Fangs for the Memories? A Survey of Pain in Snakebite Patients Does Not Support a Strong Role for Defense in the Evolution of Snake Venom Composition
Abstract
1. Introduction
“Bee stings hurt. So do wasp stings, scorpion stings, the bites of centipedes, and the venom injections of many other animals, including snakes. To inflict pain is not necessarily to the advantage of an animal that uses its venom strictly for incapacitation of prey. In fact, it may be to its disadvantage because pain may induce increased struggling on the part of the prey. But venoms are also used defensively, and it is in that context that they may derive their effectiveness largely, if not exclusively, from their pain-inducing qualities. It is principally because venoms are painful that they can function in defense”.—Eisner and Camazine [1]
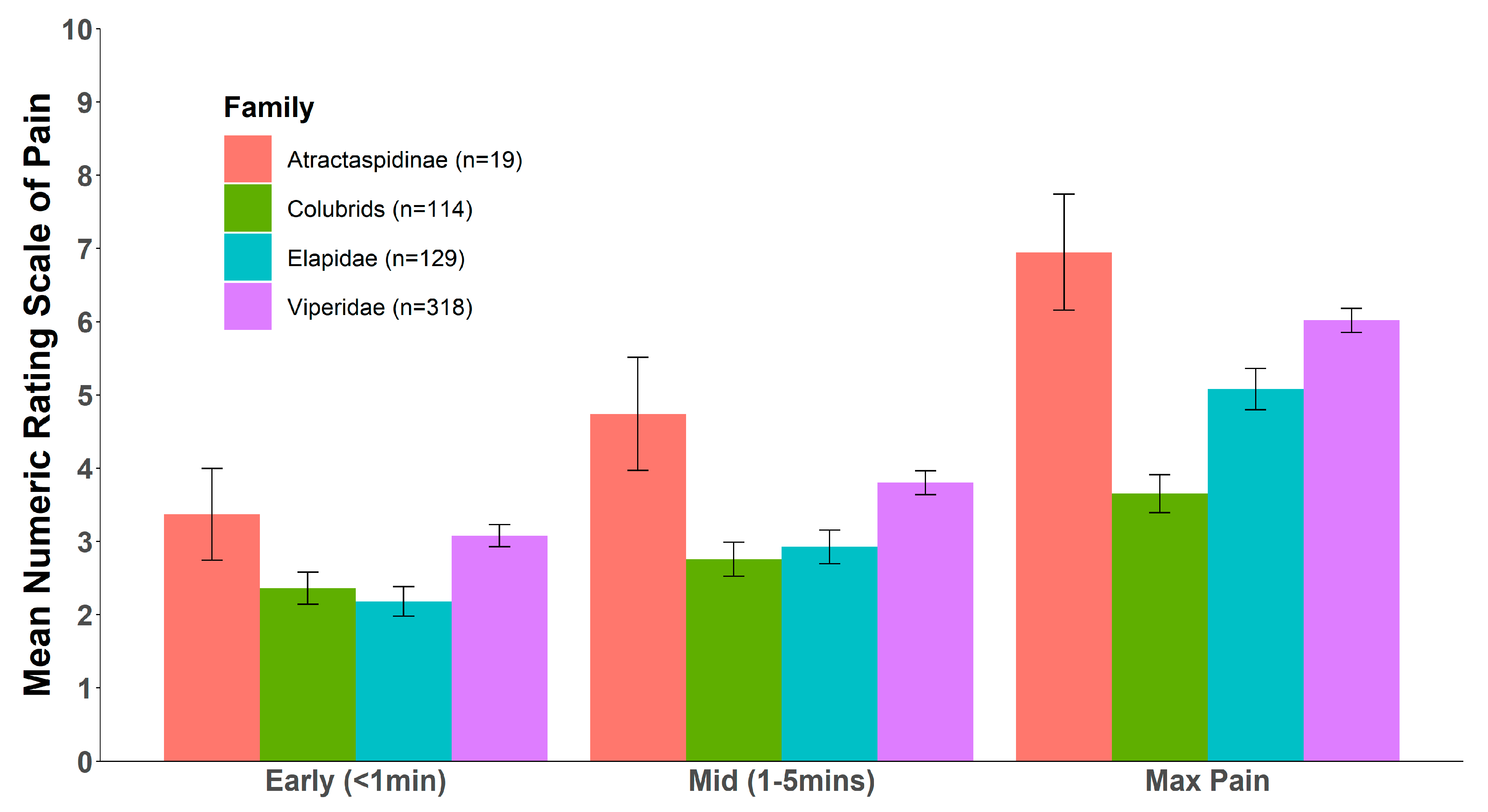
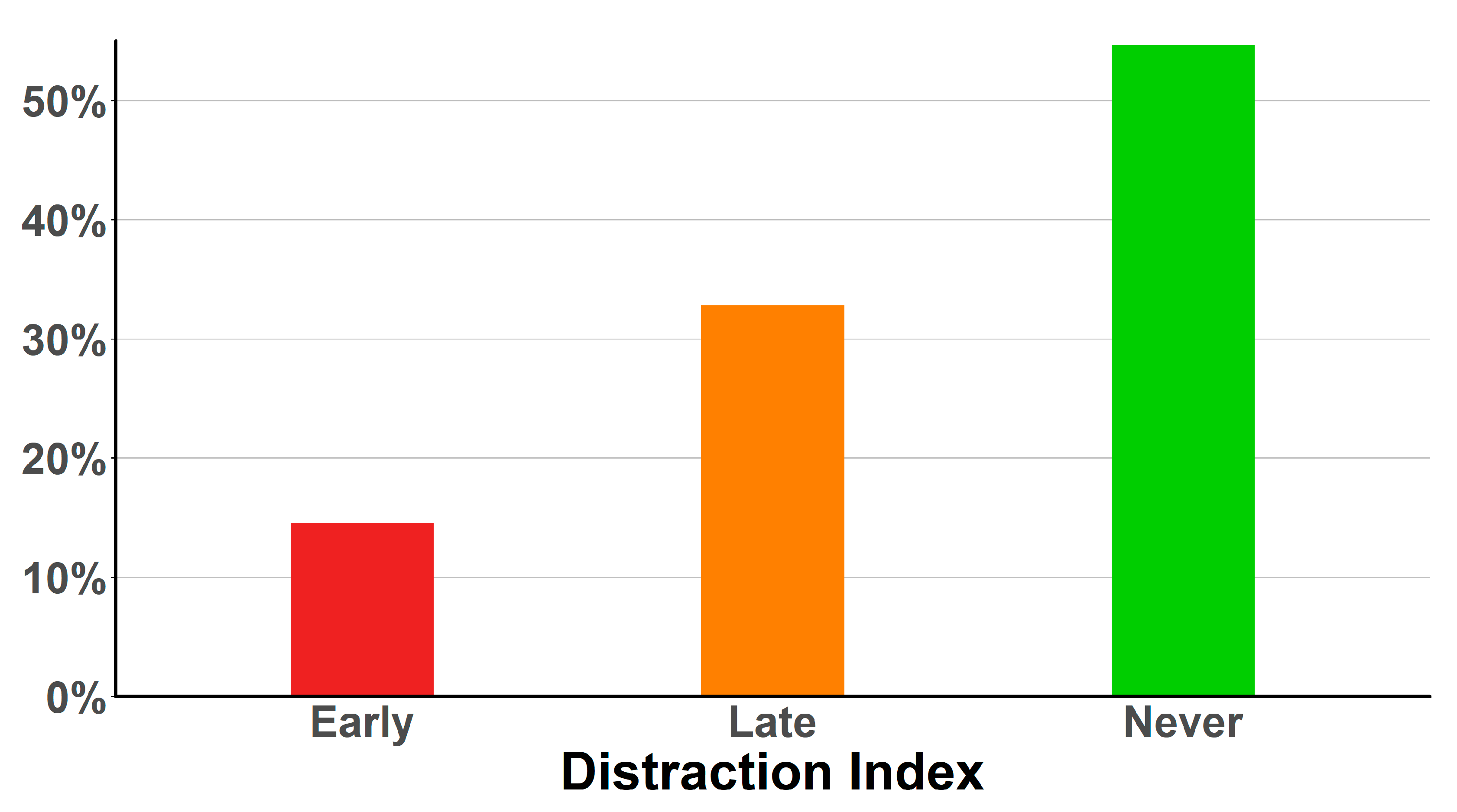
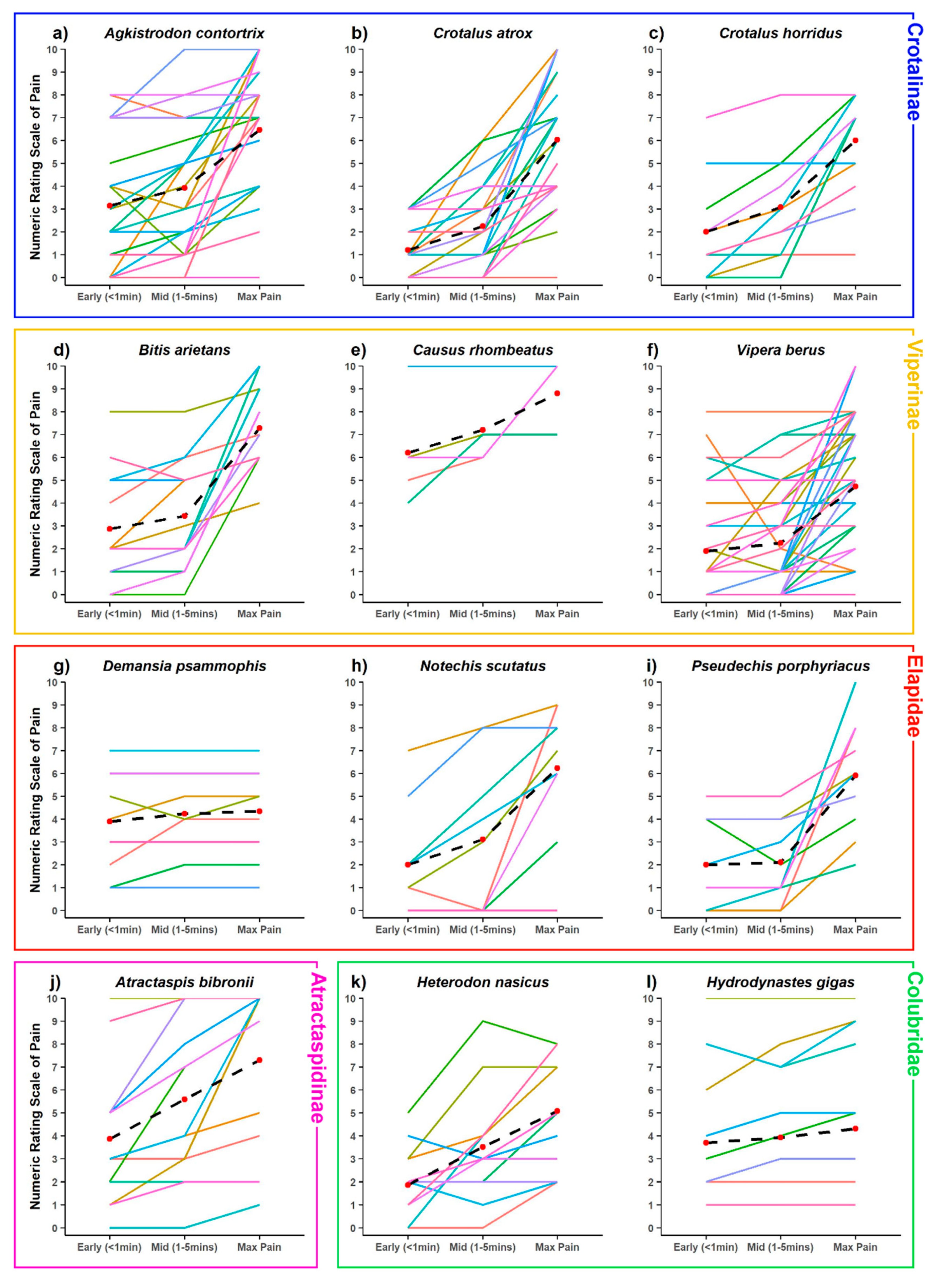
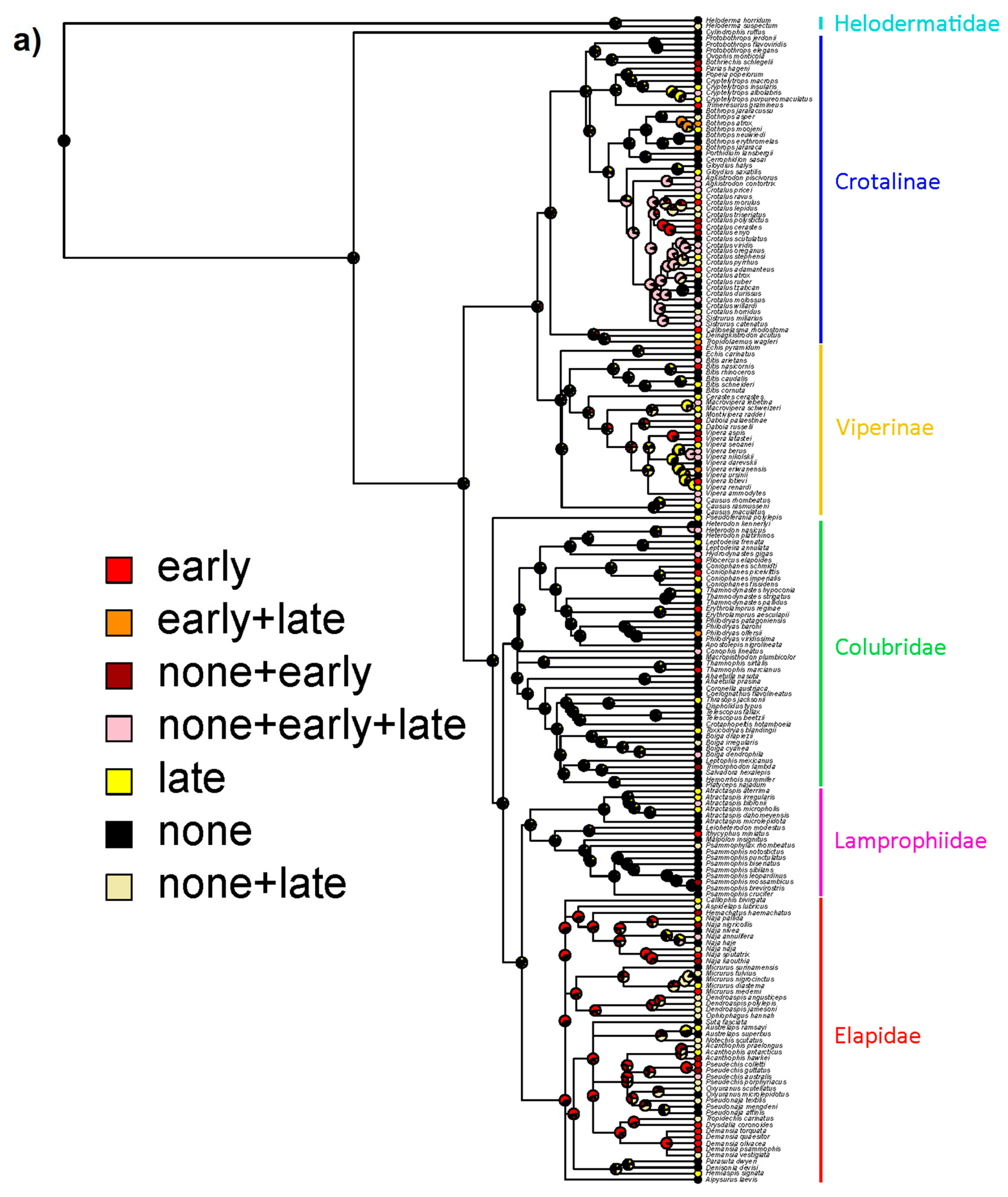
2. Results
3. Discussion
4. Materials and Methods
4.1. Questionnaire and Data Collection
4.2. Data Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eisner, T.; Camazine, S. Spider leg autotomy induced by prey venom injection: An adaptive response to “pain”? Proc. Natl. Acad. Sci. USA 1983, 80, 3382–3385. [Google Scholar] [CrossRef] [PubMed]
- Casewell, N.R.; Wüster, W.; Vonk, F.J.; Harrison, R.A.; Fry, B.G. Complex cocktails: The evolutionary novelty of venoms. Trends Ecol. Evol. 2013, 28, 219–229. [Google Scholar] [CrossRef] [PubMed]
- von Reumont, B.M.; Blanke, A.; Richter, S.; Alvarez, F.; Belidorn, C.; Jenner, R.A. The first venomous crustacean revealed by transcriptomics and functional morphology: Remipede venom glands express a unique toxin cocktail dominated by enzymes and a neurotoxin. Mol. Biol. Evol. 2013, 31, 48–58. [Google Scholar] [CrossRef] [PubMed]
- Jared, C.; Mailho-Fontana, P.L.; Antoniazzi, M.A.; Mendes, V.A.; Barbaro, K.C.; Rodrigues, M.F.; Brodie, E.D., Jr. Venomous frogs use heads as weapons. Curr. Biol. 2015, 25, 2166–2170. [Google Scholar] [CrossRef]
- Arbuckle, K. Evolutionary context of venom in animals. Evol. Venom. Anim. Toxins 2015, 1–23. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Calvete, J.J.; Habib, A.G.; Harrison, R.A.; Williams, D.J.; Warrell, D.A. Snakebite envenoming. Nat. Rev. Dis. Primers 2017, 3, 17063. [Google Scholar] [CrossRef]
- Durban, J.; Pérez, A.; Sanz, L.; Gómez, A.; Bonilla, F.; Rodríguez, S.; Chacón, D.; Sasa, M.; Angulo, Y.; Gutiérrez, J.M.; et al. Integrated “omics” profiling indicates that miRNAs are modulators of the ontogenetic venom composition shift in the Central American rattlesnake, Crotalus simus simus. BMC Genom. 2013, 14, 234. [Google Scholar] [CrossRef]
- Glenn, J.L.; Straight, R. Mojave Rattlesnake Crotalus scutulatus scutulatus venom: Variation in toxicity with geographical origin. Toxicon 1978, 16, 81–84. [Google Scholar] [CrossRef]
- Dawkins, R.; Krebs, J.R. Arms races between and within species. Proc. R. Soc. B 1979, 205, 489–511. [Google Scholar] [CrossRef]
- Daltry, J.C.; Wüster, W.; Thorpe, R.S. Diet and snake venom evolution. Nature 1996, 379, 537. [Google Scholar] [CrossRef]
- Barlow, A.; Pook, C.E.; Harrison, R.A.; Wüster, W. Coevolution of diet and prey-specific venom activity supports the role of selection in snake venom evolution. Proc. R. Soc. B 2009, 276, 2443–2449. [Google Scholar] [CrossRef] [PubMed]
- Richards, D.P.; Barlow, A.; Wüster, W. Venom lethality and diet: Differential responses of natural prey and model organisms to the venom of the saw-scaled vipers (Echis). Toxicon 2012, 59, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Starkov, V.G.; Osipov, A.V.; Utkin, Y.N. Toxicity of venoms from vipers of Pelias group to crickets Gryllus assimilis and its relation to snake entomophagy. Toxicon 2007, 49, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.L.; Mackessy, S.P. Functional basis of a molecular adaptation: Prey-specific toxic effects of venom from Sistrurus rattlesnakes. Toxicon 2009, 53, 672–679. [Google Scholar] [CrossRef]
- da Silva, N.J., Jr.; Aird, S.D. Prey specificity, comparative lethality and compositional differences of coral snake venoms. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2001, 128, 425–456. [Google Scholar] [CrossRef]
- Dashevsky, D.; Debono, J.; Rokyta, D.; Nouwens, A.; Josh, P.; Fry, B.G. Three-finger toxin diversification in the venoms of cat-eye snakes (Colubridae: Boiga). J. Mol. Evol. 2018, 86, 531–545. [Google Scholar] [CrossRef]
- Mackessy, S.P.; Sixberry, N.M.; Heyborne, W.H.; Fritts, T. Venom of the brown treesnake, Boiga irregularis: Ontogenetic shifts and taxa-specific toxicity. Toxicon 2006, 47, 537–548. [Google Scholar] [CrossRef]
- Pawlak, J.; Mackessy, S.P.; Fry, B.G.; Bhatia, M.; Mourier, G.; Fruchart-Gaillard, C.; Servent, D.; Ménez, R.; Stura, E.; Ménez, A.; et al. Denmotoxin, a three-finger toxin from the colubrid snake Boiga dendrophila (Mangrove Catsnake) with bird-specific activity. J. Biol. Chem. 2006, 281, 29030–29041. [Google Scholar] [CrossRef]
- Heyborne, W.H.; Mackessy, S.P. Identification and characterization of a taxon-specific three-finger toxin from the venom of the Green Vinesnake (Oxybelis fulgidus; family Colubridae). Biochimie 2013, 95, 1923–1932. [Google Scholar] [CrossRef]
- Modahl, C.M.; Mrinalini; Frietze, S.; Mackessy, S.P. Adaptive evolution of distinct prey-specific toxin genes in rear-fanged snake venom. Proc. R. Soc. B 2018, 285, 20181003. [Google Scholar] [CrossRef]
- Andrade, D.V.; Abe, A.S. Relationship of venom ontogeny and diet in Bothrops. Herpetologica 1999, 55, 200–204. [Google Scholar]
- Perez, J.C.; Haws, W.C.; Garcia, V.E.; Jennings, B.M. Resistance of warm-blooded animals to snake venoms. Toxicon 1978, 16, 375–383. [Google Scholar] [CrossRef]
- Poran, N.S.; Coss, R.G.; Benjamini, E.L.I. Resistance of California ground squirrels (Spermophilus beecheyi) to the venom of the northern Pacific rattlesnake (Crotalus viridis oreganus): A study of adaptive variation. Toxicon 1987, 25, 767–777. [Google Scholar] [CrossRef]
- Heatwole, H.; Poran, N.S. Resistances of sympatric and allopatric eels to sea snake venoms. Copeia 1995, 1995, 136–147. [Google Scholar] [CrossRef]
- Holding, M.L.; Biardi, J.E.; Gibbs, H.L. Coevolution of venom function and venom resistance in a rattlesnake predator and its squirrel prey. Proc. R. Soc. B 2016, 283, 20152841. [Google Scholar] [CrossRef]
- Zancolli, G.; Calvete, J.J.; Cardwell, M.D.; Greene, H.W.; Hayes, W.K.; Hegarty, M.J.; Herrmann, H.W.; Holycross, A.T.; Lannutti, D.I.; Mulley, J.F.; et al. When one phenotype is not enough: Divergent evolutionary trajectories govern venom variation in a widespread rattlesnake species. Proc. R. Soc. B 2019, 286, 20182735. [Google Scholar] [CrossRef]
- Weinstein, S.A.; Warrell, D.A.; White, J.; Keyler, D.E. “Venomous Bites from Non-Venomous Snakes: A Critical Analysis of Risk and Management of “Colubrid Snake Bites; Elsevier: London, UK, 2011; ISBN 978-0123877321. [Google Scholar]
- Greene, H.W. Tracks and Shadows, Field Biology as Art; University of California Press: Berkeley, CA, USA, 2013; ISBN 978-0520232754. [Google Scholar]
- Holding, M.L.; Drabeck, D.H.; Jansa, S.A.; Gibbs, H.L. Venom resistance as a model for understanding the molecular basis of complex coevolutionary adaptations. Integr. Comp. Biol. 2016, 56, 1032–1043. [Google Scholar] [CrossRef]
- Brodie, E.D., III. Differential avoidance of coral snake banded patterns by free-ranging avian predators in Costa Rica. Evolution 1993, 47, 227–235. [Google Scholar] [CrossRef]
- Wüster, W.; Allum, C.S.; Bjargardóttir, I.B.; Bailey, K.L.; Dawson, K.J.; Guenioui, J.; Lewis, J.; McGurk, J.; Moore, A.G.; Niskanen, M.; et al. Do aposematism and Batesian mimicry require bright colours? A test, using European viper markings. Proc. R. Soc. B 2004, 271, 2495–2499. [Google Scholar] [CrossRef]
- Smith, S.M. Innate recognition of coral snake pattern by a possible avian predator. Science 1975, 187, 759–760. [Google Scholar] [CrossRef]
- Smith, S.M. Coral snake pattern recognition and stimulus generalization by naïve great kiskadees (Aves: Tyrannidae). Nature 1977, 265, 535–536. [Google Scholar] [CrossRef]
- Greene, H.W.; McDiarmid, R.W. Coral snake mimicry: Does it occur? Science 1981, 213, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Davis Rabosky, A.R.; Cox, C.L.; Rabosky, D.L.; Title, P.O.; Holmes, I.A.; Feldman, A.; McGuire, J.A. Coral snakes predict the evolution of mimicry across New World snakes. Nat. Commun. 2017, 7, 11484. [Google Scholar] [CrossRef] [PubMed]
- Greene, H.W. Snakes: The Evolution of Mystery in Nature; University of California Press: Berkeley, CA, USA, 1997; ISBN 978-0520224872. [Google Scholar]
- Chahl, L.A.; Kirk, E.J. Toxins which produce pain. Pain 1975, 1, 3–49. [Google Scholar] [CrossRef]
- Fernandez, I.; Valladolid, G.; Varon, J.; Sternbach, G. Encounters with venomous sea-life. J. Emerg. Med. 2011, 40, 103–112. [Google Scholar] [CrossRef]
- Haddad, V.; Stolf, H.O.; Risk, J.Y.; França, F.O.; Cardoso, J.L.C. Report of 15 injuries caused by lionfish (Pterois volitans) in aquarists in Brazil: A critical assessment of the severity of envenomations. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 8. [Google Scholar] [CrossRef]
- Kizer, K.W.; McKinney, H.E.; Auerbach, P.S. Scorpaenidae envenomation: A five-year poison center experience. JAMA 1985, 253, 807–810. [Google Scholar] [CrossRef]
- Patel, M.R.; Wells, S. Lionfish envenomation of the hand. J. Hand Surg. 1993, 18, 523–525. [Google Scholar] [CrossRef]
- Vetrano, S.J.; Lebowitz, J.B.; Marcus, S. Lionfish envenomation. J. Emerg. Med. 2002, 23, 379–382. [Google Scholar] [CrossRef]
- Rowe, A.H.; Xiao, Y.; Rowe, M.P.; Cummins, T.R.; Zakon, H.H. Voltage-gated sodium channel in grasshopper mice defends against bark scorpion toxin. Science 2013, 342, 441–446. [Google Scholar] [CrossRef]
- Yang, S.; Yang, F.; Wei, N.; Hong, J.; Li, B.; Luo, L.; Rong, M.; Yarov-Yarovoy, V.; Zheng, J.; Wang, K.; et al. A pain-inducing centipede toxin targets the heat activation machinery of nociceptor TRPV1. Nat. Commun. 2015, 6, 8297. [Google Scholar] [CrossRef] [PubMed]
- Warrell, D.A. Clinical toxicology of snakebite in Africa and the Middle East/Arabian Peninsula. In Handbook of Clinical Toxicology of Animal Venoms and Poisons; Meier, J., White, J., Eds.; CRC Press: Boca Raton, FL, USA, 1995; pp. 433–492. ISBN 978-0849344893. [Google Scholar]
- Warrell, D.A. Clinical toxicology of snakebite in Asia. In Handbook of Clinical Toxicology of Animal Venoms and Poisons; Meier, J., White, J., Eds.; CRC Press: Boca Raton, FL, USA, 1995; pp. 493–594. ISBN 978-0849344893. [Google Scholar]
- Warrell, D.A. Snakebites in Central and South America: Epidemiology, clinical features, clinical management. In The Venomous Reptiles of the Western Hemisphere; Campbell, J.A., Lamar, W.W., Eds.; Cornell University Press: Ithaca, NY, USA, 2004; pp. 709–761. ISBN 978-0801441417. [Google Scholar]
- Russell, F.E.; Wainschel., J.; Carlson, R.W.; Osborne, A. Snake venom poisoning in the United States. In Animal, Plant and Microbial Toxins, Volume 2 Chemistry, Pharmacology and Immunology; Ohsaka, A., Hayashi, K., Sawai, Y., Eds.; Springer: Philadelphia, PA, USA, 1980; pp. 139–234. ISBN 978-1468408911. [Google Scholar]
- Ariaratnam, C.A.; Sheriff, M.R.; Theakston, R.D.G.; Warrell, D.A. Distinctive epidemiologic and clinical features of common krait (Bungarus caeruleus) bites in Sri Lanka. Am. J. Trop. Med. Hyg. 2008, 79, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Bohlen, C.J.; Chesler, A.T.; Sharif-Naeini, R.; Medzihradszky, K.F.; Zhou, S.; King, D.; Sánchez, E.E.; Burlingame, A.L.; Basbaum, A.I.; Julius, D. A heteromeric Texas coral snake toxin targets acid-sensing ion channels to produce pain. Nature 2011, 479, 410. [Google Scholar] [CrossRef]
- Fernández, J.; Vargas-Vargas, N.; Pla, D.; Sasa, M.; Rey-Suárez, P.; Sanz, L.; Gutiérrez, J.M.; Calvete, J.J.; Lomonte, B. Snake venomics of Micrurus alleni and Micrurus mosquitensis from the Caribbean region of Costa Rica reveals two divergent compositional patterns in New World elapids. Toxicon 2015, 107, 217–233. [Google Scholar] [CrossRef]
- Margres, M.J.; Aronow, K.; Loyacano, J.; Rokyta, D.R. The venom-gland transcriptome of the eastern coral snake (Micrurus fulvius) reveals high venom complexity in the intragenomic evolution of venoms. BMC Genom. 2013, 14, 531. [Google Scholar] [CrossRef]
- Vergara, I.; Pedraza-Escalona, M.; Paniagua, D.; Restano-Cassulini, R.; Zamudio, F.; Batista, C.V.; Possani, L.D.; Alagón, A. Eastern coral snake Micrurus fulvius venom toxicity in mice is mainly determined by neurotoxic phospholipases A2. J. Proteom. 2014, 105, 295–306. [Google Scholar] [CrossRef]
- Zhang, C.; Medzihradszky, K.F.; Sánchez, E.E.; Basbaum, A.I.; Julius, D. Lys49 myotoxin from the Brazilian lancehead pit viper elicits pain through regulated ATP release. Proc. Natl. Acad. Sci. USA 2017, 114, E2524–E2532. [Google Scholar] [CrossRef]
- Sneddon, L.U. Comparative physiology of nociception and pain. Physiology 2018, 33, 63–73. [Google Scholar] [CrossRef]
- Warrell, D.A. Commissioned article: Management of exotic snakebites. QJM Int. J. Med. 2009, 102, 593–601. [Google Scholar] [CrossRef]
- Isbister, G.K.; Brown, S.G.A.; ASP Investigators. Bites in Australian snake handlers—Australian snakebite project (ASP-15). QJM Int. J. Med. 2012, 105, 1089–1095. [Google Scholar] [CrossRef]
- Valenta, J.; Stach, Z.; Michalek, P. Exotic snake bites in the Czech Republic—Epidemiological and clinical aspects during 15-year period (1999–2013). Clin. Toxicol. 2014, 52, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Panagides, N.; Jackson, T.; Ikonomopoulou, M.; Arbuckle, K.; Pretzler, R.; Yang, D.; Ali, S.; Koludarov, I.; Dobson, J.; Sanker, B.; et al. How the cobra got its flesh-eating venom: Cytotoxicity as a defensive innovation and its co-evolution with hooding, aposematic marking, and spitting. Toxins 2017, 9, 103. [Google Scholar] [CrossRef] [PubMed]
- Sanz, L.; de Freitas-Lima, L.N.; Quesada-Bernat, S.; Graça-de-Souza, V.K.; Soares, A.M.; Calderón, L.D.A.; Calvete, J.J.; Caldeira, C.A. Comparative venomics of Brazilian coral snakes: Micrurus frontalis, Micrurus spixii spixii, and Micrurus surinamensis. Toxicon 2019, 166, 39–45. [Google Scholar] [CrossRef]
- Aird, S.; da Silva, N.; Qiu, L.; Villar-Briones, A.; Saddi, V.; Pires de Campos Telles, M.; Grau, M.; Mikheyev, A. Coralsnake venomics: Analyses of venom gland transcriptomes and proteomes of six Brazilian taxa. Toxins 2017, 9, 187. [Google Scholar] [CrossRef] [PubMed]
- Kitchens, C.S.; Van Mierop, L.H. Envenomation by the eastern coral snake (Micrurus fulvius fulvius): A study of 39 victims. J. Am. Med Assoc. 1987, 258, 1615–1618. [Google Scholar] [CrossRef]
- Sasaki, J.; Khalil, P.A.; Chegondi, M.; Raszynski, A.; Meyer, K.G.; Totapally, B.R. Coral snake bites and envenomation in children: A case series. Pediatric Emerg. Care 2014, 30, 262–265. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.L.; Borys, D.J.; Stanford, R.; Kjar, D.; Tobleman, W. Texas coral snake (Micrurus tener) bites. South. Med. J. 2007, 100, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, A.; McKelvy, A.D.; Grismer, L.L.; Bell, C.D.; Lailvaux, S.P. A species-level phylogeny of extant snakes with description of a new colubrid subfamily and genus. PLoS ONE 2016, 11, e0161070. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.G.; Scheib, H.; van der Weerd, L.; Young, B.; McNaughtan, J.; Ramjan, S.R.; Vidal, N.; Poelmann, R.E.; Norman, J.A. Evolution of an arsenal: Structural and functional diversification of the venom system in the advanced snakes (Caenophidia). Mol. Cell. Proteom. 2008, 7, 215–246. [Google Scholar] [CrossRef]
- Gopalakrishnakone, P.; Wollberg, M.; Kochva, E. The venom apparatus of sea snakes. In Sea Snake Toxinology; Gopalakrishnakone, P., Ed.; Singapore University Press: Singapore, 1994; pp. 37–65. ISBN 9789971691912. [Google Scholar]
- Li, M.; Fry, B.G.; Kini, R.M. Eggs-only diet: Its implications for the toxin profile changes and ecology of the marbled sea snake (Aipysurus eydouxii). J. Mol. Evol. 2005, 60, 81–89. [Google Scholar] [CrossRef]
- Li, M.; Fry, B.G.; Kini, R.M. Putting the brakes on snake venom evolution: The unique molecular evolutionary patterns of Aipysurus eydouxii (Marbled sea snake) phospholipase A2 toxins. Mol. Biol. Evol. 2005, 22, 934–941. [Google Scholar] [CrossRef]
- Brauer, C.; Thomsen, J.F.; Loft, I.P.; Mikkelsen, S. Can we rely on retrospective pain assessments? Am. J. Epidemiol. 2003, 157, 552–557. [Google Scholar] [CrossRef] [PubMed]
- Wüster, W.; Thorpe, R.S. Dentitional phenomena in cobras revisited: Spitting and fang structure in the Asiatic species of Naja (Serpentes: Elapidae). Herpetologica 1992, 424–434. [Google Scholar]
- Greene, H.W. Antipredator mechanisms in reptiles. In Biology of the Reptilia; Gans, C., Huey, R.B., Eds.; Alan, R. Liss: New York, NY, USA, 1988; Volume 16, pp. 1–152. ISBN 978-0-8451-4402-2. [Google Scholar]
- Martins, M. Defensive tactics in lizards and snakes: The potential contribution of the Neotropical fauna. An. Etol. 1996, 14, 185–199. [Google Scholar]
- Pough, F.H. Mimicry of vertebrates: Are the rules different? Am. Nat. 1988, 131, S67–S102. [Google Scholar] [CrossRef]
- Klopfer, P.H. An experiment on empathic learning in ducks. Am. Nat. 1957, 91, 61–63. [Google Scholar] [CrossRef]
- Gangur, A.N.; Seymour, J.E.; Liddell, M.J.; Wilson, D.; Smout, M.J.; Northfield, T.D. When is overkill optimal? Tritrophic interactions reveal new insights into venom evolution. Theor. Ecol. 2018, 11, 141–149. [Google Scholar] [CrossRef]
- Downie, W.W.; Leatham, P.A.; Rhind, V.M.; Wright, V.; Branco, J.A.; Anderson, J.A. Studies with pain rating scales. Ann. Rheum. Dis. 1978, 37, 378. [Google Scholar] [CrossRef]
- Hartrick, C.T.; Kovan, J.P.; Shapiro, S. The numeric rating scale for clinical pain measurement: A ratio measure? Pain Pract. 2003, 3, 310–316. [Google Scholar] [CrossRef]
- Williamson, A.; Hoggart, B. Pain: A review of three commonly used pain rating scales. J. Clin. Nurs. 2005, 14, 798–804. [Google Scholar] [CrossRef]
- Arbuckle, K. Phylogenetic comparative methods can provide important insights into the evolution of toxic weaponry. Toxins 2018, 10, 518. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Suleski, M.; Hedges, S.B. TimeTree: A resource for timelines, timetrees, and divergence times. Mol. Biol. Evol. 2017, 34, 1812–1819. [Google Scholar] [CrossRef] [PubMed]
- Pennell, M.W.; FitzJohn, R.G.; Cornwell, W.K. A simple approach for maximizing the overlap of phylogenetic and comparative data. Methods Ecol. Evol. 2016, 7, 751–758. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing. Version 3.6.0. 2019. Available online: https://www.R-project.org/ (accessed on 1 May 2019).
- Bollback, J.P. SIMMAP: Stochastic character mapping of discrete traits on phylogenies. BMC Bioinform. 2006, 7, 88. [Google Scholar] [CrossRef]
- Revell, L.J. Phytools: An R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 2012, 3, 217–223. [Google Scholar] [CrossRef]
- Hadfield, J.D. MCMC methods for multi-response generalized linear mixed models: The MCMCglmm R package. J. Stat. Softw. 2010, 33, 1–22. [Google Scholar] [CrossRef]
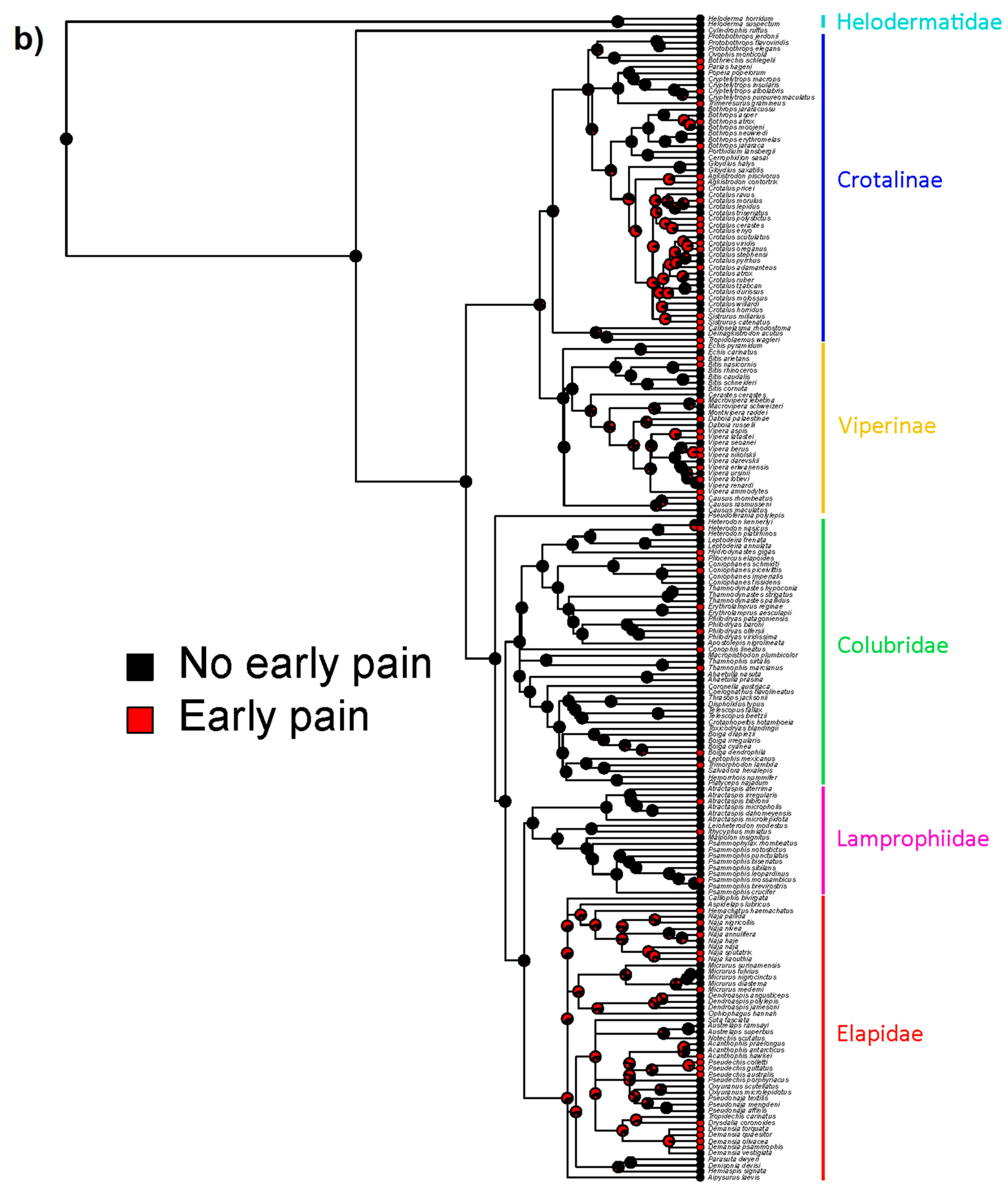
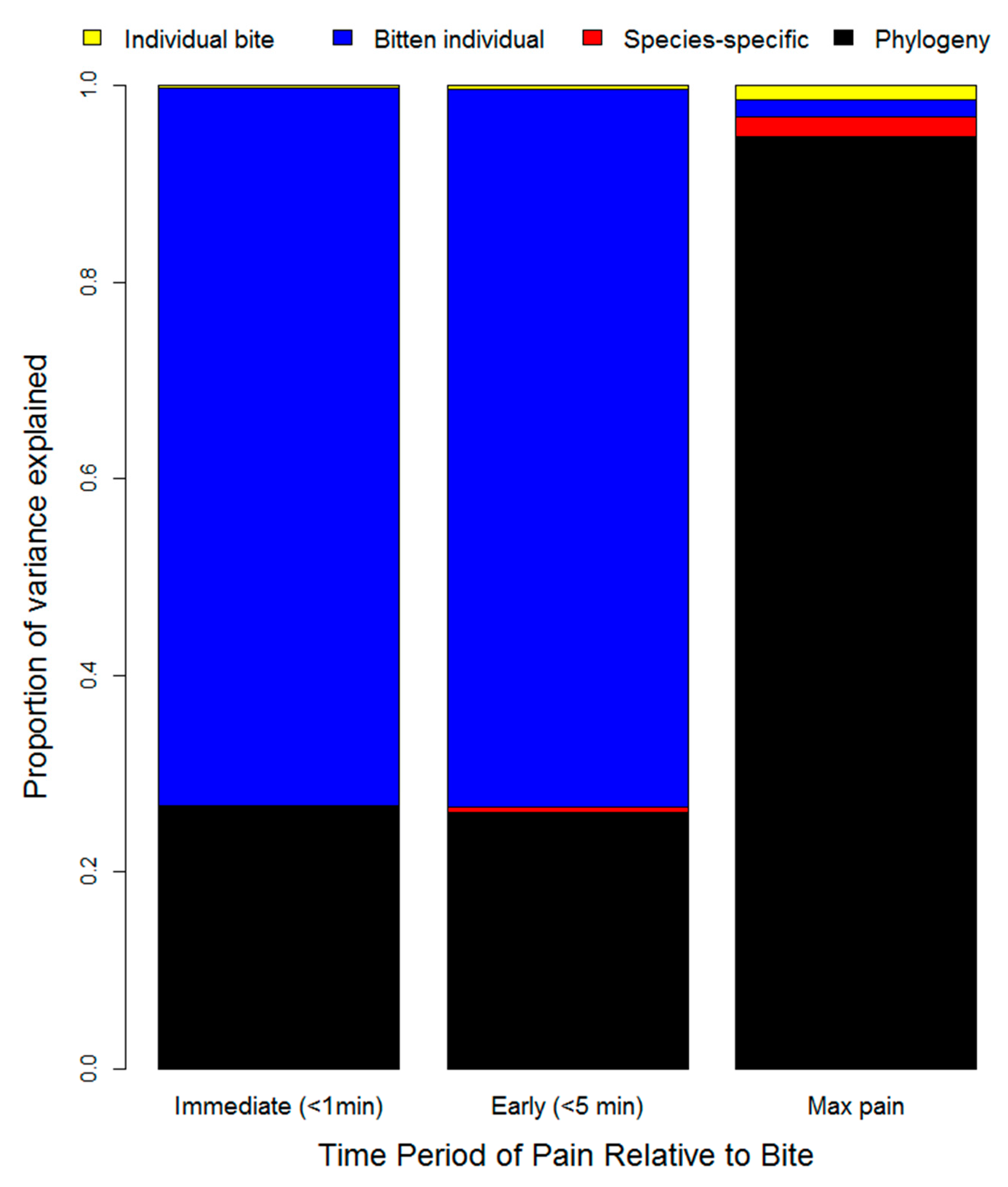
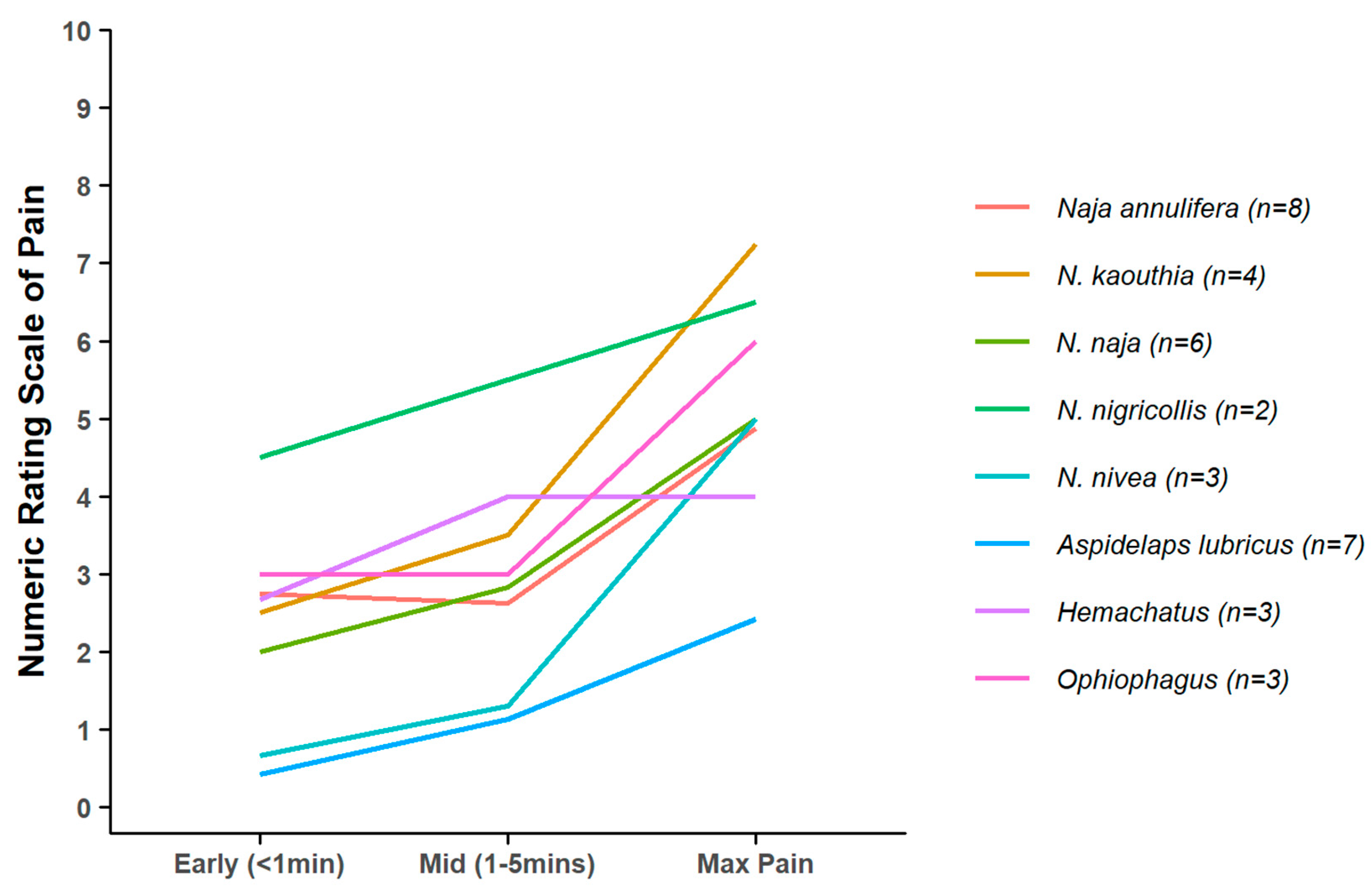
| Age (years) | Male | Female | Unreported | Total |
|---|---|---|---|---|
| Total, n (%) | 523 (89.6%) | 51 (8.7%) | 10 (1.7%) | 584 (100%) |
| 11–20, n (%) | 129 (22.1%) | 11 (1.9%) | 1 (0.2%) | 141 (24.3%) |
| 21–30, n (%) | 164 (28.1%) | 27 (4.6%) | 6 (1.0%) | 197 (33.7%) |
| 31–40, n (%) | 102 (17.5%) | 7 (1.2%) | 1 (0.2%) | 110 (18.8%) |
| 41–50, n (%) | 69 (11.8%) | 3 (0.5%) | 2 (0.3%) | 74 (12.7%) |
| 51–60, n (%) | 41 (7.0%) | 0 | 0 | 41 (7.0%) |
| ≥61, n (%) | 14 (2.4%) | 3 (0.5%) | 0 | 17 (2.9%) |
| Unreported, n (%) | 4 (0.7%) | 0 | 0 | 4 (0.7%) |
| From\To | None | None+Early | Early | Early+Late | None+Early+Late | None+Late | Late |
|---|---|---|---|---|---|---|---|
| none | - | 0.004 | 0 | 0.011 | 0.003 | 0 | 0.041 |
| none+early | 0 | - | 0.032 | 0 | 0 | 0.205 | 0 |
| early | 0 | 0.125 | - | 0 | 0 | 0 | 0 |
| early+late | 0 | 0 | 0 | - | 0 | 0.070 | 0.244 |
| none+early+late | 0.057 | 0 | 0.016 | 0 | - | 0.043 | 0.008 |
| none+late | 0.018 | 0 | 0.042 | 0 | 0 | - | 0.111 |
| late | 0.141 | 0 | 0.032 | 0.022 | 0.069 | 0 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ward-Smith, H.; Arbuckle, K.; Naude, A.; Wüster, W. Fangs for the Memories? A Survey of Pain in Snakebite Patients Does Not Support a Strong Role for Defense in the Evolution of Snake Venom Composition. Toxins 2020, 12, 201. https://doi.org/10.3390/toxins12030201
Ward-Smith H, Arbuckle K, Naude A, Wüster W. Fangs for the Memories? A Survey of Pain in Snakebite Patients Does Not Support a Strong Role for Defense in the Evolution of Snake Venom Composition. Toxins. 2020; 12(3):201. https://doi.org/10.3390/toxins12030201
Chicago/Turabian StyleWard-Smith, Harry, Kevin Arbuckle, Arno Naude, and Wolfgang Wüster. 2020. "Fangs for the Memories? A Survey of Pain in Snakebite Patients Does Not Support a Strong Role for Defense in the Evolution of Snake Venom Composition" Toxins 12, no. 3: 201. https://doi.org/10.3390/toxins12030201
APA StyleWard-Smith, H., Arbuckle, K., Naude, A., & Wüster, W. (2020). Fangs for the Memories? A Survey of Pain in Snakebite Patients Does Not Support a Strong Role for Defense in the Evolution of Snake Venom Composition. Toxins, 12(3), 201. https://doi.org/10.3390/toxins12030201






