Possibilities for the Biological Control of Mycotoxins in Food and Feed
Abstract
1. Introduction
| Mycotoxin | Main Producing Fungi | Toxic Effects | Source |
|---|---|---|---|
| Aflatoxins | Aspergillus flavus, A. parasiticus, A. aflatoxiformans | Hepatotoxicity, carcinogenicity, immunosuppression | [3] |
| Ochratoxins | Aspegillus ochraceus, Penicillium verrucosum, A. carbonarius, A. niger | Nephrotoxicity, hepatotoxicity, carcinogenicity, teratogenicity, and immunosuppression | [4] |
| Deoxynivalenol | Fusarium. graminearum (Giberella zeae), F. culmorum, F. sporotrichioides, F. tricinctum, F. Roseum, F. acuminatum | Gastrointestinal toxicity, immunodepression | [5] |
| Zearalenone | Fusarium. graminearum (Giberella zeae), F. culmorum,F. sporotrichioides, F. verticillioides (F. moniliforme), F. semitectum, F. equiseti and F. oxysporum | Reproduction toxicity | [6] |
| Fumonisins | Fusarium verticillioides, F. proliferatum | Carcinogenicity, hepatotoxicity | [7] |
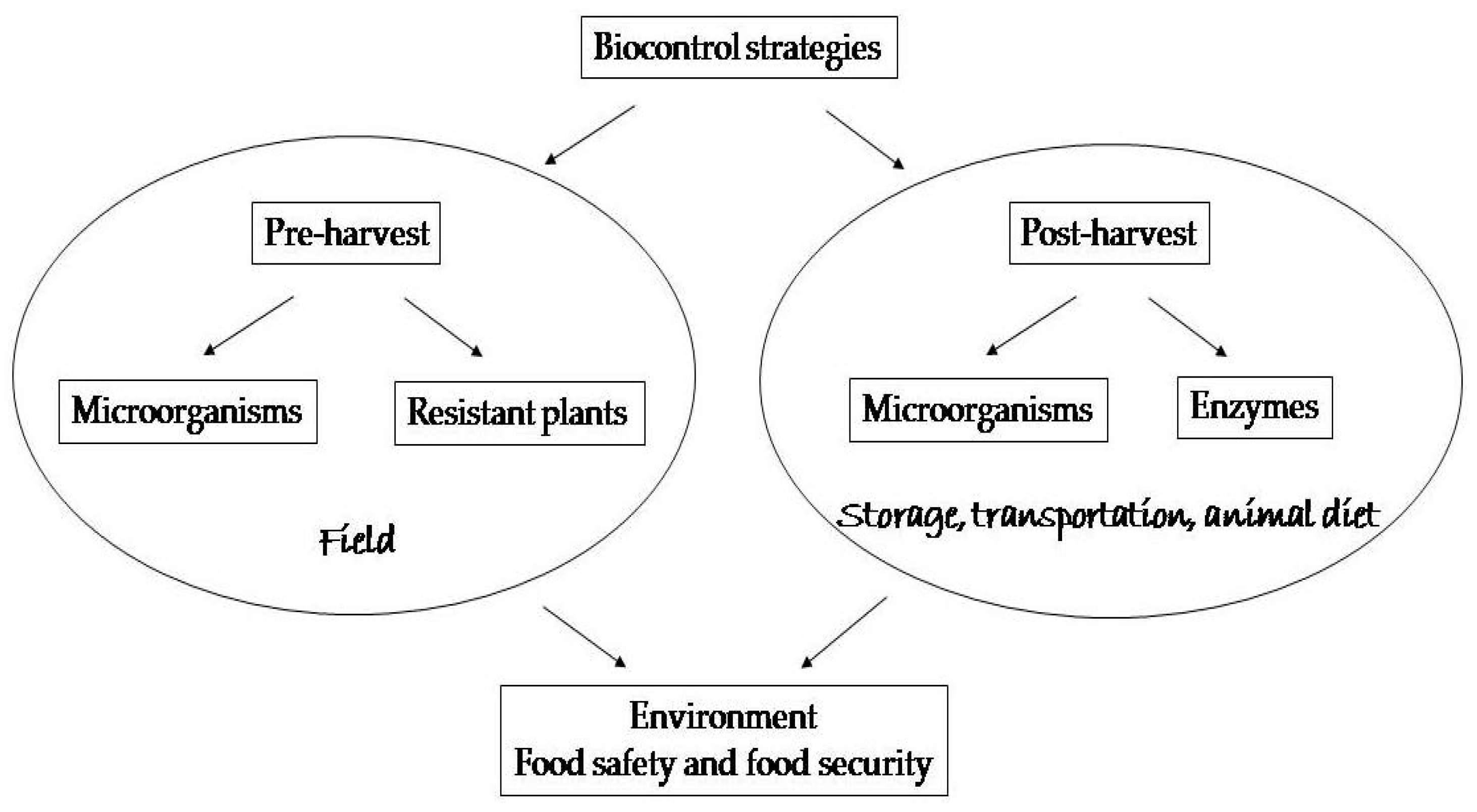
2. Pre-Harvest Biological Control
2.1. Use of Microorganisms
2.2. Use of Genetically Resistant Plants
3. Post-Harvest Biological Control
3.1. Use of Microorganisms
3.1.1. Bacteria
3.1.2. Yeast
3.1.3. Fungi
3.2. Use of Enzymes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nešić, K. Mycotoxins—Climate impact and steps to prevention based on prediction. Acta Vet. 2018, 68, 1–15. [Google Scholar] [CrossRef]
- Emmanuel, K.T.; Els, V.P.; Bart, H.; Evelyne, D.; Els, V.H.; Els, D. Carry-over of some Fusarium mycotoxins in tissues and eggs of chickens fed experimentally mycotoxin-contaminated diets. Food Chem. Toxicol. 2020, 145, 111715. [Google Scholar] [CrossRef] [PubMed]
- Benkerroum, N. Chronic and Acute Toxicities of Aflatoxins: Mechanisms of Action. Int. J. Environ. Res. Public Health 2020, 17, 423. [Google Scholar] [CrossRef]
- Heussner, A.H.; Bingle, L.E.H. Comparative Ochratoxin Toxicity: A Review of the Available Data. Toxins 2015, 7, 4253–4282. [Google Scholar] [CrossRef]
- EFSA Scientific Opinion. Risks to Human and Animal Health Related to the pre sence of deoxyni vale nol and its acetyla ted and modified forms in food and feed. EFSA J. 2017, 15, 4718. [Google Scholar] [CrossRef]
- Mahato, D.K.; Devi, S.; Pandhi, S.; Sharma, B.; Maurya, K.K.; Mishra, S.; Dhawan, K.; Selvakumar, R.; Kamle, M.; Mishra, A.; et al. Occurrence, Impact on Agriculture, Human Health, and Management Strategies of Zearalenone in Food and Feed: A Review. Toxins 2021, 13, 92. [Google Scholar] [CrossRef] [PubMed]
- Voss, K.A.; Riley, R.T. Fumonisin Toxicity and Mechanism of Action: Overview and Current Perspectives. Food Saf. 2013, 1, 2013006. [Google Scholar] [CrossRef]
- Milićević, D.; Nešić, K.; Jakšić, S. Mycotoxin Contamination of the Food Supply Chain—Implications for One Health Programme. Procedia Food Sci. 2015, 5, 187–190. [Google Scholar] [CrossRef]
- WHO—IARC. Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and My-Cotoxins. Monographs on the Evaluation of Carcinogenic Risks to Humans 1993, 56. Available online: https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Some-Naturally-Occurring-Substances-Food-Items-And-Constituents-Heterocyclic-Aromatic-Amines-And-Mycotoxins-1993 (accessed on 18 January 2021).
- WHO—IARC. Fumonisin B1. Monographs on the Evaluation of Carcinogenic Risks to Humans 2002, 82. Available online: https://publications.iarc.fr/Book-And-Report-Series/Iarc-Monographs-On-The-Identification-Of-Carcinogenic-Hazards-To-Humans/Some-Traditional-Herbal-Medicines-Some-Mycotoxins-Naphthalene-And-Styrene-2002 (accessed on 18 January 2021).
- Eskola, M.; Kos, G.; Elliott, C.T.; HajšLová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2020, 60, 2773–2789. [Google Scholar] [CrossRef]
- Mastanjević, K.; Lukinac, J.; Jukić, M.; Šarkanj, B.; Krstanović, V.; Mastanjević, K. Multi-(myco)toxins in Malting and Brewing By-Products. Toxins 2019, 11, 30. [Google Scholar] [CrossRef]
- Mehta, R.; Shetty, S.A.; Young, M.F.; Ryan, P.B.; Rangiah, K. Quantification of aflatoxin and ochratoxin contamination in animal milk using UHPLC-MS/SRM method: A small-scale study. J. Food Sci. Technol. 2021, 58, 1–12. [Google Scholar] [CrossRef]
- Souza, R.; Fernández, P.; Muela, A.; Cesio, M.V.; Heinzen, H.; Pareja, L. Development of a Methodology for the Simultaneous Analysis of Multiclass Contaminants in Milk. Food Anal. Methods 2021, 14, 1–12. [Google Scholar] [CrossRef]
- Moradi, M.; Azizi-Lalabadi, M.; Motamedi, P.; Sadeghi, E. Electrochemical determination of T 2 toxin by graphite/polyacrylonitrile nanofiber electrode. Food Sci. Nutr. 2021, 9, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Su, W.-H.; Yang, C.; Dong, Y.H.; Johnson, R.; Page, R.; Szinyei, T.; Hirsch, C.D.; Steffenson, B.J. Hyperspectral imaging and improved feature variable selection for automated determination of deoxynivalenol in various genetic lines of barley kernels for resistance screening. Food Chem. 2021, 343, 128507. [Google Scholar] [CrossRef] [PubMed]
- Horky, P.; Skalickova, S.; Caslavova, I.; Deering, A.J.; Nevrkla, P.; Slama, P.; Trojan, V.; Skladanka, J. Effect of fungicidal treatment and storage condition on content of selected mycotoxins in barley. Kvasny Prumysl 2018, 64, 212–216. [Google Scholar] [CrossRef]
- Skladanka, J.; Adam, V.; Zitka, O.; Mlejnkova, V.; Kalhotka, L.; Horky, P.; Konecna, K.; Hodulikova, L.; Knotova, D.; Balabanova, M.; et al. Comparison of Biogenic Amines and Mycotoxins in Alfalfa and Red Clover Fodder Depending on Additives. Int. J. Environ. Res. Public Health 2017, 14, 418. [Google Scholar] [CrossRef] [PubMed]
- Vaičiulienė, G.; Bakutis, B.; Jovaišienė, J.; Falkauskas, R.; Gerulis, G.; Baliukonienė, V. Origanum vulgare and Thymus vulgaris Extract Usability to Improve Silage Hygienic Quality and Reduce Mycotoxin Concentrations. J. Microbiol. Biotechnol. 2020, 30, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Nešić, K.; Ivanović, S.; Nešić, V. Fusarial Toxins: Secondary Metabolites of Fusarium Fungi. Rev. Environ. Contam. Toxicol. 2014, 228, 101–120. [Google Scholar] [CrossRef]
- Conte, G.; Fontanelli, M.; Galli, F.; Cotrozzi, L.; Pagni, L.; Pellegrini, E. Mycotoxins in Feed and Food and the Role of Ozone in Their Detoxification and Degradation: An Update. Toxins 2020, 12, 486. [Google Scholar] [CrossRef]
- Agriopoulou, S.; Stamatelopoulou, E.; Varzakas, T. Advances in Occurrence, Importance, and Mycotoxin Control Strategies: Prevention and Detoxification in Foods. Foods 2020, 9, 137. [Google Scholar] [CrossRef]
- Jouany, J. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed. Sci. Technol. 2007, 137, 342–362. [Google Scholar] [CrossRef]
- Bhattacharjee, R.; Dey, U. An overview of fungal and bacterial biopesticides to control plant pathogens/diseases. Afr. J. Microbiol. Res. 2014, 8, 1749–1762. [Google Scholar] [CrossRef]
- Mukhopadhyay, R.; Kumar, D. Trichoderma: A beneficial antifungal agent and insights into its mechanism of biocontrol potential. Egypt. J. Biol. Pest Control 2020, 30, 133. [Google Scholar] [CrossRef]
- Dorner, J.W.; Cole, R.J. Effect of application of nontoxigenic strains of Aspergillus flavus and A. parasiticus on subsequent aflatoxin contamination of peanuts in storage. J. Stored Prod. Res. 2002, 38, 329–339. [Google Scholar] [CrossRef]
- Sarrocco, S.; Vannacci, G. Preharvest application of beneficial fungi as a strategy to prevent postharvest mycotoxin contamination: A review. Crop. Prot. 2018, 110, 160–170. [Google Scholar] [CrossRef]
- Senghor, L.A.; Ortega-Beltran, A.; Atehnkeng, J.; Callicott, K.A.; Cotty, P.J.; Bandyopadhyay, R. The Atoxigenic Biocontrol Product Aflasafe SN01 Is a Valuable Tool to Mitigate Aflatoxin Contamination of Both Maize and Groundnut Cultivated in Senegal. Plant Dis. 2020, 104, 510–520. [Google Scholar] [CrossRef] [PubMed]
- Cleveland, T.E.; Dowd, P.F.; Desjardins, A.E.; Bhatnagar, D.; Cotty, P.J. United States Department of Agriculture—Agricultural Research Service research on pre-harvest prevention of mycotoxins and mycotoxigenic fungi in US crops. Pest Manag. Sci. 2003, 59, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Luongo, L.; Galli, M.; Corazza, L.; Meekes, E.; Haas, L.D.; Van Der Plas, C.L.; Köhl, J. Potential of fungal antagonists for biocontrol of Fusarium spp. in wheat and maize through competition in crop debris. Biocontrol Sci. Technol. 2005, 15, 229–242. [Google Scholar] [CrossRef]
- Sarrocco, S.; Mauro, A.; Battilani, P. Use of Competitive Filamentous Fungi as an Alternative Approach for Mycotoxin Risk Reduction in Staple Cereals: State of Art and Future Perspectives. Toxins 2019, 11, 701. [Google Scholar] [CrossRef]
- Kagot, V.; Okoth, S.; De Boevre, M.; De Saeger, S. Biocontrol of Aspergillus and Fusarium Mycotoxins in Africa: Benefits and Limitations. Toxins 2019, 11, 109. [Google Scholar] [CrossRef]
- Bacon, C.W.; Yates, I.E.; Hinton, D.M.; Meredith, F. Biological control of Fusarium moniliforme in maize. Environ. Health Perspect. 2001, 109, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Dowd, P.F. Insect Management to Facilitate Preharvest Mycotoxin Management. J. Toxicol. Toxin Rev. 2003, 22, 327–350. [Google Scholar] [CrossRef]
- Yu, J.-H.; Keller, N. Regulation of Secondary Metabolism in Filamentous Fungi. Annu. Rev. Phytopathol. 2005, 43, 437–458. [Google Scholar] [CrossRef]
- Bhatnagar, D.; Rajasekaran, K.; Payne, G.A.; Brown, R.I.; Yu, J.; Cleveland, T.E. The ‘omics’ tools: Genomics, proteomics, metabolomics and their potential for solving the aflatoxin contamination problem. World Mycotoxin J. 2008, 1, 3–12. [Google Scholar] [CrossRef]
- Wu, F. Mycotoxin Reduction in Bt Corn: Potential Economic, Health, and Regulatory Impacts. Transgenic Res. 2006, 15, 277–289. [Google Scholar] [CrossRef]
- Yu, J.; Hennessy, D.A.; Wu, F. The Impact of Bt Corn on Aflatoxin-Related Insurance Claims in the United States. Sci. Rep. 2020, 10, 10046. [Google Scholar] [CrossRef] [PubMed]
- Munkvold, G.P. Cultural and geneticapproaches tomanagingmycotoxins inmaize. Annu. Rev. Phytopathol. 2003, 41, 99–116. [Google Scholar] [CrossRef] [PubMed]
- Saharan, V.; Jain, D.; Pareek, S.; Pal, A.; Kumaraswamy, R.V.; Jakhar, S.K.; Singh, M. Viral, Fungal and Bacterial Disease Resistance in Transgenic Plants. In Advances in Plant Breeding Strategies: Agronomic, Abiotic and Biotic Stress Traits; Al-Khayri, J., Jain, S., Johnson, D., Eds.; Springer: Cham, Switzerland, 2016; pp. 627–656. [Google Scholar] [CrossRef]
- Krattinger, S.G.; Keller, B. Molecular genetics and evolution of disease resistance in cereals. New Phytol. 2016, 212, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Afshar, P.; Shokrzadeh, M.; Raeisi, S.N.; Ghorbani-HasanSaraei, A.; Nasiraii, L.R. Aflatoxins biodetoxification strategies based on probiotic bacteria. Toxicon 2020, 178, 50–58. [Google Scholar] [CrossRef]
- Taheur, F.B.; Kouidhi, B.; Al Qurashi, Y.M.A.; Salah-Abbès, J.B.; Chaieb, K. Review: Biotechnology of mycotoxins detoxification using microorganisms and enzymes. Toxicon 2019, 160, 12–22. [Google Scholar] [CrossRef]
- Schatzmayr, G.; Zehner, F.; Täubel, M.; Schatzmayr, D.; Klimitsch, A.; Loibner, A.P.; Binder, E.M. Microbiologicals for deactivating mycotoxins. Mol. Nutr. Food Res. 2006, 50, 543–551. [Google Scholar] [CrossRef]
- Umesha, S.; Manukumar, H.M.G.; Chandrasekhar, B.; Shivakumara, P.; Kumar, J.S.; Raghava, S.; Avinash, P.; Shirin, M.; Bharathi, T.R.; Rajini, S.B.; et al. Aflatoxins and food pathogens: Impact of biologically active aflatoxins and their control strategies. J. Sci. Food Agric. 2017, 97, 1698–1707. [Google Scholar] [CrossRef] [PubMed]
- Foroud, N.A.; Baines, D.; Gagkaeva, T.Y.; Thakor, N.; Badea, A.; Steiner, B.; Bürstmayr, M.; Bürstmayr, H. Trichothecenes in Cereal Grains—An Update. Toxins 2019, 11, 634. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Scientific Opinion on the safety and efficacy of micro-organism DSM 11798 when used as a technological feed additive for pigs. EFSA J. 2013, 11, 3203. [Google Scholar] [CrossRef]
- EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP); Rychen, G.; Aquilina, G.; Azimonti, G.; Bampidis, V.; Bastos, M.D.L.; Bories, G.; Chesson, A.; Cocconcelli, P.S.; Flachowsky, G.; et al. Safety and efficacy of microorganism DSM 11798 as a technological additive for all avian species. EFSA J. 2017, 15, e04676. [Google Scholar] [CrossRef] [PubMed]
- Commission Implementing Regulation (EU) No 1016/2013 of 23 October 2013 Concerning the Authorisation of a Preparation of a Micro-organism Strain DSM 11798 of the Coriobacteriaceae Family as a Feed Additive for Pigs. Available online: http://data.europa.eu/eli/reg_impl/2013/1016/oj (accessed on 6 January 2021).
- Commission Implementing Regulation (EU) 2017/930 of 31 May 2017 Concerning the Authorisation of a Preparation of a Mi-Croorganism Strain DSM 11798 of the Coriobacteriaceae Family as a Feed Additive for All Avian Species and Amending Imple-menting Regulation (EU) No 1016/2013. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32017R0930&from=EN (accessed on 6 January 2021).
- Hassan, Y.I.; Zhou, T. Addressing the mycotoxin deoxynivalenol contamination with soil-derived bacterial and enzymatic transformations targeting the C3 carbon. World Mycotoxin J. 2018, 11, 101–112. [Google Scholar] [CrossRef]
- Gao, X.; Mu, P.; Wen, J.; Sun, Y.; Chen, Q.; Deng, Y. Detoxification of trichothecene mycotoxins by a novel bacterium, Eggerthella sp. DII-9. Food Chem. Toxicol. 2018, 112, 310–319. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, X.; Li, J. Updating techniques on controlling mycotoxins—A review. Food Control 2018, 89, 123–132. [Google Scholar] [CrossRef]
- Adebo, O.A.; Kayitesi, E.; Njobeh, P.B. Reduction of Mycotoxins during Fermentation of Whole Grain Sorghum to Whole Grain Ting (a Southern African Food). Toxins 2019, 11, 180. [Google Scholar] [CrossRef]
- Muhialdin, B.J.; Saari, N.; Meor Hussin, A.S. Review on the Biological Detoxification of Mycotoxins Using Lactic Acid Bacteria to Enhance the Sustainability of Foods Supply. Molecules 2020, 25, 2655. [Google Scholar] [CrossRef]
- Perczak, A.; Goliński, P.; Bryła, M.; Waśkiewicz, A. The efficiency of lactic acid bacteria against pathogenic fungi and mycotoxins. Arch. Ind. Hyg. Toxicol. 2018, 69, 32–45. [Google Scholar] [CrossRef]
- Solis-Cruz, B.; Hernandez-Patlan, D.; Hargis, B.; Tellez, G. Control of Aflatoxicosis in Poultry Using Probiotics and Polymers. In Mycotoxins—Impact and Management Strategies; Njobeh, P.B., Stepman, F., Eds.; IntechOpen: London, UK, 2019. [Google Scholar]
- Juodeikiene, G.; Bartkiene, E.; Cernauskas, D.; Cizeikiene, D.; Zadeike, D.; Lele, V.; Bartkevics, V. Antifungal activity of lactic acid bacteria and their application for Fusarium mycotoxin reduction in malting wheat grains. LWT Food Sci. Technol. 2018, 89, 307–314. [Google Scholar] [CrossRef]
- Pfliegler, W.P.; Pusztahelyi, T.; Pócsi, I. Mycotoxins—Prevention and decontamination by yeasts. J. Basic Microbiol. 2015, 55, 805–818. [Google Scholar] [CrossRef]
- El-Tarabily, K.A.; Sivasithamparam, K. Potential of yeasts as biocontrol agents of soil-borne fungal plant pathogens and as plant growth promoters. Mycoscience 2006, 47, 25–35. [Google Scholar] [CrossRef]
- Tilocca, B.; Balmas, V.; Hassan, Z.U.; Jaoua, S.; Migheli, Q. A proteomic investigation of Aspergillus carbonarius exposed to yeast volatilome or to its major component 2-phenylethanol reveals major shifts in fungal metabolism. Int. J. Food Microbiol. 2019, 306, 108265. [Google Scholar] [CrossRef] [PubMed]
- Farbo, M.G.; Urgeghe, P.P.; Fiori, S.; Marcello, A.; Oggiano, S.; Balmas, V.; Hassan, Z.U.; Jaoua, S.; Migheli, Q. Effect of yeast volatile organic compounds on ochratoxin A-producing Aspergillus carbonarius and A. ochraceus. Int. J. Food Microbiol. 2018, 284, 1–10. [Google Scholar] [CrossRef]
- Druvefors, U. Yeast Biocontrol of Grain Spoilage Mold. Ph.D. Thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden, 2004. [Google Scholar]
- Díaz, M.A.; Pereyra, M.M.; Picón-Montenegro, E.; Meinhardt, F.; Dib, J.F. Killer Yeasts for the Biological Control of Postharvest Fungal Crop Diseases. Microorganisms 2020, 8, 1680. [Google Scholar] [CrossRef]
- Zohri, A.A.; Abdel-Kareem, M.M. Four strains of yeasts: As effective biocontrol agents against both growth and mycotoxins formation by selected 11 toxigenic fungi. GARJM 2018, 7, 132–135. [Google Scholar]
- Liu, Y.; Chang, J.; Wang, P.; Yin, Q.; Huang, W.; Liu, C.; Bai, X.; Zhu, Q.; Gao, T.; Zhou, P. Effects of Saccharomyces cerevisiae on alleviating cytotoxicity of porcine jejunal epithelia cells induced by deoxynivalenol. AMB Express 2019, 9, 137. [Google Scholar] [CrossRef]
- Mendieta, C.R.; Gómez, G.V.; Del Río, J.C.G.; Cuevas, A.C.; Arce, J.M.; Ávila, E.G. Effect of the Addition of Saccharomyces Cerevisiae Yeast Cell Walls to Diets with Mycotoxins on the Performance and Immune Responses of Broilers. J. Poult. Sci. 2018, 55, 38–46. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, M.; Wu, C.; Peng, B. Physical adsorption of patulin by Saccharomyces cerevisiae during fermentation. J. Food Sci. Technol. 2019, 56, 2326–2331. [Google Scholar] [CrossRef] [PubMed]
- Jakopović, Ž.; Čiča, K.H.; Mrvčić, J.; Pucić, I.; Čanak, I.; Frece, J.; Pleadin, J.; Stanzer, D.; Zjalic, S.; Markov, K. Properties and Fermentation Activity of Industrial Yeasts Saccharomyces cerevisiae, S. uvarum, Candida utilis and Kluyveromyces marxianus Exposed to AFB1, OTA and ZEA. Food Technol. Biotechnol. 2018, 56, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Wang, J.; Zhang, H.; Li, C.; Zhang, X. Ochratoxin A is degraded by Yarrowia lipolytica and generates non-toxic degradation products. World Mycotoxin J. 2016, 9, 269–278. [Google Scholar] [CrossRef]
- Li, X.; Tang, H.; Yang, C.; Meng, X.; Liu, B. Detoxification of mycotoxin patulin by the yeast Rhodotorula mucilaginosa. Food Control 2019, 96, 47–52. [Google Scholar] [CrossRef]
- Horn, B.W.; Dorner, J.W. Effect of nontoxigenic Aspergillus flavus and A. parasiticus on aflatoxin contamination of wounded peanut seeds inoculated with agricultural soil containing natural fungal populations. Biocontrol. Sci. Technol. 2009, 19, 249–262. [Google Scholar] [CrossRef]
- Alberts, J.F.; Lilly, M.; Rheeder, J.P.; Burger, H.-M.; Shephard, G.S.; Gelderblom, W.C.A. Technological and community-based methods to reduce mycotoxin exposure. Food Control 2017, 73, 101–109. [Google Scholar] [CrossRef]
- Hackbart, H.C.S.; Machado, A.R.; Christ-Ribeiro, A.; Prietto, L.; Badiale-Furlong, E. Reduction of aflatoxins by Rhizopus oryzae and Trichoderma reesei. Mycotoxin Res. 2014, 30, 141–149. [Google Scholar] [CrossRef]
- Błaszczyk, L.; Basińska-Barczak, A.; Ćwiek-Kupczyńska, H.; Gromadzka, K.; Popiel, D.; Stępień, Ł. Suppressive Effect of Trichoderma spp. on Toxigenic Fusarium Species. Pol. J. Microbiol. 2017, 66, 85–100. [Google Scholar] [CrossRef] [PubMed]
- Wagacha, J.; Muthomi, J. Mycotoxin problem in Africa: Current status, implications to food safety and health and possible management strategies. Int. J. Food Microbiol. 2008, 124, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pirgozliev, S.R.; Edwards, S.G.; Hare, M.C.; Jenkinson, P. Strategies for the Control of Fusarium Head Blight in Cereals. Eur. J. Plant Pathol. 2003, 109, 731–742. [Google Scholar] [CrossRef]
- Venkatesh, N.; Keller, N.P. Mycotoxins in Conversation with Bacteria and Fungi. Front. Microbiol. 2019, 10, 403. [Google Scholar] [CrossRef] [PubMed]
- Utermark, J.; Karlovsky, P. Role of Zearalenone Lactonase in Protection of Gliocladium roseum from Fungitoxic Effects of the Mycotoxin Zearalenone. Appl. Environ. Microbiol. 2006, 73, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Vekiru, E.; Hametner, C.; Mitterbauer, R.; Rechthaler, J.; Adam, G.; Schatzmayr, G.; Krska, R.; Schuhmacher, R. Cleavage of Zearalenone by Trichosporon mycotoxinivorans to a Novel Nonestrogenic Metabolite. Appl. Environ. Microbiol. 2010, 76, 2353–2359. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.; Zablotowicz, R.; Horn, B.; Phillips, N.; Johnson, B.; Jin, X.; Abel, C. Comparison of major biocontrol strains of non-aflatoxigenicAspergillus flavusfor the reduction of aflatoxins and cyclopiazonic acid in maize. Food Addit. Contam. Part A 2011, 28 Pt A, 198–208. [Google Scholar] [CrossRef]
- Okoth, S.; De Boevre, M.; Vidal, A.; Diana Di Mavungu, J.; Landschoot, S.; Kyallo, M.; Njuguna, J.; Harvey, J.; De Saeger, S. Genetic and Toxigenic Variability within Aspergillus flavus Population Isolated from Maize in Two Diverse Environments in Kenya. Front. Microbiol. 2018, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Su, R.; Yin, R.; Lai, D.; Wang, M.; Liu, Y.; Zhou, L. Detoxification of Mycotoxins through Biotransformation. Toxins 2020, 12, 121. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Paterson, R.R.; Venâncio, A. Biodegradation of Ochratoxin A for Food and Feed Decontamination. Toxins 2010, 2, 1078–1099. [Google Scholar] [CrossRef]
- Burgess, K.M.; Renaud, J.B.; McDowell, T.; Sumarah, M.W. Mechanistic Insight into the Biosynthesis and Detoxification of Fumonisin Mycotoxins. ACS Chem. Biol. 2016, 11, 2618–2625. [Google Scholar] [CrossRef]
- Chang, X.; Wu, Z.; Wu, S.; Dai, Y.; Sun, C. Degradation of ochratoxin A byBacillus amyloliquefaciensASAG1. Food Addit. Contam. Part A 2015, 32, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Dobritzsch, D.; Wang, H.; Schneider, G.; Yu, S. Structural and functional characterization of ochratoxinase, a novel mycotoxin-degrading enzyme. Biochem. J. 2014, 462, 441–452. [Google Scholar] [CrossRef]
- Hartinger, D.; Schwartz, H.; Hametner, C.; Schatzmayr, G.; Haltrich, D.; Moll, W.-D. Enzyme characteristics of aminotransferase FumI of Sphingopyxis sp. MTA144 for deamination of hydrolyzed fumonisin B1. Appl. Microbiol. Biotechnol. 2011, 91, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Heinl, S.; Hartinger, D.; Thamhesl, M.; Vekiru, E.; Krska, R.; Schatzmayr, G.; Moll, W.D.; Grabherr, R. Degradation of fumonisin B1 by the consecutive action of two bacterial enzymes. J. Biotechnol. 2010, 145, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.C.; Jackson, C.J.; Tattersall, D.B.; French, N.; Peat, T.S.; Newman, J.; Briggs, L.J.; Lapalikar, G.V.; Campbell, P.M.; Scott, C.; et al. Identification and characterization of two families of F420H2-dependent reductases from Mycobacteria that catalyse aflatoxin degradation. Mol. Microbiol. 2010, 78, 561–575. [Google Scholar] [CrossRef]
- Wang, J.; Ogata, M.; Hirai, H.; Kawagishi, H. Detoxification of aflatoxin B1 by manganese peroxidase from the white-rot fungus Phanerochaete sordida YK-624. FEMS Microbiol. Lett. 2010, 314, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Qin, X.; Guo, Y.; Zhang, Q.; Ma, Q.; Ji, C.; Zhao, L. Enzymatic degradation of deoxynivalenol by a novel bacterium, Pelagibacterium halotolerans ANSP101. Food Chem. Toxicol. 2020, 140, 111276. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Ko, T.-P.; Yang, Y.; Zheng, Y.; Chen, C.-C.; Zhu, Z.; Huang, C.-H.; Zeng, Y.-F.; Huang, J.-W.; Wang, A.H.-J.; et al. Crystal structure and substrate-binding mode of the mycoestrogen-detoxifying lactonase ZHD from Clonostachys rosea. RSC Adv. 2014, 4, 62321–62325. [Google Scholar] [CrossRef]
- Vekiru, E.; Frühauf, S.; Hametner, C.; Schatzmayr, G.; Krska, R.; Moll, W.D.; Schuhmacher, R. Isolation and characterisation of enzymatic zearalenone hydrolysis reaction products. World Mycotoxin J. 2016, 9, 353–363. [Google Scholar] [CrossRef]
- Loi, M.; Fanelli, F.; Liuzzi, V.C.; Logrieco, A.F.; Mulè, G. Mycotoxin Biotransformation by Native and Commercial Enzymes: Present and Future Perspectives. Toxins 2017, 9, 111. [Google Scholar] [CrossRef]
- Ferrara, M.; Haidukowski, M.; D’Imperio, M.; Parente, A.; De Angelis, E.; Monaci, L.; Logrieco, A.F.; Mulè, G. New insight into microbial degradation of mycotoxins during anaerobic digestion. Waste Manag. 2020, 119, 215–225. [Google Scholar] [CrossRef]
- Lyagin, I.; Efremenko, E. Enzymes for Detoxification of Various Mycotoxins: Origins and Mechanisms of Catalytic Action. Molecules 2019, 24, 2362. [Google Scholar] [CrossRef]
- Čolović, R.; Puvača, N.; Cheli, F.; Avantaggiato, G.; Greco, D.; Đuragić, O.; Kos, J.; Pinotti, L. Decontamination of Mycotoxin-Contaminated Feedstuffs and Compound Feed. Toxins 2019, 11, 617. [Google Scholar] [CrossRef]
- Regulation (EC) No 1332/2008 of the European Parliament and of the Council of 16 December 2008 on Food Enzymes and Amending Council Directive 83/417/EEC, Council Regulation (EC) No 1493/1999, Directive 2000/13/EC, Council Directive 2001/112/EC and Regulation (EC) No 258/97. Available online: http://data.europa.eu/eli/reg/2008/1332/oj (accessed on 13 January 2021).
- Commission Regulation 2015/786/EU Defining Acceptability Criteria for Detoxification Processes Applied to Products In-tended for Animal Feed as Provided for in Directive 2002/32/EC of the European Parliament and of the Council. Available online: http://data.europa.eu/eli/reg/2015/786/oj (accessed on 13 January 2021).
- Commission Implementing Regulation (EU) No 1115/2014 of 21 October 2014 concerning the Authorisation of a Preparation of Fumonisin Esterase Produced by Komagataella pastoris (DSM 26643) as a Feed Additive for Pigs. Available online: http://data.europa.eu/eli/reg_impl/2014/1115/oj (accessed on 14 January 2021).
- Commission Implementing Regulation (EU) 2017/913 of 29 May 2017 Concerning the Authorisation of a Preparation of Fumonisin Esterase Produced by Komagataella pastoris (DSM 26643) as a Feed Additive for All Avian Species. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32017R0913&from=CS (accessed on 14 January 2021).
- Commission Implementing Regulation (EU) 2018/1568 of 18 October 2018 Concerning the Authorisation of a Preparation of Fumonisin Esterase Produced by Komagataella Phaffii (DSM 32159) as a Feed Additive for All Pigs and All Poultry Species. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32018R1568&from=EN (accessed on 14 January 2021).
- EFSA. Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Safety and Efficacy of Fumonisin Esterase from Komagataella Phaffii DSM 32159 as a Feed Additive for All Animal Species. EFSA J. 2020, 18, e06207. [Google Scholar] [CrossRef]
- Regulation (EC) No 1831/2003 of the European Parliament and of the Council of 22 September 2003 on Additives for Use in Animal Nutrition. OJEU 2003, L 268, 29–43. Available online: http://data.europa.eu/eli/reg/2003/1831/oj (accessed on 27 January 2021).
| Mycotoxins | Microorganisms |
|---|---|
| Aflatoxins | Lactobacillus plantarum LOCK 0945, L. brevis LOCK 0944, L. paracasei LOCK 0920, L. kefiri, Bacillus pumilus, Bacillus subtilis ANSB060, Kazachstania servazzii, Acetobacter syzygii, Rhodococcus erythropolis, Pseudomonas putida, Mycobacterium fluoranthenivorans sp. nov. DSM 44556T, Streptomyces lividans TK 24, Saccharomyces cerevisiae, Pichia anomala, Fusarium aurantiacum strain NRRL-B-184, Pseudomonas putida, Mycobacterium fluoranthenivorans sp. nov. DSM 44556T, Streptomyces lividans TK 24, Flavobacterium aurantiacum |
| Ochratoxin A | L. acidophilus VM 20, L. bulgaricus, L. helveticus, L. rhamnosus GG, B. lichniformis, B. subtilis, Bifidobacterium animalis VM 12, Brevibacterium, Cupriavidus basilensis ŐR16, Pediococcus parvulus, B. amyloliquefaciens ASAG1, S. cerevisiae O11, S. bayanus, Yarrowia lipolytica |
| Zearalenone | B. licheniformis CK1, B. pumilus ES-21, B. subtilis, L. mucosae lm4208, L. rhamnosus, P. otitidis TH-N1, Rhodococcus, Lysinibacillus sp., Geobacillus and Tepidimicrobium |
| Trichothecenes (DON, T-2/HT-2) | Nocardioides and Devosia, Lactobacillus sakei KTU05-6, Pediococcus acidilactici KTU05-7, Pediococcus pentosaceus KTU05-8, KTU05-09 and KTU05-10, Eggerthella sp. DII-9 |
| Mycotoxin | Enzyme | Producer |
|---|---|---|
| Aflatoxin | Aflatoxin oxidase enzyme (AFO) (EC 1.1) | Armillariella tabescens |
| Peroxidase (EC 1.11.1.7) | Horseradish (Armoracia rusticana) | |
| Laccase (EC 1.10.3.2) | Trametes versicolor (Sigma-Aldrich, St. Louis, MO, USA) | |
| Laccase (EC 1.10.3.2) | Streptomyces coelicor | |
| F420H2-dependent reductases (E.C. 1.5.8) | Mycobacterium smegmatis | |
| Mn peroxidase (EC 1.11.1.7) | Pleurotus ostreatus | |
| Aflatoxin degradation enzyme | Pleurotus ostreatus | |
| Myxobacteria aflatoxin degrading enzyme (MADE) | Myxococcus fulvus ANSM068 | |
| Laccase (lac2) (EC 1.10.3.2) | Pleurotus pulmonarius (ITEM 17144) | |
| Ery4 | Pleurotus eryngii (PS419) | |
| Fumonisin | Carboxylesterase and aminotransferase (E.C. 3.1.1, E.C. 2.6.1) | Sphingomonas sp. ATCC55552 |
| Carboxylesterase B and aminotransferase (E.C. 3.1.1, E.C. 2.6.1/FJ426269.1) | Sphingopyxis sp. MTA144 | |
| Fumonisin esterase (E.C. 3.1.1.87) | Sphingopyxis sp. MTA144 | |
| Trichothecenes | Cytochrome P450 system (Ddna + Kdx + KdR) (E.C. 1.14 AB744215.1 AB744217.1) (DON; NIV) | Sphingomonas sp. strain KSM1 |
| UDP-glycosyltransferase (AC006282) | Arabidopsis thaliana | |
| Zearalenone | Laccase (EC 1.10.3.2) | Trametes versicolor (Sigma-Aldrich, USA) |
| laccase (EC 1.10.3.2) | Streptomyces coelicolor | |
| Lactono hydrolase (E.C. 3.1.1) | Clonostachys rosea | |
| 2cys-peroxiredoxin (EC 1.11.1.15) | Acinetobacter sp. SM04 | |
| Ochratoxin | Carboxypeptidase A: CPA (EC 3.4.24) | Bovine pancreas |
| Carboxypeptidase Y: CPY (EC 3.4.16) | Saccharomyces cerevisiae | |
| Lipase (EC 3.1) Protease A (EC 3.4) Amidase 2 (EC 3.5) | Aspergillus niger |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nešić, K.; Habschied, K.; Mastanjević, K. Possibilities for the Biological Control of Mycotoxins in Food and Feed. Toxins 2021, 13, 198. https://doi.org/10.3390/toxins13030198
Nešić K, Habschied K, Mastanjević K. Possibilities for the Biological Control of Mycotoxins in Food and Feed. Toxins. 2021; 13(3):198. https://doi.org/10.3390/toxins13030198
Chicago/Turabian StyleNešić, Ksenija, Kristina Habschied, and Krešimir Mastanjević. 2021. "Possibilities for the Biological Control of Mycotoxins in Food and Feed" Toxins 13, no. 3: 198. https://doi.org/10.3390/toxins13030198
APA StyleNešić, K., Habschied, K., & Mastanjević, K. (2021). Possibilities for the Biological Control of Mycotoxins in Food and Feed. Toxins, 13(3), 198. https://doi.org/10.3390/toxins13030198







