-Zelazowska, M.; Mulaw, T.B.; Bissett, J. The Trichoderma harzianum demon: Complex speciation history resulting in coexistence of hypothetical biological species, recent agamospecies and numerous relict lineages. BMC Evol. Biol. 2010, 10, 94. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From Simple to Complex Mycotoxins. Toxins 2011, 3, 802-814. https://doi.org/10.3390/toxins3070802
McCormick SP, Stanley AM, Stover NA, Alexander NJ. Trichothecenes: From Simple to Complex Mycotoxins. Toxins. 2011; 3(7):802-814. https://doi.org/10.3390/toxins3070802
Chicago/Turabian StyleMcCormick, Susan P., April M. Stanley, Nicholas A. Stover, and Nancy J. Alexander. 2011. "Trichothecenes: From Simple to Complex Mycotoxins" Toxins 3, no. 7: 802-814. https://doi.org/10.3390/toxins3070802
APA StyleMcCormick, S. P., Stanley, A. M., Stover, N. A., & Alexander, N. J. (2011). Trichothecenes: From Simple to Complex Mycotoxins. Toxins, 3(7), 802-814. https://doi.org/10.3390/toxins3070802





