T-2 and HT-2 Toxins: Toxicity, Occurrence and Analysis: A Review
Abstract
1. Introduction
2. Toxicity of T-2 and HT-2 Toxins
2.1. Toxic Effects in Animals
2.2. Toxic Effects in Humans
3. EU/UK Regulations for T-2 and HT-2 Toxins
4. T-2 and HT-2 Production

5. Worldwide Occurrence of T-2 and HT-2 in Oats and Barley
| Region | Year | Commodity | Number of Samples | Positive Samples (%) | Average (µg/kg) | Range (µg/kg) | Reference |
|---|---|---|---|---|---|---|---|
| Egypt | 2021 | Oats | 10 | 70 | 35.4 | 14.3–74.4 | [93] |
| Canada | 2016–2018 | Oats | 168 | 81 | 39.0 | 10–1155.2 | [81] |
| Croatia | 2017–2018 | Oats | 30 | 70 | 87.9 | 9.5–21.8 | [94] |
| Croatia | 2017 | Oats | 6 | 75 | 69.0 | 23–142 | [95] |
| Europe | 2013–2019 | Oats | 281 | 98 | 103.1 | 5.1–1000 | [56] |
| Lithuania | 2010-2018 | Oats | 72 | - | 182 | - | [96] |
| Finland | 2005–2006 | Oats | 804 | 100 | 348 | 25–17,451 | [90] |
| Norway | 2004–2009 | Oats | 289 | 76 | 105.2 | 10.2–658.1 | [89] |
| Sweden | 2004–2018 | Oats | 164 | - | 66.0 | - | [77] |
| UK | 2006–2008 | Oats | 303 | 85 | 450 | 10–8399 | [84] |
| Switzerland | 2013–2015 | Oats | 325 | 76 | 225.5 | 10.1–3789 | [88] |
| Ireland | 2015–2016 | Oats | 208 | 51 | 256 | 53–3405 | [78] |
| Ireland | 2020 | Oats | 202 | 62 | 138 | 5–3064 | [79] |
| Hungary | 2014–2015 | Oats | 29 | 10 | 56.0 | 50.1–69.2 | [97] |
| Scotland | 2019 | Oats | 33 | 91 | 574.4 | Nd—3474 | [85] |
| Lithuania | 2015–2018 | Oats | 62 | 100 | - | 15.7–594.6 | [98] |
| Czechia | 2022 | Oats | 52 | 92 | 6 (T-2) 27 (HT-2) | <0.2–31 (T-2) <0.2–128 (HT-2) | [99] |
| Croatia | 2017–2018 | Barley | 66 | 41 | 22.6 | 12.2–52.1 | [94] |
| Croatia | 2017 | Barley | 7 | 14.3 | 3.0 | - | [95] |
| Czechia | 2018 | Barley | 152 | - | 107.7 | 48–251.3 | [100] |
| Sweden | 2004–2018 | Barley | 177 | - | 21.0 | - | [77] |
| Czechia | 2012–2017 | Barley | 117 | 20 | - | 11.8–199.0 | [101] |
| Italy | 2011–2014 | Barley | 691 | 32 | 127.8 | 26.0–787.0 | [102] |
| Hungary | 2014–2015 | Barley | 29 | 14 | 58.0 | 52.0–79.0 | [97] |
6. Impact of Climate Change on T-2 and HT-2 Production
7. Analytical Methods
7.1. Extraction Methodologies
7.2. Sample Clean-Up
7.2.1. Solid-Phase Extraction
7.2.2. Immunoaffinity Columns
7.2.3. QuEChERS
7.2.4. Other Sample Clean-Up Techniques
7.3. Analysis
7.3.1. Rapid Diagnostics
7.3.2. Confirmatory Analysis
8. Discussion
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From Simple to Complex Mycotoxins. Toxins 2011, 3, 802–814. [Google Scholar] [CrossRef]
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- CAST. Mycotoxins: Risks in Plant, Animal and Human Systems; Task Force Report, No. 139; The Council for Agricultural Science and Technology Agricultural Science: Ames, IA, USA, 2003; Available online: https://www.cast-science.org/publication/mycotoxins-risks-in-plant-animal-and-human-systems/ (accessed on 5 April 2023).
- EC. Mycotoxins in Human Nutrition and Health; Agro-Industrial Research Division. Directorate-General XII for Scientific Research and Development: Luxembourg, 1994. [Google Scholar]
- Desjardins, A.E.; Hohn, T.M.; McCormick, S.P. Trichothecene biosynthesis in Fusarium species: Chemistry, genetics, and significance. Microbiol. Rev. 1993, 57, 595–604. [Google Scholar] [CrossRef] [PubMed]
- Plattner, R.D.; Tjarks, L.W.; Beremand, M.N. Trichothecenes accumulated in liquid culture of a mutant of Fusarium sporotrichioides NRRL 3299. Appl. Environ. Microbiol. 1989, 55, 2190–2194. [Google Scholar] [CrossRef] [PubMed]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Wannemacher, R.W.J.; Wiener, S.L. Trichothecene Mycotoxins; Zajtchuk, R., Bellamy, R., Eds., Eds.; Office of the Surgeon General at TMM Publications, Borden Institute, Walter Reed Army Medical Center: Washington, DC, USA, 1997. [Google Scholar]
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins; International Agency for Research on Cancer: Lyon, France, 1993; Volume 56, Available online: https://monographs.iarc.who.int/wp-content/uploads/2018/06/mono56.pdf (accessed on 13 January 2022).
- Adhikari, M.; Negi, B.; Kaushik, N.; Adhikari, A.; Al-Khedhairy, A.A.; Kaushik, N.K.; Choi, E.H. T-2 mycotoxin: Toxicological effects and decontamination strategies. Oncotarget 2017, 8, 33933–33952. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Jouany, J.P. Mycotoxins in feeds for ruminants; fate and effects on animals. Prod. Anim. 2002, 15, 3–16. [Google Scholar]
- Čonková, E.; Laciaková, A.; Kováč, G.; Seidel, H. Fusarial Toxins and their Role in Animal Diseases. Vet. J. 2003, 165, 214–220. [Google Scholar] [CrossRef]
- Bondy, G.S.; Pestka, J.J. Immunomodulation by fungal toxins. J. Toxicol. Environ. Health Part B Crit. Rev. 2000, 3, 109–143. [Google Scholar] [CrossRef]
- Diaz, D.E. The Mycotoxin Blue Book; Nottingham University Press: Nottingham, UK, 2005. [Google Scholar]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on the risks for animal and public health related to the presence of T-2 and HT-2 toxin in food and feed. EFSA J. 2011, 9, 2481. [Google Scholar] [CrossRef]
- Schrenk, D.; Bignami, M.; Bodin, L.; Chipman, J.K.; del Mazo, J.; Grasl-Kraupp, B.; Hogstrand, C.; Leblanc, J.C.; Nielsen, E.; Ntzani, E.; et al. Assessment of information as regards the toxicity of T-2 and HT-2 toxin for ruminants. EFSA J. 2022, 20, e07564. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.A.; Wobeser, G.A. Subacute toxic effects of dietary T-2 toxin in young mallard ducks. Can. J. Comp. Med. 1983, 47, 180–187. [Google Scholar]
- Murugesan, G.R.; Ledoux, D.R.; Naehrer, K.; Berthiller, F.; Applegate, T.J.; Grenier, B.; Phillips, T.D.; Schatzmayr, G. Prevalence and effects of mycotoxins on poultry health and performance, and recent development in mycotoxin counteracting strategies. Poult. Sci. 2015, 94, 1298–1315. [Google Scholar] [CrossRef]
- Wyatt, R.D.; Hamilton, P.B.; Burmeister, H.R. Altered feathering of chicks caused by T-2 toxin. Poult. Sci. 1975, 54, 1042–1045. [Google Scholar] [CrossRef] [PubMed]
- Raju, M.V.L.N.; Devegowda, G. Influence of esterified-glucomannan on performance and organ morphology, serum biochemistry and haematology in broilers exposed to individual and combined mycotoxicosis (aflatoxin, ochratoxin and T-2 toxin). Br. Poult. Sci. 2000, 41, 640–650. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Yu, Z.; Hou, J.; Deng, Y.; Zhou, Z.; Zhao, Z.; Cui, J. Toxicity and oxidative stress induced by T-2 toxin and HT-2 toxin in broilers and broiler hepatocytes. Food Chem. Toxicol. 2016, 87, 128–137. [Google Scholar] [CrossRef]
- Chi, M.S.; Mirocha, C.J.; Kurtz, H.F.; Weaver, G.; Bates, F.; Shimoda, W. Effects of T-2 toxin on reproductive performance and health of laying hens. Poult. Sci. 1977, 56, 628–637. [Google Scholar] [CrossRef]
- Diaz, G.J.; Julian, R.J.; Squires, E.J.; Boermans, H.J. Individual and combined effects of T-2 toxin and das in laying hens. Br. Poult. Sci. 1994, 35, 393–405. [Google Scholar] [CrossRef]
- Pinton, P.; Guzylack-Piriou, L.; Kolf-Clauw, M.; Oswald, I.P. The effect on the intestine of some fungal toxins: The trichothecenes. Curr. Immunol. Rev. 2012, 8, 193–208. [Google Scholar] [CrossRef]
- Kanora, A.; Maes, D. The role of mycotoxins in pig reproduction: A review. Vet. Med. 2009, 54, 565–576. [Google Scholar] [CrossRef]
- Harvey, R.B.; Kubena, L.F.; Elissalde, M.H.; Rottinghaus, G.E.; Corrier, D.E. Administration of ochratoxin A and T-2 toxin to growing swine. Am. J. Vet. Res. 1994, 55, 1757–1761. [Google Scholar] [PubMed]
- Harvey, R.B.; Kubena, L.F.; Huff, W.E.; Corrier, D.E.; Rottinghaus, G.E.; Phillips, T.D. Effects of treatment of growing swine with aflatoxin and T-2 toxin. Am. J. Vet. Res. 1990, 51, 1688–1693. [Google Scholar] [PubMed]
- Rafai, P.; Bata, A.; Ványi, A.; Papp, Z.; Brydl, E.; Jakab, L.; Tuboly, S.; Túry, E. Effect of various levels of T-2 toxin on the clinical status, performance and metabolism of growing pigs. Vet. Rec. 1995, 136, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Rafai, P.; Tuboly, S.; Bata, A.; Tilly, P.; Ványi, A.; Papp, Z.; Jakab, L.; Túry, E. Effect of various levels of T-2 toxin in the immune system of growing pigs. Vet. Rec. 1995, 136, 511–514. [Google Scholar] [CrossRef]
- D’Mello, J.P.F.; Placinta, C.M.; Macdonald, A.M.C. Fusarium mycotoxins: A review of global implications for animal health, welfare and productivity. Anim. Feed. Sci. Technol. 1999, 80, 183–205. [Google Scholar] [CrossRef]
- Gonkowski, S.; Gajȩcka, M.; Makowska, K. Mycotoxins and the enteric nervous system. Toxins 2020, 12, 461. [Google Scholar] [CrossRef] [PubMed]
- Kemboi, D.C.; Antonissen, G.; Ochieng, P.E.; Croubels, S.; Okoth, S.; Kangethe, E.K.; Faas, J.; Lindahl, J.F.; Gathumbi, J.K. A review of the impact of mycotoxins on dairy cattle health: Challenges for food safety and dairy production in sub-Saharan Africa. Toxins 2020, 12, 222. [Google Scholar] [CrossRef] [PubMed]
- Ogunade, I.M.; Martinez-Tuppia, C.; Queiroz, O.C.M.; Jiang, Y.; Drouin, P.; Wu, F.; Vyas, D.; Adesogan, A.T. Silage review: Mycotoxins in silage: Occurrence, effects, prevention, and mitigation. J. Dairy Sci. 2018, 101, 4034–4059. [Google Scholar] [CrossRef]
- Gallo, A.; Giuberti, G.; Frisvad, J.C.; Bertuzzi, T.; Nielsen, K.F. Review on mycotoxin issues in ruminants: Occurrence in forages, effects of mycotoxin ingestion on health status and animal performance and practical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef]
- Mann, D.D.; Buening, G.M.; Hook, B.; Osweiler, G.D. Effects of T-2 mycotoxin on bovine serum proteins. Am. J. Vet. Res. 1983, 44, 1757–1759. [Google Scholar]
- Mann, D.D.; Buening, G.M.; Osweiler, G.D.; Hook, B.S. Effect of subclinical levels of T-2 toxin on the bovine cellular immune system. Can. J. Comp. Med. 1984, 48, 308–312. [Google Scholar] [PubMed]
- Buening, G.M.; Mann, D.D.; Hook, B.; Osweiler, G.D. The effect of T-2 toxin on the bovine immune system: Cellular factors. Vet. Immunol. Immunopathol. 1982, 3, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Placinta, C.M.; D’Mello, J.P.F.; MacDonald, A.M.C. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed. Sci. Technol. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- Cheeke, P.R. Natural Toxicants in Feeds, Forages, and Poisonous Plants., 2nd ed.; Cheeke, P.R., Ed.; Interstate Publishers, Inc.: Danville, IL, USA, 1998. [Google Scholar]
- Hsu, I.C.; Smalley, E.B.; Strong, F.M.; Ribelin, W.E. Identification of T-2 toxin in moldy corn associated with a lethal toxicosis in dairy cattle. Appl. Microbiol. 1972, 24, 684–690. [Google Scholar] [CrossRef]
- Fink-Gremmels, J. The role of mycotoxins in the health and performance of dairy cows. Vet. J. 2008, 176, 84–92. [Google Scholar] [CrossRef]
- Ferreras, M.C.; Benavides, J.; García-Pariente, C.; Delgado, L.; Fuertes, M.; Muñoz, M.; García-Marín, J.F.; Pérez, V. Acute and chronic disease associated with naturally occurring T-2 mycotoxicosis in sheep. J. Comp. Pathol. 2013, 148, 236–242. [Google Scholar] [CrossRef]
- Caloni, F.; Cortinovis, C. Effects of fusariotoxins in the equine species. Vet. J. 2010, 186, 157–161. [Google Scholar] [CrossRef]
- Rahman, S.; Sharma, A.K.; Singh, N.D.; Prawez, S. Immunopathological effects of experimental T-2 mycotoxicosis in Wistar rats. Hum. Exp. Toxicol. 2021, 40, 772–790. [Google Scholar] [CrossRef]
- Jaćević, V.; Wu, Q.; Nepovimova, E.; Kuča, K. Cardiomyopathy induced by T-2 toxin in rats. Food Chem. Toxicol. 2020, 137, 111138. [Google Scholar] [CrossRef]
- Zhang, J.; Jia, H.; Wang, Q.; Zhang, Y.; Wu, W.; Zhang, H. Role of peptide YY3-36 and glucose-dependent insulinotropic polypeptide in anorexia induction by trichothecences T-2 toxin, HT-2 toxin, diacetoxyscirpenol, and neosolaniol. Toxicol. Sci. 2017, 159, 203–210. [Google Scholar] [CrossRef]
- Matejova, I.; Svobodova, Z.; Vakula, J.; Mares, J.; Modra, H. Impact of Mycotoxins on Aquaculture Fish Species: A Review. J. World Aquac. Soc. 2017, 48, 186–200. [Google Scholar] [CrossRef]
- Peraica, M.; Radić, B.; Lucić, A.; Pavlović, M. Toxic effects of mycotoxins in humans. Bull. World Health Organ. 1999, 77, 754–766. [Google Scholar]
- Peraica, M.; Richter, D.; Rašić, D. Mycotoxicoses in children. Arh. Za Hig. Rada I Toksikol. 2014, 65, 347–363. [Google Scholar] [CrossRef]
- Li, D.; Han, J.; Guo, X.; Qu, C.; Yu, F.; Wu, X. The effects of T-2 toxin on the prevalence and development of Kashin-Beck disease in China: A meta-analysis and systematic review. Toxicol. Res. 2016, 5, 731–751. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Chen, S.; Zhang, F.; Liu, Y.; Chen, F.; Li, S.; Wang, C.; Wu, Y.; Gong, Y.; Hu, M.; et al. The alteration of urinary metabolomics profiles in Kashin-Beck disease in a three consecutive year study. Mol. Omics 2022, 19, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Tucker, J.B. The “yellow rain” controversy: Lessons for arms control compliance. Nonproliferation Rev. 2001, 8, 25–42. [Google Scholar] [CrossRef]
- EU. Commission Recommendation of 27 March 2013 on the Presence of T-2 and HT-2 Toxin in Cereals and Cereal Products (2013/165/EU). 2013. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2013:091:0012:0015:EN:PDF (accessed on 17 April 2023).
- Arcella, D.; Gergelova, P.; Innocenti, M.L.; Steinkellner, H. Human and animal dietary exposure to T-2 and HT-2 toxin. EFSA J. 2017, 15, e04972. [Google Scholar] [CrossRef]
- Agrolab. Current Status of Discussions on Limit Values for Mycotoxins in Food. 2020. Available online: https://www.agrolab.com/en/news/food-news/2697-limit-values-mycotoxins.html (accessed on 20 April 2023).
- Meyer, J.C.; Hennies, I.; Wessels, D.; Schwarz, K. Survey of mycotoxins in milling oats dedicated for food purposes between 2013 and 2019 by LC–MS/MS. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2021, 38, 1934–1947. [Google Scholar] [CrossRef]
- Goswami, R.S.; Kistler, H.C. Heading for disaster: Fusarium graminearum on cereal crops. Mol. Plant Pathol. 2004, 5, 515–525. [Google Scholar] [CrossRef]
- Martínez, M.; Ramirez Albuquerque, L.D.; Dinolfo, M.I.; Biganzoli, F.; F Pinto, V.; Stenglein, S.A. Effects of Fusarium graminearum and Fusarium poae on disease parameters, grain quality and mycotoxin contamination in barley (part II). J. Sci. Food Agric. 2020, 100, 3182–3191. [Google Scholar] [CrossRef]
- Munkvold, G.P. Epidemiology of Fusarium Diseases and their Mycotoxins in Maize Ears. Eur. J. Plant Pathol. 2003, 109, 705–713. [Google Scholar] [CrossRef]
- Stȩpień, Ł.; Chełkowski, J. Fusarium head blight of wheat: Pathogenic species and their mycotoxins. World Mycotoxin J. 2010, 3, 107–119. [Google Scholar] [CrossRef]
- Ferrigo, D.; Raiola, A.; Causin, R. Fusarium toxins in cereals: Occurrence, legislation, factors promoting the appearance and their management. Molecules 2016, 21, 627. [Google Scholar] [CrossRef]
- Ismaiel, A.A.; Papenbrock, J. Mycotoxins: Producing fungi and mechanisms of phytotoxicity. Agriculture 2015, 5, 493–537. [Google Scholar] [CrossRef]
- Proctor, R.H.; McCormick, S.P.; Kim, H.-S.; Cardoza, R.E.; Stanley, A.M.; Lindo, L.; Kelly, A.; Brown, D.W.; Lee, T.; Vaughan, M.M.; et al. Evolution of structural diversity of trichothecenes, a family of toxins produced by plant pathogenic and entomopathogenic fungi. PLoS Pathog. 2018, 14, e1006946. [Google Scholar] [CrossRef] [PubMed]
- Mousavi Khaneghah, A.; Farhadi, A.; Nematollahi, A.; Vasseghian, Y.; Fakhri, Y. A systematic review and meta-analysis to investigate the concentration and prevalence of trichothecenes in the cereal-based food. Trends Food Sci. Technol. 2020, 102, 193–202. [Google Scholar] [CrossRef]
- Chen, Y.; Kistler, H.C.; Ma, Z. Fusarium graminearum Trichothecene Mycotoxins: Biosynthesis, Regulation, and Management. Annu. Rev. Phytopathol. 2019, 57, 15–39. [Google Scholar] [CrossRef]
- Suzuki, T.; Iwahashi, Y. Comprehensive Gene Expression Analysis of Type B Trichothecenes. J. Agric. Food Chem. 2012, 60, 9519–9527. [Google Scholar] [CrossRef]
- Kimura, M.; Tokai, T.; Takahashi-Ando, N.; Ohsato, S.; Fujimura, M. Molecular and genetic studies of Fusarium trichothecene biosynthesis: Pathways, genes, and evolution. Biosci. Biotechnol. Biochem. 2007, 71, 2105–2123. [Google Scholar] [CrossRef]
- Cardoza, R.E.; Malmierca, M.G.; Hermosa, M.R.; Alexander, N.J.; McCormick, S.P.; Proctor, R.H.; Tijerino, A.M.; Rumbero, A.; Monte, E.; Gutiérrez, S. Identification of loci and functional characterization of trichothecene biosynthesis genes in filamentous fungi of the genus Trichoderma. Appl. Environ. Microbiol. 2011, 77, 4867–4877. [Google Scholar] [CrossRef]
- Lee, T.; Han, Y.K.; Kim, K.H.; Yun, S.H.; Lee, Y.W. Tri13 and tri7 determine deoxynivalenol- and nivalenol-producing chemotypes of Gibberella zeae. Appl. Environ. Microbiol. 2002, 68, 2148–2154. [Google Scholar] [CrossRef]
- Lee, T.; Oh, D.W.; Kim, H.S.; Lee, J.; Kim, Y.H.; Yun, S.H.; Lee, Y.W. Identification of Deoxynivalenol- and Nivalenol-Producing Chemotypes of Gibberella zeae by Using PCR. Appl. Environ. Microbiol. 2001, 67, 2966–2972. [Google Scholar] [CrossRef] [PubMed]
- Alexander, N.J.; McCormick, S.P.; Waalwijk, C.; van der Lee, T.; Proctor, R.H. The genetic basis for 3-ADON and 15-ADON trichothecene chemotypes in Fusarium. Fungal Genet. Biol. 2011, 48, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Mahato, D.K.; Pandhi, S.; Kamle, M.; Gupta, A.; Sharma, B.; Panda, B.K.; Srivastava, S.; Kumar, M.; Selvakumar, R.; Pandey, A.K.; et al. Trichothecenes in food and feed: Occurrence, impact on human health and their detection and management strategies. Toxicon 2022, 208, 62–77. [Google Scholar] [CrossRef] [PubMed]
- Peplow, A.W.; Meek, I.B.; Wiles, M.C.; Phillips, T.D.; Beremand, M.N. Tri16 Is Required for Esterification of Position C-8 during Trichothecene Mycotoxin Production by Fusarium sporotrichioides. Appl. Environ. Microbiol. 2003, 69, 5935–5940. [Google Scholar] [CrossRef]
- Ramos-Diaz, J.M.; Sulyok, M.; Jacobsen, S.E.; Jouppila, K.; Nathanail, A.V. Comparative study of mycotoxin occurrence in Andean and cereal grains cultivated in South America and North Europe. Food Control 2021, 130, 108260. [Google Scholar] [CrossRef]
- Karlsson, I.; Mellqvist, E.; Persson, P. Temporal and spatial dynamics of Fusarium spp. and mycotoxins in Swedish cereals during 16 years. Mycotoxin Res. 2023, 39, 3–18. [Google Scholar] [CrossRef]
- De Colli, L.; De Ruyck, K.; Abdallah, M.F.; Finnan, J.; Mullins, E.; Kildea, S.; Spink, J.; Elliott, C.; Danaher, M. Natural Co-Occurrence of Multiple Mycotoxins in Unprocessed Oats Grown in Ireland with Various Production Systems. Toxins 2021, 13, 188. [Google Scholar] [CrossRef]
- Kolawole, O.; De Ruyck, K.; Greer, B.; Meneely, J.; Doohan, F.; Danaher, M.; Elliott, C. Agronomic Factors Influencing the Scale of Fusarium Mycotoxin contamination of Oats. J. Fungi 2021, 7, 965. [Google Scholar] [CrossRef]
- Van Der Fels-Klerx, H.; Stratakou, I. T-2 toxin and HT-2 toxin in grain and grain-based commodities in Europe: Occurrence, factors affecting occurrence, co-occurrence and toxicological effects. World Mycotoxin J. 2010, 3, 349–367. [Google Scholar] [CrossRef]
- Islam, M.N.; Tabassum, M.; Banik, M.; Daayf, F.; Fernando, W.G.D.; Harris, L.J.; Sura, S.; Wang, X. Naturally Occurring Fusarium Species and Mycotoxins in Oat Grains from Manitoba, Canada. Toxins 2021, 13, 670. [Google Scholar] [CrossRef]
- Imathiu, S.M.; Ray, R.V.; Back, M.; Hare, M.C.; Edwards, S.G. Agronomic practices influence the infection of an oats cultivar with Fusarium langsethiae. Acta Phytopathol. Et Entomol. Hung. 2017, 52, 15–28. [Google Scholar] [CrossRef]
- Martin, C.; Schöneberg, T.; Vogelgsang, S.; Mendes Ferreira, C.S.; Morisoli, R.; Bertossa, M.; Bucheli, T.D.; Mauch-Mani, B.; Mascher, F. Responses of oat grains to Fusarium poae and F. langsethiae infections and mycotoxin contaminations. Toxins 2018, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Edwards, S.G. Impact of agronomic and climatic factors on the mycotoxin content of harvested oats in the United Kingdom. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2017, 34, 2230–2241. [Google Scholar] [CrossRef] [PubMed]
- Daud, N.; Currie, V.; Duncan, G.; Filipe, J.A.N.; Yoshinari, T.; Stoddart, G.; Roberts, D.; Gratz, S.W. Free and Modified Mycotoxins in Organic and Conventional Oats (Avena sativa L.) Grown in Scotland. Toxins 2023, 15, 247. [Google Scholar] [CrossRef] [PubMed]
- Gavrilova, O.P.; Gagkaeva, T.Y.; Orina, A.S.; Gogina, N.N. Diversity of Fusarium Species and Their Mycotoxins in Cereal Crops from the Asian Territory of Russia. Dokl. Biol. Sci. 2023, 508, 9–19. [Google Scholar] [CrossRef]
- Edwards, S.G. Fusarium mycotoxin content of UK organic and conventional oats. Food Addit. Contam. Part A 2009, 26, 1063–1069. [Google Scholar] [CrossRef]
- Schöneberg, T.; Jenny, E.; Wettstein, F.E.; Bucheli, T.D.; Mascher, F.; Bertossa, M.; Musa, T.; Seifert, K.; Gräfenhan, T.; Keller, B.; et al. Occurrence of Fusarium species and mycotoxins in Swiss oats—Impact of cropping factors. Eur. J. Agron. 2018, 92, 123–132. [Google Scholar] [CrossRef]
- Hofgaard, I.S.; Aamot, H.U.; Torp, T.; Jestoi, M.; Lattanzio, V.M.T.; Klemsdal, S.S.; Waalwijk, C.; Van Der Lee, T.; Brodal, G. Associations between Fusarium species and mycotoxins in oats and spring wheat from farmers fields in Norway over a six-year period. World Mycotoxin J. 2016, 9, 365–378. [Google Scholar] [CrossRef]
- Hietaniemi, V.; Rämö, S.; Yli-Mattila, T.; Jestoi, M.; Peltonen, S.; Kartio, M.; Sieviläinen, E.; Koivisto, T.; Parikka, P. Updated survey of Fusarium species and toxins in Finnish cereal grains. Food Addit. Contam. Part A 2016, 33, 831–848. [Google Scholar] [CrossRef]
- Fredlund, E.; Gidlund, A.; Sulyok, M.; Börjesson, T.; Krska, R.; Olsen, M.; Lindblad, M. Deoxynivalenol and other selected fusarium toxins in swedish oats—Occurrence and correlation to specific fusarium species. Int. J. Food Microbiol. 2013, 167, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Xue, A.G.; Chen, Y.; Seifert, K.; Guo, W.; Blackwell, B.A.; Harris, L.J.; Overy, D.P. Prevalence of Fusarium species causing head blight of spring wheat, barley and oat in Ontario during 2001–2017. Can. J. Plant Pathol. 2019, 41, 392–402. [Google Scholar] [CrossRef]
- Tahoun, I.F.; Gab-Allah, M.A.; Yamani, R.N.; Shehata, A.B. Development and validation of a reliable LC-MS/MS method for simultaneous determination of deoxynivalenol and T-2 toxin in maize and oats. Microchem. J. 2021, 169, 106599. [Google Scholar] [CrossRef]
- Kiš, M.; Vulić, A.; Kudumija, N.; Šarkanj, B.; Jaki Tkalec, V.; Aladić, K.; Škrivanko, M.; Furmeg, S.; Pleadin, J. A Two-Year Occurrence of Fusarium T-2 and HT-2 Toxin in Croatian Cereals Relative of the Regional Weather. Toxins 2021, 13, 39. [Google Scholar] [CrossRef]
- Kovač, M.; Bulaić, M.; Nevistić, A.; Rot, T.; Babić, J.; Panjičko, M.; Kovač, T.; Šarkanj, B. Regulated Mycotoxin Occurrence and Co-Occurrence in Croatian Cereals. Toxins 2022, 14, 112. [Google Scholar] [CrossRef]
- Kochiieru, Y.; Mankevičienė, A.; Cesevičienė, J.; Semaškienė, R.; Dabkevičius, Z.; Janavičienė, S. The influence of harvesting time and meteorological conditions on the occurrence of Fusarium species and mycotoxin contamination of spring cereals. J. Sci. Food Agric. 2020, 100, 2999–3006. [Google Scholar] [CrossRef]
- Tima, H.; Brückner, A.; Mohácsi-Farkas, C.; Kiskó, G. Fusarium mycotoxins in cereals harvested from Hungarian fields. Food Addit. Contam. Part B Surveill. 2016, 9, 127–131. [Google Scholar] [CrossRef]
- Janavičienė, S.; Mankevičienė, A.; Kochiieru, Y.; Venslovas, E. T-2 and HT-2 toxins in harvested oat grains and their prevalence in whole grain flour during storage. Food Addit. Contam. Part A 2022, 39, 1284–1295. [Google Scholar] [CrossRef]
- Prusova, N.; Behner, A.; Dzuman, Z.; Hajslova, J.; Stranska, M. Conjugated type a trichothecenes in oat-based products: Occurrence data and estimation of the related risk. Food Control 2023, 143, 109281. [Google Scholar] [CrossRef]
- Pernica, M.; Kyralová, B.; Svoboda, Z.; Boško, R.; Brožková, I.; Česlová, L.; Benešová, K.; Červenka, L.; Běláková, S. Levels of T-2 toxin and its metabolites, and the occurrence of Fusarium fungi in spring barley in the Czech Republic. Food Microbiol. 2022, 102, 103875. [Google Scholar] [CrossRef]
- Svoboda, Z.M.; Benešová, K.R.; Běláková, S. The occurrence of the selected Fusarium mycotoxins in Czech malting barley, harvested in 2012–2017. Czech J. Food Sci. 2019, 37, 439–445. [Google Scholar] [CrossRef]
- Morcia, C.; Tumino, G.; Ghizzoni, R.; Badeck, F.; Lattanzio, V.; Pascale, M.; Terzi, V. Occurrence of Fusarium langsethiae and T-2 and HT-2 Toxins in Italian Malting Barley. Toxins 2016, 8, 247. [Google Scholar] [CrossRef]
- Medina, A.; Magan, N. Temperature and water activity effects on production of T-2 and HT-2 by Fusarium langsethiae strains from north European countries. Food Microbiol. 2011, 28, 392–398. [Google Scholar] [CrossRef]
- Xu, X.; Madden, L.V.; Edwards, S.G. Modeling the effects of environmental conditions on HT2 and T2 toxin accumulation in field oat grains. Phytopathology 2014, 104, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Kolawole, O.; Meneely, J.; Petchkongkaew, A.; Elliott, C. A review of mycotoxin biosynthetic pathways: Associated genes and their expressions under the influence of climatic factors. Fungal Biol. Rev. 2021, 37, 8–26. [Google Scholar] [CrossRef]
- Verheecke-Vaessen, C.; Garcia-Cela, E.; Lopez-Prieto, A.; Osk Jonsdottir, I.; Medina, A.; Magan, N. Water and temperature relations of Fusarium langsethiae strains and modelling of growth and T-2 and HT-2 mycotoxin production on oat-based matrices. Int. J. Food Microbiol. 2021, 348, 109203. [Google Scholar] [CrossRef]
- Edwards, S.G.; Imathiu, S.M.; Ray, R.V.; Back, M.; Hare, M.C. Molecular studies to identify the Fusarium species responsible for HT-2 and T-2 mycotoxins in UK oats. Int. J. Food Microbiol. 2012, 156, 168–175. [Google Scholar] [CrossRef]
- Benešová, K.; Boško, R.; Běláková, S.; Pluháčková, H.; Křápek, M.; Pernica, M.; Svoboda, Z. Natural contamination of Czech malting barley with mycotoxins in connection with climate variability. Food Control 2022, 140. [Google Scholar] [CrossRef]
- Edwards, S.G.; Stancic, T. Susceptibility of UK oat (Avena sativa) varieties to infection by Fusarium species and subsequent HT-2 and T-2 toxin contamination. Plant Breed. 2022. [CrossRef]
- Gil-Serna, J.; Patiño, B.; Verheecke-Vaessen, C.; Vázquez, C.; Medina, Á. Searching for the Fusarium spp. Which Are Responsible for Trichothecene Contamination in Oats Using Metataxonomy to Compare the Distribution of Toxigenic Species in Fields from Spain and the UK. Toxins 2022, 14, 592. [Google Scholar] [CrossRef]
- Kahla, A.; Verheecke-Vaessen, C.; Delpino-Deelias, M.; Gutierrez-Pozo, M.; Medina, A.; Magan, N.; Doohan, F. Acclimatisation of Fusarium langsethiae, F. poae and F. sporotrichioides to elevated CO2: Impact on fungal growth and mycotoxin production on oat-based media. Int. J. Food Microbiol. 2023, 394, 110176. [Google Scholar] [CrossRef]
- Zuo, Y.; Verheecke-Vaessen, C.; Molitor, C.; Medina, A.; Magan, N.; Mohareb, F. De novo genome assembly and functional annotation for Fusarium langsethiae. BMC Genom. 2022, 23, 158. [Google Scholar] [CrossRef]
- Magan, N.; Medina, A. Integrating gene expression, ecology and mycotoxin production by Fusarium and Aspergillus species in relation to interacting environmental factors. World Mycotoxin J. 2016, 9, 673–684. [Google Scholar] [CrossRef]
- Opoku, N.; Back, M.A.; Edwards, S.G. Susceptibility of cereal species to Fusarium langsethiae under identical field conditions. Eur. J. Plant Pathol. 2018, 150, 869–879. [Google Scholar] [CrossRef]
- Verheecke-Vaessen, C.; Lopez-Pietro, A.; Garcia-Cela, E.; Medina, A.; Magan, N. Intra-species variability in Fusarium langsethiae strains in growth and T-2/HT-2 mycotoxin production in response to climate change abiotic factors. World Mycotoxin J. 2022, 15, 27–34. [Google Scholar] [CrossRef]
- Hofgaard, I.S.; Brodal, G.; Almvik, M.; Lillemo, M.; Russenes, A.L.; Edwards, S.G.; Aamot, H.U. Different Resistance to DON versus HT2 + T2 Producers in Nordic Oat Varieties. Toxins 2022, 14, 313. [Google Scholar] [CrossRef] [PubMed]
- Whitaker, T.B. Sampling foods for mycotoxins. Food Addit. Contam. 2006, 23, 50–61. [Google Scholar] [CrossRef] [PubMed]
- EC. Commission Regulation (EC) No 401/2006 of 23 February 2006 Laying down the Methods of Sampling and Analysis for the Official Control of the Levels of Mycotoxins in Foodstuffs. 2006. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006R0401&from=EN (accessed on 2 December 2021).
- Meneely, J.P.; Ricci, F.; van Egmond, H.P.; Elliott, C.T. Current methods of analysis for the determination of trichothecene mycotoxins in food. TrAC—Trends Anal. Chem. 2011, 30, 192–203. [Google Scholar] [CrossRef]
- Yang, L.; Tu, D.; Wu, Y.; Liu, W.; Hu, Y.; Liu, T.; Tan, L.; Li, Y.; Lei, H.; Zhan, Y.; et al. Distribution and persistence of residual T-2 and HT-2 toxins from moldy feed in broiler chickens. Toxicon 2020, 178, 82–91. [Google Scholar] [CrossRef]
- Malachová, A.; Sulyok, M.; Beltrán, E.; Berthiller, F.; Krska, R. Optimization and validation of a quantitative liquid chromatography-tandem mass spectrometric method covering 295 bacterial and fungal metabolites including all regulated mycotoxins in four model food matrices. J. Chromatogr. A 2014, 1362, 145–156. [Google Scholar] [CrossRef]
- Seo, H.; Jang, S.; Jo, H.; Kim, H.; Lee, S.; Yun, H.; Jeong, M.; Moon, J.; Na, T.; Cho, H. Optimization of the quechers-based analytical method for investigation of 11 mycotoxin residues in feed ingredients and compound feeds. Toxins 2021, 13, 767. [Google Scholar] [CrossRef] [PubMed]
- Sulyok, M.; Stadler, D.; Steiner, D.; Krska, R. Validation of an LC-MS/MS-based dilute-and-shoot approach for the quantification of >500 mycotoxins and other secondary metabolites in food crops: Challenges and solutions. Anal. Bioanal. Chem. 2020, 412, 2607–2620. [Google Scholar] [CrossRef]
- Tamura, M.; Mochizuki, N.; Nagatomi, Y.; Harayama, K.; Toriba, A.; Hayakawa, K. A method for simultaneous determination of 20 fusarium toxins in cereals by high-resolution liquid chromatography-orbitrap mass spectrometry with a pentafluorophenyl column. Toxins 2015, 7, 1664–1682. [Google Scholar] [CrossRef] [PubMed]
- Arola, H.O.; Tullila, A.; Nathanail, A.V.; Nevanen, T.K. A simple and specific noncompetitive ELISA method for HT-2 toxin detection. Toxins 2017, 9, 145. [Google Scholar] [CrossRef]
- D’Agnello, P.; Vita, V.; Franchino, C.; Urbano, L.; Curiale, A.; Debegnach, F.; Iammarino, M.; Marchesani, G.; Chiaravalle, A.E.; De Pace, R. ELISA and UPLC/FLD as screening and confirmatory techniques for T-2/Ht-2 mycotoxin determination in cereals. Appl. Sci. 2021, 11, 1688. [Google Scholar] [CrossRef]
- Lehotay, S.J.; Sapozhnikova, Y.; Mol, H.G.J. Current issues involving screening and identification of chemical contaminants in foods by mass spectrometry. TrAC—Trends Anal. Chem. 2015, 69, 62–75. [Google Scholar] [CrossRef]
- Kim, H.; Baek, E.J.; Shin, B.G.; Kim, H.J.; Kim, J.E. In-house Validation of an Efficient and Rapid Procedure for the Simultaneous Determination and Monitoring of 23 Mycotoxins in Grains in Korea. Toxins 2022, 14, 457. [Google Scholar] [CrossRef]
- Nathanail, A.V.; Syvähuoko, J.; Malachová, A.; Jestoi, M.; Varga, E.; Michlmayr, H.; Adam, G.; Sieviläinen, E.; Berthiller, F.; Peltonen, K. Simultaneous determination of major type A and B trichothecenes, zearalenone and certain modified metabolites in Finnish cereal grains with a novel liquid chromatography-tandem mass spectrometric method. Anal. Bioanal. Chem. 2015, 407, 4745–4755. [Google Scholar] [CrossRef]
- De Girolamo, A.; Ciasca, B.; Pascale, M.; Lattanzio, V.M.T. Determination of Zearalenone and Trichothecenes, Including Deoxynivalenol and Its Acetylated Derivatives, Nivalenol, T-2 and HT-2 Toxins, in Wheat and Wheat Products by LC-MS/MS: A Collaborative Study. Toxins 2020, 12, 786. [Google Scholar] [CrossRef]
- Gottschalk, C.; Barthel, J.; Engelhardt, G.; Bauer, J.; Meyer, K. Occurrence of type a trichothecenes in conventionally and organically produced oats and oat products. Mol. Nutr. Food Res. 2007, 51, 1547–1553. [Google Scholar] [CrossRef]
- Klötzel, M.; Lauber, U.; Humpf, H.U. A new solid phase extraction clean-up method for the determination of 12 type A and B trichothecenes in cereals and cereal-based food by LC-MS/MS. Mol. Nutr. Food Res. 2006, 50, 261–269. [Google Scholar] [CrossRef]
- Miró-Abella, E.; Herrero, P.; Canela, N.; Arola, L.; Borrull, F.; Ras, R.; Fontanals, N. Determination of mycotoxins in plant-based beverages using QuEChERS and liquid chromatography–tandem mass spectrometry. Food Chem. 2017, 229, 366–372. [Google Scholar] [CrossRef]
- Tölgyesi, Á.; Kunsági, Z. Quantification of T-2 and HT-2 mycotoxins in cereals by liquid chromatography-multimode ionization-tandem mass spectrometry. Microchem. J. 2013, 106, 300–306. [Google Scholar] [CrossRef]
- Abdel-Rehim, M.; Pedersen-Bjergaard, S.; Abdel-Rehim, A.; Lucena, R.; Moein, M.M.; Cárdenas, S.; Miró, M. Microextraction approaches for bioanalytical applications: An overview. J. Chromatogr. A 2020, 1616, 460790. [Google Scholar] [CrossRef]
- García-Nicolás, M.; Arroyo-Manzanares, N.; Campillo, N.; Reyes-Palomo, C.; Sanz-Fernández, S.; Fenoll, J.; Rodríguez-Estévez, V.; Viñas, P. Use of polypyrrole ferrite microparticles and liquid chromatography-mass spectrometry for testing natural grass contamination by multiclass mycotoxins. Microchim. Acta 2023, 190, 178. [Google Scholar] [CrossRef] [PubMed]
- Di Marco Pisciottano, I.; Imperato, C.; Urbani, V.; Guadagnuolo, G.; Imbimbo, S.; De Crescenzo, M.; Soprano, V.; Esposito, M.; Gallo, P. T-2 and HT-2 toxins in feed and food from Southern Italy, determined by LC-MS/MS after immunoaffinity clean-up. Food Addit. Contam. Part B Surveill. 2020, 13, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V.M.T.; Ciasca, B.; Powers, S.; Visconti, A. Improved method for the simultaneous determination of aflatoxins, ochratoxin A and Fusarium toxins in cereals and derived products by liquid chromatography-tandem mass spectrometry after multi-toxin immunoaffinity clean up. J. Chromatogr. A 2014, 1354, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Mackay, N.; Marley, E.; Leeman, D.; Poplawski, C.; Donnelly, C. Analysis of Aflatoxins, Fumonisins, Deoxynivalenol, Ochratoxin A, Zearalenone, HT-2, and T-2 Toxins in Animal Feed by LC-MS/MS Using Cleanup with a Multi-Antibody Immunoaffinity Column. J. AOAC Int. 2022, 105, 1330–1340. [Google Scholar] [CrossRef] [PubMed]
- Pascale, M.; Panzarini, G.; Visconti, A. Determination of HT-2 and T-2 toxins in oats and wheat by ultra-performance liquid chromatography with photodiode array detection. Talanta 2012, 89, 231–236. [Google Scholar] [CrossRef]
- Trebstein, A.; Seefelder, W.; Lauber, U.; Humpf, H.U. Determination of T-2 and HT-2 toxins in cereals including oats after immunoaffinity cleanup by liquid chromatography and Fluoresc. detection. J. Agric. Food Chem. 2008, 56, 4968–4975. [Google Scholar] [CrossRef]
- Wilcox, J.; Donnelly, C.; Leeman, D.; Marley, E. The use of immunoaffinity columns connected in tandem for selective and cost-effective mycotoxin clean-up prior to multi-mycotoxin liquid chromatographic-tandem mass spectrometric analysis in food matrices. J. Chromatogr. A 2015, 1400, 91–97. [Google Scholar] [CrossRef]
- Annunziata, L.; Stramenga, A.; Visciano, P.; Schirone, M.; De Colli, L.; Colagrande, M.N.; Campana, G.; Scortichini, G. Simultaneous determination of aflatoxins, T-2 and HT-2 toxins, and fumonisins in cereal-derived products by QuEChERS extraction coupled with LC-MS/MS. Anal. Bioanal. Chem. 2017, 409, 5143–5155. [Google Scholar] [CrossRef] [PubMed]
- Gab-Allah, M.A.; Tahoun, I.F.; Yamani, R.N.; Rend, E.A.; Shehata, A.B. Eco-friendly and sensitive analytical method for determination of T-2 toxin and HT-2 toxin in cereal products using UPLC-MS/MS. J. Food Compos. Anal. 2022, 107, 104395. [Google Scholar] [CrossRef]
- González-Jartín, J.M.; Alfonso, A.; Sainz, M.J.; Vieytes, M.R.; Botana, L.M. Multi-detection method for mycotoxins with a modified QuEChERS extraction in feed and development of a simple detoxification procedure. Anim. Feed Sci. Technol. 2021, 272, 114745. [Google Scholar] [CrossRef]
- Nakhjavan, B.; Ahmed, N.S.; Khosravifard, M. Development of an improved method of sample extraction and quantitation of multi-mycotoxin in feed by LC-MS/MS. Toxins 2020, 12, 462. [Google Scholar] [CrossRef] [PubMed]
- Sulyok, M.; Berthiller, F.; Krska, R.; Schuhmacher, R. Development and validation of a liquid chromatography/tandem mass spectrometric method for the determination of 39 mycotoxins in wheat and maize. Rapid Commun. Mass Spectrom. 2006, 20, 2649–2659. [Google Scholar] [CrossRef] [PubMed]
- EC. Commission Decision of 12 August 2002 Implementing Council Directive 96/23/EC Concerning the Performance of Analytical Methods and the Interpretation of Results (2002/657/EC). 2002. Available online: https://op.europa.eu/en/publication-detail/-/publication/ed928116-a955-4a84-b10a-cf7a82bad858/language-en (accessed on 10 January 2022).
- Steiner, D.; Malachová, A.; Sulyok, M.; Krska, R. Challenges and future directions in LC-MS-based multiclass method development for the quantification of food contaminants. Anal. Bioanal. Chem. 2021, 413, 25–34. [Google Scholar] [CrossRef]
- Han, Q.; Sun, Y.; Ding, K.; Chen, X.; Han, T. Preparation of multitarget immunomagnetic beads based on metal–organic frameworks and their application in food samples. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2020, 1158, 122341. [Google Scholar] [CrossRef]
- Pereira, V.L.; Fernandes, J.O.; Cunha, S.C. Comparative assessment of three cleanup procedures after QuEChERS extraction for determination of trichothecenes (type A and type B) in processed cereal-based baby foods by GC-MS. Food Chem. 2015, 182, 143–149. [Google Scholar] [CrossRef]
- Zhang, L.; Lv, Q.; Zheng, Y.; Chen, X.; Kong, D.; Huang, W.; Liu, P.; Jiang, H.; Jiang, Y. A rapid and accurate method for screening T-2 toxin in food and feed using competitive AlphaLISA. FEMS Microbiol. Lett. 2021, 368, fnab029. [Google Scholar] [CrossRef]
- Maragos, C.M. Detection of T-2 Toxin in Wheat and Maize with a Portable Mass Spectrometer. Toxins 2023, 15, 222. [Google Scholar] [CrossRef] [PubMed]
- Lippolis, V.; Porricelli, A.C.R.; Mancini, E.; Ciasca, B.; Lattanzio, V.M.T.; De Girolamo, A.; Maragos, C.M.; McCormick, S.; Li, P.; Logrieco, A.F.; et al. Fluorescence polarization immunoassay for the determination of T-2 and HT-2 toxins and their glucosides in wheat. Toxins 2019, 11, 380. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Segarra-Fas, A.; Peters, J.; Zuilhof, H.; Van Beek, T.A.; Nielen, M.W.F. Multiplex surface plasmon resonance biosensing and its transferability towards imaging nanoplasmonics for detection of mycotoxins in barley. Analyst 2016, 141, 1307–1318. [Google Scholar] [CrossRef] [PubMed]
- Carballo, D.; Font, G.; Ferrer, E.; Berrada, H. Evaluation of mycotoxin residues on ready-to-eat food by chromatographic methods coupled to mass spectrometry in tandem. Toxins 2018, 10, 243. [Google Scholar] [CrossRef] [PubMed]
- De Colli, L.; Elliott, C.; Finnan, J.; Grant, J.; Arendt, E.K.; McCormick, S.P.; Danaher, M. Determination of 42 mycotoxins in oats using a mechanically assisted QuEChERS sample preparation and UHPLC-MS/MS detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2020, 1150, 122187. [Google Scholar] [CrossRef]
- Romera, D.; Mateo, E.M.; Mateo-Castro, R.; Gómez, J.V.; Gimeno-Adelantado, J.V.; Jiménez, M. Determination of multiple mycotoxins in feedstuffs by combined use of UPLC–MS/MS and UPLC–QTOF–MS. Food Chem. 2018, 267, 140–148. [Google Scholar] [CrossRef]
- Guan, X.; Feng, Y.; Suo, D.; Xiao, Z.; Wang, S.; Liang, Y.; Fan, X. Simultaneous Determination of 11 Mycotoxins in Maize via Multiple-Impurity Adsorption Combined with Liquid Chromatography-Tandem Mass Spectrometry. Foods 2022, 11, 3624. [Google Scholar] [CrossRef]
- Juan, C.; Mañes, J.; Juan-García, A.; Moltó, J.C. Multimycotoxin Analysis in Oat, Rice, Almond and Soy Beverages by Liquid Chromatography-Tandem Mass Spectrometry. Appl. Sci. 2022, 12, 3942. [Google Scholar] [CrossRef]
- Kovač, M.; Nevistić, A.; Kovač, T.; Babić, J.; Šarić, A.; Miličević, B.; Panjičko, M.; Šarkanj, B. Development and Validation of an UHPLC-MS/MS Method for the Simultaneous Determination of 11 EU-Regulated Mycotoxins in Selected Cereals. J. Fungi 2022, 8, 665. [Google Scholar] [CrossRef]
- Rakk, D.; Kukolya, J.; Škrbić, B.D.; Vágvölgyi, C.; Varga, M.; Szekeres, A. Advantages of Multiplexing Ability of the Orbitrap Mass Analyzer in the Multi-Mycotoxin Analysis. Toxins 2023, 15, 134. [Google Scholar] [CrossRef]
- Narváez, A.; Izzo, L.; Castaldo, L.; Lombardi, S.; Rodríguez-Carrasco, Y.; Ritieni, A. Multi-Mycotoxin Method Development Using Ultra-High Liquid Chromatography with Orbitrap High-Resolution Mass Spectrometry Detection in Breakfast Cereals from the Campania Region, Italy. Toxins 2023, 15, 148. [Google Scholar] [CrossRef] [PubMed]
- Krska, R.; Malachova, A.; Berthiller, F.; Van Egmond, H.P. Determination of T-2 and HT-2 toxins in food and feed: An update. World Mycotoxin J. 2014, 7, 131–142. [Google Scholar] [CrossRef]
- Nolan, P.; Auer, S.; Spehar, A.; Elliott, C.T.; Campbell, K. Current trends in rapid tests for mycotoxins. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2019, 36, 800–814. [Google Scholar] [CrossRef] [PubMed]
- Oplatowska-Stachowiak, M.; Kleintjens, T.; Sajic, N.; Haasnoot, W.; Campbell, K.; Elliott, C.T.; Salden, M. T-2 toxin/HT-2 toxin and ochratoxin A ELISAs development and in-house validation in food in accordance with the commission regulation (EU) no 519/2014. Toxins 2017, 9, 388. [Google Scholar] [CrossRef]
- Alldrick, A.J. Looking for the best compromise in rapid food mycotoxin tests: Speed, sensitivity, precision and accuracy. World Mycotoxin J. 2014, 7, 407–415. [Google Scholar] [CrossRef]
- Zachariasova, M.; Cuhra, P.; Hajslova, J. Cross-reactivity of rapid immunochemical methods for mycotoxins detection towards metabolites and masked mycotoxins: The current state of knowledge. World Mycotoxin J. 2014, 7, 449–464. [Google Scholar] [CrossRef]
- Aamot, H.U.; Hofgaard, I.S.; Brodal, G.; Elen, O.; Holen, B.; Klemsdal, S.S. Evaluation of rapid test kits for quantification of HT-2 and T-2 toxins in naturally contaminated oats. World Mycotoxin J. 2013, 6, 31–41. [Google Scholar] [CrossRef]
- Plotan, M.; Devlin, R.; Porter, J.; Benchikh, M.E.; RodRíguez, M.L.; McConnell, R.I.; Fitzgerald, S.P. The use of biochip array technology for rapid multimycotoxin screening. J. AOAC Int. 2016, 99, 878–889. [Google Scholar] [CrossRef]
- Sibanda, L.; McCallum, K.; Plotan, M.; Webb, S.; Snodgras, B.; Muenks, Q.; Porter, J.; Fitzgerald, P. Interlaboratory collaboration to determine the performance of the Randox food diagnostics biochip array technology for the simultaneous quantitative detection of seven mycotoxins in feed. World Mycotoxin J. 2022, 15, 241–250. [Google Scholar] [CrossRef]
- EC. Commission Regulation (EU) No 519/2014 of 16 May 2014 Amending Regulation (EC) No 401/2006 as Regards Methods of Sampling of Large Lots, Spices and Food Supplements, Performance Criteria for T-2, HT-2 Toxin and Citrinin and Screening Methods of Analysis. 2014. Available online: https://op.europa.eu/en/publication-detail/-/publication/5a9dcba3-dd8b-11e3-8cd4-01aa75ed71a1/language-en#:~:text=Home-,Commission%20Regulation%20 (accessed on 17 January 2023).
- Majdinasab, M.; Aissa, S.B.; Marty, J.L. Advances in colorimetric strategies for mycotoxins detection: Toward rapid industrial monitoring. Toxins 2021, 13, 13. [Google Scholar] [CrossRef]
- McNamee, S.E.; Bravin, F.; Rosar, G.; Elliott, C.T.; Campbell, K. Development of a nanoarray capable of the rapid and simultaneous detection of zearalenone, T2-toxin and fumonisin. Talanta 2017, 164, 368–376. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, J.; Mao, X.; Wu, Y.; Liu, G.; Song, L.; Li, Y.; Yang, J.; You, Y.; Cao, X. High-sensitivity chemiluminescent immunoassay investigation and application for the detection of T-2 toxin and major metabolite HT-2 toxin. J. Sci. Food Agric. 2017, 97, 818–822. [Google Scholar] [CrossRef]
- Foubert, A.; Beloglazova, N.V.; Gordienko, A.; Tessier, M.D.; Drijvers, E.; Hens, Z.; De Saeger, S. Development of a Rainbow Lateral Flow Immunoassay for the Simultaneous Detection of Four Mycotoxins. J. Agric. Food Chem. 2017, 65, 7121–7130. [Google Scholar] [CrossRef] [PubMed]
- Foubert, A.; Beloglazova, N.V.; De Saeger, S. Comparative study of colloidal gold and quantum dots as labels for multiplex screening tests for multi-mycotoxin detection. Anal. Chim. Acta 2017, 955, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Wolf, K.; Schweigert, F.J. Mycotoxin Analysis: A Focus on Rapid Methods. 2018. Available online: https://www.aflatoxinpartnership.org/sites/default/files/2018-10/Mycotoxin_Analytics_Rapid_Methods-PACA_23Feb2018_0.pdf (accessed on 17 January 2023).
- Bueno, D.; Istamboulie, G.; Muñoz, R.; Marty, J.L. Determination of Mycotoxins in Food: A Review of Bioanalytical to Analytical Methods. Appl. Spectrosc. Rev. 2015, 50, 728–774. [Google Scholar] [CrossRef]
- Meneely, J.P.; Quinn, J.G.; Flood, E.M.; Hajšlová, J.; Elliott, C.T. Simultaneous screening for T-2/HT-2 and deoxynivalenol in cereals using a surface plasmon resonance immunoassay. World Mycotoxin J. 2012, 5, 117–126. [Google Scholar] [CrossRef]
- Meneely, J.P.; Sulyok, M.; Baumgartner, S.; Krska, R.; Elliott, C.T. A rapid optical immunoassay for the screening of T-2 and HT-2 toxin in cereals and maize-based baby food. Talanta 2010, 81, 630–636. [Google Scholar] [CrossRef]
- Gupta, G.; Bhaskar, A.S.B.; Tripathi, B.K.; Pandey, P.; Boopathi, M.; Rao, P.V.L.; Singh, B.; Vijayaraghavan, R. Supersensitive detection of T-2 toxin by the in situ synthesized π-conjugated molecularly imprinted nanopatterns. An in situ investigation by surface plasmon resonance combined with electrochemistry. Biosens. Bioelectron. 2011, 26, 2534–2540. [Google Scholar] [CrossRef] [PubMed]
- Meneely, J.P.; Elliott, C.T. Rapid surface plasmon resonance immunoassays for the determination of mycotoxins in cereals and cereal-based food products. World Mycotoxin J. 2014, 7, 491–505. [Google Scholar] [CrossRef]
- Hossain, M.Z.; McCormick, S.P.; Maragos, C.M. An imaging surface plasmon resonance biosensor assay for the detection of t-2 toxin and masked t-2 toxin-3-glucoside in wheat. Toxins 2018, 10, 119. [Google Scholar] [CrossRef]
- Martinez, L.; He, L. Detection of Mycotoxins in Food Using Surface-Enhanced Raman Spectroscopy: A Review. ACS Appl. Bio Mater. 2021, 4, 295–310. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Tang, S.; Jin, Y.; Yang, C.; He, L.; Wang, J.; Chen, Y. Multiplex SERS-based lateral flow immunosensor for the detection of major mycotoxins in maize utilizing dual Raman labels and triple test lines. J. Hazard. Mater. 2020, 393, 122348. [Google Scholar] [CrossRef]
- Soleimany, F.; Jinap, S.; Faridah, A.; Khatib, A. A UPLC-MS/MS for simultaneous determination of aflatoxins, ochratoxin A, zearalenone, DON, fumonisins, T-2 toxin and HT-2 toxin, in cereals. Food Control 2012, 25, 647–653. [Google Scholar] [CrossRef]
- Donnelly, C.; Pollock, A.; Heidtmann, Y.; Marley, E. Development of an immunoaffinity column for the determination of T-2 and HT-2 toxins in cereals using liquid chromatography with fluorescence detection. ACS Symp. Ser. 2008, 1001, 276–284. [Google Scholar] [CrossRef]
- Ok, H.E.; Kang, Y.W.; Kim, M.; Chun, H.S. T-2 and HT-2 toxins in cereals and cereal-based products in South Korea. Food Addit. Contam. Part B Surveill. 2013, 6, 103–109. [Google Scholar] [CrossRef]
- Busman, M.; Maragos, C.M. Determination of T-2 and HT-2 toxins from maize by direct analysis in real time mass spectrometry. World Mycotoxin J. 2015, 8, 489–497. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Ciasca, B.; Terzi, V.; Ghizzoni, R.; McCormick, S.P.; Pascale, M. Study of the natural occurrence of T-2 and HT-2 toxins and their glucosyl derivatives from field barley to malt by high-resolution Orbitrap mass spectrometry. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 1647–1655. [Google Scholar] [CrossRef]
- Lattanzio, V.M.T.; Gatta, S.D.; Suman, M.; Visconti, A. Development and in-house validation of a robust and sensitive solid-phase extraction liquid chromatography/tandem mass spectrometry method for the quantitative determination of aflatoxins B1, B2, G 1, G2, ochratoxin A, deoxynivalenol, zearalenone, T-2 and HT-2 toxins in cereal-based foods. Rapid Commun. Mass Spectrom. 2011, 25, 1869–1880. [Google Scholar] [CrossRef]
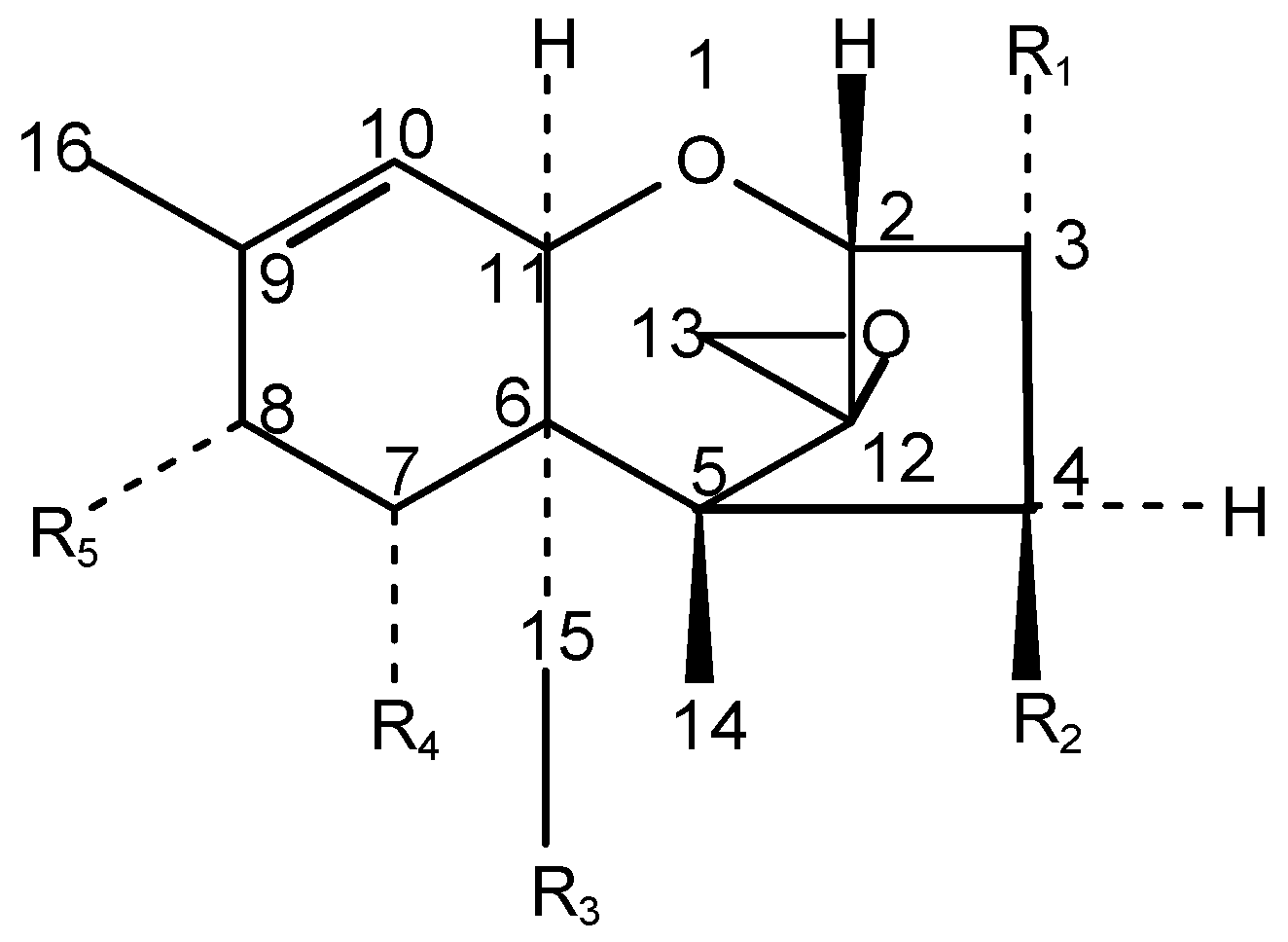
| Trichothecene | R1 | R2 | R3 | R4 | R5 |
|---|---|---|---|---|---|
| T-2 Toxin | -OH | -OCOCH3 | -OCOCH3 | -H | -OCOCH2CH(CH3)2 |
| HT-2 toxin | -OH | -OH | -OCOCH3 | -H | -OCOCH2CH(CH3)2 |
| Animal | LOAEL (µg T-2 Toxin/kg b.w. Per Day) |
|---|---|
| Pigs | 29 |
| Calves, lambs | 300 |
| Ruminants | Not identified |
| Poultry | 40 |
| Laying hens | 120 |
| Broiler chickens | 48 |
| Fattening ducks | 40 |
| Fattening turkeys | 48 |
| Rabbits | 200–500 |
| Catfish | 13 |
| Cats | Not identified |
| Horses | Not identified |
| Commodity | Indicative Levels for the Sum of T-2 and HT-2 (μg/kg) | Maximum Limits under Discussion |
|---|---|---|
| Unprocessed cereals: | ||
| Barley (including malting barley) and maize | 200 | 100 |
| Oats (with husk) | 1000 | 500 |
| Wheat, rye, other cereals | 100 | 50 |
| Cereals for direct human consumption: | ||
| Oats | 200 | 50 |
| Maize | 100 | 50 |
| Other cereals | 50 | 20 |
| Cereal products for human consumption: | ||
| Oat bran and flaked oats | 200 | 50 |
| Cereal bran except oat bran, oat milling products other than oat bran and flaked oats, and maize milling products | 100 | 50 |
| Other cereal milling products | 50 | 20 |
| Breakfast cereals including formed cereal flakes | 75 | 20 |
| Bread (including small bakery wares), pastries, biscuits, cereal snacks, pasta | 25 | 10 |
| Cereal-based foods for infants and young children | 15 | 10 |
| Cereal products for feed and compound feed: | ||
| Oat milling products (husks) | 2000 | |
| Other cereal products | 500 | |
| Compound feed, except feed for cats | 250 |
| Detection Platform Used | Matrices Analysed | Extraction and Clean-Up Method | Analyte(s) Included | LOD/LOQ [T-2/HT-2 Only] | Type of Analysis | Reference |
|---|---|---|---|---|---|---|
| LC-MS/MS: Shimadzu LC–MS 8050 triple-quadrupole MS equipped with a Nexera X2 UHPLC (Shimadzu, Kyoto City, Japan) | Feed ingredients and compound feed | QuEChERS: Extraction with 10 mL ACN, then 10 mL Water (10% formic) followed by dSPE: C18 and PSA | 11 mycotoxins including T-2 and HT-2 toxins | Not detailed | Confirmatory/quantitative | [121] |
| LC-HRMS: Ultimate 3000 LC coupled to a Q-Exactive™ Orbitrap MS (Thermo Fisher Scientific, Waltham, MA, USA) | Cereals (corn, wheat and barley) | QuEChERS + SPE: Extraction with Water:ACN (50:50, v/v) containing 2% formic acid, followed by QuEChERS + Multistep 229 Ochra Cartridge | 20 fusarium toxins including T-2 and HT-2 toxins | LOQ: 5 µg/kg for HT-2 and T-2 toxins | Confirmatory/quantitative | [123] |
| 1ELISA: Ridascreen T-2/HT-2 R3805 (R-Biopharma) with a Multiskan™ FC microplate photometer reader (85 and 100% cross-reactivity with antibody used, respectively) 2UPLC-FLD: Acquity UPLC H-Class Bio System coupled to a FLR detector (Waters, Milford, MA, USA) | Cereals (barley, malting barley, maize, oats, wheat and rye) | 1ELISA: Extraction buffer only 2IAC: Extraction with 90% MeOH(aq) | T-2 and HT-2 toxins | 1LOD: 75 µg/kg (sum of T-2/HT-2) 2LOQ: 29 and 19 µg/kg (T-2 and HT-2 respectively) | 1Screening/qualitative 2Confirmatory /quantitative | [125] |
| GC-MS: Agilent GC 6890 equipped with an inert 5973 N mass selective detector with EI ionisation | Processed cereal-based baby foods | QuEChERS: Extraction with 15 mL water, then 10 mL can followed by either: 1. dSPE: C18 and PSA 2. IAC 3. MultiSep 226 clean-up column (SPE) | 12 mycotoxins including T-2 and HT-2 toxins | LOD: 6.4 and 6.76 µg/kg (HT-2 and T-2 respectively) LOQ: 21.1 and 22.3 µg/kg (HT-2 and T-2 respectively) NB: Method 1 only | Screening/qualitative | [150] |
| UPLC-PDA: Acquity UPLC® system equipped with a PDA detector (Waters) | Oats and wheat | IAC: Extraction with 90% MeOH(aq) | T-2 and HT-2 toxins only | LOD: 8 µg/kg for both T-2 and HT-2 in both matrices | Confirmatory/quantitative | [139] |
| ELISA: Competitive AlphaLISA using a SpectraMax I3 Microplate reader (Molecular Devices) (No cross-reactivity with the antibody used specified) | Flour, cornmeal and formulated feed. | LLE: Extraction with 70% MeOH(aq) then dilution with 70% MeOH(aq) (No further clean-up) | T-2 and HT-2 toxins only | LOD: 0.03 ng/mL for T-2 and HT-2 toxins | Screening/qualitative | [151] |
| APCI-MS: TD-APCI-MS (Portable MS) | White wheat, red wheat, and yellow dent maize | SPE: Extraction with ACN:Water (84:16, v/v) then MycoSep 225 clean-up column | T-2 and HT-2 toxins only | LOD: 28 and 20 µg/kg for T-2 in wheat. LOD: 2.0, 1.5 and 0.9 mg/kg for HT-2 in wheat and maize respectively. | Screening/qualitative | [152] |
| FPIA: HT-2 specific antibody using a Sentry® 100 portable reader (Diachemix Corporation) (80% cross-reactivity for T-2 and both T-2/HT-2 glucosides) | Wheat | LLE: Extraction with MeOH:Water (9:1, v/v) or water only followed by filtration | T-2, HT-2 and their glucosides | LOD and LOQ: 10 and 15 µg/kg respectively for all analytes | Screening/qualitative | [153] |
| iSPR: Mult17mmuneimmuno-assay with nanostructured iSPR chips. Performed on a Biacore 3000 SPR instrument (GE Healthcare) (76% cross-reactivity for HT-2) | Barley | LLE: Extraction with 80% MeOH(aq) then dilution with HBS-EP buffer to 20% MeOH(aq) | T-2 and HT-2 toxins only (76% cross-reactivity for HT-2) | LOD: 26 µg/kg (T-2 only) | Screening/qualitative | [154] |
| GC-MS/MS: Agilent 7890A coupled. with an Agilent 7000A QqQ MS with inter electron-impact ion source LC-MS/MS: Agilent 1200 LC system coupled to a 3200 QTRAP® QqQ MS (ABSCIEX, Framingham, MA, USA) | Various commodities including: acereals, blegumes, cfish, dvegetables and emeats. | QuEChERS: Extraction with 10 mL water (2% formic) then 10 mL ACN, followed by QuEChERS (dSPE: C18) NB: Derivatisation of sample before GC-MS analysis | 26 mycotoxins including T-2 and HT-2 toxins | a, b, c, eLOD: 0.75 and 0.15 µg/kg (T-2 and HT-2 respectively) dLOD: 0.75 µg/kg for T-2 and HT-2 toxins | Confirmatory/quantitative | [155] |
| LC-MS/MS: 1100 series micro-LC (Agilent) coupled to a QTrap QqQ MS (ABSCIEX) | Cereals and cereal-based food | IAC: Extraction with water followed by MeOH. Samples filtered then diluted with PBS. | 26 mycotoxins including T-2 and HT-2 toxins | LOQ: 5 µg/kg for T-2 and HT-2 toxins | Confirmatory/quantitative | [137] |
| LC-HRMS: Exactive™ Orbitrap MS equipped with a heated ESI source (HESI II) coupled to an Accela HPLC system (Thermo Fisher, Waltham, MA, USA) | Barley including malt | SPE: Extraction with ACN:Water (84:16, v/v) then MycoSep 225 clean-up column | T-2, HT-2 and their glucosides (T2G and HT2G) | LOQ: 5 µg/kg for T-2 and HT-2 toxins; 0.1 µg/kg for T2G and HT2G | Confirmatory/quantitative | [137] |
| LC-MS/MS: API 3000 QqQ MS (Applied Biosystems) coupled to a model series 200 HPLC system (Perkin Elmer, Waltham, MA, USA) | Cereal-derived products | QuEChERS: Extraction with 10 mL of water (0.1% formic) then 10 mL ACN. (No dSPE used) | 8 mycotoxins including T-2 and HT-2 toxins | LOD: 1.3 µg/kg for T-2 and HT-2 toxins LOQ: 2.5 µg/kg for T-2 and HT-2 toxins | Confirmatory/quantitative | [142] |
| LC-MS/MS: Acquity UHPLC system coupled to a Waters Quattro Premier XE QqQ MS with ESI probe (Waters) | Oats | QuEChERS-based protocol: Extraction with 10 mL of a 1% aqueous acetic acid solution then 10 mL ACN. (No dSPE used) | 42 mycotoxins including T-2, HT-2, T2G and T2-3OH | LODs: 12.1 and 17.2 µg/kg (T-2 and HT-2 respectively) 4.1 and 6.5 µg/kg (T2-3OH and T2G respectively) | Confirmatory/quantitative | [78,156] |
| LC-MS/MS: A 1290 Infinity UHPLC system coupled to a 6460 QqQ MS (Agilent, Santa Clara, CA, USA) | Seven raw materials and eight animal feeds | QuEChERS-based protocol: Extraction with 10 mL of a 2% aqueous acetic acid solution then 10 mL ACN. (No dSPE used) | 22 mycotoxins including T-2 and HT-2 | LOQ: 2.7 and 14.3 µg/kg (T-2 and HT-2 toxins respectively) | Confirmatory/quantitative | [144] |
| LC-MS/MS: A 1290 Infinity UHPLC system coupled to a 6495 QqQ MS (Agilent) | Oats | SPE: Extraction with 20 mL ACN:Water:Acetic Acid (79:20:1, v/v/v), then SPE using a MycoSpinTM 400 SPE cartridge. | 16 mycotoxins including T-2 and HT-2 | LOQ: 5 µg/kg for T-2 and HT-2 toxins | Confirmatory/quantitative | [56] |
| 1LC-MS/MS: ACQUITY UPLC™ system coupled to an ACQUITY TQD tandem quadrupole MS 2LC-HRMS: ACQUITY UPLC™ system coupled to a Triple TOF 5600 System (AB SCIEX, Framingham, MA, USA) | Feed samples: swine, sheep, poultry, cattle, equine, aquaculture and feed materials | DnS: Extraction with 8 mL ACN:Water:Formic Acid (80:20:1, v/v/v) then filtered | 15 mycotoxins including T-2 and HT-2 | LOD: 12.5 µg/kg for T-2 and HT-2 toxins (matrix not specified) | 1Confirmatory/quantitative 2Screening | [157] |
| LC-MS/MS: QTRAP 6500+ ultra-HPLC-MS/MS instrument equipped with an ESI source (AB SCIEX) | Maize | Multiple-impurity adsorption purification (MIcan) Extraction with 70% ACN(aq) then MIA added to an aliquot for purification | 11 mycotoxins including T-2 and HT-2 | LOD: 0.2 and 0.8 µg/kg for T-2 and HT-2 toxins | Confirmatory/quantitative | [158] |
| LC-MS/MS: HPLC Nanospace SI-2 (Shieido, Tokyo, Japan) coupled to a QTRAP®4500 mass spectrometry system (SCIEX, Darmstadt, Germany) | Wheat, oat and barley | QuEChERS-based protocol: Extraction with 10 mL of distilled water then 10 mL of 5% formic acid in ACN (No dSPE used) | 23 mycotoxins including T-2 and HT-2 | LOD: T-2: 0.24 µg/kg for oats and barley HT-2: 0.16 µg/kg for barley and 0.09 µg/kg for oats. LOQ: T-2: 0.79 V and 0.81 V for barley and oats HT-2: 0.52 µg/kg and 0.31 µg/kg in barley and oats | Confirmatory/quantitative | [127] |
| LC-MS/MS: Agilent 1200 LC binary pump chromatograph and autosampler, coupled to 3200 QTRAP® AB SCIEX (Applied Biosystems, Foster City, CA, USA) | Oat, rice, soy and almond drinks | SPE: Extraction with 5 mL ACN then SPE using a STRATA® C18-E column. | 16 mycotoxins including T-2 and HT-2 | LOD (oat drink): T-2: 0.8 µg/kg HT-2: 1.1 µg/kg LOQ (oat drink): T-2: 4.2 µg/kg HT-2: 5.3 µg/kg | Confirmatory/quantitative | [159] |
| LC-MS/MS: UHPLC (Acquity H-Class, Waters, Milford, MA, USA) coupled to a triple quadruple mass spectrometer (XEVO TQD, Milford, MA, USA) | Maize, wheat and barley | DnS: Extraction with acetonitrile/water/formic acid (79:20:1, v/v/v) then a dilution using water/acetonitrile/formic acid (79:20:1, v/v/v) | 11 mycotoxins including T-2 and HT-2 | LOD (barley): T-2 and HT-2: 3 µg/kg LOQ (barley): T-2 and HT-2: 10 µg/kg | Confirmatory/quantitative | [160] |
| LC-MS/MS: Sciex QTRAP 4500 tandem quadrupole mass spectrometer coupled to an ExionLCTM AC LC system | Animal feed | IAC: Extraction with 20mL ACN–H2O– HCOOH (79:20:1, v/v/v) followed by IAC clean-up using 11+Myco MS-PREP® | 11 mycotoxins including T-2 and HT-2 | LOD: T-2 and HT-2: 0.2 µg/kg and 1 µg/kg LOQ: T-2 and HT-2: 0.7 µg/kg and 3 µg/kg | Confirmatory/quantitative | [138] |
| LC-HRMS: Dionex UltiMate 3000 UHPLC coupled to a Thermo Scientific Q-Exactive Plus Orbitrap mass spectrometer (Thermo Scientific, San Jose, CA, USA) | Corn and wheat | QuEChERS-based protocol: Extraction with 4 mL of MeCN:H2O:acetic acid (79:20:1, v/v/v) (No dSPE used) | 11 mycotoxins including T-2 and HT-2 | LOD (corn): T-2: 4.8 µg/kg HT-2: 43 µg/kg LOQ (corn): T-2: 10 µg/kg HT-2: 64 µg/kg LOD (wheat): T-2: 11 µg/kg HT-2: 14.5 µg/kg LOQ (wheat): T-2: 15 µg/kg HT-2: 20 µg/kg | Confirmatory/quantitative | [161] |
| LC-MS/MS: HPLC (Agilent Technologies) coupled to an Agilent G6410A triple quadrupole (QqQ) mass spectrometer | Grass | Dispersive magnetic solid-phase extraction (DMSPE): Extraction with 10 mL distilled water containing 2% m/v NaCl and 400 μL Fe3O4@ PPy microcomposite suspension | 13 mycotoxins including T-2 and HT-2 | LOD: T-2: 5.3 µg/kg HT-2: 11 µg/kg LOQ: T-2: 17 µg/kg HT-2: 37 µg/kg | Confirmatory/quantitative | [135] |
| LC-HRMS: Dionex UltiMate® 3000 system UPLC coupled to Q-Exactive Orbitrap | Breakfast cereals | Extraction with 10 mL of ACN containing 0.1% formic acid followed by salting out with NaCl | 24 mycotoxins including T-2 and HT-2 | LOQ: T-2: 0.39 µg/kg HT-2: 0.78 µg/kg | Confirmatory/quantitative | [162] |
| Manufacturer | Kit | Matrix | Analytical Method | Detection Method | Extraction Solvent | Limit of Detection (LOD) (µg/kg) | Antibody Cross-Reactivity Profile | Test Time (Incubation Following Sample Preparation) |
|---|---|---|---|---|---|---|---|---|
| Aokin AG (Berlin, Germany) | Aokin Mycontrol T2/HT2 | Oats, wheat, corn, other grains | FPIA | Quantitative | Methanol based SPE clean-up | Not detailed | Not detailed | 15 min |
| Charm Sciences Inc. (Lawrence, MA, USA) | ROSA T-2 and HT-2 Quantitative Test | Barley, corn, corn gluten meal, oat groats, sorghum soybean meal, wheat, wheat flour | LFD | Quantitative | 70% methanol | Not detailed | Not detailed | 10 min |
| Elabscience Inc. (Houston, TX, USA) | T-2(T-2 Toxin) Lateral Flow Assay Kit | Cereals, feed | LFD | Qualitative | Ethyl acetate | 10 µg/kg | Not detailed | 5 min |
| Elabscience Inc. (Houston, TX, USA) | T-2(T-2 Toxin) ELISA Kit | Beans, corn, oats, peanuts, feed | ELISA | Quantitative | 60% methanol | 0.05 µg/kg | Not detailed | 45 min |
| Elabscience Inc. (Houston, TX, USA) | T-2(T-2 Toxin) ELISA Kit | Cereals, feed | ELISA | Quantitative | 70% methanol | 1 µg/kg | T2: 100% ZEN: 59% HT2: <1% | 20 min |
| Envirologix Inc. (Portland, ME, USA) | QuickTox Kit for QuickScan T-2/HT-2 Flex | Corn | LFD | Quantitative | Extraction buffer | 25–50 µg/kg | AFB1: <1% DON: <1% FB1: <1% OTA: <1% ZEN: <1% | 5 min |
| Eurofins Tecna Laboratories (Luxembourg City, Luxembourg) | B ZERO T2 | Cereals, feed | ELISA | Quantitative | 70% methanol and 4% NaCl | 25 µg/kg Oats: 40 µg/kg | T2: 100% HT2: 72% | 20 min |
| Eurofins Tecna Laboratories (Luxembourg City, Luxembourg) | Celer T2 | Cereals, feed | ELISA | Quantitative | 70% methanol and 4% NaCl | 25 µg/kg Oats: 40 µg/kg | T2: 100% HT2: 72% DON: <1% | 20 min |
| Hygiena LLC (Camarillo, CA, USA) | Helica™ T-2 Toxin ELISA | Cereals, feed | ELISA | Quantitative | 70% methanol | 12.5 µg/kg | T2: 100% HT2: 3% | 30 min |
| Neogen Corporation (Lansing, MI, USA) | Reveal® Q+ MAX for T-2/HT-2 | Wheat, oats, corn | LFD | Quantitative | Aqueous extraction | 50 µg/kg | Not detailed | 5 min |
| Neogen Corporation (Lansing, MI, USA) | Reveal® Q+ for T-2/HT-2 | Corn, corn products | LFD | Quantitative | Water | 50 µg/kg | Not detailed | 6 min |
| Neogen Corporation (Lansing, MI, USA) | Veratox® for T-2/HT-2 | Barley, corn, corn flour, corn gluten, corn steep, DDGS wet cake, oats, oat hulls (whole), rice (brown), rice flour (white), rice gluten, rice hulls, rye, pea fibre, potato (white), soy, soybean meal, tapioca, wheat, wheat bran, wheat flour, wheat gluten | ELISA | Quantitative | 70% methanol | 25 µg/kg | T2: 100% HT2: 100% | 10 min |
| R-Biopharm AG (Darmstadt, Germany) | RIDA®QUICK T-2/HT-2 RQS ECO | Oats, wheat, corn | LFD | Quantitative | Aqueous extraction buffer | 50 µg/kg | Not detailed | 5 min |
| R-Biopharm AG (Darmstadt, Germany) | RIDASCREEN® T-2/HT-2 Toxin | Oats, corn, barley, wheat | ELISA | Quantitative | Water-based extraction | 12 µg/kg | T2: 100% HT2: 85% T2 Triol: <0.5% T2 Tetraol: <0.5% | 45 min |
| R-Biopharm AG (Darmstadt, Germany) | RIDASCREEN® T-2 Toxin | Cereals, feed | ELISA | Quantitative | 84% acetonitrile | 3.5–56 µg/kg | T2: 100% HT2: 7% Acetyl T2: <114% Iso T2: 2% | 90 min |
| R-Biopharm AG (Darmstadt, Germany) | RIDASCREEN®FAST T-2 Toxin | Cereals, feed | ELISA | Quantitative | 70% methanol | <20 µg/kg | Not detailed | 15 min |
| Romer Labs Diagnostic GmbH (Tulln, Austria) | AgraQuant® T-2 Toxin ELISA test | Grains, cereals, other commodities | ELISA | Quantitative | 70% methanol | 10 µg/kg | Not detailed | 15 min |
| Perkin Elmer Inc. (Waltham, MA, USA) | AuroFlow™ AQ T-2/HT-2 Strip Test | Corn, wheat | LFD | Quantitative | Water-based | 50 µg/kg | Not detailed | 5 min |
| MaxSignal® T-2 ELISA Kit | Milk, dried meat, dried fish, seed, feed, cereal | ELISA | Quantitative | Not detailed | 10 µg/kg | Not detailed | <30 min | |
| Vicam LP (Milford, MA, USA) | T2/HT2-V AQUA | Grains, feed | LFD | Quantitative | Water-based extraction | 10 µg/kg | Not detailed | 5 min |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meneely, J.; Greer, B.; Kolawole, O.; Elliott, C. T-2 and HT-2 Toxins: Toxicity, Occurrence and Analysis: A Review. Toxins 2023, 15, 481. https://doi.org/10.3390/toxins15080481
Meneely J, Greer B, Kolawole O, Elliott C. T-2 and HT-2 Toxins: Toxicity, Occurrence and Analysis: A Review. Toxins. 2023; 15(8):481. https://doi.org/10.3390/toxins15080481
Chicago/Turabian StyleMeneely, Julie, Brett Greer, Oluwatobi Kolawole, and Christopher Elliott. 2023. "T-2 and HT-2 Toxins: Toxicity, Occurrence and Analysis: A Review" Toxins 15, no. 8: 481. https://doi.org/10.3390/toxins15080481
APA StyleMeneely, J., Greer, B., Kolawole, O., & Elliott, C. (2023). T-2 and HT-2 Toxins: Toxicity, Occurrence and Analysis: A Review. Toxins, 15(8), 481. https://doi.org/10.3390/toxins15080481





