The Black Aspergillus Species of Maize and Peanuts and Their Potential for Mycotoxin Production
Abstract
:1. Introduction
2. The Black Aspergillus Species
| Speciesa | Hostb | Ochratoxin | Biologically active metabolites |
|---|---|---|---|
| A. niger var. nigerc | Maize, peanuts, grapes, and grape products, coffee, tea, beans, spicesd | +i | Fumonisin B2 |
| Fumonisin B4 | |||
| Nigragilline, Malforminf | |||
| A. carbonarius | Grapes, java coffee bean | + | Carbonaronesg |
| A. tubingensis | Arabica coffee beans | + | Malformin, nigranillin |
| A. brasiliensis | Grapes | - | Malformin |
| A. acidus | Raisins | - | Uk |
| A. ibericus | Grapes | - | Uk |
| A homomorphus: | Soil, nh | - | Secalonic acidh |
| A. ellipticus | Soil, nh | - | Terphenyllinf |
| A. aculeatinus | Arabica coffee bean | - | Uk |
| A. aculeatus | Green Coffee bean | - | Secalonic acid, Aspergillusol Ag |
| A. japonicus | Grapes, maize, peanut | + | Cycloclavin |
| A. uvarum | Healthy grapes | - | Secalonic acid |
| A. piperis | Black pepper | - | Aflavininese |
| A. sclerotiicarbonarius | Robusta coffee bean | - | Uk |
| A. sclerotioniger | Coffe bean | Aflavinines | |
| A. heteromorphus | Soil, nh | - | Uk |
3. Mycotoxins Produced by Black Aspergillus sp.
3.1. Ochratoxins

| Strain Numbera | Location | Species |
|---|---|---|
| RRC 453 | Peanut, South Georgia | A. niger |
| RRC 454 | Peanut, South Georgia | A. foetidus |
| RRC 455 | Peanut, South Georgia | A. niger |
| RRC 456 | Peanut, South Georgia | A. niger |
| RRC 457 | Peanut, South Georgia | A. niger |
| RRC 458 | Peanut, South Georgia | A. foetidus |
| RRC 459 | Peanut, South Georgia | A. niger |
| RRC 460 | Peanut, South Georgia | A. japonicus |
| RRC 475 | Peanut slurries, Dawson, Georgia | A. niger |
| RRC 476 | Peanut slurries, Dawson, Georgia | A. niger |
| RRC 477 | Peanut slurries, Dawson, Georgia | A. foetidus |
| RRC 478 | Peanut slurries, Dawson, Georgia | A. niger |
| RRC 479 | Peanut slurries, Dawson, Georgia | A. japonicus |
| RRC 480 | Peanut slurries, Dawson, Georgia | A. niger |
| RRC 481 | Peanut slurries, Dawson, Georgia | A. niger |
| RRC 482 | Maize kernels, Midwestern, USA | A. niger |
| RRC 483 | Maize kernels, Midwestern, USA | A. niger |
| RRC 484 | Maize kernels, Midwestern, USA | A. niger |
| RRC 485 | Maize kernels, Midwestern, USA | A. niger |
| RRC 486 | Maize, kernels, Midwestern, USA | A. niger |
| RRC 487 | Maize kernels, Midwestern, USA | A. niger |
| RRC 488 | Maize kernels, Midwestern, USA | A. niger |
| RRC 489 | Maize kernels, Midwestern, USA | A. niger |
| RRC 490 | Maize kernels, Midwestern, USA | A. niger |
| RRC 493 | Maize kernels, Midwestern, USA | A. niger |
| RRC 494 | Maize kernels, Midwestern, USA | A. niger |
| RRC 495 | Maize kernels, Midwestern, USA | A. niger |
| RRC 497 | Maize kernels, Midwestern, USA | A. niger |
| RRC 500 | Maize kernels, Midwestern, USA | A. niger |
| RRC 501 | Maize kernels, Midwestern, USA | A. niger |
| RRC 503 | Maize kernels, Midwestern, USA | A. niger |
| RRC 504 | Maize kernels, Midwestern, USA | A. niger |
| RRC 507 | Maize kernels, Midwestern, USA | A. foetidus |
| RRC 510 | Maize kernels, Midwestern, USA | A. niger |
3.2. Ochratoxin contaminated products and producing species
3.3. Fumonisins
| Aspergillus Species | % Isolation Frequencya | |
|---|---|---|
| Maize | Peanuts | |
| A. niger | 95 | 67 |
| A. foetidus | - | 20 |
| A. japonicus | 5 | 13 |
3.4. Other mycotoxins
4. Host Associations and Plant Pathology
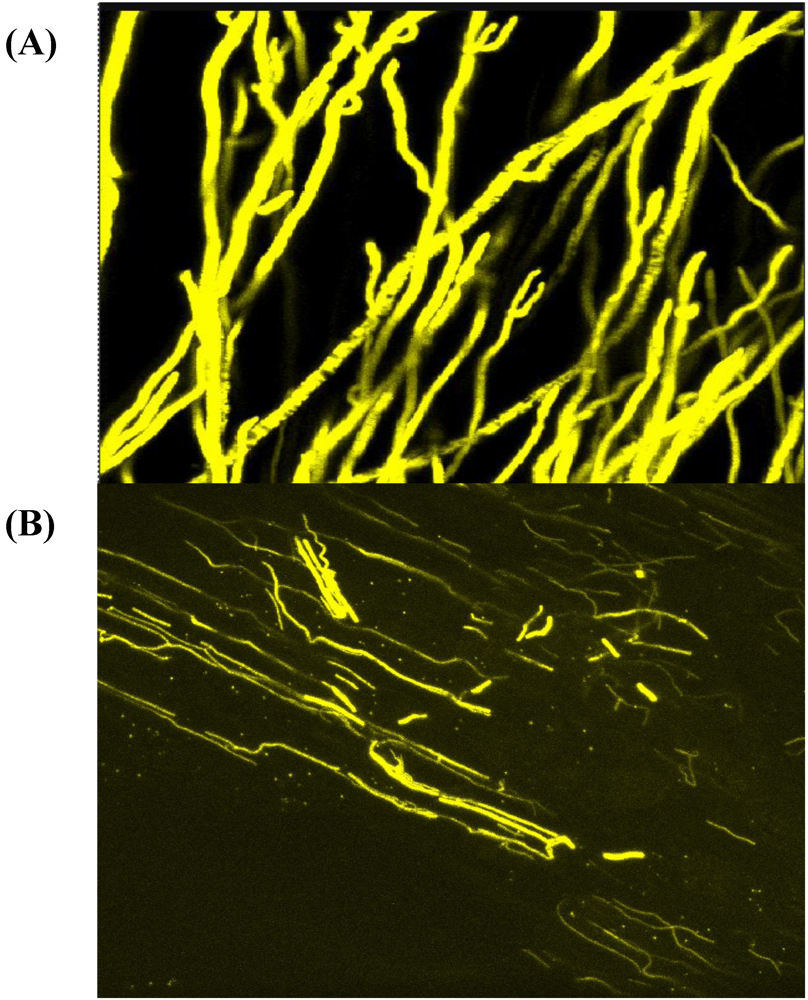
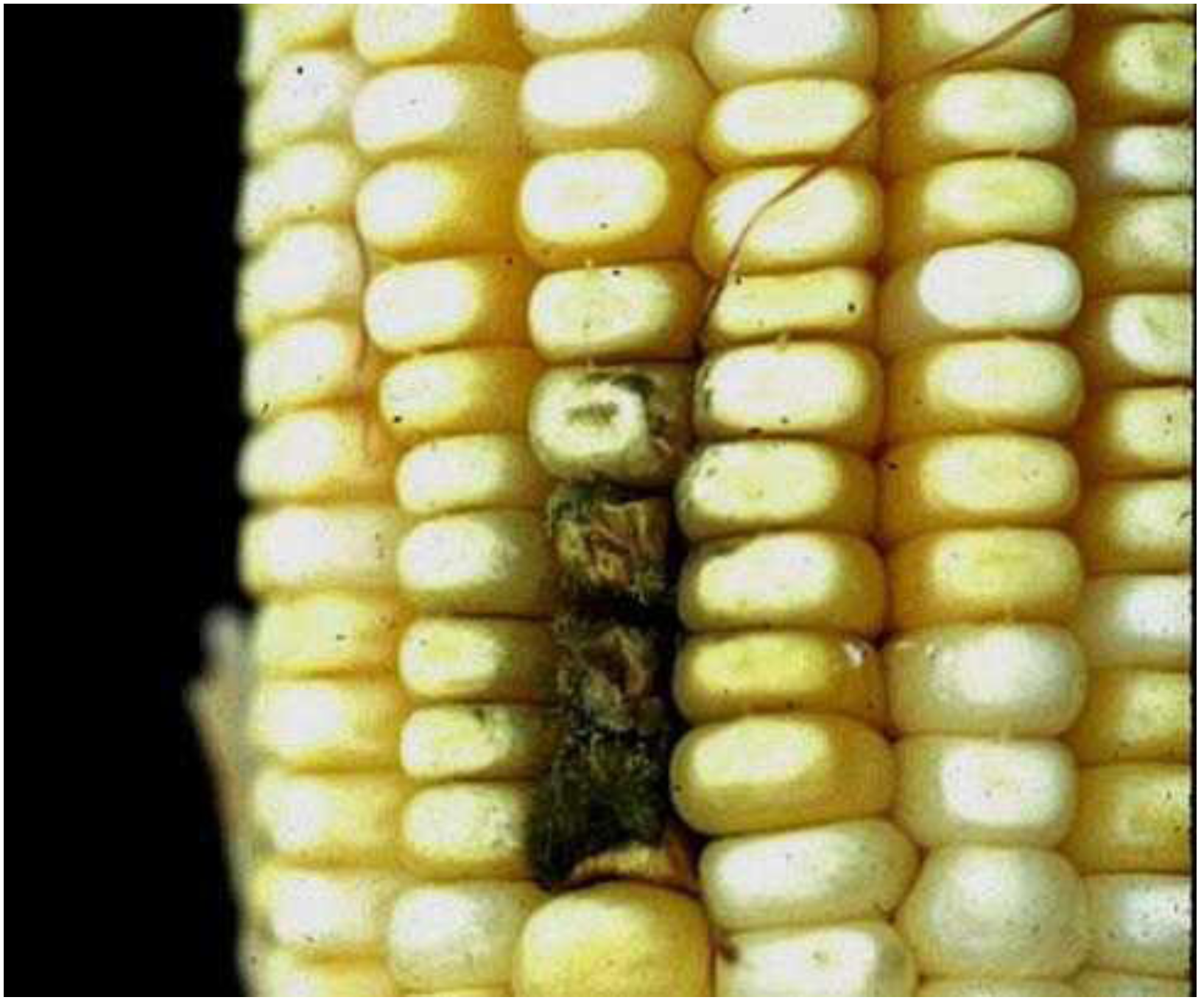

5. Conclusions
References
- Micheli, P.A. Nova Plantarum General Juxta Tournefortii Methodum Disposita; Typis Bernardi Paperinii: Florence, Italy, 1729. [Google Scholar]
- Link, H.F. Observationes in Ordines Plantarum Naturales; Gesellschaft Naturforschender Freunde zu: Berlin, Germany, 1809; Volume 3, pp. 1–42. [Google Scholar]
- Forgacs, J.; Carll, W.T. Mycotoxicoses. Adv. Vet. Sci. 1962, 7, 273–382. [Google Scholar]
- Allcroft, R.C.R.B.; Sargent, K.; O'Kelly, J. A toxic factor in Brazilian groundnut meal. Vet. Rec. 1961, 73, 428. [Google Scholar]
- Sargeant, K.; Sheridan, A.; O'Kelly, J.; Carnaghan, R.B.A. Toxicity assocaited with certain samples of groundnuts. Nature 1961, 192, 1096. [Google Scholar]
- van der Merve, K.J.; Steyn, P.S.; Fourie, L. The constitution of ochratoxin A, B, and C, metabolites of Aspergillus ochraceus Wilh. J. Chem. Soc. 1965, 7083–7088. [Google Scholar]
- Hamilton, P.B.; Huff, W.E.; Harris, J.R.; Wyatt, R.D. Natural occurrences of ochratoxicoses in poultry. Poult. Sci. 1982, 61, 1832–1841. [Google Scholar] [PubMed]
- Abarca, M.L.; Bragulat, M.R.; Castella, G.; Cabanes, F.J. Ochratoxin A production by strains of Aspergillus niger var. niger. Appl. Environ. Microbiol. 1994, 60, 2650–2652. [Google Scholar] [PubMed]
- Klich, M.A. Biogeography of Aspergillus species in soil and litter. Mycologia 2002, 94, 21–27. [Google Scholar] [PubMed]
- Raper, K.B.; Fennell, D.I. Aspregillus niger Group. In The Genus Aspergillus; Raper, K.B., Fennell, D.I., Eds.; The Williams & Wilkins Co.: Baltimore, USA, 1965; Volume Chapter 16, pp. 293–344. [Google Scholar]
- Samson, R.A.; Varga, J. Molecular systematics of Aspergillus and its teleomophs. In Aspergillus: Molecular Biology and Genomics; Machida, M., Gomi, K., Eds.; Caister Academic Press: Tsukuba, Ibaraki, Japan, 2010; pp. 20–25. [Google Scholar]
- Abarca, M.L.F.; Accensi, F.; Cano, J.; Cabanes, F.J. Taxonomy and significance of black aspergilli. Anton. Van Leeuwen. 2004, 86, 33–49. [Google Scholar]
- Accensi, F.; Abarca, M.L.; Cano, J.; Figuera, L.; Cabanes, F.J. Distribution of ochratoxin A producing strains in the A. niver aggregate. Antonie Van Leeuwenhoek 2001, 79, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Samson, R.A.; Houbraken, J.; kuijpers, A.; Frank, J.M.; Frisvad, J.C. New ochratoxin or sclerotium producing species in Aspergillus section Nigra. Stud. Mycol. 2004, 50, 45–61. [Google Scholar]
- Palencia, E.R.; Klich, M.A.; Glenn, A.E.; Bacon, C.W. Use of a rep PCR system to predict species in the Aspergillus section Nigri. J. Microbiol. Methods 2009, 79, 1–7. [Google Scholar] [PubMed]
- Magnoli, C.; Hallak, C.; Astoreca, A.; Ponsone, L.; Chiacchiera, S.; Dalcero, A.M. Occurrence of ochratoxin A-producing fungi in commercial corn kernels in Argentina. Mycopathologia 2006, 161, 53–58. [Google Scholar] [PubMed]
- Frischbier, C.T.; Richtesteiger, R. Bildung von oxalsaure durch Aspergillus niger in Brot and in der streu. S. Veterinark. 1941, 53, 391. [Google Scholar]
- Wilson, B.J.; Wilson, C.H. Oxalate formation in moldy feedstuffs as a possible factor in livestock toxic disease. Am. J. Vet. Res. 1961, 21, 261–269. [Google Scholar]
- International Agency on Research on Cancer. Some naturally occurring substances: Food items and constitutents, heterocyclic aromatic amines and mycotoxins. IRRC, 1993. [Google Scholar]
- Castegnaro, M.; Canadas, T.; Vrabcheva, T.; Petkova-Bocharova, T.; Chernozemsky, I.N.; Pfohl-Leszkowicz, P. Balkan endemic nephropathy: Role of ochratoxin A through biomarkers. Mol. Nutri. Food Res. 2005, 50, 519–529. [Google Scholar]
- Lambert, D.; Padfield, P.J.; McLaughlin, S.; Cannell, S.; O'Neill, C.A. Ochratoxin A displaces claudins from detergent resistant membrane microdomains. Biochem. Biophys. Res. Commun. 2007, 358, 632–636. [Google Scholar] [PubMed]
- Shotwell, O.L.; Hesseltine, C.W.; Goulden, M.L. Ochratoxin A: Occurrence as natural contaminant of a corn sample. Appl. Microbiol. 1981, 17, 765–766. [Google Scholar]
- Accensi, F.; Abarca, M.L.; Cano, J.; Figuera, L.; Cabanes, F.J. Ochratoxin A producing strains in the Aspergillus niger aggregate. Antonie Van Leeuwenhoek 2001, 79, 365–370. [Google Scholar] [PubMed]
- Bennett, J.W.; Klich, M.A. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [PubMed]
- Rosa, C.A.R.; Ribeiro, J.M.M.; Fraga, M.J.; Gattuso, M.A.; Cavaglieri, L.R.; Magnoli, C.E.; Dalcero, A.M.; Lopes, C.W.G. Mycoflora of poultry feeds and ochratoxin-producing ability of isolated Aspergillus and Penicillium species. Vet. Microbiol. 2006, 113, 89–96. [Google Scholar] [PubMed]
- Dalcero, A.; Magnoli, C.; Hallak, C.; Chiacchiera, S.M.; Palacio, G.; Rosa, C.A.R. Detection of ochratoxin A in animal feeds and capacity to produce this mycotoxin by Aspergillus section Nigri in Argentina. Food Addit. Contam. 2002, 19, 1065–1072. [Google Scholar] [PubMed]
- Puntaric, D.; Bosnir, J.; Smit, Z.; Skes, I.; Baklaic, Z. Ochratoxin A in corn and wheat: Georgraphical association with endemic nephropathy. Croatian Med. J. 2001, 42, 175–180. [Google Scholar]
- Kuiper-Goodman, T. Risk assessment of ochratoxin A residues in food. IARC Sci. Publ. 1991, 115, 307–320. [Google Scholar] [PubMed]
- Domijan, A.M.; Peraica, M.; Fuchs, R.; Lucic, A.; Radic, B.; Balija, M. Ochratoxin A in blood of healthy population in Zagrab. Arh. Hig. Rada. Tokskol. 1999, 50, 263–271. [Google Scholar]
- Rosa, C.A.R.; Ribeiro, J.M.M.; Fraga, M.J.; Gatti, M.; Cavaglieri, L.R.; Magnoli, C.E.; Dalcero, A.M.; Lopes, C.W.G. Mycoflora of poultry feeds and ochratoxin-producing ability of isolated Aspergillus and Penicillium species. Vet. Microbiol. 2006, 113, 89–96. [Google Scholar] [PubMed]
- Hajjaji, A.; El Otmani, M.; Bouya, D.; Bouseta, A.; Mathieu, F.; Collin, S.; Lebrihi, A. Occurrence of mycotoxins (Ochratoxin A, deoxynivalenol) and toxigenic fungi in Moroccan wheat grains: Impact of ecological factors on the growth and ochratoxin A production. Mol. Nutri. Food Res. 2006, 50, 494–499. [Google Scholar] [CrossRef]
- Accensi, F.; Cano, J.; Figuera, L.; Abaca, M.L.; Cabanes, F.J. New PCR method to differentiate species in the Aspergillus niger aggregate. FEMS Microbiol. Lett. 1999, 180, 191–196. [Google Scholar] [PubMed]
- Medina, A.; Mateo, R.; Lopez-Ocana, L.; Valle-Algarra, F.M. Study of Spanish grape microbiota and ochratoxin A production by isolates of Aspergillus tubingensis and other members of Aspergillus section nigri. Appl. Environ. Microbiol. 2005, 71, 4696–4702. [Google Scholar] [PubMed]
- Frisvad, J.C.; Smedsgaard, J.; Samson, R.A.; Larsen, T.O.; Trane, U. Fumonisin B2 Production by Aspergillus niger. J. Agric. Food Chem. 2007, 55, 9727–9732. [Google Scholar] [PubMed]
- Frisvad, J.C.; Frank, J.M.; Houbraken, J.A.M.P.; Kuijpers, A.F.A.; Samson, R.A. New ochratoxin A producing species of Aspergillus section circumdati. Stud. Mycol. 2004, 50, 23–24. [Google Scholar]
- Nielsen, K.F.; Mogensen, J.M.; Larsen, T.O.; Frisvad, J.C. Review of secondary metabolites and mycotoxins from the Aspergillus group. Anal. Bioanal. Chem. 2009, 395, 1225–1242. [Google Scholar] [PubMed]
- Mansson, M.; Klejnstrup, M.L.; Phipps, R.K.; Nielsen, K.F.; Frisvad, J.C.; Gotfredsen, C.H.; Larsen, T.O. Isolation and NMR chracterization of fumonisin B2 and a new fumonisin B6 from Aspergillus niger. J. Agric. Food Chem. 2010, 58, 949–953. [Google Scholar] [PubMed]
- Magan, N.; Aldrich, C.G. Conditions of formation of ochratoxin A in drying, transport and in different commodities. Food Addit. Contam. 2005, 1, 10–16. [Google Scholar]
- Bacon, C.W.; Burdick, D. The growth of fungi in broiler houses. Poult. Sci. 1977, 56, 653–661. [Google Scholar] [PubMed]
- Bacon, C.W.; Sweeny, J.G.; Robbins, J.D.; Burdick, D. Production of penicillic acid and ochratoxin A on poultry feed and capacity to produce this mycotoxin in Aspergillus section Nigri in Argentina. Appl. Microbiol. 1973, 26, 155–160. [Google Scholar] [PubMed]
- Balcero, A.; Magnoli, C.; Hallak, C.; Ciacchiera, G.; Palacio, G.; Rosa, C.A.R. Detection of ochratoxin A in animal feed and capacity to produce this mycotoxin in Aspergillus section nigri in Argentina. Food Addit.Contam. 2002, 19, 1065–1072. [Google Scholar] [PubMed]
- Gelderblom, W.C.A.; Jaskiewicz, K.; Marasas, W.F.O.; Thiel, P.G.; Horak, R.M.; Vleggar, R.; Kriek, N.P.J. Fumonisins--novel mycotoxins with cancer-promoting activity produced by Fusarium moniliforme. Appl. Environ. Microbiol. 1988, 54, 1806–1811. [Google Scholar] [PubMed]
- Riley, R.T.; Norred, W.P.; Bacon, C.W. Fungal toxins in foods: Recent concerns. Annu. Rev. Nutr. 1993, 13, 167–189. [Google Scholar] [PubMed]
- Noonim, P.; Wahakarnchanakul, W.; Nielsen, K.F.; Frisvad, K.F.; Samson, R.A. Fumonisin B2 production by Aspergillus niger in Thai coffee beans. Food Addit. Contam. Part A 2009, 26, 94–100. [Google Scholar] [CrossRef]
- Lindenfelser, L.A.; Ciegler, A. Penicillic acid production in submerged culture. Appl. Environ. Microbiol. 1977, 34, 553–556. [Google Scholar] [PubMed]
- Ciegler, A. Bioproduction of ochratoxin and penicillic acid by members of the Aspergillus ochraceus group. Can. J. Microbiol. 1972, 18, 631–636. [Google Scholar] [PubMed]
- Birkinshaw, J.H.; Oxforn, A.E.; Raistrick, H. Studies in the biochemistry of microorganisms. XLVIII. Penicillic acid, a metabolic products of Penicillium puberculum Banier and P. cyclopium Westling. Biochem. J. 1936, 30, 394–411. [Google Scholar] [PubMed]
- Kubena, L.F.; Phillips, T.D.; Witzel, D.A.; Heidelbaugh, N.D. Toxicity of ochratoxin A and penicillic acid to chicks. Bull. Environ. Contam. Toxicol. 1984, 32, 711–716. [Google Scholar] [PubMed]
- Sansing, G.; Lillehoe, E.; Detroy, R.; Muller, M. Synergistic toxic effects of citrinum, ochratoxin A, and penicillic acid in mice. Toxicon 1976, 14, 213–220. [Google Scholar] [PubMed]
- Shepherd, E.; Philips, T.; Joiner, G.; Kubena, L.; Heidelbaugh, N. Ochratoxin A and Penicillic acid interaction in mice. J. Environ. Sci. Health 1981, B16, 557–573. [Google Scholar]
- Jamaji, K.; Fukushi, Y.; Hashidoko, Y.; Tahara, S. Penicillium frequentans isolated from Picea glehnii seedling roots as a possible biological control agent against damping-off. Ecol. Res. 2005, 20, 103–107. [Google Scholar]
- Cole, R.J.; Cox, R.H. Handbook of toxic fungal metabolites; Academic Press: New York, NY, USA, 1981. [Google Scholar]
- Heatley, N.G.; Philpot, F.J. The routine examination for antibiotics produced by moulds. J. Gen. Microbiol. 1947, 1, 232–237. [Google Scholar] [PubMed]
- Rasmussen, T.B.; Givskov, M. Quorum sensing inhibitors: A bargan of effects. Microbiology 2006, 152, 1325–1340. [Google Scholar]
- Rasmussen, T.B.; Skindersoe, M.E.; Bjarnsholt, T.; Phipps, R.K.; Christensen, K.B.; Jensen, P.O.; Andersen, J.B.; Larsen, T.O.; Hentzer, M.; Hoiby, N.; Givskov, M. Identity and effects of quorum-sensing inhibitors produced by Penicillium species. Microbiology 2005, 151, 1325–1340. [Google Scholar] [PubMed]
- Uroz, S.; Dessaux, Y.; Oger, P. Quorum sensing and quorum quenching: The yin and yang of bacterial communication. Chembiochem 2009, 10, 205–216. [Google Scholar] [PubMed]
- Liaqat, I.; Bachmann, T.R.; Sabri, A.N.; Edyvean, G.J.; Biggs, C.A. Investigating the effect of patulin, penicillic acid and EDTA on biofilm formation of isolates from dental unit water lines. Appl. Microbiol. Biotechnol. 2008, 81, 349–358. [Google Scholar] [PubMed]
- Rasch, M.; Rasmussen, T.B.; Andersen, J.B.; Persson, T.; Givskov, J.; Givskov, M.; Gram, L. Well-known quorum sensing inhibitors do not affect bacterial quorum sensing-regulated bean sprout spoilage. J. Appl. Microbiol. 2007, 102, 826–837. [Google Scholar] [PubMed]
- Pitt, J.I. Xerophilic Fungi and the Spoilage of Foods of Plant Origin; Duckworth, R.B., Ed.; Academic press: New York, NY, USA, 1975; pp. 273–307. [Google Scholar]
- Pozzi, C.R.; Correa, B.; Gambale, W.; Paula, C.A.; Chancon-Reche, N.O.; Carlos, M.; Meirelles, C.A. Postharvest and stored corn in Brazil: Mycoflora interactions, abiotic factors and mycotoxin occurrence. Food Addit. Contam. 1995, 12, 313–319. [Google Scholar] [PubMed]
- Magnoli, C.E.; Saenz, M.A.; Chiacchiera, S.M.; Dalcero, A.M. Natural occurrence of Fusarium species and fumonisin-production by toxigenic strains isolated from poultry feeds in Argentina. Mycopathologia 1999, 145, 35–41. [Google Scholar] [PubMed]
- Hayden, N.J.; Maude, R.B. The role of seed-borne Aspergillus niger in transmission of black mould of onion. Plant Pathol. 1992, 41, 573–581. [Google Scholar]
- Leong, S.L.; Hocking, A.D.; Scott, E.S. Aspergillus species producing ochratoxin A: Isolation from vineyard soils and infection of Semillo bunches in Australia. J. Appl. Microbiol. 2007, 102, 124–133. [Google Scholar] [PubMed]
- Griffin, C.J.; Smith, E.P.; Robinson, T.J. Population patterns of Aspergillus flavus group and A. niger group in field soils. Soil Biol. Biochem. 2001, 33, 253–257. [Google Scholar] [CrossRef]
- Palencia, E.R.; Glenn, A.E.; Bacon, C.W. Colonization of maize seedlings under drought conditions by two ochratoxin A producer species within the section A. section Nigri. Phytopathology 2009, 99, S99. [Google Scholar]
- Burgess, L.M.; Knight, T.E.; Tesoriero, L.; VaPhan, H.T. Common Diseases of Some Economically Important Crops; Australian Centre for International Agricultural Research: Canberra, Australia, 2009; Volume 11, pp. 166–167. [Google Scholar]
- Marin, S.; Sanchis, V.; Rull, F.; Ramos, A.J.; Magan, N. Collinization of maize grain by Fusarium moniliforme and Fusarium proliferatum in the presence of competing fungi and their impact on fumonisin production. J. Food Prot. 1998, 61, 1489–1496. [Google Scholar] [PubMed]
- Palacios-Cabrera, H.; Tanikawi, M.H.; Hashimoto, J.M.; de Menezes, H.C. Growth of Aspergillus ochraceus, A. carbonarium and A. niger on culture media at different water activites and temperatures. Brazilian J. Microbiol. 2005, 36, 24–28. [Google Scholar] [CrossRef]
- Glenn, A.E.; Zitomer, N.C.; Zimeri, A.M.; Williams, L.D.; Riley, R.T. Transformation-mediated complementation of a FUM gene cluster deletion in Fusarium verticilioides restores both fumonisin production and pathogenicity on maize seedlings. Mol. Plant Microbe Interact. 2008, 21, 87–97. [Google Scholar] [PubMed]
- Scudamore, K.A.; Livesey, C.T. Occurrence and significance of mycotoxins in forage crops and silage: A review. J. Sci. Food Agric. 1998, 77, 1–17. [Google Scholar]
- Park, J.W.; Kim, M.A.; Shon, D.H.; Kim, Y.B. Natural co-occurrence of aflatoxin B1, fumonisin B1 and ochratoxin A in barley and corn food from Korea. Food Addit. Contam. 2002, 19, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- Axxiz, N.H.; Youssef, A.Y.; El-Fouly, M.Z.; Moussa, L.A. Contamination of some common medicinal plant samples and spices by fungi and their mycotoxins. Bot. Bull. Acad. Sin. 1998, 39, 278–285. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Palencia, E.R.; Hinton, D.M.; Bacon, C.W. The Black Aspergillus Species of Maize and Peanuts and Their Potential for Mycotoxin Production. Toxins 2010, 2, 399-416. https://doi.org/10.3390/toxins2040399
Palencia ER, Hinton DM, Bacon CW. The Black Aspergillus Species of Maize and Peanuts and Their Potential for Mycotoxin Production. Toxins. 2010; 2(4):399-416. https://doi.org/10.3390/toxins2040399
Chicago/Turabian StylePalencia, Edwin R., Dorothy M. Hinton, and Charles W. Bacon. 2010. "The Black Aspergillus Species of Maize and Peanuts and Their Potential for Mycotoxin Production" Toxins 2, no. 4: 399-416. https://doi.org/10.3390/toxins2040399
APA StylePalencia, E. R., Hinton, D. M., & Bacon, C. W. (2010). The Black Aspergillus Species of Maize and Peanuts and Their Potential for Mycotoxin Production. Toxins, 2(4), 399-416. https://doi.org/10.3390/toxins2040399




