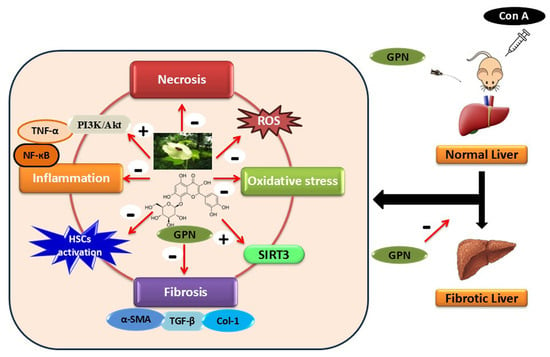
SIRT3, NF-κB/TNF-α and PI3K/Akt Pathways Mediate the Hepatoprotective Activity of Gossypin Against Concanavalin A-Induced Hepatic Fibrosis
Abstract
1. Introduction
2. Results
2.1. GPN Opposed the Con A-Induced Elevation in Serum Biomarkers of Liver Function
2.2. GPN Attenuated Con A-Induced Hepatic Histopathological Lesions
2.3. GPN Ameliorated Hepatic Levels of TGF-β1 and Col-1 as Well as Hepatic Immunohistochemical Expression of α-SMA in Con A-Administered Mice
2.4. GPN Influenced Hepatic Redox Status in Con A-Challenged Mice
2.5. GPN Enhanced Hepatic Protein and Gene Expression of SIRT3, in Con A-Administered Mice
2.6. GPN Alleviated NF-κB/TNF-α Pathway in Con A-Administered Mice
2.7. GPN Enhanced Protein and Gene Expression of PI3K and Akt in the Hepatic Tissue of Con A-Challenged Mice
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals
4.3. Experimental Design
4.4. Assessment of Liver Functions
4.5. Histopathological Evaluation and Grading
4.6. Assessment of Hepatic Oxidative Stress Biomarkers
4.7. Assessment of Hepatic Tissue Levels of Transforming Growth Factor Beta 1 (TGF-β1), Tumor Necrosis Factor-Alpha (TNF-α), Collagen Type 1 (Col-1), and Sirtuin 3 (SIRT3) by ELISA
4.8. Assessment of Hepatic Tissue Levels of Phosphoinositide-3-Kinase (p-PI3K) and Protein Kinase B (p-Akt) by Western Blotting
4.9. Assessment of Hepatic SIRT-3, PI3K, and Akt Gene Expression by RT-PCR
4.10. Immunohistochemical Analysis
4.11. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| α-SMA | Alpha-smooth muscle actin |
| AKT | Protein Kinase B |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| CLI | Chronic liver injury |
| Col-1 | Collagen type 1 |
| Con A | Concanavalin A |
| ECM | Extracellular matrix |
| GPN | Gossypin |
| HSCs | hepatic stellate cells |
| LDH | Lactate dehydrogenase |
| MDA | Malondialdehyde |
| NF-κB | Nuclear factor kappa-B |
| PI3K | Phosphoinositide 3-kinase |
| SIRT3 | Sirtuin 3 |
| TAC | Total antioxidant capacity |
| TGF-β | Transforming growth factor-beta |
| TNF-α | Tumor necrosis factor-alpha |
References
- Poynard, T.; Mathurin, P.; Lai, C.-L.; Guyader, D.; Poupon, R.; Tainturier, M.-H.; Myers, R.P.; Muntenau, M.; Ratziu, V.; Manns, M. A comparison of fibrosis progression in chronic liver diseases. J. Hepatol. 2003, 38, 257–265. [Google Scholar] [CrossRef]
- Fisher, K.; Vuppalanchi, R.; Saxena, R. Drug-induced liver injury. Arch. Pathol. Lab. Med. 2015, 139, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Hora, S.; Wuestefeld, T. Liver injury and regeneration: Current understanding, new approaches, and future perspectives. Cells 2023, 12, 2129. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Lee, Y.A.; Fujiwara, N.; Ybanez, M.; Allen, B.; Martins, S.; Fiel, M.I.; Goossens, N.; Chou, H.-I.; Hoshida, Y. A simple diet-and chemical-induced murine NASH model with rapid progression of steatohepatitis, fibrosis and liver cancer. J. Hepatol. 2018, 69, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.-D.; Zhou, J.; Chen, E.-Q. Molecular mechanisms and potential new therapeutic drugs for liver fibrosis. Front. Pharmacol. 2022, 13, 787748. [Google Scholar] [CrossRef]
- Parola, M.; Pinzani, M. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues. Mol. Aspects Med. 2019, 65, 37–55. [Google Scholar] [CrossRef]
- Brenner, D.A. Molecular pathogenesis of liver fibrosis. Trans. Am. Clin. Climatol. Assoc. 2009, 120, 361. [Google Scholar]
- Zhang, Y.E. Non-Smad pathways in TGF-β signaling. Cell Res. 2009, 19, 128–139. [Google Scholar] [CrossRef]
- Shamsan, E.; Almezgagi, M.; Gamah, M.; Khan, N.; Qasem, A.; Chuanchuan, L.; Haining, F. The role of PI3k/AKT signaling pathway in attenuating liver fibrosis: A comprehensive review. Front. Med. 2024, 11, 1389329. [Google Scholar] [CrossRef]
- Nabekura, T.; Matsuo, S.; Shibuya, A. Concanavalin-A-Induced Acute Liver Injury in Mice. Curr. Protoc. 2024, 4, e1117. [Google Scholar] [CrossRef]
- Li, S.-L.; Cao, R.; Hu, X.-F.; Xiong, P.; Zhao, G.-Y.; Xie, Y.-N.; Wang, Z.-M.; Li, Y.-K.; Yang, B.; Yang, J. Daidzein ameliorated concanavalin A-induced liver injury through the Akt/GSK-3β/Nrf2 pathway in mice. Ann. Transl. Med. 2021, 9, 1228. [Google Scholar] [CrossRef]
- Feng, Q.; Yao, J.; Zhou, G.; Xia, W.; Lyu, J.; Li, X.; Zhao, T.; Zhang, G.; Zhao, N.; Yang, J. Quantitative proteomic analysis reveals that arctigenin alleviates concanavalin A-induced hepatitis through suppressing immune system and regulating autophagy. Front. Immunol. 2018, 9, 1881. [Google Scholar] [CrossRef]
- Kunnumakkara, A.B.; Nair, A.S.; Ahn, K.S.; Pandey, M.K.; Yi, Z.; Liu, M.; Aggarwal, B.B. Gossypin, a pentahydroxy glucosyl flavone, inhibits the transforming growth factor beta-activated kinase-1-mediated NF-κB activation pathway, leading to potentiation of apoptosis, suppression of invasion, and abrogation of osteoclastogenesis. Blood J. Am. Soc. Hematol. 2007, 109, 5112–5121. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Shen, X.; Tong, C.; Zhang, S.; Chen, Q.; Li, Y.; Li, S. Gossypin: A flavonoid with diverse pharmacological effects. Chem. Biol. Drug Des. 2023, 101, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, M.A.; Amer, N.M.; Abdallah, H.M.; Saleh, M.S. A comprehensive tool in recycling plant-waste of Gossypium barbadense L agricultural and industrial waste extracts containing gossy pin and gossypol: Hepatoprotective, anti-inflammatory and antioxidant effects. Plant Methods 2024, 20, 54. [Google Scholar] [CrossRef] [PubMed]
- Kweon, Y.-O.; Goodman, Z.D.; Dienstag, J.L.; Schiff, E.R.; Brown, N.A.; Burkhardt, E.; Schoonhoven, R.; Brenner, D.A.; Fried, M.W. Decreasing fibrogenesis: An immunohistochemical study of paired liver biopsies following lamivudine therapy for chronic hepatitis B. J. Hepatol. 2001, 35, 749–755. [Google Scholar] [CrossRef]
- Guido, M.; Rugge, M.; Chemello, L.; Leandro, G.; Fattovich, G.; Giustina, G.; Cassaro, M.; Alberti, A. Liver stellate cells in chronic viral hepatitis: The effect of interferon therapy. J. Hepatol. 1996, 24, 301–307. [Google Scholar] [CrossRef]
- Nakatani, K.; Seki, S.; Kawada, N.; Kitada, T.; Yamada, T.; Sakaguchi, H.; Kadoya, H.; Ikeda, K.; Kaneda, K. Expression of SPARC by activated hepatic stellate cells and its correlation with the stages of fibrogenesis in human chronic hepatitis. Virchows Arch. 2002, 441, 466–474. [Google Scholar] [CrossRef]
- Mann, D.; Smart, D. Transcriptional regulation of hepatic stellate cell activation. Gut 2002, 50, 891–896. [Google Scholar] [CrossRef]
- Wu, Q.-J.; Zhang, T.-N.; Chen, H.-H.; Yu, X.-F.; Lv, J.-L.; Liu, Y.-Y.; Liu, Y.-S.; Zheng, G.; Zhao, J.-Q.; Wei, Y.-F. The sirtuin family in health and disease. Signal Transduct. Target. Ther. 2022, 7, 402. [Google Scholar] [CrossRef]
- Zhang, J.; Xiang, H.; Liu, J.; Chen, Y.; He, R.-R.; Liu, B. Mitochondrial Sirtuin 3: New emerging biological function and therapeutic target. Theranostics 2020, 10, 8315. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, M.; Wang, J.; Ji, K.; Wang, Y.; Sun, X.; Xu, C.; Wang, Q.; He, N.; Song, H. NMN ameliorated radiation induced damage in NRF2-deficient cell and mice via regulating SIRT6 and SIRT7. Free. Radic. Biol. Med. 2022, 193, 342–353. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Wang, A.; Li, D.; Wang, Y.; Lin, L. Pinocembrin from Penthorum chinense Pursh suppresses hepatic stellate cells activation through a unified SIRT3-TGF-β-Smad signaling pathway. Toxicol. Appl. Pharmacol. 2018, 341, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Sharawy, M.H.; El-Kashef, D.H.; Shaaban, A.A.; El-Agamy, D.S. Anti-fibrotic activity of sitagliptin against concanavalin A-induced hepatic fibrosis. Role of Nrf2 activation/NF-κB inhibition. Int. Immunopharmacol. 2021, 100, 108088. [Google Scholar] [CrossRef]
- Cinar, I.; Yayla, M.; Toktay, E.; Binnetoğlu, D. Effects of gossypin on acetaminophen-induced hepatotoxicity in mice. Trak. Univ. J. Nat. Sci. 2024, 25. [Google Scholar] [CrossRef]
- Elfeky, M.G.; Mantawy, E.M.; Gad, A.M.; Fawzy, H.M.; El-Demerdash, E. Mechanistic aspects of antifibrotic effects of honokiol in Con A-induced liver fibrosis in rats: Emphasis on TGF-β/SMAD/MAPK signaling pathways. Life Sci. 2020, 240, 117096. [Google Scholar] [CrossRef]
- Ding, W.; Zhou, D.; Zhang, S.; Qian, J.; Yang, L.; Tang, L. Trimetazidine inhibits liver fibrosis and hepatic stellate cell proliferation and blocks transforming growth factor-β (TGFβ)/Smad signaling in vitro and in vivo. Bioengineered 2022, 13, 7147–7156. [Google Scholar] [CrossRef]
- Darwish, S.F.; El-Bakly, W.M.; El-Naga, R.N.; Awad, A.S.; El-Demerdash, E. Antifibrotic mechanism of deferoxamine in concanavalin A induced-liver fibrosis: Impact on interferon therapy. Biochem. Pharmacol. 2015, 98, 231–242. [Google Scholar] [CrossRef]
- Serrya, M.S.; Zaghloul, M.S. Mycophenolate mofetil attenuates concanavalin A-induced acute liver injury through modulation of TLR4/NF-κB and Nrf2/HO-1 pathways. Pharmacol. Rep. 2020, 72, 945–955. [Google Scholar] [CrossRef]
- Mohamed, G.A.; Ibrahim, S.R.; El-Agamy, D.S.; Elsaed, W.M.; Sirwi, A.; Asfour, H.Z.; Koshak, A.E.; Elhady, S.S. Cucurbitacin E glucoside alleviates concanavalin A-induced hepatitis through enhancing SIRT1/Nrf2/HO-1 and inhibiting NF-ĸB/NLRP3 signaling pathways. J. Ethnopharmacol. 2022, 292, 115223. [Google Scholar] [CrossRef]
- Yazğan, Y.; Yazğan, B. Gossypin regulated doxorubicin-induced oxidative stress and inflammation in H9c2 cardiomyocyte cells. Med. Rec. 2024, 6, 44–49. [Google Scholar] [CrossRef]
- Huang, H.; Wang, J.; Hussain, S.A.; Gangireddygari, V.S.R.; Fan, Y. Gossypin exert lipopolysaccharide induced lung inflammation via alteration of Nrf2/HO-1 and NF-κB signaling pathway. Environ. Toxicol. 2023, 38, 1786–1799. [Google Scholar] [CrossRef]
- Liu, J.; Li, D.; Zhang, T.; Tong, Q.; Ye, R.D.; Lin, L. SIRT3 protects hepatocytes from oxidative injury by enhancing ROS scavenging and mitochondrial integrity. Cell Death Dis. 2017, 8, e3158. [Google Scholar] [CrossRef]
- Zhang, T.; Fang, Z.; Linghu, K.G.; Liu, J.; Gan, L.; Lin, L. Small molecule-driven SIRT3-autophagy-mediated NLRP3 inflammasome inhibition ameliorates inflammatory crosstalk between macrophages and adipocytes. Br. J. Pharmacol. 2020, 177, 4645–4665. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Dou, X.; Wang, Z.; Shi, K.; Wang, Z.; Ding, C.; Sang, X.; Zhong, X.; Shao, M.; Han, X. SIRT3: A potential therapeutic target for liver fibrosis. Pharmacol. Ther. 2024, 257, 108639. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.K. Does the interdependence between oxidative stress and inflammation explain the antioxidant paradox? Oxid. Med. Cell. Longev. 2016, 2016, 5698931. [Google Scholar] [CrossRef] [PubMed]
- Seki, E.; Schwabe, R.F. Hepatic inflammation and fibrosis: Functional links and key pathways. Hepatology 2015, 61, 1066–1079. [Google Scholar] [CrossRef]
- Li, J.; Wang, T.; Liu, P.; Yang, F.; Wang, X.; Zheng, W.; Sun, W. Hesperetin ameliorates hepatic oxidative stress and inflammation via the PI3K/AKT-Nrf2-ARE pathway in oleic acid-induced HepG2 cells and a rat model of high-fat diet-induced NAFLD. Food Funct. 2021, 12, 3898–3918. [Google Scholar] [CrossRef]
- Abraldes, J.G.; Tarantino, I.; Turnes, J.; Garcia-Pagan, J.C.; Rodés, J.; Bosch, J. Hemodynamic response to pharmacological treatment of portal hypertension and long-term prognosis of cirrhosis. Hepatology 2003, 37, 902–908. [Google Scholar] [CrossRef]
- Maddika, S.; Ande, S.R.; Panigrahi, S.; Paranjothy, T.; Weglarczyk, K.; Zuse, A.; Eshraghi, M.; Manda, K.D.; Wiechec, E.; Los, M. Cell survival, cell death and cell cycle pathways are interconnected: Implications for cancer therapy. Drug Resist. Updates 2007, 10, 13–29. [Google Scholar] [CrossRef]
- Yang, F.-J.; He, Y.-H.; Zhou, J.-H. Fenofibrate pre-treatment suppressed inflammation by activating phosphoinositide 3 kinase/protein kinase B (PI3K/Akt) signaling in renal ischemia-reperfusion injury. J. Huazhong Univ. Sci. Technol. Med. Sci. 2015, 35, 58–63. [Google Scholar] [CrossRef]
- Mao, Y.; Wang, J.; Yu, F.; Cheng, J.; Li, H.; Guo, C.; Fan, X. Ghrelin reduces liver impairment in a model of concanavalin A-induced acute hepatitis in mice. Drug Des. Dev. Ther. 2015, 9, 5385–5396. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.-M.; Li, X.-L.; Zhu, Y.; Shi, R.; Wang, Z.-J.; Xiao, J.-P.; Wang, D.-G. Diosmin ameliorates renal fibrosis through inhibition of inflammation by regulating SIRT3-mediated NF-κB p65 nuclear translocation. BMC Complement. Med. Ther. 2024, 24, 29. [Google Scholar] [CrossRef] [PubMed]
- Zuo, H.-J.; Wang, P.-X.; Ren, X.-Q.; Shi, H.-L.; Shi, J.-S.; Guo, T.; Wan, C.; Li, J.-J. Gastrodin Regulates PI3K/AKT-Sirt3 Signaling Pathway and Proinflammatory Mediators in Activated Microglia. Mol. Neurobiol. 2024, 61, 2728–2744. [Google Scholar] [CrossRef] [PubMed]
- Salah, M.M.; Ashour, A.A.; Abdelghany, T.M.; Abdel-Aziz, A.-A.H.; Salama, S.A. Pirfenidone alleviates concanavalin A-induced liver fibrosis in mice. Life Sci. 2019, 239, 116982. [Google Scholar] [CrossRef]
- Elazab, S.T.; Hsu, W.H. Ferulic acid ameliorates concanavalin A-induced hepatic fibrosis in mice via suppressing TGF-β/smad signaling. Toxicol. Appl. Pharmacol. 2024, 492, 117099. [Google Scholar] [CrossRef]
- Shay, J.W. Using Antibodies: A Laboratory Manual. In Vitro Cell. Dev. Biol. 1999, 35, 425. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]








| Gene | Probe |
|---|---|
| SIRT3 | 5-TGCACGGTCTGTCGAAGGTC-3 5-A TGTCAGGTTTCACAACGCCAGTA-3 |
| PI3K | 5-AACACAGAAGACCAATACTC-3 5-TTCGCCATCTACCACTAC-3 |
| Akt | 5-GTGGCAAGATGTGTATGAG-3 5-CTGGCTGAGTAGGAGAAC-3 |
| b-actin | 5-CCT GGAGAAGAGCTATGAGC-3 5-A CAGGATTCCATACCCAGG-3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Alrawili, H.M.; Elshal, M.; Serrya, M.S.; El-Agamy, D.S. SIRT3, NF-κB/TNF-α and PI3K/Akt Pathways Mediate the Hepatoprotective Activity of Gossypin Against Concanavalin A-Induced Hepatic Fibrosis. Toxins 2026, 18, 74. https://doi.org/10.3390/toxins18020074
Alrawili HM, Elshal M, Serrya MS, El-Agamy DS. SIRT3, NF-κB/TNF-α and PI3K/Akt Pathways Mediate the Hepatoprotective Activity of Gossypin Against Concanavalin A-Induced Hepatic Fibrosis. Toxins. 2026; 18(2):74. https://doi.org/10.3390/toxins18020074
Chicago/Turabian StyleAlrawili, Hani M., Mahmoud Elshal, Marwa S. Serrya, and Dina S. El-Agamy. 2026. "SIRT3, NF-κB/TNF-α and PI3K/Akt Pathways Mediate the Hepatoprotective Activity of Gossypin Against Concanavalin A-Induced Hepatic Fibrosis" Toxins 18, no. 2: 74. https://doi.org/10.3390/toxins18020074
APA StyleAlrawili, H. M., Elshal, M., Serrya, M. S., & El-Agamy, D. S. (2026). SIRT3, NF-κB/TNF-α and PI3K/Akt Pathways Mediate the Hepatoprotective Activity of Gossypin Against Concanavalin A-Induced Hepatic Fibrosis. Toxins, 18(2), 74. https://doi.org/10.3390/toxins18020074