Reducing Ochratoxin A Content in Grape Pomace by Different Methods
Abstract
1. Introduction
2. Results and Discussion
2.1. Effects of Thermal Pressure Treatment on the OTA Contents of Grape Pomace
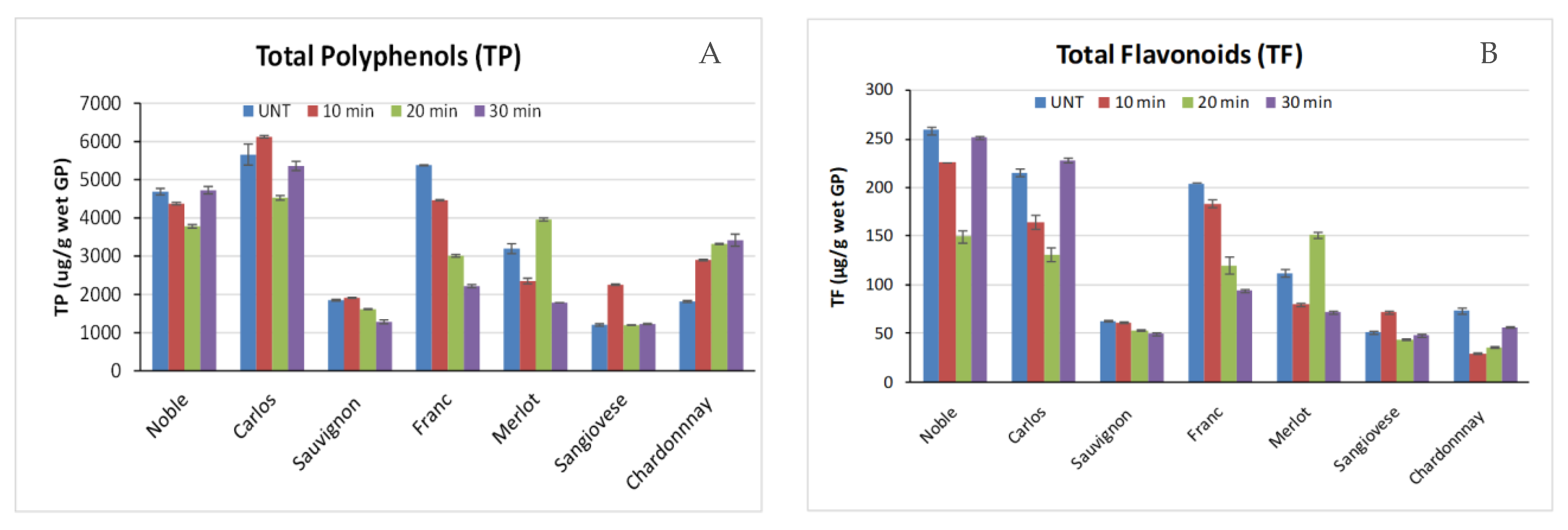
2.2. Effects of Thermal Pressure Treatment on Polyphenol Contents of Grape Pomace
2.3. Effects of Acid Treatments on OTA Content in GP
2.4. Effects of Baking on OTA Contents
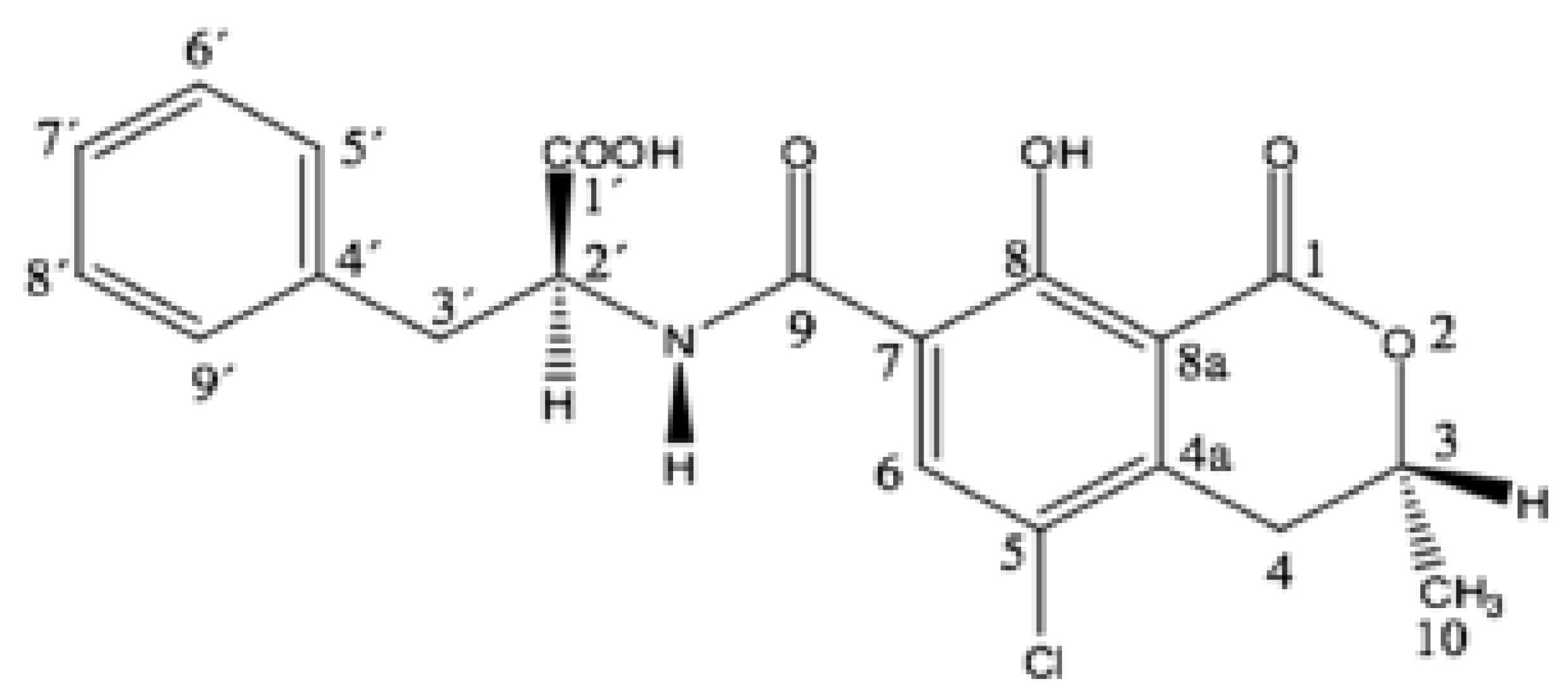
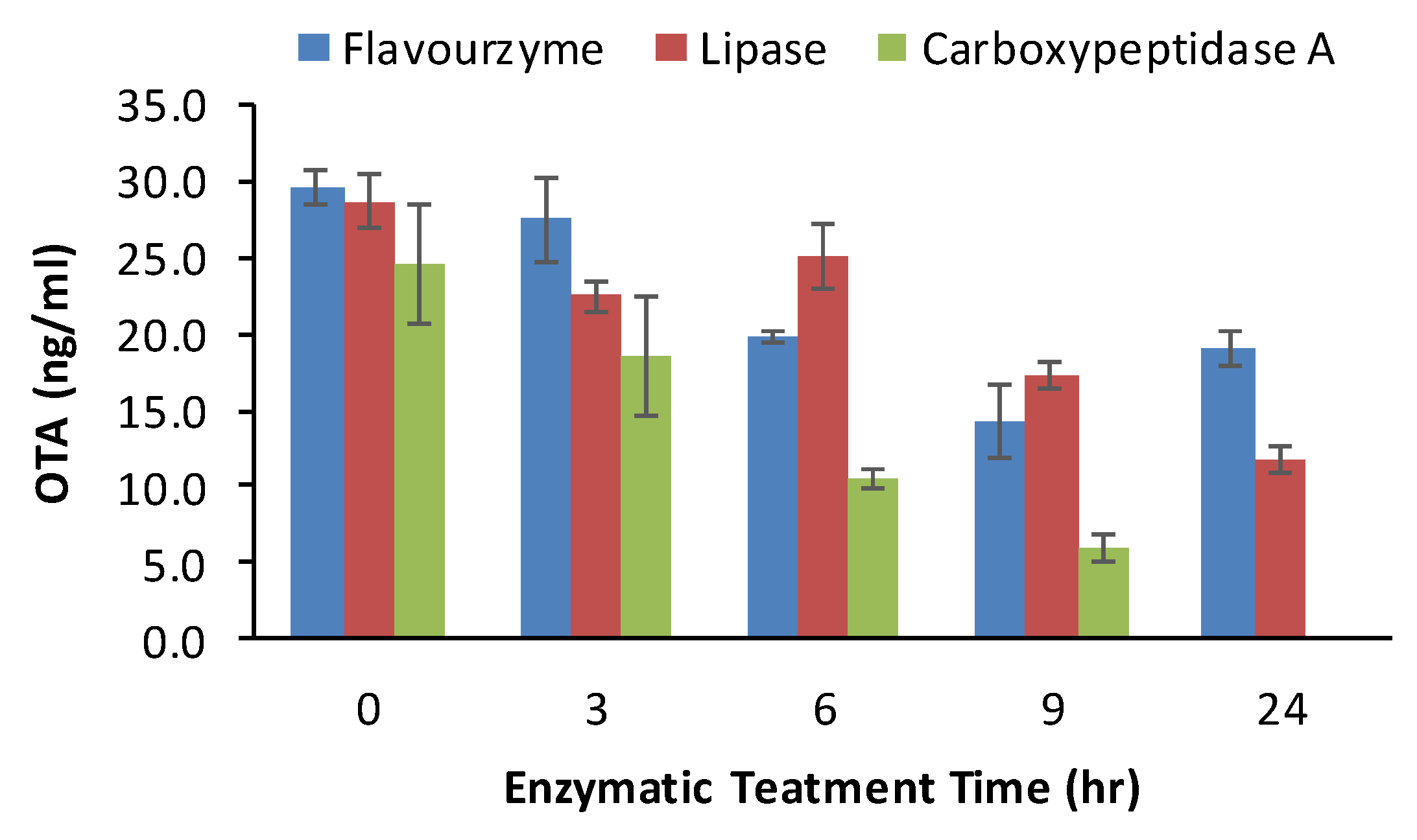
2.5. Effects of Enzymatic Treatment on OTA Content in Grape Pomace
3. Conclusion and Implication
4. Materials and Methods
4.1. Materials
4.2. Treatments of Grape Pomace and OTA Extraction
4.2.1. Thermal Pressure Treatment
4.2.2. Acid Treatment
4.2.3. Baking
4.2.4. Enzymatic Treatment
4.3. OTA Determination
4.4. Polyphenol Extraction and Analysis
4.5. Data Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Varga, J.; Kozakiewicz, Z. Ochratoxin A in grapes and grape derived products. Trends Food Sci. Tech. 2006, 17, 72–81. [Google Scholar] [CrossRef]
- Dachery, B.; Manfroi, V.; Berleze, J.K.; Welke, J.E. Occurrence of ochratoxin A in grapes, juices and wines and risk assessment related to this mycotoxin exposure. Ciência Rural 2016, 46, 176–183. [Google Scholar] [CrossRef]
- Amézqueta, S.; Schorr-Galindo, S.; Murillo-Arbizu, M.; González-Peñas, E.; López de Cerain, A.; Guiraud, J.P. OTA-producing fungi in foodstuffs: A review. Food Control 2012, 26, 259–268. [Google Scholar] [CrossRef]
- Smith, I.; Yu, J. Nutritional and sensory quality of bread containing different cultivars and quantities of grape pomace. EC Nutr. 2015, 2, 291–301. [Google Scholar]
- Yu, J.; Smith, I.; Chen, G. Physical and chemical properties of corn-based extrudates containing grape pomace. SDRP J. Food Sci. Technol. 2018, 3, 516–526. [Google Scholar] [CrossRef]
- Maman, R.; Yu, J. Particle size and chemical composition of grape seed flour and their effects on the characteristics of cookies. J. Food Res. 2019, 8, 111–121. [Google Scholar] [CrossRef]
- Ishida, K.; Kishi, Y.; Oishi, K.; Hirooka, H.; Kumagai, H. Effects of feeding polyphenol-rich winery wastes on digestibility, nitrogen utilization, ruminal fermentation, antioxidant status and oxidative stress in wethers. Anim. Sci. J. 2015, 86, 260–269. [Google Scholar] [CrossRef]
- Hassan, Y.I.; Kosir, V.; Yin, X.; Ross, K.; Diarra, M.S. Grape pomace as a promising antimicrobial alternative in feed: A critical review. J. Agric. Food Chem. 2019, 67, 9705–9718. [Google Scholar] [CrossRef]
- Ianni, A.; Di Maio, G.; Pittia, P.; Grotta, L.; Perpetuini, G.; Tofalo, R.; Cichelli, A.; Martino, G. Chemical-nutritional quality and oxidative stability of milk and dairy products obtained from Friesian cows fed with a dietary supplementation of dried grape pomace. J. Sci. Food Agric. 2019, 99, 3635–3643. [Google Scholar] [CrossRef]
- Karlton-Senaye, B.D.; Yu, J.; Williams, L.L. Morphological and molecular characterization of ochratoxin A producing black Aspergillus from grape pomace. J. Food Res. 2015, 4, 39–50. [Google Scholar] [CrossRef]
- Yu, J.; Smith, I.; Karlton-Senaye, B.; Mikiashvili, N.; Williams, L. Impacts of different drying methods on mold viability and ochratoxin A content of grape pomace. Int. J. Appl. Agric. Sci. 2018, 4, 35–42. [Google Scholar] [CrossRef]
- Heussner, A.H.; Bingle, L.E.H. Comparative Ochratoxin Toxicity: A Review of the Available Data. Toxins Basel 2015, 7, 4253–4282. [Google Scholar] [CrossRef] [PubMed]
- Battacone, G.; Nudda, A.; Pulina, G. Effects of Ochratoxin A on livestock production. Toxins 2010, 2, 1796–1824. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.I.; Wild, C.P.; Baan, R.A.; Gelderblom, W.C.A.; Miller, J.D.; Riley, R.T.; Wu, F. Improving Public Health through Mycotoxin Control; International Agency for Research on Cancer: Lyon, France, 2012; pp. 31–38, 59–85. [Google Scholar]
- Bui-Klimke, T.R.; Wu, F. Ochratoxin A and human health risk: A review of the evidence. Crit. Rev. Food Sci. Nutr. 2015, 55, 1860–1869. [Google Scholar] [CrossRef]
- EFSA. Scientific opinion on the risk assessment of ochratoxin A in food. EFSA J. 2020, 18, 6113. [Google Scholar] [CrossRef]
- EC 2006. European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs; European Commission: Brussels, Belgium, 2006; p. 5. [Google Scholar]
- USDA-FAS. USDA Foreign Agricultural Service. China Releases Standard for Maximum Levels of Mycotoxins in Foods. 2018. Available online: https://www.fas.usda.gov/data/china-china-releases-standard-maximum-levels-mycotoxins-foods (accessed on 8 May 2020).
- Kolakowski, B.; O’Rourke, S.M.; Bietlot, H.P.; Kurz, K.; Aweryn, B. Ochratoxin A concentrations in a variety of grain-based and non–grain-based foods on the Canadian retail market from 2009 to 2014. J. Food Prot. 2016, 79, 2143–2159. [Google Scholar] [CrossRef]
- Budavari, S.; O’Neil, M.J.; Smith, A.; Heckelman, P.E.; Kinneary, J.F. The Merk Index, 12th ed.; Merck & Co., Inc.: White House Station, NJ, USA, 1996; p. 1159. [Google Scholar]
- Malir, F.; Ostry, V.; Pfohl-Leszkowicz, A.; Malir, J.; Toman, J. Ochratoxin A: 50 Years of Research. Toxins 2016, 8, 191. [Google Scholar] [CrossRef]
- Bullerman, L.B.; Bianchini, A. Stability of mycotoxins during food processing. Int. J. Food Microbiol. 2007, 119, 40–146. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Sueck, F.; Hemp, V.; Specht, J.; Torres, O.; Cramer, B.; Humpf, H.U. Occurrence of the Ochratoxin A Degradation Product 2′R-Ochratoxin A in Coffee and Other Food: An Update. Toxins Basel 2019, 11, 329. [Google Scholar] [CrossRef]
- Deberghes, P.; Betbeder, A.M.; Boisard, F.; Blanc, R.; Delaby, J.F.; Krivobok, S.; Steiman, R.; Seigle-Murandi, F.; Creppy, E. Detoxification of ochratoxin A, a food contaminant: Prevention of growth of Aspergillus ochraceus and its production of ochratoxin A. Mycotoxin Res. 1995, 11, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, C.; Zhang, B.; Zhou, Z.; Shen, Y.; Liao, X.; Yang, J.; Wang, Y.; Li, X.; Li, Y.; et al. Advances in Biodetoxification of Ochratoxin A–A Review of the Past Five Decades. Front Microbiol. 1918, 9, 1386. [Google Scholar] [CrossRef] [PubMed]
- Scudamore, K.A. Prevention of ochratoxin A in commodities and likely effects of processing fractionation and animal feeds. Food Addit. Contam. 2005, 22, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Mikiashvili, N.; Liang, C. Deoxynivalenol and Ochratoxin A in North Carolina grown organic wheat grains. J. Food Saf. 2019, 39, e12687. [Google Scholar] [CrossRef]
- Khoury, A.; Atoui, A. Ochratoxin A: General overview and actual molecular status. Toxins 2010, 2, 461–493. [Google Scholar] [CrossRef] [PubMed]
- Cramer, B.; Königs, M.; Humpf, H.U. Identification and in vitro cytotoxicity of ochratoxin A degradation products formed during coffee roasting. J. Agric. Food Chem. 2008, 56, 5673–5681. [Google Scholar] [CrossRef]
- Bittner, A.; Cramer, B.; Harrer, H.; Humpf, H.U. Structure elucidation and in vitro cytotoxicity of ochratoxin α amide, a new degradation product of ochratoxin A. Mycotoxin Res. 2015, 31, 83–90. [Google Scholar] [CrossRef]
- Park, J.W.; Chung, S.H.; Lee, C.; Kim, Y.B. Fate of ochratoxin A during cooking of naturally contaminated polished rice. J. Food Prot. 2005, 68, 2107–2111. [Google Scholar] [CrossRef]
- Boudra, H.; Le Bars, P.; Le Bars, J. Thermostability of ochratoxin A in wheat under two moisture conditions. Appl. Environ. Microbiol. 1995, 61, 1156–1158. [Google Scholar] [CrossRef]
- Friedman, M.; Jürgens, H.S. Effect of pH on the stability of plant phenolic compounds. J. Agric. Food Chem. 2000, 48, 2101–2110. [Google Scholar] [CrossRef]
- Zeng, L.; Ma, M.; Li, C.; Luo, L. Stability of tea polyphenols solution with different pH at different temperatures. Int. J. Food Prop. 2017, 20, 1–18. [Google Scholar] [CrossRef]
- Honda, S.; Ishida, R.; Hidaka, K.; Masuda, T. Stability of Polyphenols under Alkaline Conditions and the Formation of a Xanthine Oxidase Inhibitor from Gallic Acid in a Solution at pH 7.4. Food Sci. Technol. 2019, 25, 123–129. [Google Scholar] [CrossRef]
- Trivedi, A.B.; Dol, E.; Kitabatake, N. Detoxification of Ochratoxin A on heating under acidic and alkaline conditions. Biosci. Biotech. Biochem. 1992, 56, 741–745. [Google Scholar] [CrossRef]
- Dahal, S.; Lee, H.J.; Gu, K.; Ryu, D. Heat stability of Ochratoxin A in an aqueous buffered model system. J. Food Prot. 2016, 79, 1748–1752. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Madhyastha, S.; Marquardt, R.R.; Li, S.; Vodela, J.K.; Frohlich, A.A.; Kemppainen, B.W. Toxicity of ochratoxin A, its opened lactone form and several of its analogs: Structure-Activity relationships. Toxicol. Appl. Pharmacol. 1996, 137, 182–192. [Google Scholar] [CrossRef]
- Vidal, A.; Morales, H.; Sanchis, V.; Ramos, A.J.; Marín, S. Stability of DON and OTA during the breadmaking process and determination of process and performance criteria. Food Control 2014, 40, 234–242. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Paterson, R.R.M.; Venâncio, A. Biodegradation of ochratoxin A for food and feed decontamination. Toxins 2010, 2, 1078–1099. [Google Scholar] [CrossRef]
- Yu, J.; Me, Y.; Ji, S. In vitro evaluation of grape seed polyphenol extract on the digestibility of macronutrient. J. Health Sci. 2016, 4, 167–176. [Google Scholar]
- Abrunhosa, L.; Paterson, R.R.M.; Venâncio, A. Degradation of ochratoxin A by proteases and by a crude enzyme of Aspergillus niger. Food Biotechnol. 2006, 20, 231–242. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu re-agent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Yang, J.; Martinson, T.E.; Liu, R.H. Phytochemical profiles and antioxidant activities of wine grapes. Food Chem. 2009, 116, 332–339. [Google Scholar] [CrossRef]



| Variety of Grape Pomace | OTA Reduction (%) | ||
|---|---|---|---|
| 10 min | 20 min | 30 min | |
| Muscadine Noble | 21.64 | 65.77 | 43.77 |
| Muscadine Carlos | 62.93 | 79.98 | 52.03 |
| Cabernet Sauvignon | 67.60 | 63.29 | 52.57 |
| Cabernet Franc | 47.89 | 70.95 | 59.46 |
| Merlot | 47.49 | 37.64 | 61.87 |
| Sangiovese | 19.21 | 61.03 | 78.01 |
| Chardonnay | 55.71 | 62.23 | 50.97 |
| Treatment | Cabernet Franc | Reduction (%) | Cabernet Sauvignon | Reduction (%) | Chardonnay | Reduction (%) |
|---|---|---|---|---|---|---|
| Control | 25.11 ± 3.15 | 0.00 | 14.15 ± 0.79 | 0.00 | 21.68 ± 2.59 | 0.00 |
| AA | 10.09 ± 3.22 | 59.82 | 12.07 ± 2.57 | 14.68 | 8.36 ± 4.53 | 61.42 |
| CA | 10.84 ± 0.31 | 56.84 | 6.12 ± 5.44 | 56.78 | 17.29 ± 1.91 | 20.23 |
| HCl | 23.07 ± 0.12 | 8.12 | 5.27 ± 2.72 | 62.77 | 16.27 ± 0.73 | 24.95 |
| LA | 26.20 ± 0.42 | −4.35 | 4.64 ± 0.54 | 67.23 | 11.62 ± 3.29 | 46.39 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, J.; Smith, I.N.; Mikiashvili, N. Reducing Ochratoxin A Content in Grape Pomace by Different Methods. Toxins 2020, 12, 424. https://doi.org/10.3390/toxins12070424
Yu J, Smith IN, Mikiashvili N. Reducing Ochratoxin A Content in Grape Pomace by Different Methods. Toxins. 2020; 12(7):424. https://doi.org/10.3390/toxins12070424
Chicago/Turabian StyleYu, Jianmei, Ivy N. Smith, and Nona Mikiashvili. 2020. "Reducing Ochratoxin A Content in Grape Pomace by Different Methods" Toxins 12, no. 7: 424. https://doi.org/10.3390/toxins12070424
APA StyleYu, J., Smith, I. N., & Mikiashvili, N. (2020). Reducing Ochratoxin A Content in Grape Pomace by Different Methods. Toxins, 12(7), 424. https://doi.org/10.3390/toxins12070424




