Food Poisoning and Staphylococcus aureus Enterotoxins
Abstract
:1. Staphylococcal Food Poisoning
2. Staphylococcus aureus Enterotoxins
| Toxin | Molecular Mass (kDa) | Emetic Activity | Crystal Structure Solved | Gene | Accessory genetic element |
|---|---|---|---|---|---|
| SEA | 27.1 | yes | yes | sea | ΦSa3ms, ΦSa3mw, Φ252B, ΦNM3, ΦMu50a |
| SEB | 28.4 | yes | yes | seb | pZA10, SaPI3 |
| SEC | 27.5–27.6 | yes | yes | sec | SaPIn1, SaPIm1, SaPImw2, SaPIbov1 |
| SED | 26.9 | yes | yes | sed | pIB485-like |
| SEE | 26.4 | yes | no | see | ΦSa b |
| SEG | 27.0 | yes | yes | seg | egc1 (vSaβ I); egc2 (vSaβ III); egc3; egc4 |
| SEH | 25.1 | yes | yes | seh | MGEmw2/mssa476 seh/∆seo |
| SEI | 24.9 | weak | yes | sei | egc1 (vSaβ I); egc2 (vSaβ III) ); egc3 |
| SElJ | 28.5 | nd | no | selj | pIB485-like; pF5 |
| SElK | 26.0 | nd | yes | selk | ΦSa3ms, ΦSa3mw, SaPI1, SaPI3, SaPIbov1, SaPI5 |
| SElL | 26.0 | no a | no | sell | SaPIn1, SaPIm1, SaPImw2, SaPIbov1 |
| SElM | 24.8 | nd | no | selm | egc1 (vSaβ I); egc2 (vSaβ III) |
| SElN | 26.1 | nd | no | seln | egc1 (vSaβ I); egc2 (vSaβ III); egc3; egc4 |
| SElO | 26.7 | nd | no | selo | egc1 (vSaβ I); egc2 (vSaβ III); egc3; egc4; MGEmw2/mssa476 seh/∆seo |
| SElP | 27.0 | nd a | no | selp | ΦN315, ΦMu3A |
| SElQ | 25.0 | no | no | selq | ΦSa3ms, ΦSa3mw, SaPI1, SaPI3, SaPI5 |
| SER | 27.0 | yes | no | ser | pIB485-like; pF5 |
| SES | 26.2 | yes | no | ses | pF5 |
| SET | 22.6 | weak | no | set | pF5 |
| SElU | 27.1 | nd | no | selu | egc2 (vSaβ III); egc3 |
| SElU2 (SEW) | nd | nd | no | selu2 | egc4 |
| SElV | nd | nd | no | selv | egc4 |
2.1. Nomenclature
2.2. Structure
| Group | SEs and SEls |
|---|---|
| Group 1 | SEA, SED, SEE, (SEH), SElJ, SElN, SElO, SElP, SES |
| Group 2 | SEB, SEC, SEG, SER, SElU, SElU2 |
| Group 3 | SEI, SElK, SElL, SElM, SElQ, SElV |
| Group 4 | SET |
| (Group 5) | (SEH) |
2.3. Mode of Action
2.4. Enterotoxin Gene Location
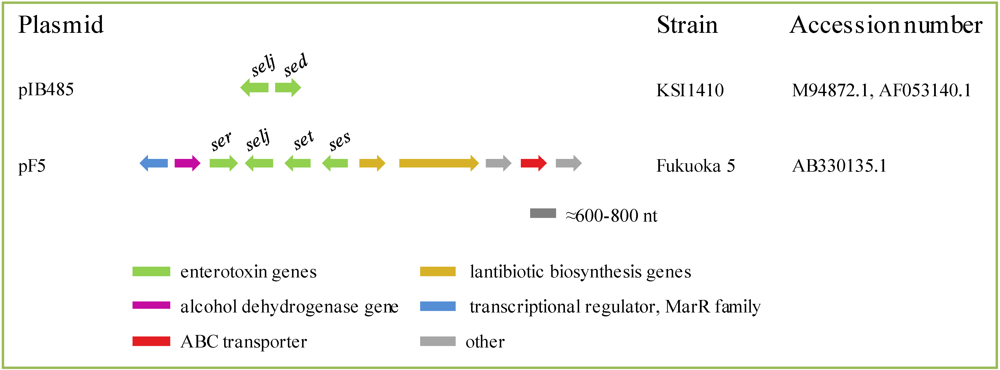
2.4.1. Plasmids
2.4.2. Prophages
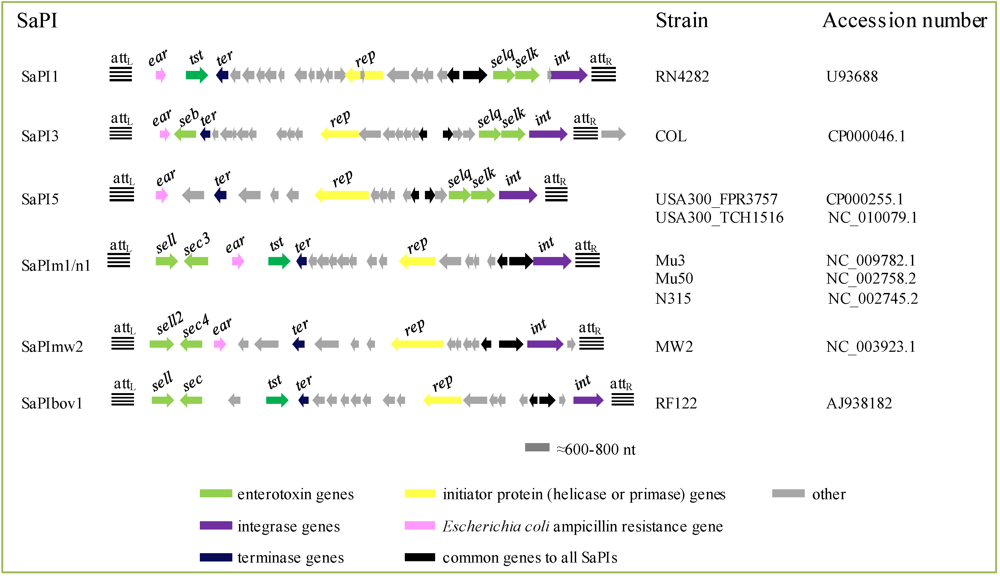
2.4.3. Staphylococcus aureus Pathogenicity Islands

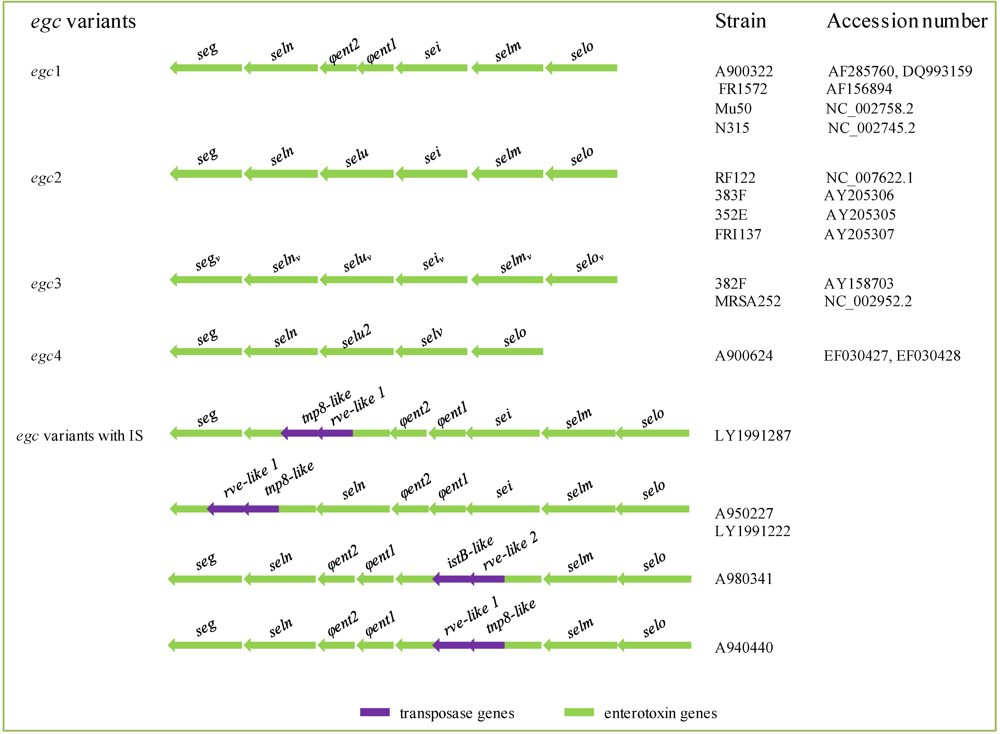
2.4.4. vSa Genomic Islands

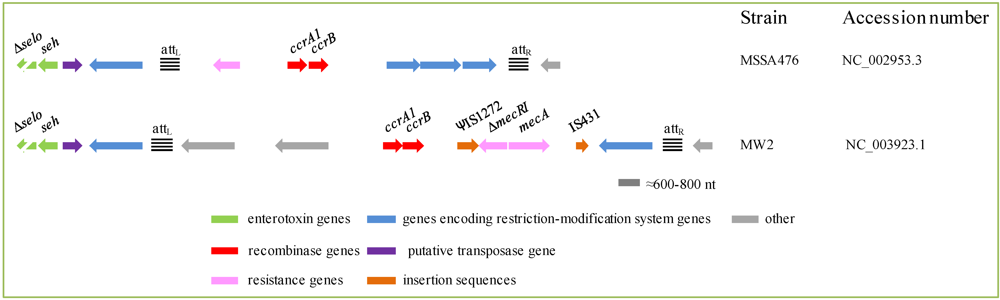
2.4.5. Enterotoxin Genes in the Proximity of the Staphylococcal Cassette Chromosome
2.5. Staphylococcal Enterotoxins and Food Poisoning Outbreaks
3. Conclusions
Acknowledgements
References
- Dinges, M.M.; Orwin, P.M.; Schlievert, P.M. Exotoxins of Staphylococcus aureus. Clin. Microbiol. Rev. 2000, 13, 16–34. [Google Scholar]
- Le Loir, Y.; Baron, F.; Gautier, M. Staphylococcus aureus and food poisoning. Genet. Mol. Res. 2003, 2, 63–76. [Google Scholar]
- Balaban, N.; Rasooly, A. Staphylococcal enterotoxins. Int. J. Food Microbiol. 2000, 61, 1–10. [Google Scholar]
- Murray, R.J. Recognition and management of Staphylococcus aureus toxin-mediated disease. Intern. Med. J. 2005, 2, S106–S119. [Google Scholar]
- Tranter, H.S. Foodborne staphylococcal illness. Lancet 1990, 336, 1044–1046. [Google Scholar]
- Kluytmans, J.A.J.W.; Wertheim, H.F.L. Nasal carriage of Staphylococcus aureus and prevention of nosocomial infections. Infection 2005, 33, 3–8. [Google Scholar]
- Stewart, G.C. Staphylococcus aureus. In Foodborne pathogens: Microbiology and Molecular Biology; Fratamico, P.M., Bhunia, A.K., Smith, J.L., Eds.; Caister Academic Press: Norfolk, UK, 2005; pp. 273–284. [Google Scholar]
- Tamarapu, S.; McKillip, J.L.; Drake, M. Development of a multiplex polymerase chain reaction assay for detection and differentiation of Staphylococcus aureus in dairy products. J. Food Prot. 2001, 64, 664–668. [Google Scholar]
- Wieneke, A.A.; Roberts, D.; Gilbert, R.J. Staphylococcal food poisoning in the United Kingdom, 1969–1990. Epidemiol. Infect. 1993, 110, 519–531. [Google Scholar]
- Qi, Y.; Miller, K.J. Effect of low water activity on staphylococcal enterotoxin A and B biosynthesis. J. Food Prot. 2000, 63, 473–478. [Google Scholar]
- Scott, W.J. Water relations of Staphylococcus aureus at 30 degrees C. Aust. J. Biol. Sci. 1953, 6, 549–564. [Google Scholar]
- Anonymous. The community summary report on trends and sources of zoonoses, zoonotic agents, antimicrobial resistance and foodborne outbreaks in the European Union in 2006. EFSA J. 2007, 130, 1–310. [Google Scholar]
- Chiang, Y.C.; Liao, W.W.; Fan, C.M.; Pai, W.Y.; Chiou, C.S.; Tsen, H.Y. PCR detection of staphylococcal enterotoxins (SEs) N, O, P, Q, R, U, and survey of SE types in Staphylococcus aureus isolates from food-poisoning cases in Taiwan. Int. J. Food Microbiol. 2008, 121, 66–73. [Google Scholar]
- Delmas, G.; Le Querrec, F.; Weill, F.-X.; Gallay, A.; Espié, E.; Haeghebaert, S.; Vaillant, V. Les toxi-infections alimentaires. In Surveillance nationale des maladies infectieuses 2001–2003; Institut de Veille Sanitaire: Saint-Maurice, France, 2005; pp. 1–10. [Google Scholar]
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-related illness and death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar]
- Betley, M.J.; Borst, D.W.; Regassa, L.B. Staphylococcal enterotoxins, toxic shock syndrome toxin and streptococcal pyrogenic exotoxins: a comparative study of their molecular biology. Chem. Immunol. 1992, 55, 1–35. [Google Scholar]
- Bergdoll, M.S. Staphylococcal intoxications. In Foodborne Infections and Intoxications; Reimann, H., Bryan, F.L., Eds.; Academic Press Inc: New York, NY, USA, 1979; pp. 443–494. [Google Scholar]
- Czop, J.K.; Bergdoll, M.S. Staphylococcal enterotoxin synthesis during the exponential, transitional, and stationary growth phases. Infect. Inmun. 1974, 9, 229–235. [Google Scholar]
- Derzelle, S.; Dilasser, F.; Duquenne, M.; Deperrois, V. Differential temporal expression of the staphylococcal enterotoxins genes during cell growth. Food Microbiol. 2009, 26, 896–904. [Google Scholar]
- Otero, A.; García, M.L.; García, M.C.; Moreno, B.; Bergdoll, M.S. Production of staphylococcal enterotoxins C1 and C2 and thermonuclease throughout the growth cycle. Appl. Environ. Microbiol. 1990, 56, 555–559. [Google Scholar]
- Larkin, E.A.; Carman, R.J.; Krakauer, T.; Stiles, B.G. Staphylococcus aureus: the toxic presence of a pathogen extraordinaire. Curr. Med. Chem. 2009, 16, 4003–4019. [Google Scholar]
- Bergdoll, M.S. Enterotoxins. In Staphylococci and Staphylococcal Infections; Easman, C.S.F., Adlam, C., Eds.; Academic Press Inc: London, UK, 1983; Volume 2, pp. 559–598. [Google Scholar]
- Evenson, M.L.; Hinds, M.W.; Bernstein, R.S.; Bergdoll, M.S. Estimation of human dose of staphylococcal enterotoxin A from a large outbreak of staphylococcal food poisoning involving chocolate milk. Int. J. Food. Microbiol. 1988, 7, 311–316. [Google Scholar]
- Schantz, E.J.; Roessler, W.G.; Wagman, J.; Spero, L.; Dunnery, D.A.; Bergdoll, M.S. Purification of staphylococcal enterotoxin B. Biochemistry 1965, 4, 1011–1016. [Google Scholar]
- Fleischer, B.; Mittrücker, H.W.; Metzroth, B.; Braun, M.; Hartwig, U. Mitogenic toxins as MHC class II-dependent probes for T cell antigen receptors. Behring Inst. Mitt. 1991, 88, 170–176. [Google Scholar]
- Marrack, P.; Kappler, J. The staphylococcal enterotoxins and their relatives. Science 1990, 248, 1066–1068. [Google Scholar]
- Petersson, K.; Pettersson, H.; Skartved, N.J.; Walse, B.; Forsberg, G. Staphylococcal enterotoxin H induces V alpha-specific expansion of T cells. J. Immunol. 2003, 170, 4148–4154. [Google Scholar]
- Thomas, D.Y.; Jarraud, S.; Lemercier, B.; Cozon, G.; Echasserieau, K.; Etienne, J.; Gougeon, M.L.; Lina, G.; Vandenesch, F. Staphylocccal enterotoxin-like toxins U2 and V, two new staphylococcal superantigens arising from recombination within the enterotoxin gene cluster. Infect. Immun. 2006, 74, 4724–4734. [Google Scholar]
- Thomas, D.; Chou, S.; Dauwalder, O.; Lina, G. Diversity in Staphylococcus aureus enterotoxins. Chem. Immunol. Allergy 2007, 93, 24–41. [Google Scholar]
- Lina, G.; Bohach, G.A.; Nair, S.P.; Hiramatsu, K.; Jouvin-Marche, E.; Mariuzza, R. Standard nomenclature for the superantigens expressed by Staphylococcus. J. Infect. Dis. 2004, 189, 2334–2336. [Google Scholar]
- Bergdoll, M.S.; Surgalla, M.J.; Dack, G.M. Staphylococcal enterotoxin: Identification of a specific precipitating antibody with enterotoxin neutralizing property. J. Immunol. 1959, 83, 334–338. [Google Scholar]
- Bergdoll, M.S.; Borja, C.R.; Avena, R.M. Identification of a new enterotoxin as enterotoxin C. J. Bacteriol. 1965, 90, 1481–1485. [Google Scholar]
- Bergdoll, M.S.; Borja, C.R.; Robbins, R.N.; Weiss, K.F. Identification of enterotoxin E. Infect. Immun. 1971, 4, 593–595. [Google Scholar]
- Casman, E.P. Further serological studies of staphylococcal enterotoxin. J. Bacteriol. 1960, 79, 849–856. [Google Scholar]
- Casman, E.P.; Bennett, R.W.; Dorsey, A.E.; Issa, J.A. Identification of a fourth staphylococcal enterotoxin, enterotoxin D. J. Bacteriol. 1967, 94, 1875–1882. [Google Scholar]
- Jarraud, S.; Peyrat, M.A.; Lim, A.; Tristan, A.; Bes, M.; Mougel, C.; Etienne, J.; Vandenesch, F.; Bonneville, M.; Lina, G. egc, a highly prevalent operon of enterotoxin gene, forms a putative nursery of superantigens in Staphylococcus aureus. J. Immunol. 2001, 166, 669–677. [Google Scholar]
- Letertre, C.; Perelle, S.; Dilasser, F.; Fach, P. Identification of a new putative enterotoxin SEU encoded by the egc cluster of Staphylococcus aureus. J. Appl. Microbiol. 2003, 95, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Munson, S.H.; Tremaine, M.T.; Betley, M.J.; Welch, R.A. Identification and characterization of staphylococcal enterotoxin types G and I from Staphylococcus aureus. Infect Immun. 1998, 66, 3337–3348. [Google Scholar]
- Omoe, K.; Imanishi, K.; Hu, D.L.; Kato, H.; Fugane, Y.; Abe, Y.; Hamaoka, S.; Watanabe, Y.; Nakane, A.; Uchiyama, T.; Shinagawa, K. Characterization of novel staphylococcal enterotoxin-like toxin type P. Infect. Immun. 2005, 73, 5540–5546. [Google Scholar]
- Ono, H.K.; Omoe, K.; Imanishi, K.; Iwakabe, Y.; Hu, D.L.; Kato, H.; Saito, N.; Nakane, A.; Uchiyama, T.; Shinagawa, K. Identification and characterization of two novel staphylococcal enterotoxins, types S and T. Infect. Immun. 2008, 76, 4999–5005. [Google Scholar]
- Orwin, P.M.; Leung, D.Y.; Donahue, H.L.; Novick, R.P.; Schlievert, P.M. Biochemical and biological properties of staphylococcal enterotoxin K. Infect. Immun. 2001, 69, 360–366. [Google Scholar]
- Orwin, P.M.; Leung, D.Y.; Tripp, T.J.; Bohach, G.A.; Earhart, C.A.; Ohlendorf, D.H.; Schlievert, P.M. Characterization of a novel staphylococcal enterotoxin-like superantigen, a member of the group V subfamily of pyrogenic toxins. Biochemistry 2002, 41, 14033–14040. [Google Scholar]
- Orwin, P.M.; Fitzgerald, J.R.; Leung, D.Y.; Gutierrez, J.A.; Bohach, G.A.; Schlievert, P.M. Characterization of Staphylococcus aureus enterotoxin L. Infect. Immun. 2003, 71, 2916–2919. [Google Scholar]
- Su, Y.C.; Wong, A.C. Identification and purification of a new staphylococcal enterotoxin, H. Appl. Environ. Microbiol. 1995, 61, 1438–1443. [Google Scholar]
- Zhang, S.; Iandolo, J.J.; Stewart, G.C. The enterotoxin D plasmid of Staphylococcus aureus encodes a second enterotoxin determinant (sej). FEMS Microbiol. Lett. 1998, 168, 227–233. [Google Scholar]
- Bergdoll, M.S.; Crass, B.A.; Reiser, R.F.; Robbins, R.N.; Davis, J.P. A new staphylococcal enterotoxin, enterotoxin F, associated with toxic-shock-syndrome Staphylococcus aureus isolates. Lancet 1981, 5, 1017–1021. [Google Scholar]
- Reiser, R.F.; Robbins, R.N.; Khoe, G.P.; Bergdoll, M.S. Purification and some physicochemical properties of toxic-shock toxin. Biochemistry 1983, 22, 3907–3912. [Google Scholar]
- Couch, J.L.; Soltis, M.T.; Betley, M.J. Cloning and nucleotide sequence of the type E staphylococcal enterotoxin gene. J. Bacteriol. 1988, 170, 2954–2960. [Google Scholar]
- Uchiyama, T.; Imanishi, K.; Miyoshi-Akiyama, T.; Kato, H. Staphylococcal superantigens and the diseases they cause. In The Comprehensive Sourcebook of Bacterial Protein Toxins, 3rd; Alouf, J.E., Popoff, M.R., Eds.; Academic Press: Burlington, VT, USA, 2006; pp. 830–843. [Google Scholar]
- Fraser, J.D.; Proft, T. The bacterial superantigen and superantigen-like proteins. Immunol. Rev. 2008, 225, 226–243. [Google Scholar]
- Baker, H.M.; Basu, I.; Chung, M.C.; Caradoc-Davies, T.; Fraser, J.D.; Baker, E.N. Crystal structures of the staphylococcal toxin SSL5 in complex with sialyl Lewis X reveal a conserved binding site that shares common features with viral and bacterial sialic acid binding proteins. J. Mol. Biol. 2007, 374, 1298–1308. [Google Scholar]
- Chung, M.C.; Wines, B.D.; Baker, H.; Langley, R.J.; Baker, E.N.; Fraser, J.D. The crystal structure of staphylococcal superantigen-like protein 11 in complex with sialyl Lewis X reveals the mechanism for cell binding and immune inhibition. Mol. Microbiol. 2007, 66, 1342–1355. [Google Scholar]
- Langley, R.; Wines, B.; Willoughby, N.; Basu, I.; Proft, T.; Fraser, J.D. The staphylococcal superantigen-like protein 7 binds IgA and complement C5 and inhibits IgA-Fc alpha RI binding and serum killing of bacteria. J. Immunol. 2005, 174, 2926–2933. [Google Scholar]
- Ramsland, P.A.; Willoughby, N.; Trist, H.M.; Farrugia, W.; Hogarth, P.M.; Fraser, J.D.; Wines, B.D. Structural basis for evasion of IgA immunity by Staphylococcus aureus revealed in the complex of SSL7 with Fc of human IgA1. Proc. Natl. Acad. Sci. USA 2007, 104, 15051–15056. [Google Scholar]
- Williams, R.J.; Ward, J.M.; Henderson, B.; Poole, S.; O'Hara, B.P.; Wilson, M.; Nair, S.P. Identification of a novel gene cluster encoding staphylococcal exotoxin-like proteins: Characterization of the prototypic gene and its protein product, SET1. Infect. Immun. 2000, 68, 4407–4415. [Google Scholar]
- Wines, B.D.; Willoughby, N.; Fraser, J.D.; Hogarth, P.M. A competitive mechanism for staphylococcal toxin SSL7 inhibiting the leukocyte IgA receptor, Fc alphaRI, is revealed by SSL7 binding at the C alpha2/C alpha3 interface of IgA. J. Biol. Chem. 2006, 281, 1389–1393. [Google Scholar]
- Acharya, K.R.; Passalacquam, E.F.; Jones, E.Y.; Harlos, K.; Stuart, D.I.; Brehm, R.D.; Tranter, H.S. Structural basis of superantigen action inferred from crystal structure of toxic-shock syndrome toxin-1. Nature 1994, 367, 94–97. [Google Scholar]
- Prasad, G.S.; Earhart, C.A.; Murray, D.L.; Novick, R.P.; Schlievert, P.M.; Ohlendorf, D.H. Structure of toxic shock syndrome toxin 1. Biochemistry 1993, 32, 13761–13766. [Google Scholar]
- Fernández, M.M.; Bhattacharya, S.; De Marzi, M.C.; Brown, P.H.; Kerzic, M.; Schuck, P.; Mariuzza, R.A.; Malchiodi, E.L. Superantigen natural affinity maturation revealed by the crystal structure of staphylococcal enterotoxin G and its binding to T-cell receptor Vbeta8.2. Proteins 2007, 68, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Günther, S.; Varma, A.K.; Moza, B.; Kasper, K.J.; Wyatt, A.W.; Zhu, P.; Rahman, A.K.; Li, Y.; Mariuzza, R.A.; McCormick, J.K.; Sundberg, E.J. A novel loop domain in superantigens extends their T cell receptor recognition site. J. Mol. Biol. 2007, 371, 210–221. [Google Scholar]
- Håkansson, M.; Petersson, K.; Nilsson, H.; Forsberg, G.; Björk, P.; Antonsson, P.; Svensson, L.A. The crystal structure of staphylococcal enterotoxin H: implication for binding properties to MHC class II and TcR molecules. J. Mol. Biol. 2000, 302, 527–537. [Google Scholar]
- Papageorgiou, A.C.; Acharya, K.R.; Shapiro, R.; Passalacqua, E.F.; Brehm, R.D.; Tranter, H.S. Crystal structure of the superantigen enterotoxin C2 from Staphylococcus aureus reveals a zinc-binding site. Structure 1995, 3, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, A.C.; Tranter, H.S.; Acharya, K.R. Crystal structure of microbial superantigen staphylococcal enterotoxin B at 1.5 A resolution: Implications for superantigen recognition by MHC class II molecules and T-cell receptors. J. Mol. Biol. 1998, 277, 61–79. [Google Scholar] [CrossRef] [PubMed]
- Schad, E.M.; Zaitseva, I.; Zaitsev, V.N.; Dohlsten, M.; Kalland, T.; Schlievert, P.M.; Ohlendorf, D.H.; Svensson, L.A. Crystal structure of the superantigen staphylococcal enterotoxin type A. EMBO J. 1995, 14, 3292–3301. [Google Scholar]
- Singh, B.R.; Fu, F.N.; Ledoux, D.N. Crystal and solution structures of superantigenic staphylococcal enterotoxins compared. Nat. Struct. Biol. 1994, 1, 358–360. [Google Scholar]
- Sundström, M.; Hallén, D.; Svensson, A.; Schad, E.; Dohlsten, M.; Abrahmsén, L. The Co-crystal structure of staphylococcal enterotoxin type A with Zn2+ at 2.7 A resolution. Implications for major histocompatibility complex class II binding. J. Biol. Chem. 1996, 271, 32212–32216. [Google Scholar] [PubMed]
- Sundström, M.; Abrahmsén, L.; Antonsson, P.; Mehindate, K.; Mourad, W.; Dohlsten, M. The crystal structure of staphylococcal enterotoxin D type D reveals Zn2+ mediated homodimersation. EMBO J. 1996, 15, 6832–6840. [Google Scholar]
- Swaminathan, S.; Furey, W.; Pletcher, J.; Sax, M. Crystal structure of staphylococcal enterotoxin B, a superantigen. Nature 1992, 359, 801–806. [Google Scholar]
- Swaminathan, S.; Furey, W.; Pletcher, J.; Sax, M. Residues defining V beta specificity in staphylococcal enterotoxins. Nat. Struct. Biol. 1995, 8, 680–686. [Google Scholar]
- McCormick, J.K.; Yarwood, J.M.; Schlievert, P.M. Toxic shock syndrome and bacterial superantigens: An update. Annu. Rev. Microbiol. 2001, 55, 77–104. [Google Scholar]
- Mitchell, D.T.; Levitt, D.G.; Schlievert, P.M.; Ohlendorf, D.H. Structural evidence for the evolution of pyrogenic toxin superantigens. J. Mol. Evol. 2000, 51, 520–531. [Google Scholar]
- Murzin, A.G. OB(oligonucleotide/oligosaccharide binding)-fold: Common structural and functional solution for non-homologous sequences. EMBO J. 1993, 12, 861–867. [Google Scholar]
- Hovde, C.J.; Marr, J.C.; Hoffmann, M.L.; Hackett, S.P.; Chi, Y.I.; Crum, K.K.; Stevens, D.L.; Stauffacher, C.V.; Bohach, G.A. Investigation of the role of the disulphide bond in the activity and structure of staphylococcal enterotoxin C1. Mol. Microbiol. 1994, 13, 897–909. [Google Scholar]
- Wang, X.; Xu, M.; Cai, Y.; Yang, H.; Zhang, H.; Zhang, C. Functional analysis of the disulphide loop mutant of staphylococcal enterotoxin C2. Appl. Microbiol. Biotechnol. 2009, 82, 861–871. [Google Scholar]
- Alber, G.; Hammer, D.K.; Fleischer, B. Relationship between enterotoxic- and T lymphocyte-stimulating activity of staphylococcal enterotoxin B. J. Immunol. 1990, 144, 4501–4506. [Google Scholar]
- Stelma, G.N.; Bergdoll, M.S. Inactivation of staphylococcal enterotoxin A by chemical modification. Biochem. Biophys. Res. Commun. 1982, 105, 121–126. [Google Scholar]
- Hoffman, M.; Tremaine, M.; Mansfield, J.; Betley, M. Biochemical and mutational analysis of the histidine residues of staphylococcal enterotoxin A. Infect. Immun. 1996, 64, 885–890. [Google Scholar]
- Harris, T.O.; Grossman, D.; Kappler, J.W.; Marrack, P.; Rich, R.R.; Betley, M.J. Lack of complete correlation between emetic and T-cell-stimulatory activities of staphylococcal enterotoxins. Infect. Immunol. 1993, 61, 3175–3183. [Google Scholar]
- Sugiyama, H.; Hayama, T. Abdominal viscera as site of emetic action for staphylococcal enterotoxin in monkey. J. Infect. Dis. 1965, 115, 330–336. [Google Scholar]
- Hu, D.L.; Zhu, G.; Mori, F.; Omoe, K.; Okada, M.; Wakabayashi, K.; Kaneko, S.; Shinagawa, K.; Nakane, A. Staphylococcal enterotoxin induces emesis through increasing serotonin release in intestine and it is downregulated by cannabinoid receptor 1. Cell. Microbiol. 2007, 9, 2267–2277. [Google Scholar]
- Shupp, J.W.; Jett, M.; Pontzer, C.H. Identification of a transcytosis epitope on staphylococcal enterotoxins. Infect. Immun. 2002, 70, 2178–2186. [Google Scholar]
- Alber, G.; Scheuber, P.H.; Reck, B.; Sailer-Kramer, B.; Hartmann, A.; Hammer, D.K. Role of substance P in immediate-type skin reactions induced by staphylococcal enterotoxin B in unsensitized monkeys. J. Allergy Clin. Immunol. 1989, 6, 880–885. [Google Scholar]
- Jett, M.; Brinkley, W.; Neill, R.; Gemski, P.; Hunt, R. Staphylococcus aureus enterotoxin B challenge of monkeys: Correlation of plasma levels of arachidonic acid cascade products with occurrence of illness. Infect. Immun. 1990, 58, 3494–3499. [Google Scholar]
- Scheuber, P.H.; Denzlinger, C.; Wilker, D.; Beck, G.; Keppler, D.; Hammer, D.K. Cysteinyl leukotrienes as mediators of staphylococcal enterotoxin B in the monkey. Eur. J. Clin. Invest. 1987, 17, 455–459. [Google Scholar]
- Shanahan, F.; Denburg, J.A.; Fox, J.; Bienenstock, J.; Befus, D. Mast cell heterogeneity: Effects of neuroenteric peptides on histamine release. J. Inmunol. 1985, 135, 1331–13337. [Google Scholar]
- Scheuber, P.H.; Golecki, J.R.; Kickhöfen, B.; Scheel, D.; Beck, G.; Hammer, D.K. Skin reactivity of unsensitized monkeys upon challenge with staphylococcal enterotoxin B: A new approach for investigating the site of toxin action. Infect. Immun. 1985, 50, 869–876. [Google Scholar]
- Holmberg, S.D.; Blake, P.A. Staphylococcal food poisoning in the United States: New facts and old misconceptions. JAMA 1984, 251, 487–489. [Google Scholar]
- Palmer, E.D. The morphologic consequences of acute exogenous (staphylococcic) gastroenteritis of the gastric mucosa. Gastroenterology 1951, 19, 462–475. [Google Scholar]
- Kent, T.H. Staphylococcal enterotoxin gastroenteritis in rhesus monkeys. Am. J. Pathol. 1966, 48, 387–407. [Google Scholar]
- Sheehan, D.G.; Jervis, H.R.; Takeuchi, A.; Sprinz, H. The effect of staphylococcal enterotoxin on the epithelial mucosubstance of the small intestine of rhesus monkeys. Am. J. Pathol. 1970, 60, 1–18. [Google Scholar]
- Sullivan, R. Effects of enterotoxin B on intestinal transport in vitro. Proc. Soc. Exp. Biol. Med. 1969, 131, 1159–1162. [Google Scholar]
- Hamad, A.R.; Marrack, P.; Kappler, J.W. Transcytosis of staphylococcal superantigen toxins. J. Exp. Med. 1997, 185, 1447–1454. [Google Scholar]
- Omoe, K.; Hu, D.L.; Takahashi-Omoe, H.; Nakane, A.; Shinagawa, K. Identification and characterization of a new staphylococcal enterotoxin-related putative toxin encoded by two kinds of plasmids. Infect. Immun. 2003, 71, 6088–6094. [Google Scholar]
- Bayles, K.W.; Iandolo, J.J. Genetic and molecular analyses of the gene encoding staphylococcal enterotoxin D. J. Bacteriol. 1989, 171, 4799–4806. [Google Scholar]
- Fueyo, J.M.; Mendoza, M.C.; Martín, M.C. Enterotoxins and toxic shock syndrome toxin in Staphylococcus aureus recovered from human nasal carriers and manually handled foods: Epidemiological and genetic findings. Microbes Infect. 2005, 7, 187–194. [Google Scholar]
- Goerke, C.; Pantucek, R.; Silva Holtfreter, S.; Berit Schulte, B.; Manuel Zink, M.; Grumann, D.; Bröker, B.M.; Doskar, J.; Wolz, C. Diversity of prophages in dominant Staphylococcus aureus clonal lineages. J. Bacteriol. 2009, 191, 3462–3468. [Google Scholar]
- de Haas, C.J.; Veldkamp, K.E.; Peschel, A.; Weerkamp, F.; Van Wamel, W.J.; Heezius, E.C.; Poppelier, M.J.; Van Kessel, K.P.; van Strijp, J.A. Chemotaxis inhibitory protein of Staphylococcus aureus, a bacterial antiinflammatory agent. J. Exp. Med. 2004, 199, 687–695. [Google Scholar]
- Rooijakkers, S.H.; Ruyken, M.; Roos, A.; Daha, M.R.; Presanis, J.S.; Sim, R.B.; van Wamel, W.J.; van Kessel, K.P.; van Strijp, J.A. Immune evasion by a staphylococcal complement inhibitor that acts on C3 convertases. Nat. Immunol. 2005, 6, 920–927. [Google Scholar]
- Ji, Y.; Yin, D.; Fox, B.; Holmes, D.J.; Payne, D.; Rosenberg, M. Validation of antibacterial mechanism of action using regulated antisense RNA expression in Staphylococcus aureus. FEMS Microbiol. Lett. 2004, 231, 177–184. [Google Scholar]
- Rooijakkers, S.H.; van Wamel, W.J.; Ruyken, M.; van Kessel, K.P.; van Strijp, J.A. Anti-opsonic properties of staphylokinase. Microbes Infect. 2005, 7, 476–484. [Google Scholar]
- van Wamel, W.J.; Rooijakkers, S.H.; Ruyken, M.; van Kessel, K.P.; van Strijp, J.A. The innate immune modulators staphylococcal complement inhibitor and chemotaxis inhibitory protein of Staphylococcus aureus are located on beta-hemolysin-converting bacteriophages. J. Bacteriol. 2006, 188, 1310–1315. [Google Scholar]
- Coleman, D.C.; Sullivan, D.J.; Russell, R.J.; Arbuthnott, J.P.; Carey, B.F.; Pomeroy, H.M. Staphylococcus aureus bacteriophages mediating the simultaneous lysogenic conversion of beta-lysin, staphylokinase and enterotoxin A: Molecular mechanism of triple conversion. J. Gen. Microbiol. 1989, 135, 1679–1697. [Google Scholar]
- Lindsay, J.A.; Ruzin, A.; Ross, H.F.; Kurepina, N.; Novick, R.P. The gene for toxic shock toxin is carried by a family of mobile pathogenicity islands in Staphylococcus aureus. Mol. Microbiol. 1998, 29, 527–543. [Google Scholar]
- Tallent, S.M.; Langston, T.B.; Moran, R.G.; Christie, G.E. Transducing particles of Staphylococcus aureus pathogenicity island SaPI1 are comprised of helper phage-encoded proteins. J. Bacteriol. 2007, 189, 7520–7524. [Google Scholar]
- Novick, R.P.; Subedi, A. The SaPIs: mobile pathogenicity islands of Staphylococcus. Chem. Immunol. Allergy 2007, 93, 42–57. [Google Scholar]
- Sumby, P.; Waldor, M.K. Transcription of the toxin genes present within the Staphylococcal phage phiSa3ms is intimately linked with the phage's life cycle. J. Bacteriol. 2003, 185, 6841–6851. [Google Scholar]
- Baba, T.; Takeuchi, F.; Kuroda, M.; Yuzawa, H.; Aoki, K.; Oguchi, A.; Nagai, Y.; Iwama, N.; Asano, K.; Naimi, T.; Kuroda, H.; Cui, L.; Yamamoto, K.; Hiramatsu, K. Genome and virulence determinants of high virulence community-acquired MRSA. Lancet 2002, 359, 1819–1827. [Google Scholar]
- Baba, T.; Bae, T.; Schneewind, O.; Takeuchi, F.; Hiramatsu, K. Genome sequence of Staphylococcus aureus strain Newman and comparative analysis of staphylococcal genomes: polymorphism and evolution of two major pathogenicity islands. J. Bacteriol. 2008, 190, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Waldron, D.E.; Lindsay, J.A. Sau1: A novel lineage-specific type I restriction-modification system that blocks horizontal gene transfer into Staphylococcus aureus and between S. aureus isolates of different lineages. J. Bacteriol. 2006, 188, 5578–5585. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, J.R.; Reid, S.D.; Ruotsalainen, E.; Tripp, T.J.; Liu, M.; Cole, R.; Kuusela, P.; Schlievert, P.M.; Järvinen, A.; Musser, J.M. Genome diversification in Staphylococcus aureus: Molecular evolution of a highly variable chromosomal region encoding the staphylococcal exotoxin-like family of proteins. Infect. Immun. 2003, 71, 2827–2838. [Google Scholar]
- Bestebroer, J.; Poppelier, M.J.; Ulfman, L.H.; Lenting, P.J.; Denis, C.V.; van Kessel, K.P.; van Strijp, J.A.; de Haas, C.J. Staphylococcal superantigen-like 5 binds PSGL-1 and inhibits P-selectin-mediated neutrophil rolling. Blood 2007, 109, 2936–2943. [Google Scholar]
- Fitzgerald, J.R.; Monday, S.R.; Foster, T.J.; Bohach, G.A.; Hartigan, P.J.; Meaney, W.J.; Smith, C.J. Characterization of putative pathogenicity island from bovine Staphylococcus aureus encoding multiple superantigens. J. Bacteriol. 2001, 183, 63–70. [Google Scholar]
- Monday, S.R.; Bohach, G.A. Genes encoding staphylococcal enterotoxins G and I are linked and separated by DNA related to other staphylococcal enterotoxins. J. Nat. Toxins 2001, 10, 1–8. [Google Scholar]
- Collery, M.M.; Smyth, D.S.; Tumilty, J.J.; Twohig, J.M.; Smyth, C.J. Associations between enterotoxin gene cluster types egc1, egc2 and egc3, agr types, enterotoxin and enterotoxin-like gene profiles, and molecular typing characteristics of human nasal carriage and animal isolates of Staphylococcus aureus. J. Med. Microbiol. 2009, 58, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Holden, M.T.; Feil, E.J.; Lindsay, J.A.; Peacock, S.J.; Day, N.P.; Enright, M.C.; Foster, T.J.; Moore, C.E.; Hurst, L.; Atkin, R.; Barron, A.; Bason, N.; Bentley, S.D.; Chillingworth, C.; Chillingworth, T.; Churcher, C.; Clark, L.; Corton, C.; Cronin, A.; Doggett, J.; Dowd, L.; Feltwell, T.; Hance, Z.; Harris, B.; Hauser, H.; Holroyd, S.; Jagels, K.; James, K.D.; Lennard, N.; Line, A.; Mayes, R.; Moule, S.; Mungall, K.; Ormond, D.; Quail, M.A.; Rabbinowitsch, E.; Rutherford, K.; Sanders, M.; Sharp, S.; Simmonds, M.; Stevens, K.; Whitehead, S.; Barrell, B.G.; Spratt, B.G.; Parkhill, J. Complete genomes of two clinical Staphylococcus aureus strains: Evidence for the rapid evolution of virulence and drug resistance. Proc. Natl. Acad. Sci. USA 2004, 101, 9786–9791. [Google Scholar]
- Collery, M.M.; Smyth, C.J. Rapid differentiation of Staphylococcus aureus isolates harbouring egc loci with pseudogenes psient1 and psient2 and the selu or seluv gene using PCR-RFLP. J. Med. Microbiol. 2007, 56, 208–216. [Google Scholar]
- Omoe, K.; Hu, D.L.; Takahashi-Omoe, H.; Nakane, A.; Shinagawa, K. Comprehensive analysis of classical and newly described staphylococcal superantigenic toxin genes in Staphylococcus aureus isolates. FEMS Microbiol. Lett. 2005, 246, 191–198. [Google Scholar]
- Noto, M.J.; Archer, G.L. A subset of Staphylococcus aureus strains harboring staphylococcal cassette chromosome mec (SCCmec) type IV is deficient in CcrAB-mediated SCCmec excision. Antimicrob. Agents Chemother. 2006, 50, 2782–2788. [Google Scholar]
- Bergdoll, M.S. Monkey feeding test for staphylococcal enterotoxin. Meth. Enzymol. 1988, 165, 324–333. [Google Scholar]
- Holmberg, S.D.; Blake, P.A. Staphylococcal food poisoning in the United States. New facts and old misconceptions. JAMA 1984, 251, 487–489. [Google Scholar] [PubMed]
- Wieneke, A.A.; Roberts, D.; Gilbert, R.J. Staphylococcal food poisoning in the United Kingdom, 1969–90. Epidemiol. Infect. 1993, 110, 519–531. [Google Scholar]
- Kérouanton, A.; Hennekinne, J.A.; Letertre, C.; Petit, L.; Chesneau, O.; Brisabois, A.; De Buyser, M.L. Characterization of Staphylococcus aureus strains associated with food poisoning outbreaks in France. Int. J. Food Microbiol. 2007, 115, 369–375. [Google Scholar]
- Schmid, D.; Fretz, R.; Winter, P.; Mann, M.; Höger, G.; Stöger, A.; Ruppitsch, W.; Ladstätter, J.; Mayer, N.; de Martin, A.; Allerberger, F. Outbreak of staphylococcal food intoxication after consumption of pasteurized milk products, June 2007, Austria. Wien. Klin. Wochenschr. 2009, 121, 125–131. [Google Scholar]
- Casman, E.P. Staphylococal enterotoxin. Ann. N.Y. Acad. Sci. 1965, 128, 124–131. [Google Scholar]
- Veras, J.F.; do Carmo, L.S.; Tong, L.C.; Shupp, J.W.; Cummings, C.; Dos Santos, D.A.; Cerqueira, M.M.; Cantini, A.; Nicoli, J.R.; Jett, M. A study of the enterotoxigenicity of coagulase-negative and coagulase-positive staphylococcal isolates from food poisoning outbreaks in Minas Gerais, Brazil. Int. J. Infect. Dis. 2008, 12, 410–415. [Google Scholar]
- Cha, J.O.; Lee, J.K.; Jung, Y.H.; Yoo, J.I.; Park, Y.K.; Kim, B.S.; Lee, Y.S. Molecular analysis of Staphylococcus aureus isolates associated with staphylococcal food poisoning in South Korea. J. Appl. Microbiol. 2006, 101, 864–871. [Google Scholar]
- Shimizu, A.; Fujita, M.; Igarashi, H.; Takagi, M.; Nagase, N.; Sasaki, A.; Kawano, J. Characterization of Staphylococcus aureus coagulase type VII isolates from staphylococcal food poisoning outbreaks (1980–1995) in Tokyo, Japan, by pulsed field gel electrophoresis. J. Clin. Microbiol. 2000, 38, 3746–3749. [Google Scholar]
- Asao, T.; Kumeda, Y.; Kawai, T.; Shibata, T.; Oda, H.; Haruki, K.; Nakazawa, H.; Kozaki, S. An extensive outbreak of staphylococcal food poisoning due to low-fat milk in Japan: estimation of enterotoxin A in the incriminated milk and powdered skim milk. Epidemiol. Infect. 2003, 130, 33–40. [Google Scholar]
- Kitamoto, M.; Kito, K.; Niimi, Y.; Shoda, S.; Takamura, A.; Hiramatsu, T.; Akashi, T.; Yokoi, Y.; Hirano, H.; Hosokawa, M.; Yamamoto, A.; Agata, N.; Hamajima, N. Food poisoning by Staphylococcus aureus at a university festival. Jpn. J. Infect. Dis. 2009, 62, 242–243. [Google Scholar]
- Jones, T.F.; Kellum, M.E.; Porter, S.S.; Bell, M.; Schaffner, W. An outbreak of community-acquired foodborne illness caused by methicillin-resistant Staphylococcus aureus. Emerg. Infect. Dis. 2002, 8, 82–84. [Google Scholar]
- McLauchlin, J.; Narayanan, G.L.; Mithani, V.; O’Neil, G. The detection of enterotoxins and toxic schock síndrome toxin genes in Staphylococcus aureus by polymerase chain reaction. J. Food Prot. 2000, 63, 479–488. [Google Scholar]
- Morris, C.A.; Conway, H.D.; Everall, P.H. Food poisoning due to staphylococcal enterotoxin E. Lancet 1972, 2, 1375–1376. [Google Scholar]
- Ostyn, A.; De Buyser, M.L.; Guillier, F.; Groult, J.; Felix, B.; Salah, S.; Delmas, G.; Hennekinne, J.A. First evidence of a food poisoning outbreak due to staphylococcal enterotoxin type E, France, 2009. Euro. Surveill. 2010, 15, pii. 19528. [Google Scholar] [PubMed]
- Su, Y.C.; Wong, A.C. Detection of staphylococcal enterotoxin H by an enzyme-linked immunosorbent assay. J. Food Prot. 1996, 59, 327–330. [Google Scholar]
- Omoe, K.; Ishikawa, M.; Shimoda, Y.; Hu, D.L.; Ueda, S.; Shinagawa, K. Detection of seg, seh, and sei genes in Staphylococcus aureus isolates and determination of the enterotoxin productivities of S. aureus isolates harboring seg, seh, or sei genes. J. Clin. Microbiol. 2002, 40, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Tamate, N.; Yamaguchi, K.; Makino, S. Mass outbreak of food poisoning disease caused by small amounts of staphylococcal enterotoxins A and H. Appl. Environ. Microbiol. 2005, 71, 2793–2795. [Google Scholar]
- Jørgensen, H.J.; Mathisen, T.; Løvseth, A.; Omoe, K.; Qvale, K.S.; Loncarevic, S. An outbreak of staphylococcal food poisoning caused by enterotoxin H in mashed potato made with raw milk. FEMS Microbiol. Lett. 2005, 252, 267–272. [Google Scholar]
- Pereira, M.L.; do Carmo, L.S.; dos Santos, E.J.; Pereira, J.L.; Bergdoll, M.S. Enterotoxin H in staphylococcal food poisoning. J. Food Prot. 1996, 59, 559–561. [Google Scholar]
- Abe, J.; Ito, Y.; Onimaru, M.; Kohsaka, T.; Takeda, T. Characterization and distribution of a new enterotoxin-related superantigen produced by Staphylococcus aureus. Microbiol. Immunol. 2000, 44, 79–88. [Google Scholar]
- Bania, J.; Dabrowska, A.; Bystron, J.; Korzekwa, K.; Chrzanowska, J.; Molenda, J. Distribution of newly described enterotoxin-like genes in Staphylococcus aureus from food. Int. J. Food Microbiol. 2006, 108, 36–41. [Google Scholar]
- Blaiotta, G.; Ercolini, D.; Pennacchia, C.; Fusco, V.; Casaburi, A.; Pepe, O.; Villani, F. PCR detection of staphylococcal enterotoxin genes in Staphylococcus spp. strains isolated from meat and dairy products. Evidence for new variants of seG and seI in S. aureus AB-8802. J. Appl. Microbiol. 2004, 97, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, H.J.; Mørk, T.; Caugant, D.A.; Kearns, A.; Rørvik, L.M. Genetic variation among Staphylococcus aureus strains from Norwegian bulk milk. Appl. Environ. Microbiol. 2005, 71, 8352–8361. [Google Scholar]
- Martin, M.C.; Fueyo, J.M.; González-Hevia, M.A.; Mendoza, M.C. Genetic procedures for identification of enterotoxigenic strains of Staphylococcus aureus from three food poisoning outbreaks. Int. J. Food Microbiol. 2004, 94, 279–286. [Google Scholar]
- Rosec, J.P.; Gigaud, O. Staphylococcal enterotoxin genes of classical and new types detected by PCR in France. Int. J. Food Microbiol. 2002, 77, 61–70. [Google Scholar]
- Hwang, S.Y.; Kim, S.H.; Jang, E.J.; Kwon, N.H.; Park, Y.K.; Koo, H.C.; Jung, W.K.; Kim, J.M.; Park, Y.H. Novel multiplex PCR for the detection of the Staphylococcus aureus superantigen and its application to raw meat isolates in Korea. Int. J. Food Microbiol. 2007, 117, 99–105. [Google Scholar]
- Naik, S.; Smith, F.; Ho, J.; Croft, N.M.; Domizio, P.; Price, E.; Sanderson, I.R.; Meadows, N.J. Staphylococcal enterotoxins G and I, a cause of severe but reversible neonatal enteropathy. Clin. Gastroenterol. Hepatol. 2008, 6, 251–254. [Google Scholar]
- Argudín, M.A.; Mendoza, M.C.; Méndez, F.J.; Martín, M.C.; Guerra, B.; Rodicio, M.R. Clonal complexes and diversity of exotoxin gene profiles in methicillin-resistant and methicillin-susceptible Staphylococcus aureus isolates from patients in a Spanish hospital. J. Clin. Microbiol. 2009, 47, 2097–2105. [Google Scholar]
- Fueyo, J.M.; Mendoza, M.C.; Alvarez, M.A.; Martín, M.C. Relationships between toxin gene content and genetic background in nasal carried isolates of Staphylococcus aureus from Asturias, Spain. FEMS Microbiol. Lett. 2005, 243, 447–454. [Google Scholar]
- Fueyo, J.M.; Mendoza, M.C.; Rodicio, M.R.; Muñiz, J.; Alvarez, M.A.; Martín, M.C. Cytotoxin and pyrogenic toxin superantigen gene profiles of Staphylococcus aureus associated with subclinical mastitis in dairy cows and relationships with macrorestriction genomic profiles. J. Clin. Microbiol. 2005, 43, 1278–1284. [Google Scholar]
- Dauwalder, O.; Thomas, D.; Ferry, T.; Debard, A.L.; Badiou, C.; Vandenesch, F.; Etienne, J.; Lina, G.; Monneret, G. Comparative inflammatory properties of staphylococcal superantigenic enterotoxins SEA and SEG: implications for septic shock. J. Leukoc. Biol. 2006, 80, 753–758. [Google Scholar]
- Dauwalder, O.; Pachot, A.; Cazalis, M.A.; Paye, M.; Faudot, C.; Badiou, C.; Mougin, B.; Vandenesch, F.; Etienne, J.; Lina, G.; Monneret, G. Early kinetics of the transcriptional response of human leukocytes to staphylococcal superantigenic enterotoxins A and G. Microb. Pathog. 2009, 47, 171–176. [Google Scholar]
- Mempel, M.; Lina, G.; Hojka, M.; Schnopp, C.; Seidl, H.P.; Schäfer, T.; Ring, J.; Vandenesch, F.; Abeck, D. High prevalence of superantigens associated with the egc locus in Staphylococcus aureus isolates from patients with atopic eczema. Eur. J. Clin. Microbiol. Infect. Dis. 2003, 22, 306–309. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Argudín, M.Á.; Mendoza, M.C.; Rodicio, M.R. Food Poisoning and Staphylococcus aureus Enterotoxins. Toxins 2010, 2, 1751-1773. https://doi.org/10.3390/toxins2071751
Argudín MÁ, Mendoza MC, Rodicio MR. Food Poisoning and Staphylococcus aureus Enterotoxins. Toxins. 2010; 2(7):1751-1773. https://doi.org/10.3390/toxins2071751
Chicago/Turabian StyleArgudín, María Ángeles, María Carmen Mendoza, and María Rosario Rodicio. 2010. "Food Poisoning and Staphylococcus aureus Enterotoxins" Toxins 2, no. 7: 1751-1773. https://doi.org/10.3390/toxins2071751
APA StyleArgudín, M. Á., Mendoza, M. C., & Rodicio, M. R. (2010). Food Poisoning and Staphylococcus aureus Enterotoxins. Toxins, 2(7), 1751-1773. https://doi.org/10.3390/toxins2071751




