Daily Headache in Chronic Migraine Is a Predictive Factor of Response in Patients Who Had Completed Three Sessions of OnabotulinumtoxinA
Abstract
:1. Introduction
2. Results
2.1. General Characteristics
2.2. Treatment Response
2.2.1. Total Sample
2.2.2. Primary Endpoint
2.3. Secondary Endpoint
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Study Design and Data Sources
5.2. Eligibility Criteria
5.3. Procedure
5.4. Variables
5.5. Endpoints
5.5.1. Primary Endpoint
5.5.2. Secondary Endpoints
5.6. Ethical Aspects
5.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [CrossRef]
- Bigal, M.E.; Rapoport, A.M.; Lipton, R.B.; Tepper, S.J.; Sheftell, F.D. Assessment of migraine disability using the migraine disability assessment (MIDAS) questionnaire: A comparison of chronic migraine with episodic migraine. Headache 2003, 43, 336–342. [Google Scholar] [CrossRef]
- Silberstein, S.D. Preventive Migraine Treatment. Continuum (Minneap Minn) 2015, 21, 973–989. [Google Scholar] [CrossRef]
- Ezpeleta, D.; Pozo-Rosich, P. Guías Diagnósticas y Terapéuticas de la Sociedad Española de Neurología. Guía Oficial de Práctica Clínica en Cefaleas; Editorial Luzan 5: Madrid, Spain, 2015; Volume 53. [Google Scholar]
- Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Aurora, S.K.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F.; PREEMPT Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Pooled results from the double-blind, randomized, placebo-controlled phases of the PREEMPT clinical program. Headache 2010, 50, 921–936. [Google Scholar] [CrossRef]
- Aurora, S.K.; Winner, P.; Freeman, M.C.; Spierings, E.L.; Heiring, J.O.; DeGryse, R.E.; VanDenburgh, A.M.; Nolan, M.E.; Turkel, C.C. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache 2011, 51, 1358–1373. [Google Scholar] [CrossRef]
- Silberstein, S.D.; Dodick, D.W.; Aurora, S.K.; Diener, H.C.; DeGryse, R.E.; Lipton, R.B.; Turkel, C.C. Per cent of patients with chronic migraine who responded per onabotulinumtoxinA treatment cycle: PREEMPT. J. Neurol. Neurosurg. Psychiatry 2015, 86, 996–1001. [Google Scholar] [CrossRef] [PubMed]
- Gago-Veiga, A.B.; Santos-Lasaosa, S.; Cuadrado, M.L.; Guerrero, Á.L.; Irimia, P.; Láinez, J.M.; Leira, R.; Pascual, J.; Sanchez Del Río, M.; Viguera, J.; et al. Evidence and experience with onabotulinumtoxinA in chronic migraine: Recommendations for daily clinical practice. Neurologia 2019, 34, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Blumenfeld, A.M.; Varon, S.F.; Wilcox, T.K.; Buse, D.C.; Kawata, A.K.; Manack, A.; Goadsby, P.J.; Lipton, R.B. Disability, HRQoL and resource use among chronic and episodic migraineurs: Results from the International Burden of Migraine Study (IBMS). Cephalalgia 2011, 31, 301–315. [Google Scholar] [CrossRef] [PubMed]
- Lake, A.E., 3rd. Chronic daily headache in the absence of medication overuse: Is daily or continuous pain more treatment-resistant than chronic daily headache with pain-free days? Curr. Pain Headache Rep. 2005, 9, 4–6. [Google Scholar] [CrossRef]
- Martínez-Pías, E.; García-Azorín, D.; Trigo-López, J.; Sierra, A.; Guerrero-Peral, A.L. Chronic migraine with daily headache. Literature review. Rev. Neurol. 2021, 72, 133–140. [Google Scholar] [CrossRef]
- Aurora, S.K.; Dodick, D.W.; Turkel, C.C.; DeGryse, R.E.; Silberstein, S.D.; Lipton, R.B.; Diener, H.C.; Brin, M.F. PREEMPT 1 Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 1 trial. Cephalalgia 2010, 30, 793–803. [Google Scholar] [CrossRef]
- Diener, H.C.; Dodick, D.W.; Aurora, S.K.; Turkel, C.C.; DeGryse, R.E.; Lipton, R.B.; Silberstein, S.D.; Brin, M.F. PREEMPT 2 Chronic Migraine Study Group. OnabotulinumtoxinA for treatment of chronic migraine: Results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia 2010, 30, 804–814. [Google Scholar] [CrossRef]
- Detke, H.C.; Goadsby, P.J.; Wang, S.; Friedman, D.I.; Selzler, K.J.; Aurora, S.K. Galcanezumab in chronic migraine: The randomized, double-blind, placebo-controlled REGAIN study. Neurology 2018, 91, e2211–e2221. [Google Scholar] [CrossRef] [Green Version]
- Lipton, R.B.; Goadsby, P.J.; Smith, J.; Schaeffler, B.A.; Biondi, D.M.; Hirman, J.; Pederson, S.; Allan, B.; Cady, R. Efficacy and safety of eptinezumab in patients with chronic migraine: PROMISE-2. Neurology 2020, 94, e1365–e1377. [Google Scholar] [CrossRef] [Green Version]
- Silberstein, S.D.; Dodick, D.W.; Bigal, M.E.; Yeung, P.P.; Goadsby, P.J.; Blankenbiller, T.; Grozinski-Wolff, M.; Yang, R.; Ma, Y.; Aycardi, E. Fremanezumab for the Preventive Treatment of Chronic Migraine. N. Engl. J. Med. 2017, 377, 2113–2122. [Google Scholar] [CrossRef] [PubMed]
- Tepper, S.; Ashina, M.; Reuter, U.; Brandes, J.L.; Doležil, D.; Silberstein, S.; Winner, P.; Leonardi, D.; Mikol, D.; Lenz, R. Safety and efficacy of erenumab for preventive treatment of chronic migraine: A randomised, double-blind, placebo-controlled phase 2 trial. Lancet Neurol. 2017, 16, 425–434. [Google Scholar] [CrossRef]
- Tinsley, A.; Rothrock, J.F. What Are We Missing in the Diagnostic Criteria for Migraine? Curr. Pain Headache Rep. 2018, 22, 84. [Google Scholar] [CrossRef]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018, 38, 1–211. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Dhaliwal, H.P.; Kukreja, R.V.; Singh, B.R. The Botulinum Toxin as a Therapeutic Agent: Molecular Structure and Mechanism of Action in Motor and Sensory Systems. Semin. Neurol. 2016, 36, 10–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burstein, R.; Blumenfeld, A.M.; Silberstein, S.D.; Manack Adams, A.; Brin, M.F. Mechanism of Action of OnabotulinumtoxinA in Chronic Migraine: A Narrative Review. Headache 2020, 60, 1259–1272. [Google Scholar] [CrossRef]
- Gfrerer, L.; Raposio, E.; Ortiz, R.; Austen, W.G., Jr. Surgical treatment of migraine headache: Back to the future. Plast Reconstr. Surg. 2018, 142, 1036–1045. [Google Scholar] [CrossRef]
- Aurora, S.K.; Brin, M.F. Chronic Migraine: An Update on Physiology, Imaging, and the Mechanism of Action of Two Available Pharmacologic Therapies. Headache 2017, 57, 109–125. [Google Scholar] [CrossRef]
- Jackson, J.L.; Kuriyama, A.; Hayashino, Y. Botulinum toxin A for prophylactic treatment of migraine and tension headaches in adults: A meta-analysis. JAMA 2012, 307, 1736–1745. [Google Scholar] [CrossRef]
- Ornello, R.; Ahmed, F.; Negro, A.; Miscio, A.M.; Santoro, A.; Alpuente, A.; Russo, A.; Silvestro, M.; Cevoli, S.; Brunelli, N.; et al. Early Management of OnabotulinumtoxinA Treatment in Chronic Migraine: Insights from a Real-Life European Multicenter Study. Pain Ther. 2021, in press. [Google Scholar] [CrossRef]
- Ahmed, F.; Gaul, C.; García-Moncó, J.C.; Sommer, K.; Martelletti, P. REPOSE Principal Investigators. An open-label prospective study of the real-life use of onabotulinumtoxinA for the treatment of chronic migraine: The REPOSE study. J. Headache Pain 2019, 20, 26. [Google Scholar] [CrossRef] [Green Version]
- Khalil, M.; Zafar, H.W.; Quarshie, V.; Ahmed, F. Prospective analysis of the use of OnabotulinumtoxinA (BOTOX) in the treatment of chronic migraine; real-life data in 254 patients from Hull, U.K. J. Headache Pain 2014, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Blumenfeld, A.M.; Stark, R.J.; Freeman, M.C.; Orejudos, A.; Manack Adams, A. Long-term study of the efficacy and safety of OnabotulinumtoxinA for the prevention of chronic migraine: COMPEL study. J. Headache Pain 2018, 19, 13. [Google Scholar] [CrossRef] [Green Version]
- García-Azorin, D.; Santos-Lasaosa, S.; Gago-Veiga, A.B.; Viguera Romero, J.; Guerrero-Peral, A.L. Real world preventative drug management of migraine among Spanish neurologists. J. Headache Pain 2019, 20, 19. [Google Scholar] [CrossRef] [Green Version]
- Caronna, E.; Gallardo, V.J.; Hernández-Beltrán, N.; Torres-Ferrus, M.; Pozo-Rosich, P. OnabotulinumtoxinA: An Effective Tool in the Therapeutic Arsenal for Chronic Migraine with Medication Overuse. Front. Neurol. 2018, 9, 808. [Google Scholar] [CrossRef] [PubMed]
- Young, W.B.; Ivan Lopez, J.; Rothrock, J.F.; Orejudos, A.; Manack Adams, A.; Lipton, R.B.; Blumenfeld, A.M. Effects of onabotulinumtoxinA treatment in chronic migraine patients with and without daily headache at baseline: Results from the COMPEL Study. J. Headache Pain 2019, 20, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaul, C.; Brömstrup, J.; Fritsche, G.; Diener, H.C.; Katsarava, Z. Evaluating integrated headache care: A one-year follow-up observational study in patients treated at the Essen headache centre. BMC Neurol. 2011, 11, 124. [Google Scholar] [CrossRef] [Green Version]
- Luconi, R.; Bartolini, M.; Taffi, R.; Vignini, A.; Mazzanti, L.; Provinciali, L.; Silvestrini, M. Prognostic significance of personality profiles in patients with chronic migraine. Headache 2007, 47, 1118–1124. [Google Scholar] [CrossRef]
- Alpuente, A.; Gallardo, V.J.; Torres-Ferrús, M.; Álvarez-Sabin, J.; Pozo-Rosich, P. Short and Mid-Term Predictors of Response to OnabotulinumtoxinA: Real-Life Experience Observational Study. Headache 2020, 60, 677–685. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Headache Classification Committee; Olesen, J.; Bousser, M.G.; Diener, H.C.; Dodick, D.; First, M.; Goadsby, P.J.; Göbel, H.; Lainez, M.J.; Lance, J.W.; et al. New appendix criteria open for a broader concept of chronic migraine. Cephalalgia 2006, 26, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808. [Google Scholar] [CrossRef] [Green Version]
- Pedraza, M.I.; de la Cruz, C.; Ruiz, M.; López-Mesonero, L.; Martínez, E.; de Lera, M.; Guerrero, Á.L. OnabotulinumtoxinA treatment for chronic migraine: Experience in 52 patients treated with the PREEMPT paradigm. Springerplus 2015, 4, 176. [Google Scholar] [CrossRef] [Green Version]
- Santos, S.; Pozo-Rosich, P. Manual de Práctica Clínica en Cefaleas. Recomendaciones Diagnóstico-Terapéuticas de la Sociedad Española de Neurología; Editorial Luzan 5: Madrid, Spain, 2020. [Google Scholar]
- Tassorelli, C.; Diener, H.C.; Dodick, D.W.; Silberstein, S.D.; Lipton, R.B.; Ashina, M.; Becker, W.J.; Ferrari, M.D.; Goadsby, P.J.; Pozo-Rosich, P.; et al. Guidelines of the International Headache Society for controlled trials of preventive treatment of chronic migraine in adults. Cephalalgia 2018, 38, 815–832. [Google Scholar] [CrossRef]
- Domínguez, C.; Pozo-Rosich, P.; Torres-Ferrús, M.; Hernández-Beltrán, N.; Jurado-Cobo, C.; González-Oria, C.; Santos, S.; Monzón, M.J.; Latorre, G.; Álvaro, L.C.; et al. OnabotulinumtoxinA in chronic migraine: Predictors of response. A prospective multicentre descriptive study. Eur. J. Neurol. 2018, 25, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Eross, E.J.; Gladstone, J.P.; Lewis, S.; Rogers, R.; Dodick, D.W. Duration of migraine is a predictor for response to botulinum toxin type A. Headache 2005, 45, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Ornello, R.; Guerzoni, S.; Baraldi, C.; Evangelista, L.; Frattale, I.; Marini, C.; Tiseo, C.; Pistoia, F.; Sacco, S. Sustained response to onabotulinumtoxin A in patients with chronic migraine: Real-life data. J. Headache Pain 2020, 21, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiano di Cola, F.; Caratozzolo, S.; Liberini, P.; Rao, R.; Padovani, A. Response Predictors in Chronic Migraine: Medication Overuse and Depressive Symptoms Negatively Impact Onabotulinumtoxin-A Treatment. Front. Neurol. 2019, 10, 678. [Google Scholar] [CrossRef] [PubMed]
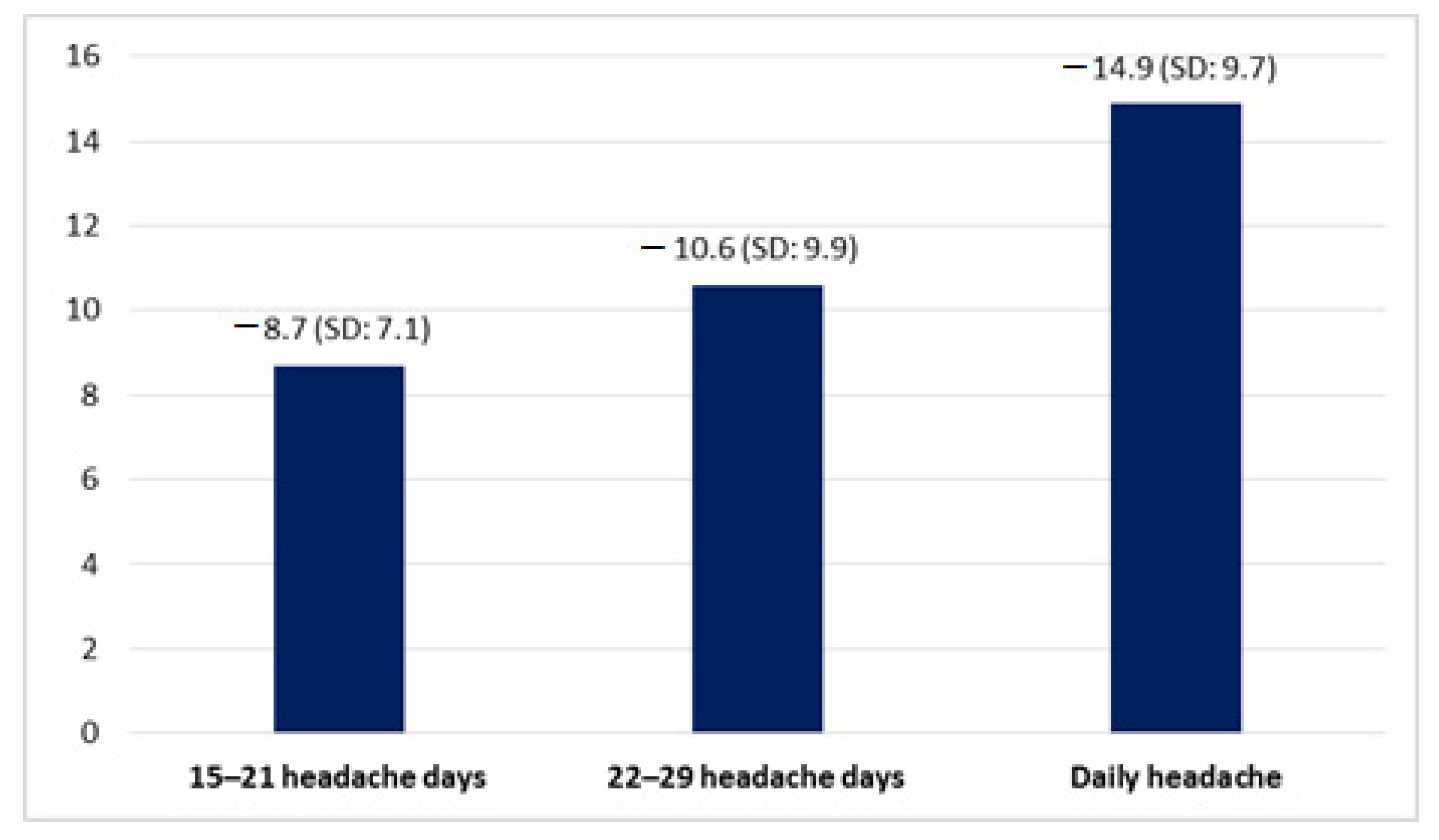

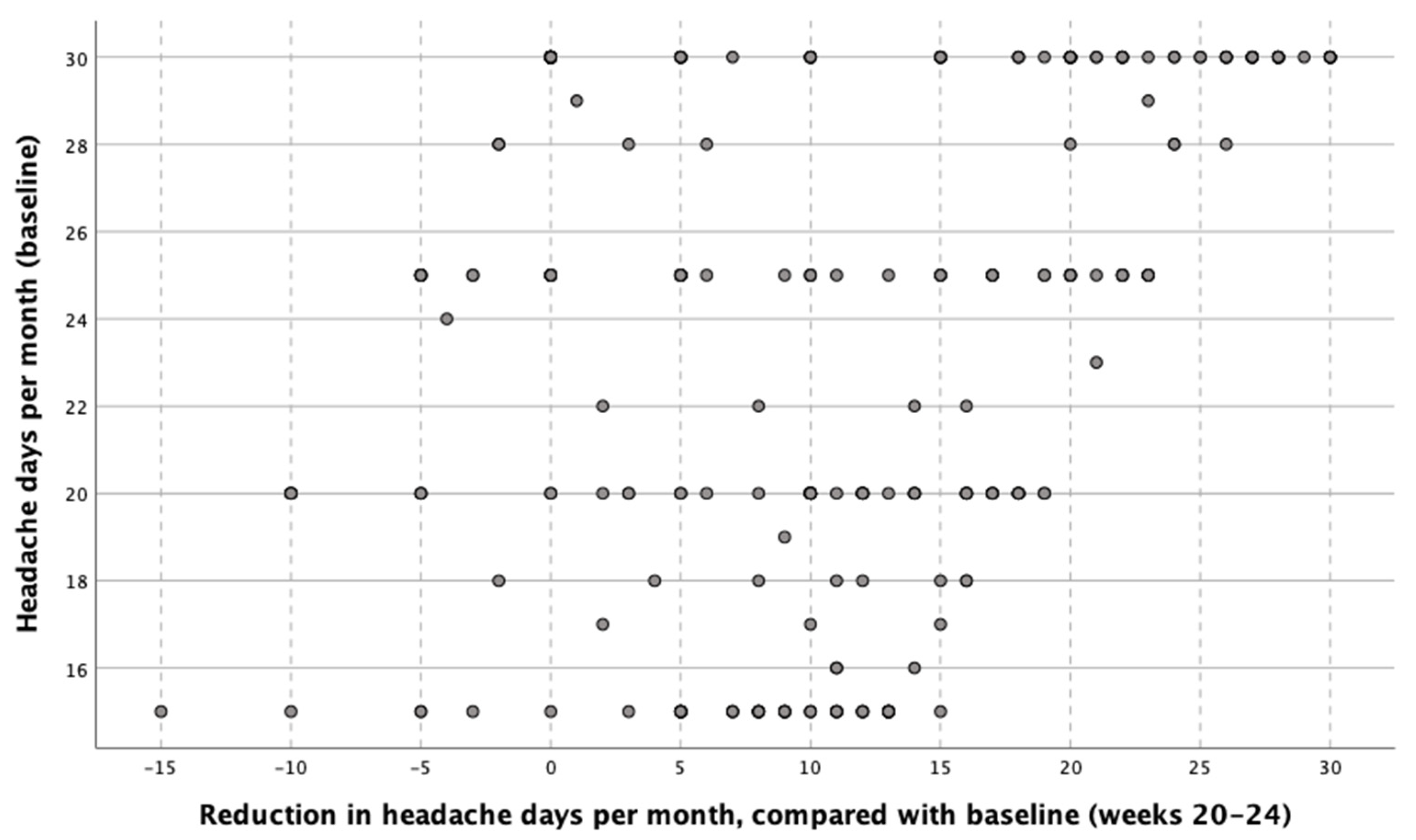
| Variable | Total Sample (n = 273) | Patients with 15–21 Headache Days/Month (n = 120) | Patients with 22–29 Headache Days/Month (n = 64) | Patients with Daily Headache (n = 89) | p-Value |
|---|---|---|---|---|---|
| 50% response rate (N) | 166 (60.8%) | 82 (68.3%) | 31 (48.4%) | 53 (59.6%) | (p = 0.030) |
| 75% response rate N (%) | 81 (29.7%) | 33 (27.5%) | 20 (31.3%) | 28 (31.5%) | (p = 0.785) |
| Reduction of headache days per month (days) | 11.2 (SD 9.7) | 8.7 (SD: 7.1) | 10.6 (SD: 9.9) | 14.9 (SD: 9.7) | (p < 0.001) |
| Reduction of severe headache days per month (days) | 6.6 (SD: 8.2) | 4.9 (SD: 6.5) | 7.3 (SD: 7.6) | 8.4 (SD: 10.2) | (p = 0.008) |
| Reduction of days of acute medication days use per month (days) | 8.1 (SD: 10.2) | 6.0 (SD: 7.9) | 8.8 (SD: 11.2) | 10.4 (SD: 11.6) | (p = 0.006) |
| Reduction of days of triptan use per month (days) | 2.1 (SD: 7.2) | 2.2 (SD: 7.1) | 3.8 (SD: 7.3) | 0.8 (SD: 7.3) | (p = 0.040) |
| Variable | Analysis | OR | IC 95% | p-Value |
|---|---|---|---|---|
| Age at the time of the first OnabotA session | Univariate Multivariate | −0.017 | (−0.060)–(0.026) | 0.436 |
| Age of migraine onset | Univariate Multivariate | 0.095 0.080 | (−0.028)–(0.218) (−0.064)–(0.224) | 0.130 0.275 |
| Years with migraine before OnabotA treatment | Univariate Multivariate | −0.025 −0.007 | (−0.123)–(0.073) (−0.122)–(0.108) | 0.615 0.902 |
| Months with CM before OnabotA treatment. | Univariate Multivariate | −0.013 −0.033 | (−0.039)–(0.012) (−0.059)–(0.007) | 0.312 0.013 |
| Sex | Univariate Multivariate | −1.040 | (−4.374)–(2.294) | 0.540 |
| Headache days per month | Univariate Multivariate | 0.474 0.540 | 0.278–0.670 0.333–0.746 | p < 0.001 p < 0.001 |
| Medication overuse | Univariate Multivariate | 0.454 −0.509 | (−2.302)–(3.210) (−3.225)–(2.206) | 0.746 0.712 |
| Number of previous preventive treatments | Univariate Multivariate | −0.047 −0.012 | (−0.766)–(0.673) (−0.703)–(0.678) | 0.898 0.972 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Pías, E.; Guerrero, Á.L.; Sierra, Á.; Trigo, J.; García-Azorín, D. Daily Headache in Chronic Migraine Is a Predictive Factor of Response in Patients Who Had Completed Three Sessions of OnabotulinumtoxinA. Toxins 2021, 13, 432. https://doi.org/10.3390/toxins13060432
Martínez-Pías E, Guerrero ÁL, Sierra Á, Trigo J, García-Azorín D. Daily Headache in Chronic Migraine Is a Predictive Factor of Response in Patients Who Had Completed Three Sessions of OnabotulinumtoxinA. Toxins. 2021; 13(6):432. https://doi.org/10.3390/toxins13060432
Chicago/Turabian StyleMartínez-Pías, Enrique, Ángel L. Guerrero, Álvaro Sierra, Javier Trigo, and David García-Azorín. 2021. "Daily Headache in Chronic Migraine Is a Predictive Factor of Response in Patients Who Had Completed Three Sessions of OnabotulinumtoxinA" Toxins 13, no. 6: 432. https://doi.org/10.3390/toxins13060432
APA StyleMartínez-Pías, E., Guerrero, Á. L., Sierra, Á., Trigo, J., & García-Azorín, D. (2021). Daily Headache in Chronic Migraine Is a Predictive Factor of Response in Patients Who Had Completed Three Sessions of OnabotulinumtoxinA. Toxins, 13(6), 432. https://doi.org/10.3390/toxins13060432





