Llama-Derived Single Domain Antibodies Specific for Abrus Agglutinin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Abrax
2.3. Library Construction and Selection
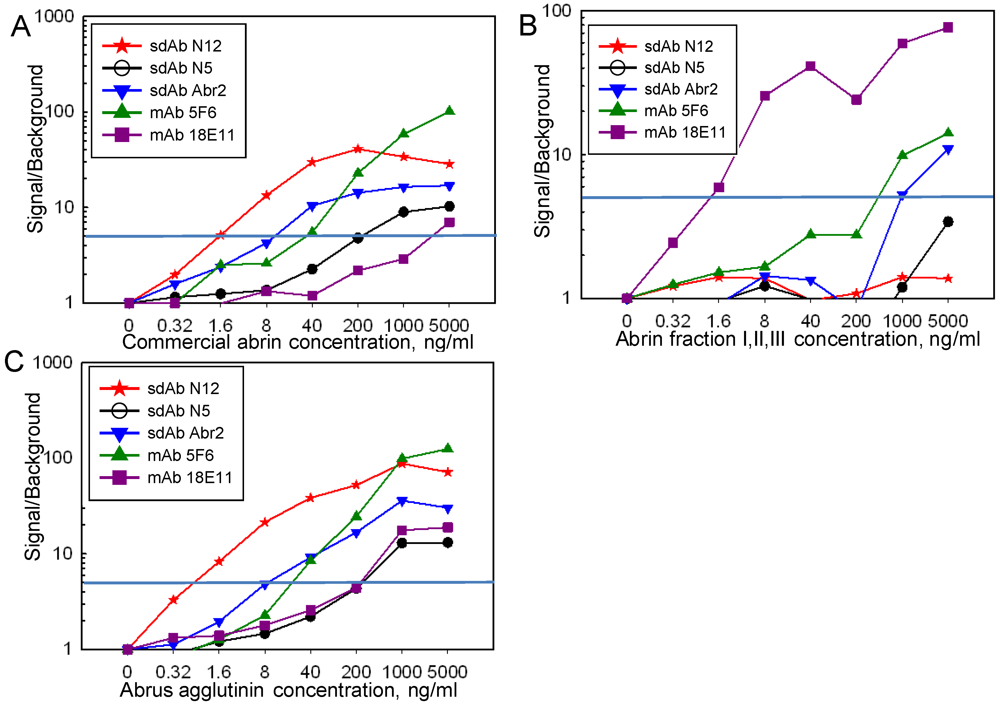
2.4. Luminex Assays
2.5. Surface Plasmon Resonance (SPR)
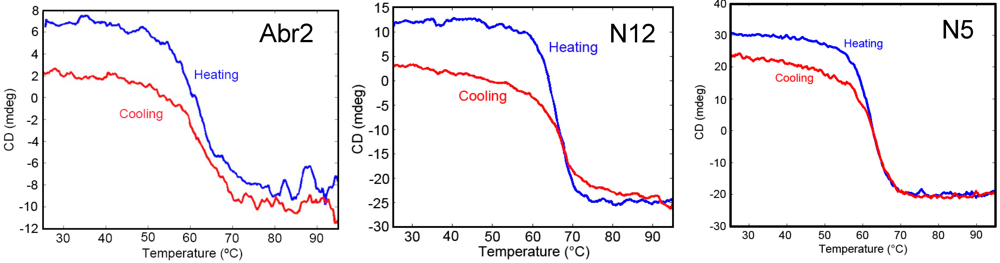
2.6. Circular Dichroism (CD)
3. Results and Discussion
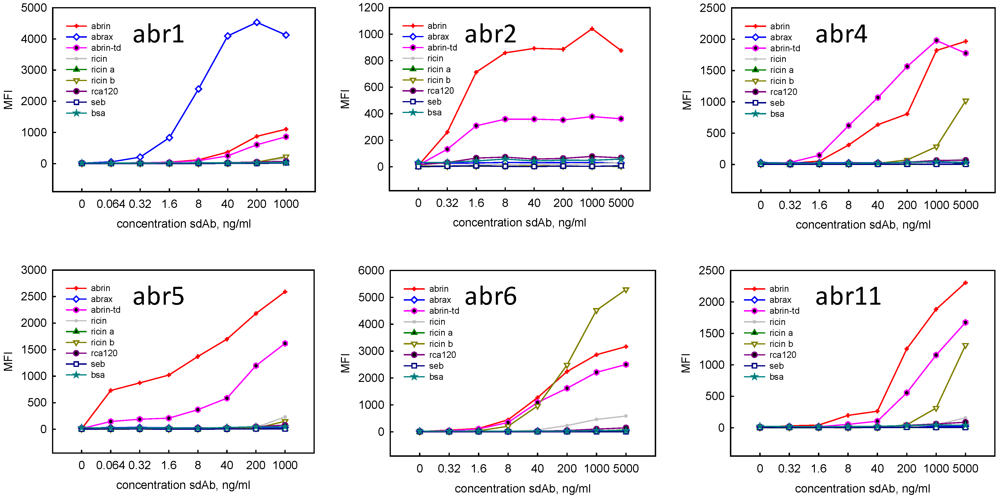
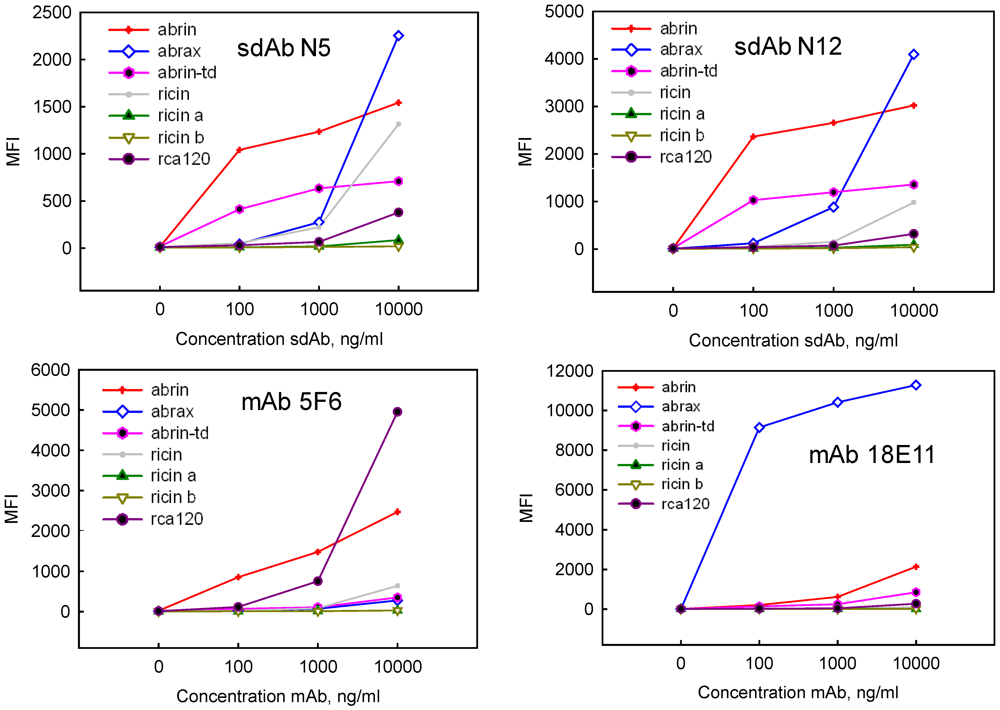
| Binder | Commercial Abrin | Abrax | ||||
|---|---|---|---|---|---|---|
| on, 1/Ms | off, 1/s | Kd, M a | on, 1/Ms | off, 1/s | Kd, M a | |
| mAb 5F6 | 1.5 × 105 | 6.1 × 10−4 | 4.0 × 10−9 | NB | NB | NB |
| mAb 18E11 | 2.0 × 105 | 1.9 × 10−2 | 9.3 × 10−8 | 1.5 × 105 | 3.4 × 102 | 2.2 × 10−7 |
| Abr1 | NB | NB | NB | 1.8 × 105 | 4.79 × 105 | 2.64 × 10−10 |
| Abr2 | 2.2 × 106 | 3.2 × 10−5 | 1.5 × 10−11 | NB | NB | NB |
| Abr4 | 4.2 × 105 | 2.7 × 10−4 | 6.5 × 10−10 | NB | NB | NB |
| N5 | 1.6 × 105 | 3.1 × 10−5 | 1.9 × 10−10 | NB | NB | NB |
| N12 | 3.7 × 106 | 4.5 × 10−6 | 1.2 × 10−12 | NB | NB | NB |
| Abr6 | 8.3 × 104 | 1.1 × 10−4 | 1.3 × 10−9 | NB | NB | NB |
| 3E | 3.3 × 105 | 2.4 × 10−4 | 7.4 × 10−10 | NB | NB | NB |
| 10C | 8.6 × 105 | 9.7 × 10−4 | 1.1 × 10−9 | NB | NB | NB |
| Binder | Abrin frac I | Abrus Agglutinin | ||||
|---|---|---|---|---|---|---|
| on, 1/Ms | off, 1/s | Kd, M a | on, 1/Ms | off, 1/s | Kd, M a | |
| mAb 5F6 | 1.6 × 105 | 12.1 × 10−4 | 1.3 × 10−9 | 1.5 × 105 | 6.3 × 10−4 | 4.3 × 10−9 |
| mAb 18E11 | 1.2 × 105 | 1.1 × 10−2 | 8.5 × 10−8 | 1.8 × 105 | 1.6 × 10−2 | 9.0 × 10−8 |
| Abr1 | NB | NB | NB | NB | NB | NB |
| Abr2 | NB | NB | NB | 1.1 × 106 | 1.6 × 10−5 | 1.4 × 10−11 |
| Abr4 | 1.5 × 105 | 1.6 × 10−4 | 1.1 × 10−9 | 5.1 × 105 | 4.9 × 10−4 | 9.5 × 10−10 |
| N5 | NB | NB | NB | 1.8 × 105 | 5.3 × 10−5 | 2.9 × 10−10 |
| N12 | NB | NB | NB | 1.6 × 106 | 4.2 × 10−6 | 2.6 × 10−12 |
| Abr6 | 4.0 × 104 | 2.1 × 10−4 | 5.2 × 10−9 | 9.1 × 104 | 2.6 × 10−5 | 2.9 × 10−10 |
| 3E | 1.2 × 105 | 1.6 × 10−4 | 1.3 × 10−9 | 5.3 × 105 | 4.2 × 10−4 | 7.8 × 10−10 |
| 10C | 3.2 × 105 | 6.5 × 10−4 | 2.0 × 10−9 | 1.2 × 106 | 8.1 × 10−4 | 6.6 × 10−10 |
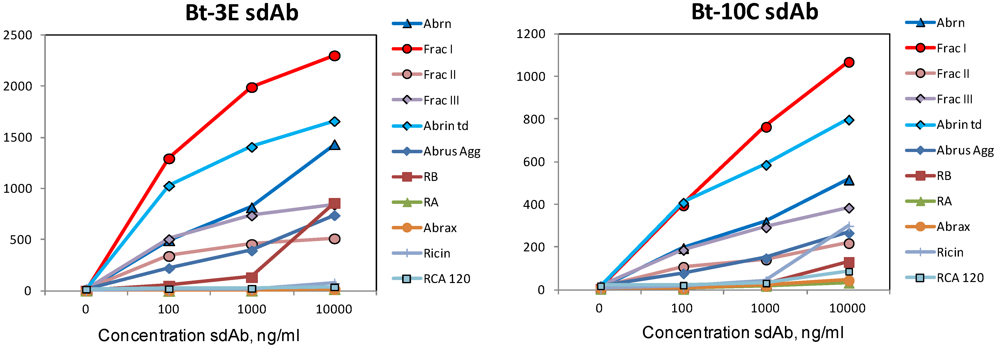
4. Summary
Acknowledgments
Conflict of Interest
Supplementary Files
References
- Endo, Y.; Mitsui, K.; Motizuki, M.; Tsurugi, K. The mechanism of action of ricin and related toxic lectins on eukaryotic ribosomes-The site and characteristics of the modification in 28-S ribosoman-RNA caued by the toxins. J. Biol. Chem. 1987, 262, 5908–5912. [Google Scholar]
- Kozlov, Y.V.; Sudarkina, O.Y.; Kurmanova, A.G. Ribosome-inactivating lectins of plants. Mol. Biol. 2006, 40, 711–723. [Google Scholar]
- Stirpe, F.; Battelli, M.G. Ribosome-inactivating proteins: Progress and problems. Cell. Mol. Life Sci. 2006, 63, 1850–1866. [Google Scholar]
- Robertus, J.D.; Monzingo, A.F. The structure of ribosome inactivating proteins. Mini-Rev. Med. Chem. 2004, 4, 477–486. [Google Scholar] [PubMed]
- Wood, K.A.; Lord, J.M.; Wawrzynczak, E.J.; Piatak, M. Preproabrin-Genomic cloning, characterization and the expression of the A-chain in Escheria-coli. Eur. J. Biochem. 1991, 198, 723–732. [Google Scholar] [PubMed]
- Kimura, M.; Sumizawa, T.; Funatsu, G. The complete amino-acid sequences of the B-chains of Abrin-A and Abrin-B, toxic proteins from the seeds of Abrus-precatorius. Biosci. Biotechnol. Biochem. 1993, 57, 166–169. [Google Scholar]
- Battelli, M.G. Cytotoxicity and toxicity to animals and humans of ribosome-inactivating proteins. Mini-Rev. Med. Chem. 2004, 4, 513–521. [Google Scholar] [PubMed]
- Garber, E.A.E.; Walker, J.L.; O’Brien, T.W. Detection of abrin in food using enzyme-linked immunosorbent assay and electrochemiluminescence technologies. J. Food Prot. 2008, 71, 1868–1874. [Google Scholar]
- Stirpe, F.; Barbieri, L.; Battelli, M.G.; Soria, M.; Lappi, D.A. Ribosome-inactivating proteins from plants-present status and future prospects. Bio-Technology 1992, 10, 405–412. [Google Scholar] [PubMed]
- Hegde, R.; Maiti, T.K.; Podder, S.K. Purification and characteruzation of 3 toxins and agglutinins from Abrus-precatorius seed by using lactamyl-sepharose affinity-chromatography. Anal. Biochem. 1991, 194, 101–109. [Google Scholar] [PubMed]
- Hegde, R.; Karande, A.A.; Podder, S.K. The variants of the protein toxins abrin and ricin-A useful guide to understanding the processing events in the toxin transport. Eur. J. Biochem. 1993, 215, 411–419. [Google Scholar]
- Hegde, R.; Podder, S.K. Studies on the variants of the protein toxins ricin and abrin. Eur. J. Biochem. 1992, 204, 155–164. [Google Scholar]
- Hegde, R.; Podder, S.K. A- and B-subunit variant distribution in the holoprotein variants of protein toxin abrin: Variants of abrins I and, III have constant toxic A subunits and variant lectin B subunits. Arch. Biochem. Biophys. 1997, 344, 75–84. [Google Scholar]
- Liu, C.L.; Tsai, C.C.; Lin, S.C.; Wang, L.I.; Hsu, C.I.; Hwang, M.J.; Lin, J.Y. Primary structure and function analysis of the Abrus precatorius agglutinin A chain by site-directed mutagenesis-Pro(199) of amphiphilic alpha-helix H impairs protein synthesis inhibitory activity. J. Biol. Chem. 2000, 275, 1897–1901. [Google Scholar] [PubMed]
- Bagaria, A.; Surendranath, K.; Ramagopal, U.A.; Ramakumar, S.; Karande, A.A. Structure-function analysis and insights into the reduced toxicity of Abrus precatorius agglutinin I in relation to abrin. J. Biol. Chem. 2006, 281, 34465–34474. [Google Scholar] [PubMed]
- Garber, E.A.E. Toxicity and detection of ricin and abrin in beverages. J. Food Prot. 2008, 71, 1875–1883. [Google Scholar]
- Garber, E.A.E.; Venkateswaran, K.V.; O’Brien, T.W. Simultaneous multiplex detection and confirmation of the proteinaceous toxins abrin, ricin, botulinum toxins, and staphylococcus enterotoxins A, B, and C in . J. Agric. Food Chem. 2010, 58, 6600–6607. [Google Scholar]
- Zhou, H.Y.; Zhou, B.; Ma, H.Z.; Carney, C.; Janda, K.D. Selection and characterization of human monoclonal antibodies against Abrin by phage display. Bioorg. Med. Chem. Lett. 2007, 17, 5690–5692. [Google Scholar]
- Yang, W.; Li, X.B.; Liu, G.W.; Zhang, B.B.; Zhang, Y.; Kong, T.; Tang, J.J.; Li, D.N.; Wang, Z. A colloidal gold probe-based silver enhancement immunochromatographic assay for the rapid detection of abrin-a. Biosens. Bioelectron. 2011, 26, 3710–3713. [Google Scholar]
- Mu, X.H.; Zhou, Z.Q.; Tong, Z.Y.; Liu, B.; Hao, L.Q. Detection of Abrin by Piezoelectric Immunosensor Based on Biotin-Avidin System. Chin. J. Anal. Chem. 2009, 37, 1499–1502. [Google Scholar]
- Pauly, D.; Kirchner, S.; Stoermann, B.; Schreiber, T.; Kaulfuss, S.; Schade, R.; Zbinden, R.; Avondet, M.A.E.; Dorner, M.B.; Dorner, B.G. Simultaneous quantification of five bacterial and plant toxins from complex matrices using a multiplexed fluorescent magnetic suspension assay. Analyst 2009, 134, 2028–2039. [Google Scholar]
- Tang, J.J.; Yu, T.; Guo, L.; Xie, J.W.; Shao, N.S.; He, Z.K. In vitro selection of, DNA aptamer against abrin toxin and aptamer-based abrin direct detection. Biosens. Bioelectron. 2007, 22, 2456–2463. [Google Scholar] [CrossRef] [PubMed]
- Hamerscasterman, C.; Atarhouch, T.; Muyldermans, S.; Robinson, G.; Hamers, C.; Songa, E.B.; Bendahman, N.; Hamers, R. Naturally-occurring antibodies devoid of light-chains. Nature 1993, 363, 446–448. [Google Scholar]
- Greenberg, A.S.; Avila, D.; Hughes, M.; Hughes, A.; McKinney, E.C.; Flajnik, M.F. A new antigen receptor gene family that undergoes rearrangement and extensive somatic diversification in sharks. Nature 1995, 374, 168–173. [Google Scholar]
- Perez, JM.J.; Renisio, J.G.; Prompers, J.J.; van Platerink, C.J.; Cambillau, C.; Darbon, H.; Frenken, L.G.J. Thermal unfolding of a llama antibody fragment: A two-state reversible process. Biochemistry 2001, 40, 74–83. [Google Scholar]
- van der Linden, RH.J.; Frenken, LG.J.; de Geus, B.; Harmsen, M.M.; Ruuls, R.C.; Stok, W.; de Ron, L.; Wilson, S.; Davis, P.; Verrips, C.T. Comparison of physical chemical properties of llama V-HH antibody fragments and mouse monoclonal antibodies. Biochim. Biophys. Acta-Protein Struct. Mol. Enzymol. 1999, 1431, 37–46. [Google Scholar] [CrossRef]
- Dona, V.; Urrutia, M.; Bayardo, M.; Alzogaray, V.; Goldbaum, F.A.; Chirdo, F.G. Single domain antibodies are specially suited for quantitative determination of gliadins under denaturing conditions. J. Agric. Food Chem. 2010, 58, 918–926. [Google Scholar]
- Wesolowski, J.; Alzogaray, V.; Reyelt, J.; Unger, M.; Juarez, K.; Urrutia, M.; Cauerhff, A.; Danquah, W.; Rissiek, B.; Scheuplein, F.; Schwarz, N.; Adriouch, S.; Boyer, O.; Seman, M.; Licea, A.; Serreze, D.V.; Goldbaum, F.A.; Haag, F.; Koch-Nolte, F. Single domain antibodies: Promising experimental and therapeutic tools in infection and immunity. Med. Microbiol. Immunol. 2009, 198, 157–174. [Google Scholar]
- Muyldermans, S.; Baral, T.N.; Retarnozzo, V.C.; de Baetselier, P.; de Genst, E.; Kinne, J.; Leonhardt, H.; Magez, S.; Nguyen, V.K.; Revets, H.; Rothbauer, U.; Stijemans, B.; Tillib, S.; Wernery, U.; Wyns, L.; Hassanzadeh-Ghassabeh, G.; Saerens, D. Camelid immunoglobulins and nanobody technology. Vet. Immunol. Immunopathol. 2009, 128, 178–183. [Google Scholar]
- de Marco, A. Biotechnological applications of recombinant single-domain antibody fragments. Microb. Cell Fact. 2011, 10, 44–1. [Google Scholar]
- Smallshaw, J.E.; Firan, A.; Fulmer, J.R.; Ruback, S.L.; Ghetie, V.; Vitetta, E.S. A novel recombinant vaccine which protects mice against ricin intoxication. Vaccine 2002, 20, 3422–3427. [Google Scholar]
- Ghahroudi, M.A.; Desmyter, A.; Wyns, L.; Hamers, R.; Muyldermans, S. Selection and identification of single domain antibody fragments from camel heavy-chain antibodies. FEBS Lett. 1997, 414, 521–526. [Google Scholar]
- Goldman, E.R.; Anderson, G.P.; Liu, J.L.; Delehanty, J.B.; Sherwood, L.J.; Osborn, L.E.; Cummins, L.B.; Hayhurst, A. Facile generation of heat-stable antiviral and antitoxin single domain antibodies from a semisynthetic llama library. Anal. Chem. 2006, 78, 8245–8255. [Google Scholar]
- Sherwood, L.J.; Osborn, L.E.; Carrion, R., Jr.; Patterson, J.L.; Hayhurst, A. Rapid assembly of sensitive antigen-capture assays for Marburg virus, using in vitro selection of llama single-domain antibodies, at biosafety level. J. Infect. Dis. 2007, 2, S213–S219. [Google Scholar]
- Anderson, G.P.; Matney, R.; Liu, J.L.; Hayhurst, A.; Goldman, E.R. Multiplexed fluid array screening of phage displayed anti-ricin single domain antibodies for rapid assessment of specificity. Biotechniques 2007, 43, 806–811. [Google Scholar]
- Corpet, F. Multiple sequence alignment with hierarchical-clustering. Nucleic Acids Res. 1988, 16, 10881–10890. [Google Scholar]
- Anderson, G.P.; Liu, J.L.; Hale, M.L.; Bernstein, R.D.; Moore, M.; Swain, M.D.; Goldman, E.R. Development of Antiricin Single Domain Antibodies Toward Detection and Therapeutic Reagents. Anal. Chem. 2008, 80, 9604–9611. [Google Scholar]
- Goldman, E.R.; Anderson, G.P.; Conway, J.; Sherwood, L.J.; Fech, M.; Vo, B.; Liu, J.L.; Hayhurst, A. Thermostable Llama Single Domain Antibodies for Detection of Botulinum A Neurotoxin Complex. Anal. Chem. 2008, 80, 8583–8591. [Google Scholar]
- Anderson, G.P.; Bernstein, R.D.; Swain, M.D.; Zabetakis, D.; Goldman, E.R. Binding kinetics of antiricin single domain antibodies and improved detection using a B chain specific binder. Anal. Chem. 2010, 82, 7202–7207. [Google Scholar]
- Lauwereys, M.; Ghahroudi, M.A.; Desmyter, A.; Kinne, J.; Holzer, W.; De Genst, E.; Wyns, L.; Muyldermans, S. Potent enzyme inhibitors derived from dromedary heavy-chain antibodies. EMBO J. 1998, 17, 3512–3520. [Google Scholar]
- Swain, M.D.; Anderson, G.P.; Zabetakis, D.; Bernstein, R.D.; Liu, J.L.; Sherwood, L.J.; Hayhurst, A.; Goldman, E.R. Llama-derived single-domain antibodies for the detection of botulinum A neurotoxin. Anal. Bioanal. Chem. 2010, 398, 339–348. [Google Scholar]
- Sehgal, P.; Khan, M.; Kumar, O.; Vijayaraghayan, R. Purification, characterization and toxicity profile of ricin isoforms from castor beans. Food Chem. Toxicol. 2010, 48, 3171–3176. [Google Scholar]
- Berry, J.D. Rational monoclonal antibody development to emerging pathogens, biothreat agents and agents of foreign animal disease: The antigen scale. Vet. J. 2005, 170, 193–211. [Google Scholar]
- Conway, J.O.; Sherwood, L.J.; Collazo, M.T.; Garza, J.A.; Hayhurst, A. Llama single domain antibodies specific for the 7 botulinum neurotoxin serotypes as heptaplex immunoreagents. PLoS One 2010, 5, e8818–1. [Google Scholar]
- Tremblay, J.M.; Kuo, C.L.; Abeijon, C.; Sepulveda, J.; Oyler, G.; Hu, X.B.; Jin, M.M.; Shoemaker, C.B. Camelid single domain antibodies (VHHs) as neuronal cell intrabody binding agents and inhibitors of Clostridium botulinum neurotoxin (BoNT) proteases. Toxicon 2010, 56, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.B.; Thompson, A.A.; Fan, Y.F.; Lou, J.L.; Conrad, F.; Ho, M.F.; Pires-Alves, M.; Wilson, B.A.; Stevens, R.C.; Marks, J.D. A Single-domain llama antibody potently inhibits the enzymatic activity of botulinum neurotoxin by binding to the non-catalytic alpha-exosite binding region. J. Mol. Biol. 2010, 397, 1106–1118. [Google Scholar]
- Thanongsaksrikul, J.; Srimanote, P.; Maneewatch, S.; Choowongkomon, K.; Tapchaisri, P.; Makino, S.; Kurazono, H.; Chaicumpa, W. A V H H that neutralizes the zinc metalloproteinase activity of botulinum neurotoxin type A. J. Biol. Chem. 2010, 285, 9657–9666. [Google Scholar]
- Surendranath, K.; Karande, A.A. A neutralizing antibody to the A chain of abrin inhibits Abrin toxicity both in vitro and in vivo. Clin. Vaccine Immunol. 2008, 15, 737–743. [Google Scholar] [CrossRef] [PubMed]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goldman, E.R.; Anderson, G.P.; Zabetakis, D.; Walper, S.; Liu, J.L.; Bernstein, R.; Calm, A.; Carney, J.P.; O’Brien, T.W.; Walker, J.L.; et al. Llama-Derived Single Domain Antibodies Specific for Abrus Agglutinin. Toxins 2011, 3, 1405-1419. https://doi.org/10.3390/toxins3111405
Goldman ER, Anderson GP, Zabetakis D, Walper S, Liu JL, Bernstein R, Calm A, Carney JP, O’Brien TW, Walker JL, et al. Llama-Derived Single Domain Antibodies Specific for Abrus Agglutinin. Toxins. 2011; 3(11):1405-1419. https://doi.org/10.3390/toxins3111405
Chicago/Turabian StyleGoldman, Ellen R., George P. Anderson, Dan Zabetakis, Scott Walper, Jinny L. Liu, Rachael Bernstein, Alena Calm, James P. Carney, Thomas W. O’Brien, Jennifer L. Walker, and et al. 2011. "Llama-Derived Single Domain Antibodies Specific for Abrus Agglutinin" Toxins 3, no. 11: 1405-1419. https://doi.org/10.3390/toxins3111405
APA StyleGoldman, E. R., Anderson, G. P., Zabetakis, D., Walper, S., Liu, J. L., Bernstein, R., Calm, A., Carney, J. P., O’Brien, T. W., Walker, J. L., & Garber, E. A. E. (2011). Llama-Derived Single Domain Antibodies Specific for Abrus Agglutinin. Toxins, 3(11), 1405-1419. https://doi.org/10.3390/toxins3111405





