Botulinum Toxin Treatment for Cancer-Related Disorders: A Systematic Review
Abstract
1. Introduction
2. Search Design and Search Results
3. Results
4. Cancer-Related Pain
5. Pain Following Surgery and/or Radiation
6. Botulinum Toxin Therapy after Esophagectomy and for Gastroparesis
7. Esophageal Stricture (ES)
8. Parotidectomy and Parotid Radiation
9. First Bite Syndrome
10. Discussion
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rossetto, O.; Pirazzini, M.; Fabris, F.; Montecucco, C. Botulinum Neurotoxins: Mechanism of Action. Handb. Exp. Pharmacol. 2021, 263, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Pirazzini, M.; Montecucco, C.; Rossetto, O. Toxicology and pharmacology of botulinum and tetanus neurotoxins: An update. Arch. Toxicol. 2022, 96, 1521–1539. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, J. Botulinum toxin: State of the art. Mov. Disord. 2017, 32, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.A.; Schütz, S.G.; Simpson, D.M. Botulinum toxin for neuropathic pain and spasticity: An overview. Pain Manag. 2014, 4, 129–151. [Google Scholar] [CrossRef] [PubMed]
- Matak, I.; Tékus, V.; Bölcskei, K.; Lacković, Z.; Helyes, Z. Involvement of substance P in the antinociceptive effect of botulinum toxin type A: Evidence from knockout mice. Neuroscience 2017, 358, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lian, Y.; Zhang, H.; Xie, N.; Chen, Y. CGRP Plasma Levels Decrease in Classical Trigeminal Neuralgia Patients Treated with Botulinum Toxin Type A: A Pilot Study. Pain Med. 2020, 21, 1611–1615. [Google Scholar] [CrossRef]
- Lacković, Z. Botulinum Toxin and Pain. Handb. Exp. Pharmacol. 2021, 263, 251–264. [Google Scholar] [CrossRef]
- Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Aurora, S.K.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F. OnabotulinumtoxinA for treatment of chronic migraine: Pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache 2010, 50, 921–936. [Google Scholar] [CrossRef]
- Babcock, M.S.; Foster, L.; Pasquina, P.; Jabbari, B. Treatment of pain attributed to plantar fasciitis with botulinum toxin a: A short-term, randomized, placebo-controlled, double-blind study. Am. J. Phys. Med. Rehabil. 2005, 84, 649–654. [Google Scholar] [CrossRef]
- Yuan, R.Y.; Sheu, J.J.; Yu, J.M.; Chen, W.T.; Tseng, I.J.; Chang, H.H.; Hu, C.J. Botulinum toxin for diabetic neuropathic pain: A randomized double-blind crossover trial. Neurology 2009, 72, 1473–1478. [Google Scholar] [CrossRef]
- Xiao, L.; Mackey, S.; Hui, H.; Xong, D.; Zhang, Q.; Zhang, D. Subcutaneous injection of botulinum toxin a is beneficial in postherpetic neuralgia. Pain Med. 2010, 11, 1827–1833. [Google Scholar] [CrossRef] [PubMed]
- Attal, N.; de Andrade, D.C.; Adam, F.; Ranoux, D.; Teixeira, M.J.; Galhardoni, R.; Raicher, I.; Üçeyler, N.; Sommer, C.; Bouhassira, D. Safety and efficacy of repeated injections of botulinum toxin A in peripheral neuropathic pain (BOTNEP): A randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2016, 15, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Jabbari, B. Botulinum Toxin Treatment of Pain Disorders, 2nd ed.; Springer Nature: Berlin/Heidelberg, Germany, 2022. [Google Scholar]
- Kim, S.I.; Choi, Y. Botulinum Toxin Injection for Intractable Pain in Cancer Patients With Psoas Muscle Invasion. J. Pain Symptom Manag. 2022, 63, e441–e444. [Google Scholar] [CrossRef]
- Nam, K.E.; Kim, J.S.; Hong, B.Y.; Sul, B.; Choi, H.; Jun, S.Y.; Lim, S.H. Botulinum Toxin Type A Injection for Neuropathic Pain in a Patient With a Brain Tumor: A Case Report. Ann. Rehabil. Med. 2017, 41, 1088–1092. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mittal, S.O.; Jabbari, B. Botulinum Neurotoxins and Cancer-A Review of the Literature. Toxins 2020, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Van Daele, D.J.; Finnegan, E.M.; Rodnitzky, R.L.; Zhen, W.; McCulloch, T.M.; Hoffman, H.T. Head and neck muscle spasm after radiotherapy: Management with botulinum toxin A injection. Arch. Otolaryngol. Head Neck Surg. 2002, 128, 956–959. [Google Scholar] [CrossRef] [PubMed]
- Vasan, C.W.; Liu, W.C.; Klussmann, J.P.; Guntinas-Lichius, O. Botulinum toxin type A for the treatment of chronic neck pain after neck dissection. Head Neck 2004, 26, 39–45. [Google Scholar] [CrossRef]
- Wittekindt, C.; Liu, W.C.; Preuss, S.F.; Guntinas-Lichius, O. Botulinum toxin A for neuropathic pain after neck dissection: A dose-finding study. Laryngoscope 2006, 116, 1168–1171. [Google Scholar] [CrossRef]
- Hartl, D.M.; Cohen, M.; Juliéron, M.; Marandas, P.; Janot, F.; Bourhis, J. Botulinum toxin for radiation-induced facial pain and trismus. Otolaryngol. Head Neck Surg. 2008, 138, 459–463. [Google Scholar] [CrossRef]
- Stubblefield, M.D.; Levine, A.; Custodio, C.M.; Fitzpatrick, T. The role of botulinum toxin type A in the radiation fibrosis syndrome: A preliminary report. Arch. Phys. Med. Rehabil. 2008, 89, 417–421. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Kim, D.K.; Chiang, P.H.; Chancellor, M.B. Bladder botulinum toxin A injection can benefit patients with radiation and chemical cystitis. BJU Int. 2008, 102, 704–706. [Google Scholar] [CrossRef] [PubMed]
- Vuong, T.; Waschke, K.; Niazi, T.; Richard, C.; Parent, J.; Liberman, S.; Mayrand, S.; Loungnarath, R.; Stein, B.; Devic, S. The value of Botox-A in acute radiation proctitis: Results from a phase I/II study using a three-dimensional scoring system. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Mittal, S.; Machado, D.G.; Jabbari, B. OnabotulinumtoxinA for treatment of focal cancer pain after surgery and/or radiation. Pain Med. 2012, 13, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Bach, C.A.; Wagner, I.; Lachiver, X.; Baujat, B.; Chabolle, F. Botulinum toxin in the treatment of post-radiosurgical neck contracture in head and neck cancer: A novel approach. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2012, 129, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Rostami, R.; Mittal, S.O.; Radmand, R.; Jabbari, B. Incobotulinum Toxin-A Improves Post-Surgical and Post-Radiation Pain in Cancer Patients. Toxins 2016, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- De Groef, A.; Devoogdt, N.; Van Kampen, M.; Nevelsteen, I.; Smeets, A.; Neven, P.; Geraerts, I.; Dams, L.; Van der Gucht, E.; Debeer, P. Effectiveness of Botulinum Toxin A for Persistent Upper Limb Pain after Breast Cancer Treatment: A Double-Blinded Randomized Controlled Trial. Arch. Phys. Med. Rehabil. 2018, 99, 1342–1351. [Google Scholar] [CrossRef] [PubMed]
- Mailly, M.; Benzakin, S.; Chauvin, A.; Brasnu, D.; Ayache, D. Douleurs post-radiques après radiothérapie pour cancer des voies aérodigestives superieures: Traitement par injections de toxine botulique A Radiation-induced head and neck pain: Management with botulinum toxin a injections. Cancer Radiother. 2019, 23, 312–315. [Google Scholar] [CrossRef] [PubMed]
- Dang, S.; Shinn, J.R.; Sowder, J.; Ries, W.R.; Stephan, S.J. Peri-Incisional Botulinum Toxin Therapy for Treatment of Intractable Head Pain After Lateral Skull Base Surgery: A Case Series. Headache 2019, 59, 1624–1630. [Google Scholar] [CrossRef]
- Wang, L.; Lei, Q.S.; Liu, Y.Y.; Song, G.J.; Song, C.L. A case report of the beneficial effects of botulinum toxin type A on Raynaud phenomenon in a patient with lung cancer. Medicine 2016, 95, e5092. [Google Scholar] [CrossRef]
- Konradsson, M.; Nilsson, M. Delayed emptying of the gastric conduit after esophagectomy. J. Thorac. Dis. 2019, 11, S835–S844. [Google Scholar] [CrossRef]
- Kent, M.S.; Pennathur, A.; Fabian, T.; McKelvey, A.; Schuchert, M.J.; Luketich, J.D.; Landreneau, R.J. A pilot study of botulinum toxin injection for the treatment of delayed gastric emptying following esophagectomy. Surg. Endosc. 2007, 21, 754–757. [Google Scholar] [CrossRef] [PubMed]
- Cerfolio, R.J.; Bryant, A.S.; Canon, C.L.; Dhawan, R.; Eloubeidi, M.A. Is botulinum toxin injection of the pylorus during Ivor Lewis [corrected] esophagogastrectomy the optimal drainage strategy? J. Thorac. Cardiovasc. Surg. 2009, 137, 565–572. [Google Scholar] [CrossRef]
- Martin, J.T.; Federico, J.A.; McKelvey, A.A.; Kent, M.S.; Fabian, T. Prevention of delayed gastric emptying after esophagectomy: A single center’s experience with botulinum toxin. Ann. Thorac. Surg. 2009, 87, 1708–1713; discussion 1713–1714. [Google Scholar] [CrossRef]
- Bagheri, R.; Fattahi, S.H.; Haghi, S.Z.; Aryana, K.; Aryanniya, A.; Akhlaghi, S.; Riyabi, F.N.; Sheibani, S. Botulinum toxin for prevention of delayed gastric emptying after esophagectomy. Asian Cardiovasc. Thorac. Ann. 2013, 21, 689–692. [Google Scholar] [CrossRef] [PubMed]
- Antonoff, M.B.; Puri, V.; Meyers, B.F.; Baumgartner, K.; Bell, J.M.; Broderick, S.; Krupnick, A.S.; Kreisel, D.; Patterson, G.A.; Crabtree, T.D. Comparison of pyloric intervention strategies at the time of esophagectomy: Is more better? Ann. Thorac. Surg. 2014, 97, 1950–1957; discussion 1957–1958. [Google Scholar] [CrossRef] [PubMed]
- Eldaif, S.M.; Lee, R.; Adams, K.N.; Kilgo, P.D.; Gruszynski, M.A.; Force, S.D.; Pickens, A.; Fernandez, F.G.; Luu, T.D.; Miller, D.L. Intrapyloric botulinum injection increases postoperative esophagectomy complications. Ann. Thorac. Surg. 2014, 97, 1959–1964; discussion 1964–1965. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, H.F.; Broderick, R.C.; Harnsberger, C.R.; Divo, F.A.; Coker, A.M.; Jacobsen, G.R.; Sandler, B.J.; Bouvet, M.; Horgan, S. Intraoperative Endoscopic Botox Injection During Total Esophagectomy Prevents the Need for Pyloromyotomy or Dilatation. J. Laparoendosc. Adv. Surg. Tech. A 2016, 26, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Stewart, C.L.; Wilson, L.; Hamm, A.; Bartsch, C.; Boniface, M.; Gleisner, A.; Mitchell, J.D.; Weyant, M.J.; Meguid, R.; Gajdos, C.; et al. Is Chemical Pyloroplasty Necessary for Minimally Invasive Esophagectomy? Ann. Surg. Oncol. 2017, 24, 1414–1418. [Google Scholar] [CrossRef]
- Giugliano, D.N.; Berger, A.C.; Meidl, H.; Pucci, M.J.; Rosato, E.L.; Keith, S.W.; Evans, N.R.; Palazzo, F. Do intraoperative pyloric interventions predict the need for postoperative endoscopic interventions after minimally invasive esophagectomy? Dis. Esophagus 2017, 30, 1–8. [Google Scholar] [CrossRef]
- Marchese, S.; Qureshi, Y.A.; Hafiz, S.P.; Dawas, K.; Turner, P.; Mughal, M.M.; Mohammadi, B. Intraoperative Pyloric Interventions during Oesophagectomy: A Multicentre Study. J. Gastrointest. Surg. 2018, 22, 1319–1324. [Google Scholar] [CrossRef]
- Tham, J.C.; Nixon, M.; Ariyarathenam, A.V.; Humphreys, L.; Berrisford, R.; Wheatley, T.; Sanders, G. Intraoperative pyloric botulinum toxin injection during Ivor-Lewis gastroesophagectomy to prevent delayed gastric emptying. Dis. Esophagus 2019, 32, doy112. [Google Scholar] [CrossRef] [PubMed]
- Bhutani, M.S.; Ejaz, S.; Cazacu, I.M.; Singh, B.S.; Shafi, M.; Stroehlein, J.R.; Mehran, R.J.; Walsh, G.; Vaporciyan, A.; Swisher, S.G.; et al. Endoscopic Intrapyloric Botulinum Toxin Injection with Pyloric Balloon Dilation for Symptoms of Delayed Gastric Emptying after Distal Esophagectomy for Esophageal Cancer: A 10-Year Experience. Cancers 2022, 14, 5743. [Google Scholar] [CrossRef] [PubMed]
- Urschel, J.D.; Blewett, C.J.; Young, J.E.; Miller, J.D.; Bennett, W.F. Pyloric drainage (pyloroplasty) or no drainage in gastric reconstruction after esophagectomy: A meta-analysis of randomized controlled trials. Dig. Surg. 2002, 19, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Lew, R.J.; Kochman, M.L. A review of endoscopic methods of esophageal dilation. J. Clin. Gastroenterol. 2002, 35, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Hordijk, M.L.; Siersema, P.D.; Tilanus, H.W.; Kuipers, E.J. Electrocautery therapy for refractory anastomotic strictures of the esophagus. Gastrointest. Endosc. 2006, 63, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Lu, Z.; Linghu, E.; Yang, Y.; Yang, J.; Wang, S.; Yan, B.; Song, J.; Zhou, X.; Wang, X.; et al. Prevention of esophageal strictures after endoscopic submucosal dissection with the injection of botulinum toxin type A. Gastrointest. Endosc. 2016, 84, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Chen, H.; Chen, M.; Ding, C.; Zhang, G.; Si, X. Comparison of endoscopic injection of botulinum toxin and steroids immediately after endoscopic submucosal dissection to prevent esophageal stricture: A prospective cohort study. J. Cancer 2021, 12, 5789–5796. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xia, W.; Tian, L.; Zhu, B.; Chen, M.; Si, X.; Lin, S.; Gong, Y. Comparison of statins with steroids and botulinum toxin A in the prevention of benign strictures after esophageal endoscopic submucosal dissection: A retrospective cohort study. Surg. Endosc. 2023, 37, 4328–4337. [Google Scholar] [CrossRef]
- Svensson, E.; Zvara, P.; Qvist, N.; Hagander, L.; Möller, S.; Rasmussen, L.; Schrøder, H.D.; Hejbøl, E.K.; Bjørn, N.; Petersen, S.; et al. The Effect of Botulinum Toxin Type A Injections on Stricture Formation, Leakage Rates, Esophageal Elongation, and Anastomotic Healing Following Primary Anastomosis in a Long- and Short-Gap Esophageal Atresia Model—A Protocol for a Randomized, Controlled, Blinded Trial in Pigs. Int. J. Surg. Protoc. 2021, 25, 171–177. [Google Scholar] [CrossRef]
- Usui, Y.; Ono, S. Impact of botulinum toxin A injection on esophageal anastomosis in a rabbit model. Pediatr. Surg. Int. 2016, 32, 881–886. [Google Scholar] [CrossRef]
- Linder, T.E.; Huber, A.; Schmid, S. Frey’s syndrome after parotidectomy: A retrospective and prospective analysis. Laryngoscope 1997, 107, 1496–1501. [Google Scholar] [CrossRef] [PubMed]
- Bjerkhoel, A.; Trobbe, O. Frey’s syndrome: Treatment with botulinum toxin. J. Laryngol. Otol. 1997, 111, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Laccourreye, O.; Muscatelo, L.; Naude, C.; Bonan, B.; Brasnu, D. Botulinum toxin type A for Frey’s syndrome: A preliminary prospective study. Ann. Otol. Rhinol. Laryngol. 1998, 107, 52–55. [Google Scholar] [CrossRef] [PubMed]
- von Lindern, J.J.; Niederhagen, B.; Bergé, S.; Hägler, G.; Reich, R.H. Frey syndrome: Treatment with type A botulinum toxin. Cancer 2000, 89, 1659–1663. [Google Scholar] [CrossRef] [PubMed]
- Cavalot, A.L.; Palonta, F.; Preti, G.; Nazionale, G.; Ricci, E.; Staffieri, A.; Di Girolamo, S.; Cortesina, G. [Post-parotidectomy Frey’s syndrome. Treatment with botulinum toxin type A]. Acta Otorhinolaryngol. Ital. 2000, 20, 187–191. [Google Scholar]
- Vargas, H.; Galati, L.T.; Parnes, S.M. A pilot study evaluating the treatment of postparotidectomy sialoceles with botulinum toxin type A. Arch. Otolaryngol. Head Neck Surg. 2000, 126, 421–424. [Google Scholar] [CrossRef][Green Version]
- Küttner, C.; Tröger, M.; Dempf, R.; Eckardt, A. [Effectiveness of botulinum toxin A in the treatment of gustatory sweating]. Nervenarzt 2001, 72, 787–790. [Google Scholar] [CrossRef]
- Eckardt, A.; Kuettner, C. Treatment of gustatory sweating (Frey’s syndrome) with botulinum toxin A. Head Neck 2003, 25, 624–628. [Google Scholar] [CrossRef]
- Nolte, D.; Gollmitzer, I.; Loeffelbein, D.J.; Hölzle, F.; Wolff, K.D. [Botulinum toxin for treatment of gustatory sweating. A prospective randomized study]. Mund. Kiefer Gesichtschir 2004, 8, 369–375. [Google Scholar] [CrossRef]
- Marchese-Ragona, R.; Marioni, G.; Restivo, D.A.; Staffieri, A. The role of botulinum toxin in postparotidectomy fistula treatment. A technical note. Am. J. Otolaryngol. 2006, 27, 221–224. [Google Scholar] [CrossRef]
- Pomprasit, M.; Chintrakarn, C. Treatment of Frey’s syndrome with botulinum toxin. J. Med. Assoc. Thai 2007, 90, 2397–2402. [Google Scholar] [PubMed]
- Marchese, M.R.; Almadori, G.; Giorgio, A.; Paludetti, G. Post-surgical role of botulinum toxin-A injection in patients with head and neck cancer: Personal experience. Acta Otorhinolaryngol. Ital. 2008, 28, 13–16. [Google Scholar] [PubMed]
- Martos Díaz, P.; Bances del Castillo, R.; Mancha de la Plata, M.; Naval Gías, L.; Martínez Nieto, C.; Lee, G.Y.; Muñoz Guerra, M. Clinical results in the management of Frey’s syndrome with injections of Botulinum toxin. Med. Oral Patol. Oral Cir. Bucal 2008, 13, E248–E252. [Google Scholar] [PubMed]
- Cantarella, G.; Berlusconi, A.; Mele, V.; Cogiamanian, F.; Barbieri, S. Treatment of Frey’s syndrome with botulinum toxin type B. Otolaryngol. Head Neck Surg. 2010, 143, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Laskawi, R.; Winterhoff, J.; Köhler, S.; Kottwitz, L.; Matthias, C. Botulinum toxin treatment of salivary fistulas following parotidectomy: Follow-up results. Oral Maxillofac. Surg. 2013, 17, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Steffen, A.; Hasselbacher, K.; Heinrichs, S.; Wollenberg, B. Botulinum toxin for salivary disorders in the treatment of head and neck cancer. Anticancer. Res. 2014, 34, 6627–6632. [Google Scholar] [PubMed]
- Melville, J.C.; Stackowicz, D.J.; Jundt, J.S.; Shum, J.W. Use of Botox (OnabotulinumtoxinA) for the Treatment of Parotid Sialocele and Fistula After Extirpation of Buccal Squamous Cell Carcinoma with Immediate Reconstruction Using Microvascular Free Flap: A Report of 3 Cases. J. Oral Maxillofac. Surg. 2016, 74, 1678–1686. [Google Scholar] [CrossRef]
- Marchese, M.R.; Di Cesare, T.; De Corso, E.; Petracca, M.; Oliveto, G.; Almadori, G. Botulinum Neurotoxin A in the Treatment of Pharyngocutaneous Fistula after Salvage Surgery in Head and Neck Cancer Patients: Our Preliminary Results. Curr. Oncol. 2022, 29, 7099–7105. [Google Scholar] [CrossRef]
- Mueller, J.; Langbein, T.; Mishra, A.; Baum, R.P. Safety of High-Dose Botulinum Toxin Injections for Parotid and Submandibular Gland Radioprotection. Toxins 2022, 14, 64. [Google Scholar] [CrossRef]
- Laccourreye, O.; Werner, A.; Garcia, D.; Malinvaud, D.; Tran Ba Huy, P.; Bonfils, P. First bite syndrome. Eur. Ann. Otorhinolaryngol. Head Neck Dis. 2013, 130, 269–273. [Google Scholar] [CrossRef]
- Shaikh, N.E.; Jafary, H.A.; Behnke, J.W.; Turner, M.T. Botulinum toxin A for the treatment of first bite syndrome-a systematic review. Gland Surg. 2022, 11, 1251–1263. [Google Scholar] [CrossRef] [PubMed]
- Tighe, A.P.; Schiavo, G. Botulinum neurotoxins: Mechanism of action. Toxicon 2013, 67, 87–93. [Google Scholar] [CrossRef]
- Matak, I.; Bölcskei, K.; Bach-Rojecky, L.; Helyes, Z. Mechanisms of Botulinum Toxin Type A Action on Pain. Toxins 2019, 11, 459. [Google Scholar] [CrossRef] [PubMed]
- Durham, P.L.; Cady, R.; Cady, R. Regulation of calcitonin gene-related peptide secretion from trigeminal nerve cells by botulinum toxin type A: Implications for migraine therapy. Headache 2004, 44, 35–42; discussion 42–43. [Google Scholar] [CrossRef] [PubMed]
- Purkiss, J.; Welch, M.; Doward, S.; Foster, K. Capsaicin-stimulated release of substance P from cultured dorsal root ganglion neurons: Involvement of two distinct mechanisms. Biochem. Pharmacol. 2000, 59, 1403–1406. [Google Scholar] [CrossRef] [PubMed]
- Rapp, D.E.; Turk, K.W.; Bales, G.T.; Cook, S.P. Botulinum toxin type a inhibits calcitonin gene-related peptide release from isolated rat bladder. J. Urol. 2006, 175, 1138–1142. [Google Scholar] [CrossRef] [PubMed]
- Lucioni, A.; Bales, G.T.; Lotan, T.L.; McGehee, D.S.; Cook, S.P.; Rapp, D.E. Botulinum toxin type A inhibits sensory neuropeptide release in rat bladder models of acute injury and chronic inflammation. BJU Int. 2008, 101, 366–370. [Google Scholar] [CrossRef]
- Hong, B.; Yao, L.; Ni, L.; Wang, L.; Hu, X. Antinociceptive effect of botulinum toxin A involves alterations in AMPA receptor expression and glutamate release in spinal dorsal horn neurons. Neuroscience 2017, 357, 197–207. [Google Scholar] [CrossRef]
- Cui, M.; Khanijou, S.; Rubino, J.; Aoki, K.R. Subcutaneous administration of botulinum toxin A reduces formalin-induced Pain. Pain 2004, 107, 125–133. [Google Scholar] [CrossRef]
- Shin, M.C.; Wakita, M.; Xie, D.J.; Yamaga, T.; Iwata, S.; Torii, Y.; Harakawa, T.; Ginnaga, A.; Kozaki, S.; Akaike, N. Inhibition of Membrane Na(+) Channels by A Type Botulinum Toxin at Femtomolar Concentrations in Central and Peripheral Neurons. J. Pharmacol. Sci. 2012, 118, 33–42. [Google Scholar] [CrossRef]
- Waxman, S.G.; Cummins, T.R.; Dib-Hajj, S.D.; Black, J.A. Voltage-gated sodium channels and the molecular pathogenesis of pain: A review. J. Rehabil. Res. Dev. 2000, 37, 517–528. [Google Scholar] [PubMed]
- Filipović, B.; Matak, I.; Bach-Rojecky, L.; Lacković, Z. Central action of peripherally applied botulinum toxin type A on pain and dural protein extravasation in rat model of trigeminal neuropathy. PLoS ONE 2012, 7, e29803. [Google Scholar] [CrossRef] [PubMed]
- Favre-Guilmard, C.; Auguet, M.; Chabrier, P.E. Different antinociceptive effects of botulinum toxin type A in inflammatory and peripheral polyneuropathic rat models. Eur. J. Pharmacol. 2009, 617, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Bach-Rojecky, L.; Salković-Petrisić, M.; Lacković, Z. Botulinum toxin type A reduces pain supersensitivity in experimental diabetic neuropathy: Bilateral effect after unilateral injection. Eur. J. Pharmacol. 2010, 633, 10–14. [Google Scholar] [CrossRef] [PubMed]
- Drinovac Vlah, V.; Bach-Rojecky, L.; Lacković, Z. Antinociceptive action of botulinum toxin type A in carrageenan-induced mirror Pain. J. Neural Transm. 2016, 123, 1403–1413. [Google Scholar] [CrossRef]
- Grenda, T.; Grenda, A.; Krawczyk, P.; Kwiatek, K. Botulinum toxin in cancer therapy-current perspectives and limitations. Appl. Microbiol. Biotechnol. 2022, 106, 485–495. [Google Scholar] [CrossRef]
- Karsenty, G.; Rocha, J.; Chevalier, S.; Scarlata, E.; Andrieu, C.; Zouanat, F.Z.; Rocchi, P.; Giusiano, S.; Elzayat, E.A.; Corcos, J. Botulinum toxin type A inhibits the growth of LNCaP human prostate cancer cells in vitro and in vivo. Prostate 2009, 69, 1143–1150. [Google Scholar] [CrossRef]
- Bandala, C.; Cortés-Algara, A.L.; Mejía-Barradas, C.M.; Ilizaliturri-Flores, I.; Dominguez-Rubio, R.; Bazán-Méndez, C.I.; Floriano-Sánchez, E.; Luna-Arias, J.P.; Anaya-Ruiz, M.; Lara-Padilla, E. Botulinum neurotoxin type A inhibits synaptic vesicle 2 expression in breast cancer cell lines. Int. J. Clin. Exp. Pathol. 2015, 8, 8411–8418. [Google Scholar]
- Rust, A.; Leese, C.; Binz, T.; Davletov, B. Botulinum neurotoxin type C protease induces apoptosis in differentiated human neuroblastoma cells. Oncotarget 2016, 7, 33220–33228. [Google Scholar] [CrossRef]
- Shebl, R.I. Anti-cancer Potential of Captopril and Botulinum Toxin Type-A and Associated p53 Gene Apototic Stimulating Activity. Iran. J. Pharm. Res. 2019, 18, 1967–1977. [Google Scholar] [CrossRef]
- Ulloa, F.; Gonzàlez-Juncà, A.; Meffre, D.; Barrecheguren, P.J.; Martínez-Mármol, R.; Pazos, I.; Olivé, N.; Cotrufo, T.; Seoane, J.; Soriano, E. Blockade of the SNARE protein syntaxin 1 inhibits glioblastoma tumor growth. PLoS ONE 2015, 10, e0119707. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Manzoni, A.; Florentin, D.; Fisher, W.; Ding, Y.; Lee, M.; Ayala, G. Biologic effect of neurogenesis in pancreatic cancer. Hum. Pathol. 2016, 52, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Coarfa, C.; Florentin, D.; Putluri, N.; Ding, Y.; Au, J.; He, D.; Ragheb, A.; Frolov, A.; Michailidis, G.; Lee, M.; et al. Influence of the neural microenvironment on prostate cancer. Prostate 2018, 78, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Ansiaux, R.; Baudelet, C.; Cron, G.O.; Segers, J.; Dessy, C.; Martinive, P.; De Wever, J.; Verrax, J.; Wauthier, V.; Beghein, N.; et al. Botulinum toxin potentiates cancer radiotherapy and chemotherapy. Clin. Cancer Res. 2006, 12, 1276–1283. [Google Scholar] [CrossRef]

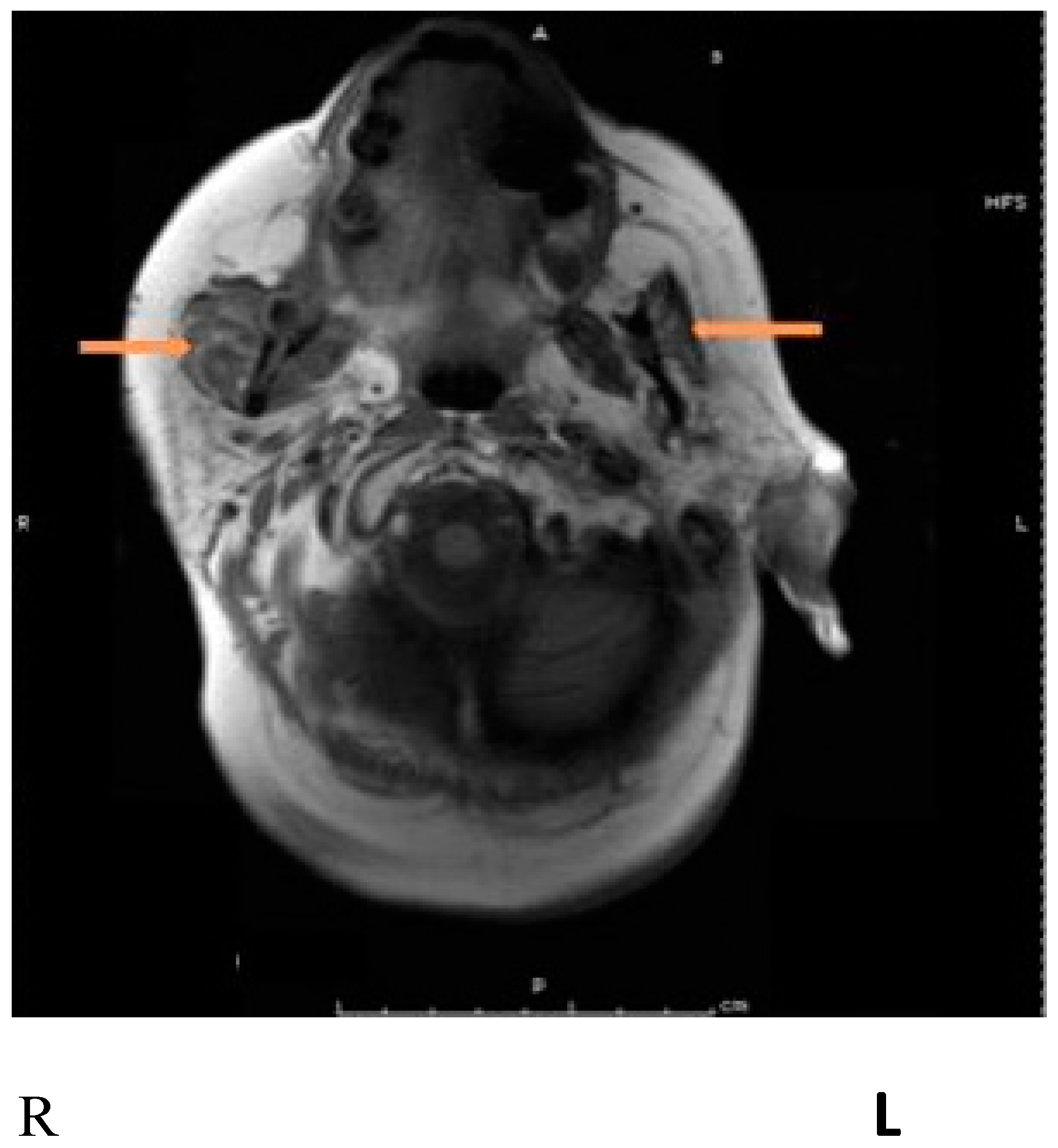
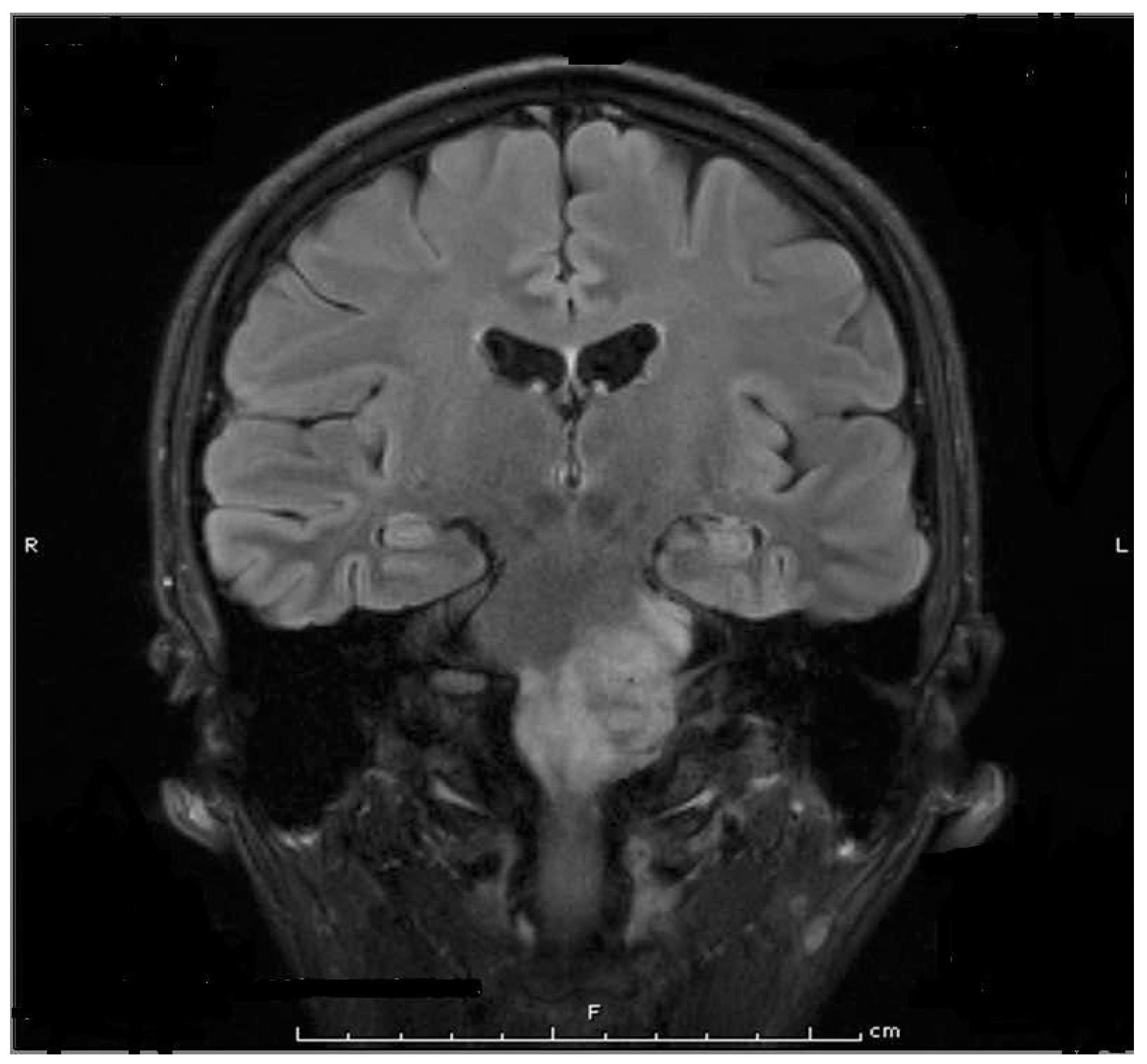
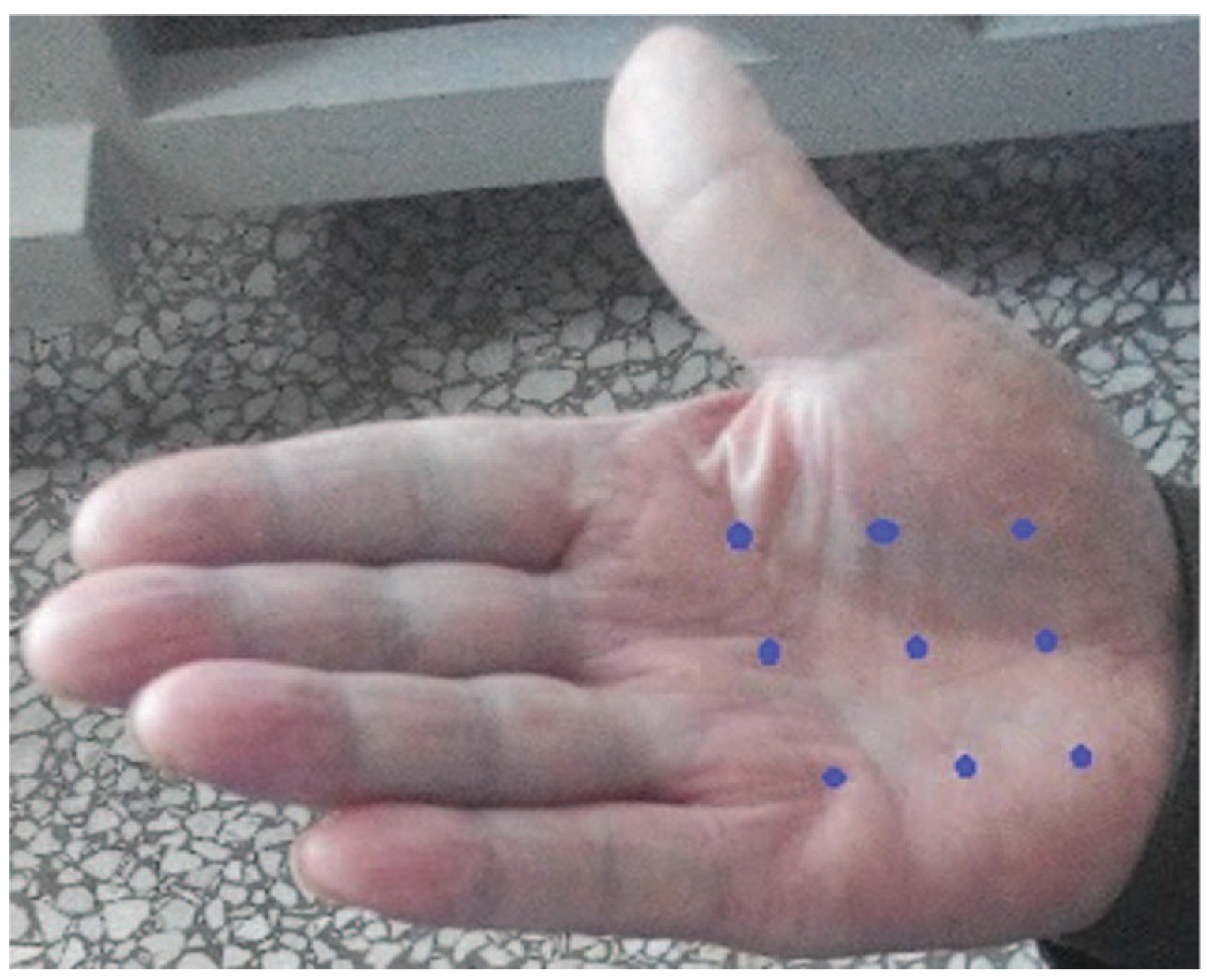
| Authors, Date | #Pts Study Type | Toxin | Dose Units | Treatment | Type of Cancer | Primary Outcome | Result |
|---|---|---|---|---|---|---|---|
| Van Deale et al., 2002 [17] | 6, Retro | onaA | 20–25 | Radiation or Chemotherapy | Head and neck | Pain (VAS) | Complete pain relief in 4 of 6 Pts; Significant improvement in quality of life using SF36, EQ-5D scales |
| Vasan et al., 2004 [18] | 16, Pros | aboA | 100–320 | Surgery | Head and neck | Pain (VAS-days 3 and 4 wks), Global Quality of Life (GQL) | Significant pain reduction (p = 0.05) Quality of life improved (p = 0.7) |
| Wittekind et al., 2006 [19] | 23, Pros | OnaA | 60–120; 160–240 | Radiation, Surgery | Head and neck | Pain (VAS) At 28 wks | Significant reduction in pain at lower dose (<0.05) |
| Hartl et al., 2008 [20] | 19, Pros | onaA aboA | 50 250 | Chemotherapy radiation | Head and neck | Pain: (VAS) Function: At 4 wks | Both improved Pain (p =0.02) Function (p = 0.04) |
| Stublefied et al., 2008 [21] | 23, Retro | onaA | 25–200 | Radiation Surgery | Head, neck, breast | Pain (VAS) | Pain Improved in 85% of Pts |
| Chuang et al., 2008 [22] | 8, Retro | onaA | 100 | Radiation | Bladder Prostate | Pain (VAS) | Pain average dropped from level 8 to 2 |
| Voung et al., 2010 [23] | 15, Can 20 Con Pros | onaA | 100 unit | Radiation | Pelvic cancer | Pain(VAS) | Significant difference from control (p < 0.02) |
| Mittal et al., 2012 [24] | 7, Retro | onaA | 100 | Radiation Surgery | Head, neck, breast | Pain (VAS), PGIC at 4 weeks | VAS 6 of 7 improved: p < 0.05 PGIC: 6 of 7, very satisfied QoL; 6 of 7 improved (p < 0.05) |
| Bach et al., 2012 [25] | 9, Pros | onaA | 100–400 | Radiation and Surgery | Head and neck | Pain (VAS) and FDSNP at 4 weeks | Both pain and FDSNP improved (p < 0.01) |
| Rostami et al., 2014 [26] | 12, Pros | incoA | 100 | Radiation and Surgery | Head, neck, breast | Pain (VAS) and PGIC at week 6 | Both VAS (p < 0.05) PGIC: very satisfied QoL in 38% (p < 0.05) |
| Degroef et al., 2018 [27] | 50, Db-pc | 0naA | 100 | During surgery | Breast cancer | Pain (VAS) at 3 and 6 months | At 6 months, 40% in the intervention group and 60% in control group still had pectoral pain (p > 0.05) |
| Mailly et al., 2019 [28] | 16, Retro | incoA aboA | 20– 40 | Radiation and surgery | Head and neck | Pain (VAS) | VAS improved p < 0.01 in 11 pts |
| Dang et al., 2019 [29] | 3, Retro | onaA | 20–60 | surgery | Base of skull schwannoma | Pain at the site of skull incision | Headaches subsided |
| Authors and Date | Number of Patients, Study Type | Toxin | Dose in Units, Injected Site | Procedure | Pathology | Results |
|---|---|---|---|---|---|---|
| Kent et al., 2007 [32] | 15 12 injected during esophagectomy 3 shortly after surgery; Retro | onaA | 200 (Anterior pylorus at 4 points) | During esophagectomy | Not specified | No patient injected with Botox during esophagectomy developed DGE. |
| Cerfolio et al., 2009 [33] | 150, No intervention vs pyloroplasty vs. BoNT-A Retro | OnaA | 100 (Divided into 4 injections into pylorus) | During esophagectomy | Adeno and squamous cell carcinoma | Incidence of DGE in no intervention and pyloroplasty: 96%; in OnaA: 56% (p = 0.05) |
| Martin et al., 2009 [34] | 45, Intrapyloric injection of Botox during pyloroplasty; Pro | onaA | 200 (Into anterior wall of pyrolus) | After esophagectomy | Not specified | 96% of patients showed no evidence of DGE (3 months or more) |
| Bagheri et al., 2013 [35] | 60, Pyloroplasty vs. Botox injection; Pros | onaA | 100 (Lower section of pylorus) | After esophagectomy | Thoracic, gastric, esophageal cancers | Incidence of DGE in onaA injected group 10% vs. 26% in pyloroplasty group |
| Antonoff et al., 2014 [36] | 293, Pyloroplasty vs. Botox with dilation; Retro | onaA | 200 (Different parts of pyrolus) | After esophagectomy | Different cancers | Both interventions were superior to no intervention in preventing DGE. No difference between the two interventions, but pyloroplasty group had two serious side effects, |
| Eldaif et al., 2014 [37] | 322: 86% with esophageal cancer, Pyloroplasty vs. pylomyotomy vs. onaA; Retro | onaA | 100 (Divided into 4 separate pyloric Injections) | After esophagectomy Evaluated for DGE between 5–7 postoperative day | Not specified Majority had preoperative radiation | Botox injection decreased operative time but did not change the incidence of DGE |
| Fuchs et al., 2016 [38] | 41, BoNT-A injection vs. no intervention Retro | onaA | 200 (Divided into 4 injections into pylorus | During esophagectomy | Adeno and squamous cell carsinomas | DGE Botox: 0 Non-intervention: 8 p < 0.05 |
| Stewart et al., 2017 [39] | 71, Intraoperative injection of BoNT-A compared with no intervention Retro | onaA | Not mentioned | During esophagectomy | Cancer Type not specified | No difference between onaA injected and no intervention group in duration of jejunostomy tube use |
| Giugliano et al., 2017 [40] | 146, BoNT-A injection compared with no intervention; Retro | BoNT-A | Not mentioned | During esophagectomy | Cancer, 91.8% adenocarcinoma | No difference between BoNT group and no intervention |
| Marchese et al., 2019 [41] | 90, Pyloroplasty vs. onaA injection vs. no therapy; Pros | onaA | 200 (Divided into 4 injections pylorus) | During esophagectomy | Not mentioned | Incidence of DGE was the same in all three groups |
| Tham et al., 2019 [42] | 228, Botulinum toxin injection versus no intervention Retro | onaA | Not mentioned | During esophagectomy | Most adenocarcinoma | No difference between Botox injection and no intervention |
| Bhutani et al., 2022 [43] | 21 with DGE after esophagostomy treated with BoNT injection and ballooning | onaA | 100 units (Divided into 4 injections into pylorus) | Treating post–esophagectomy DGE | Esophageal cancer | 85% reported improvement by more than 50% of DGE symptoms |
| Authors | Design | Number of Patients | Clinical Problem | Injection Site | Toxin and Dose in Units | Result |
|---|---|---|---|---|---|---|
| Wen et al., 2016 [47] | Pro | 67 | Prevention of ES after esophageal submucosal dissection | Esophagus | Ona-A 100 | Decrease in ES, decrease in esophageal dilation |
| Zhou et al., 2021 [48] | Pro | 78 | Prevention of ES after esophageal submucosal dissection | Esophagus | Ona-A 100 | Decrease in ES, particularly in patients who had entire circumference mucosal defect. |
| Wang et al., 2023 [49] | Retro | 204 | Prevention of ES after esophageal submucosal dissection | Esophagus | Ona-A 100 | Decrease in ES in patients who received statins or BoNT injection |
| Authors | Design | Number of Patients | Clinical Problem | Injection Site | Toxin and Dose in Units | Result |
|---|---|---|---|---|---|---|
| Bjerkhoel et al., 1997 [53] | Pro | 15 | GH after parotidectomy | Face | OnaA 17–62.5 | Total cessation of facial sweating in 13 patients |
| Laccourreye et al., 1998 [54] | Pro | 14 | GH after parotidectomy | Face | OnaA 25–88 | All showed total cessation of sweating |
| Von Lindern et al., 2000 [55] | Retro | 7 | GH after parotidectomy | Face | OnaA | Sweating stopped after BoNT injection |
| Cavalot et al., 2000 [56] | Pro | 40 | GH after parotidectomy | Face | OnaA, 2.5/cm | 100% response in severe group, 72% response in moderate group |
| Vargas et al., 2000 [57] | Pro | 4 | Post-parotidectomy sialocele pain | Parotid gland | OnaA, 30–50 | Total resolution within 4 weeks in all patients |
| Kuttner et al., 2001 [58] | Retro | 8 | GH after parotidectomy | Face | BoNT-A, 0.5/cm | Stopped facial sweating within one week |
| Eckardt et al., 2003 [59] | Retro | 33 | GH after parotidectomy | Face | OnaA, 16–80 | Facial sweating disappeared within a week after injections |
| Nolte et al., 2004 [60] | Pro | 20 | Gustatory sweating after parotidectomy | Facial | OnaA, 3/cm | Total cessation of sweating for 12 months |
| Marchese-Ragona et al., 2006 [61] | Retro | 3 | Post-parotidectomy fistula | Parotid gland | OnaA, 15–20 | Complete healing of fistula with follow-ups of 12, 18, and 14 months |
| Pomprasit et al., 2007 [62] | Pro | 9 | GH after Parotidectomy | Face | OnaA, 10.6 | Sweating stopped in 5 pts and was reduced in 4 pts |
| Machese et al., 2008 [63] | Retro | 8 | Head and neck cancer: 6 sialorrhea; 1 1 fistula; 1sialocele | Parotid gland | AboA, 100 | Fistulas healed. Sialorrhea stopped |
| Martos Dias et al., 2008 [64] | Retro | 10 | GH after parotidectomy | Face | OnaA, 38 | Sweating stopped |
| Hatrl et al., 2008 [20] | Retro | 7 | GH after parotidectomy | Face | OnaA 50 Abo-A 250 | Sweating and quality of life improved |
| Cantarella and Barbieri, 2010 [65] | Retro | 7 | GH after parotidectomy | Face | RimaB, 2200 | Cessation of sweating in 6 of 7 patients 4 weeks after injection |
| Laskawi et al., 2013 [66] | Retro | 10 | Post-parotidectomy fistula | Parotid gland | OnaA, 30–50 | Treated within 6 weeks of surgery: fistulas healed in 9 of 10 patients |
| Steffen et al., 2014 [67] | Rro | 25 | Head and neck cancer: FHS: (19), Fistula (6) | Parotid gland | OnaA and incoA, Parotid: 30 UM: 20 | FHS: 11 of 19 improved Fistula: 4 of 6 healed |
| Melville et al., 2016 [68] | Pro | 3 | Buccal squamous cell carcinoma; parotid sialocele and fistula | Parotid gland | onaA, 50–70 | In all three, fistula and sialocele healed, and serous drainage stopped |
| Marchese et al., 2022 [69] | Retro | 12 All had CT and RT | Cancer of larynx and pharynx, sialocele/fistula | Parotid gland | onaA, 80 into each gland | 54% of the patients had closure of fistula within days |
| Mueller et al., 2022 [70] | Pro | 10 | Prostate cancer Post Ac-PSMA therapy | face | IncoA, 6 injections, 30 u per injection point | Those injected by BoNT showed a mean of 29% gland destruction after two cycles of Ac-PSMA treatment compared 60–70% seen in those who did not receive BoNT injection |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Safarpour, D.; Jabbari, B. Botulinum Toxin Treatment for Cancer-Related Disorders: A Systematic Review. Toxins 2023, 15, 689. https://doi.org/10.3390/toxins15120689
Safarpour D, Jabbari B. Botulinum Toxin Treatment for Cancer-Related Disorders: A Systematic Review. Toxins. 2023; 15(12):689. https://doi.org/10.3390/toxins15120689
Chicago/Turabian StyleSafarpour, Delaram, and Bahman Jabbari. 2023. "Botulinum Toxin Treatment for Cancer-Related Disorders: A Systematic Review" Toxins 15, no. 12: 689. https://doi.org/10.3390/toxins15120689
APA StyleSafarpour, D., & Jabbari, B. (2023). Botulinum Toxin Treatment for Cancer-Related Disorders: A Systematic Review. Toxins, 15(12), 689. https://doi.org/10.3390/toxins15120689





