The Degradation of Deoxynivalenol by Using Electrochemical Oxidation with Graphite Electrodes and the Toxicity Assessment of Degradation Products
Abstract
1. Introduction
2. Results and Discussion
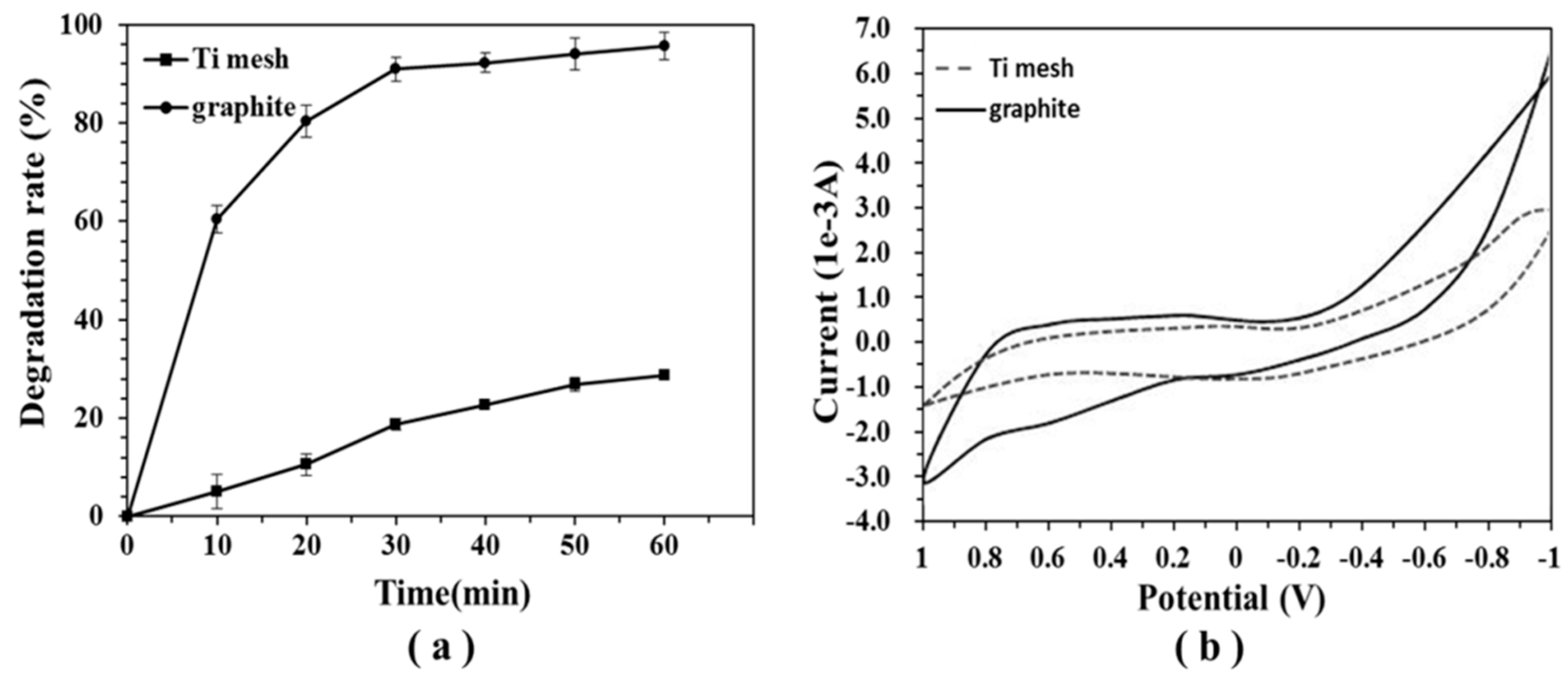
2.1. The Efficiency of Different Electrodes on DON Degradation
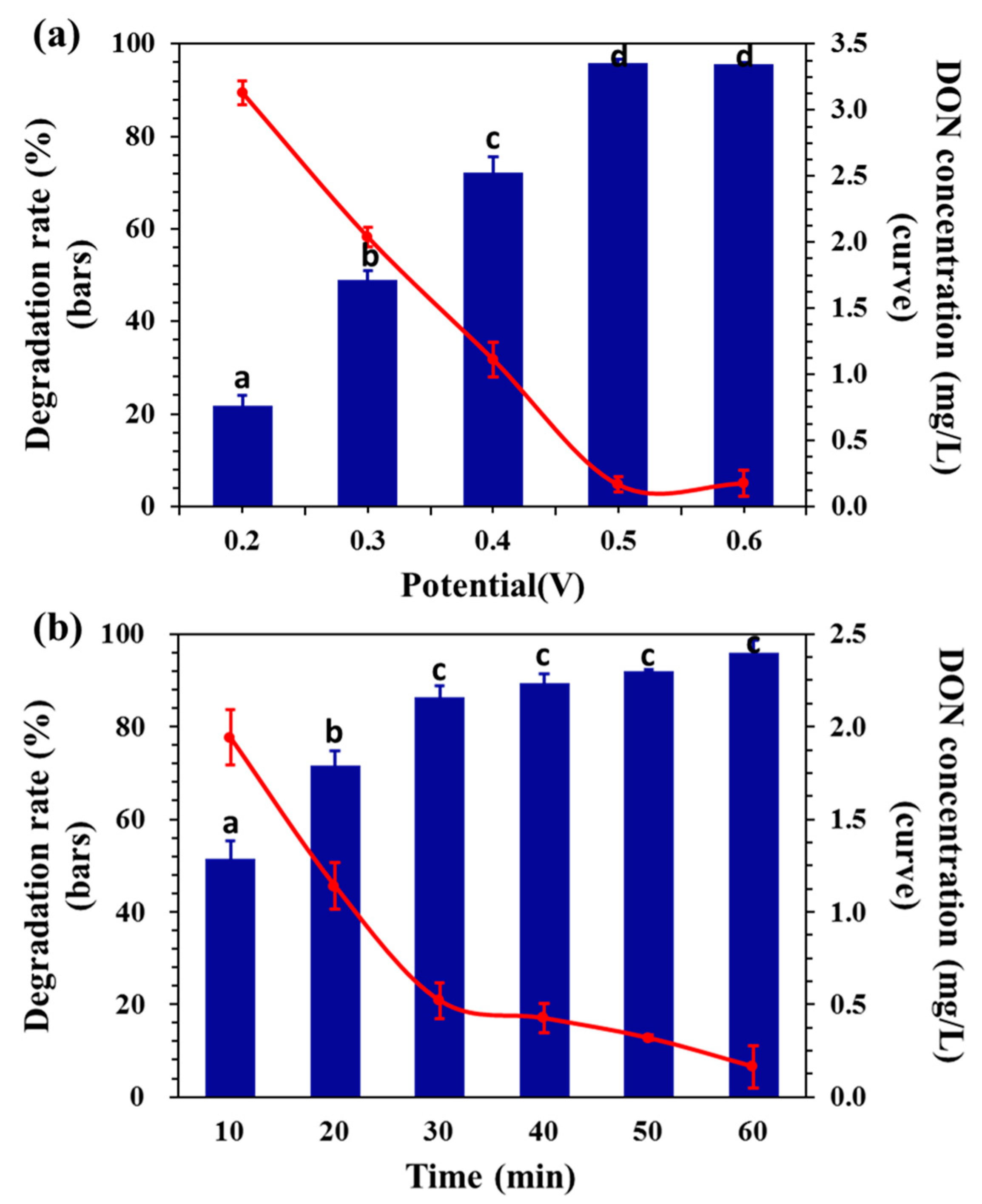
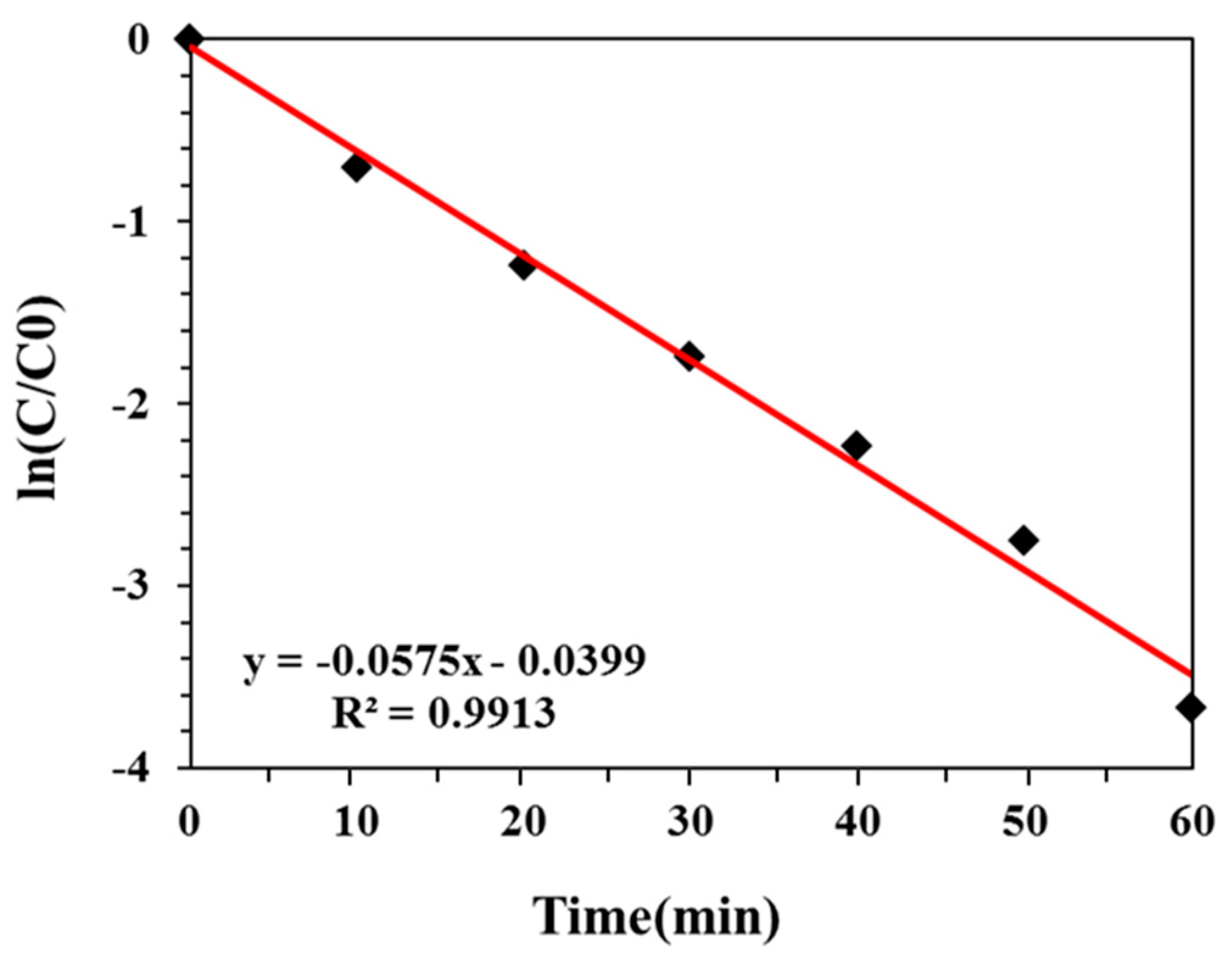
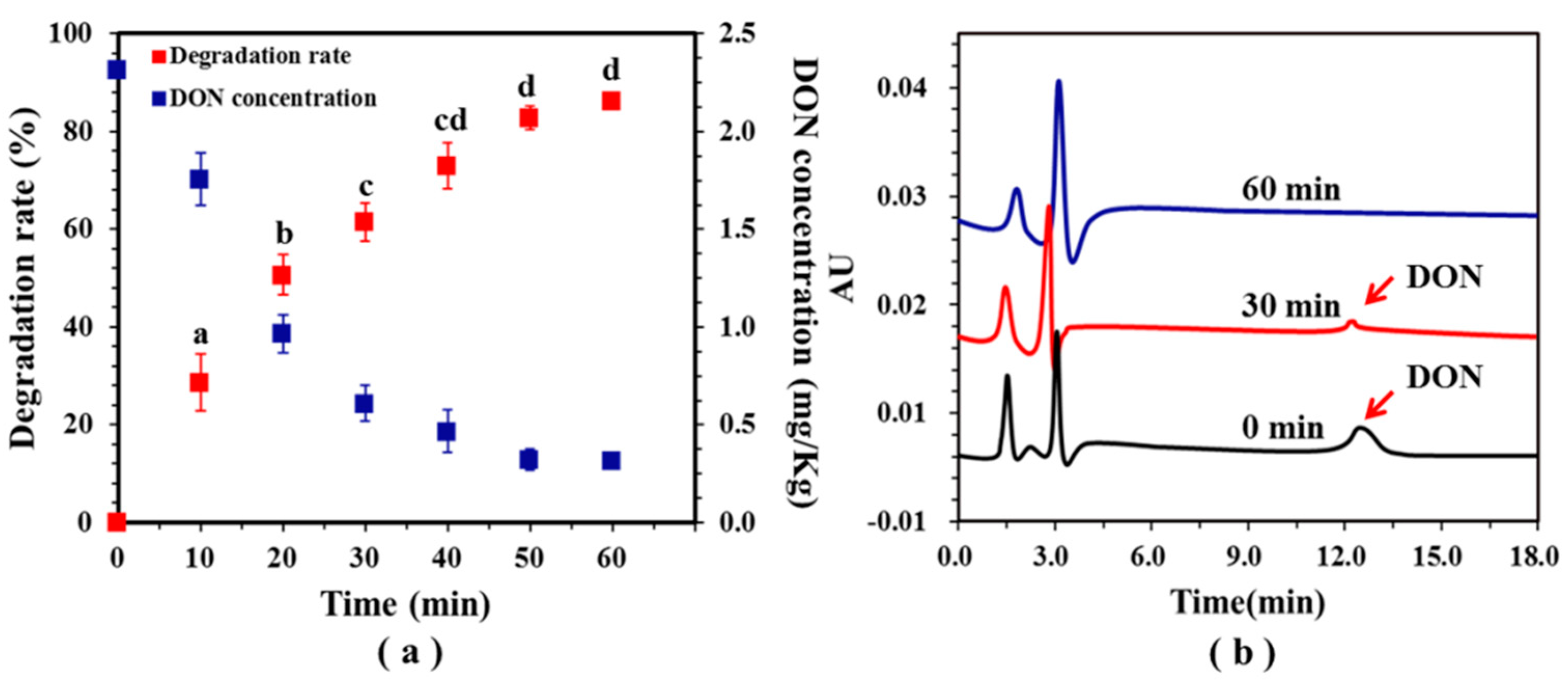
2.2. Effect of Potential on DON Degradation
- C = concentration of DON at time t (mg/L);
- C0 = initial concentration of DON (mg/L);
- k = thermal degradation rate constant (min−1);
- t = processing time (min).
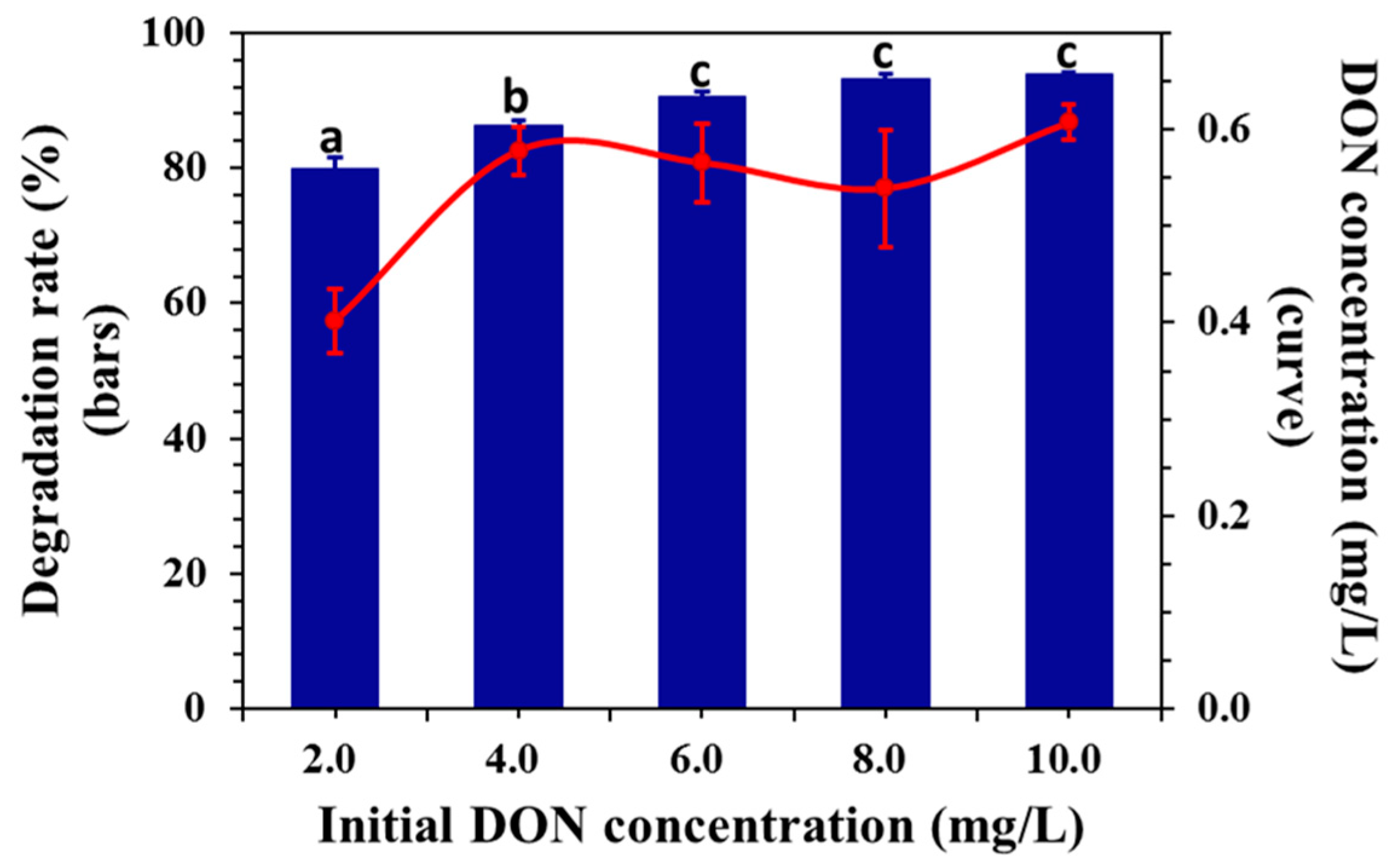
2.3. Effect of Initial Concentration on DON Degradation
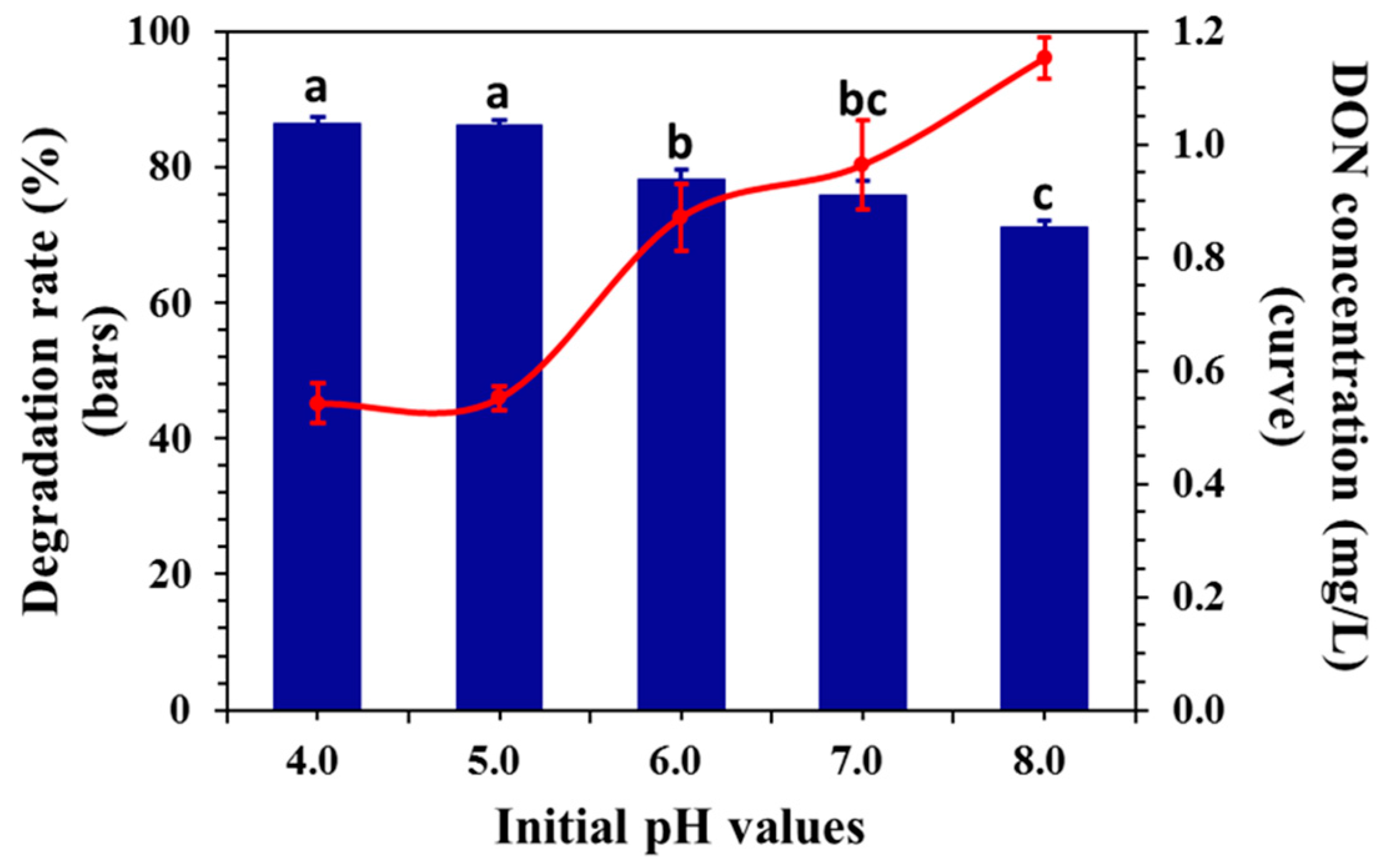
2.4. Effect of Initial pH on DON Degradation
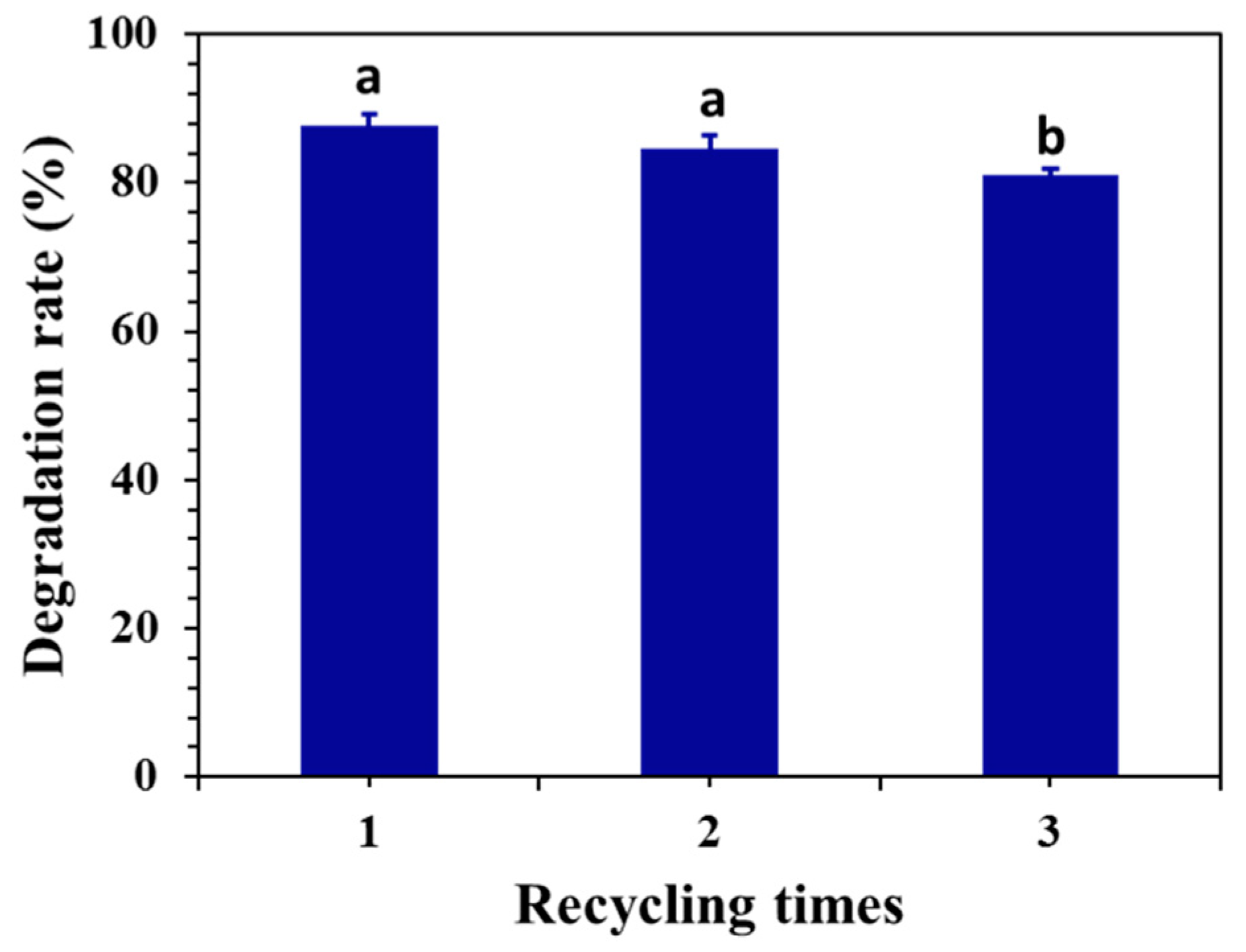
2.5. Electrolytic Stability and SEM of the Graphite Anode
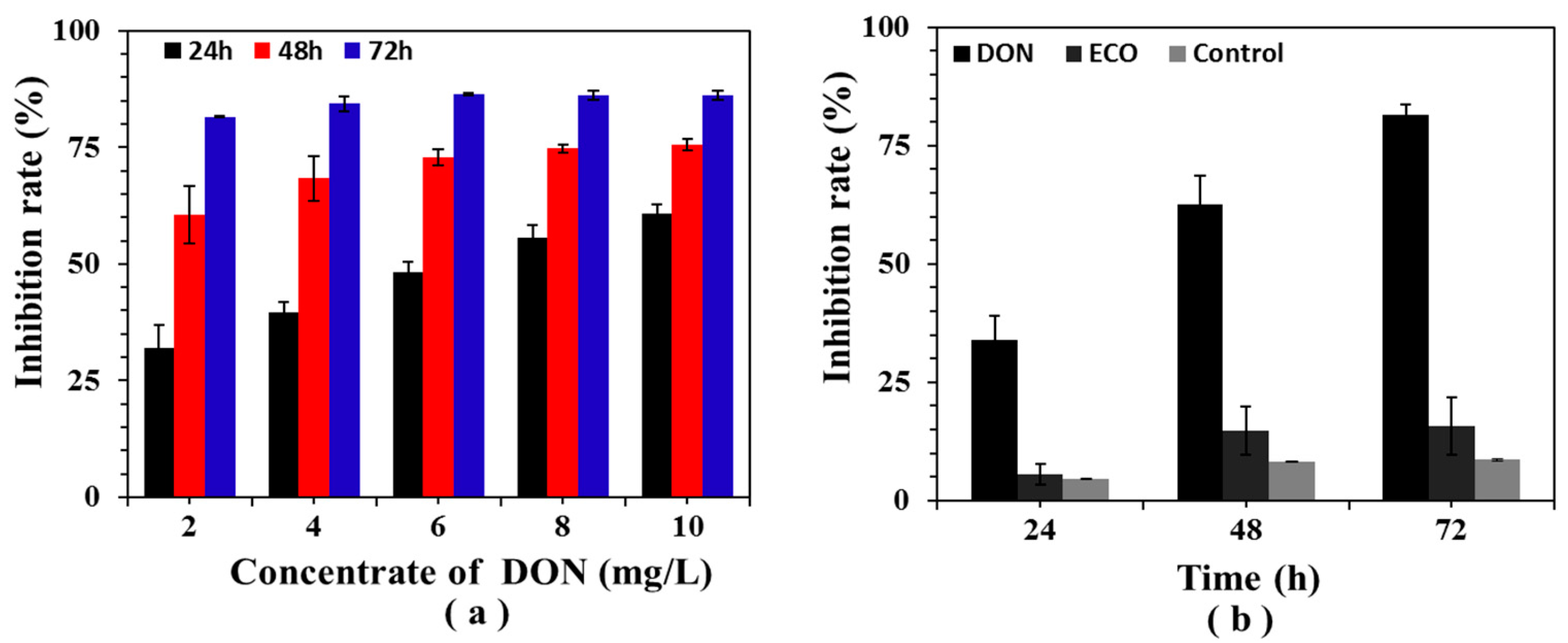
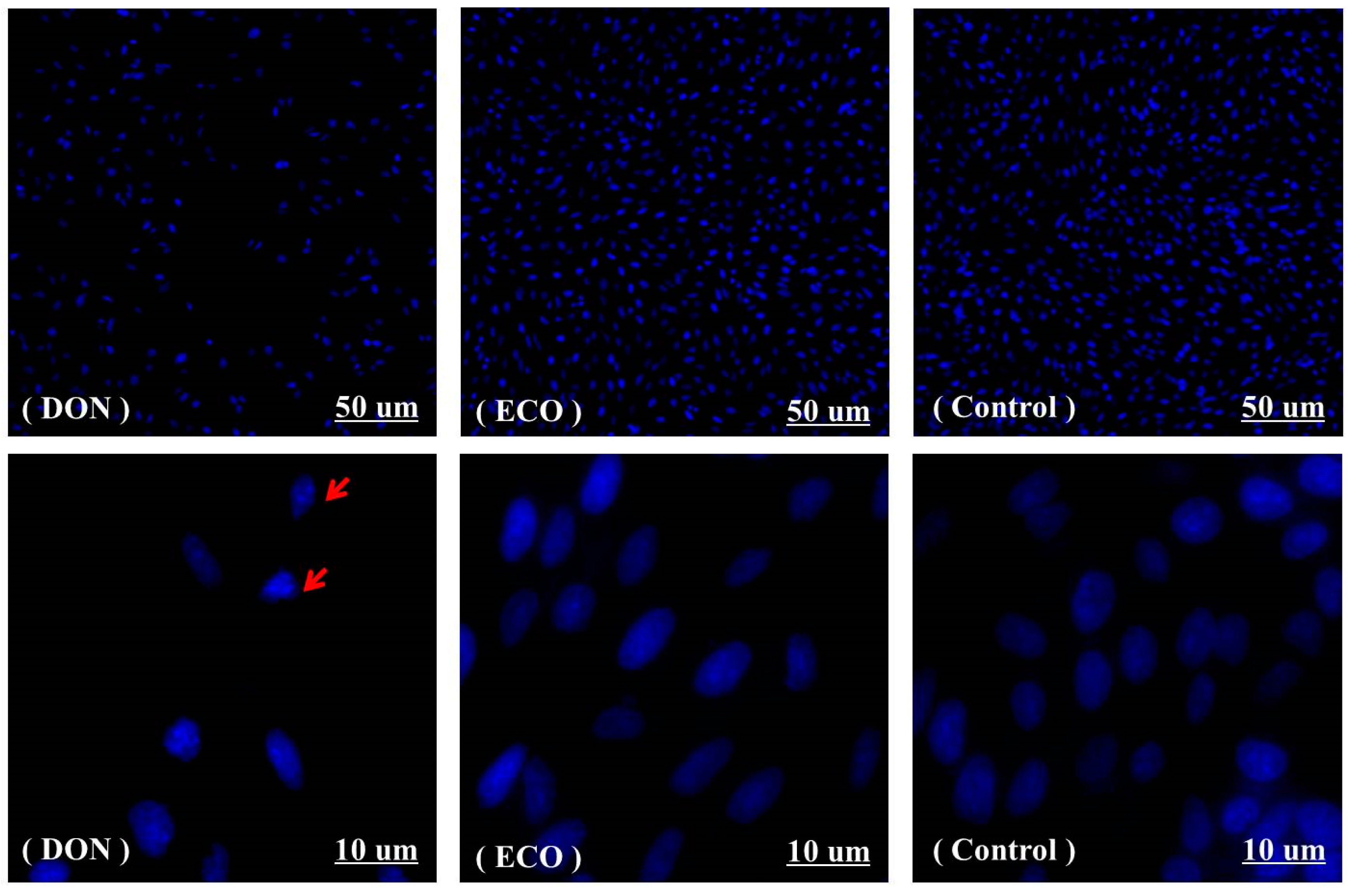
2.6. Cytotoxicity Assay of DON and Its Electrochemically Oxidized Products
2.7. Electrochemical Oxidation of DON in WDGS
3. Materials and Methods
3.1. Materials
3.2. Experimental Setup
3.3. The Efficiency of Different Electrode Materials on the Degradation Rate of DON
3.4. Electroche of DON with the Graphite Electrode
3.5. Electrolytic Stability of the Graphite Electrode
3.6. Cytotoxicity Assay of DON and Its Electrochemically Oxidized Products
- In: Inhibition ratio;
- EO: Optical density(OD) value of the experimental groups;
- CO: OD value of the blank groups.
3.7. DAPI Staining
3.8. Electrochemical Oxidation of WDGS
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sulyok, M.; Berthiller, F.; Krska, R.; Schuhmacher, R. Development and validation of a liquid chromatography/tandem mass spectrometric method for the determination of 39 mycotoxins in wheat and maize. Rapid Commun. Mass Spectrom. 2006, 20, 2649–2659. [Google Scholar] [CrossRef] [PubMed]
- Placinta, C.M.; D’Mello, J.P.F.; Macdonald, A.M.C. A review of worldwide contamination of cereal grains and animal feed with Fusarium mycotoxins. Anim. Feed. Sci. Technol. 1999, 78, 21–37. [Google Scholar] [CrossRef]
- Parry, D.W.; Jenkinson, P.; McLeod, L. Fusarium ear blight (SCAB) in small-grain cereals—A review. Plant. Pathol. 2007, 44, 207–238. [Google Scholar] [CrossRef]
- Heyndrickx, E.; Sioen, I.; Huybrechts, B.; Callebaut, A.; De Henauw, S.; De Saeger, S. Human biomonitoring of multiple mycotoxins in the Belgian population: Results of the BIOMYCO study. Environ. Int. 2015, 84, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J. Toxicological mechanisms and potential health effects of deoxynivalenol and nivalenol. World Mycotoxin J. 2010, 3, 323–347. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Wang, S.B.; Wang, R.G.; Zhang, W.; Wang, P.L.; Su, X.O. Phosphoproteome Analysis Reveals the Molecular Mechanisms Underlying Deoxynivalenol-Induced Intestinal Toxicity in IPEC-J2 Cells. Toxins 2016, 8, 270. [Google Scholar] [CrossRef] [PubMed]
- Awad, W.; Ghareeb, K.; Böhm, J.; Zentek, J. The Toxicological Impacts of the Fusarium Mycotoxin, Deoxynivalenol, in Poultry Flocks with Special Reference to Immunotoxicity. Toxins 2013, 5, 912–925. [Google Scholar] [CrossRef]
- Pestka, J.; Smolinski, A.T. Deoxynivalenol: Toxicology and Potential Effects on Humans. J. Toxicol. Environ. Health Part B 2005, 8, 39–69. [Google Scholar] [CrossRef]
- Miao, Y.; Liangkai, C.; Zhao, P.; Di, W.; Yadong, S.; Hanyin, W.; Ping, Y.; Hong, Y.; Andreas, N.; Liegang, L. Embryotoxicity Caused by DON-Induced Oxidative Stress Mediated by Nrf2/HO-1 Pathway. Toxins 2017, 9, 188. [Google Scholar] [CrossRef]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar] [CrossRef]
- Alsheikh-Ali, A.A.; Trikalinos, T.A.; Ruthazer, R.; Terrin, N.; Wong, J.B.; Sarnak, M.J.; III, N.A.M.E.; Kent, D.M. Risk of arrhythmic and nonarrhythmic death in patients with heart failure and chronic kidney disease. Am. Hear. J. 2011, 161, 204–209.e1. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.W.S.; Jacobs, S.; Lee, B. Gastrointestinal dysfunction among intensive care unit patients. Crit. Care Med. 1987, 15, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Toor, S.S.; Rosendahl, L.; Nielsen, M.P.; Glasius, M.; Rudolf, A.; Iversen, S.B. Continuous production of bio-oil by catalytic liquefaction from wet distiller’s grain with solubles (WDGS) from bio-ethanol production. Biomass- Bioenergy 2012, 36, 327–332. [Google Scholar] [CrossRef]
- Arias, R.P.; Unruhsnyder, L.J.; Scholljegerdes, E.J.; Baird, A.N.; Johnson, K.D.; Buckmaster, D.; Lemenager, R.P.; Lake, S.L. Effects of feeding corn modified wet distillers grain plus solubles co-ensiled with direct-cut forage on feedlot performance, carcass characteristics, and diet digestibility of finishing steers. J. Anim. Sci. 2012, 90, 3574–3583. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, Y.; Caupert, J. Survey of mycotoxins in U.S. distiller’s dried grains with solubles from 2009 to 2011. J. Agric. Food Chem. 2012, 60, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Awad, W.A.; Ghareeb, K.; Böhm, J.; Zentek, J. Decontamination and detoxification strategies for the Fusarium mycotoxin deoxynivalenol in animal feed and the effectiveness of microbial biodegradation. Food Additives & Contaminants. Food Addit. Contam. 2010, 27, 510–520. [Google Scholar] [CrossRef]
- Bai, X.; Sun, C.; Liu, D.; Luo, X.; Li, D.; Wang, J.; Wang, N.; Chang, X.; Zong, R.; Zhu, Y. Photocatalytic degradation of deoxynivalenol using graphene/ZnO hybrids in aqueous suspension. Appl. Catal. B Environ. 2017, 204, 11–20. [Google Scholar] [CrossRef]
- Park, B.J.; Takatori, K.; Sugita-Konishi, Y.; Kim, I.-H.; Lee, M.-H.; Han, D.-W.; Chung, K.-H.; Hyun, S.O.; Park, J.-C. Degradation of mycotoxins using microwave-induced argon plasma at atmospheric pressure. Surf. Coatings Technol. 2007, 201, 5733–5737. [Google Scholar] [CrossRef]
- Bretz, M.; Beyer, M.; Cramer, B.; Anja Knecht, A.; Humpf, H.U. Thermal Degradation of the Fusarium Mycotoxin Deoxynivalenol. J. Agric. Food Chem. 2006, 54, 6445–6451. [Google Scholar] [CrossRef]
- Sun, C.; Ji, J.; Wu, S.; Sun, C.; Pi, F.; Zhang, Y.; Tang, L.; Sun, X. Saturated aqueous ozone degradation of deoxynivalenol and its application in contaminated grains. Food Control. 2016, 69, 185–190. [Google Scholar] [CrossRef]
- Sirés, I.; Brillas, E.; Oturan, M.A.; Rodrigo, M.A.; Panizza, M. Electrochemical advanced oxidation processes: today and tomorrow. A review. Environ. Sci. Pollut. Res. 2014, 21, 8336–8367. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Huitle, C.A.; Brillas, E. Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: A general review. Appl. Catal. B: Environ. 2009, 87, 105–145. [Google Scholar] [CrossRef]
- Moreira, F.C.; Boaventura, R.A.R.; Brillas, E.; Vilar, V.J.P. Electrochemical advanced oxidation processes: A review on their application to synthetic and real wastewaters. Appl. Catal. B Environ. 2017, 202, 217–261. [Google Scholar] [CrossRef]
- Scialdone, O. Electrochemical oxidation of organic pollutants in water at metal oxide electrodes: A simple theoretical model including direct and indirect oxidation processes at the anodic surface. Electrochimica Acta 2009, 54, 6140–6147. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Ferro, S. Electrochemical oxidation of organic pollutants for the wastewater treatment: direct and indirect processes. Chem. Soc. Rev. 2006, 35, 1324–1340. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Segura, S.; Brillas, E. Mineralization of the recalcitrant oxalic and oxamic acids by electrochemical advanced oxidation processes using a boron-doped diamond anode. Water Res. 2011, 45, 2975–2984. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Segura, S.; Brillas, E.; Cornejo-Ponce, L.; Salazar, R. Effect of the Fe3+/Cu2+ ratio on the removal of the recalcitrant oxalic and oxamic acids by electro-Fenton and solar photoelectro-Fenton. Solar Energy 2016, 124, 242–253. [Google Scholar] [CrossRef]
- Chaplin, B. Critical review of electrochemical advanced oxidation processes for water treatment applications. Environ. Sci. Process. Impacts 2014, 16, 1182–1203. [Google Scholar] [CrossRef]
- Dominguez, C.M.; Oturan, N.; Romero, A.; Santos, A.; Oturan, M.A. Lindane degradation by electrooxidation process: Effect of electrode materials on oxidation and mineralization kinetics. Water Res. 2018, 135, 220–230. [Google Scholar] [CrossRef]
- Wang, H.; Ren, Z.J. Bioelectrochemical metal recovery from wastewater: A review. Water Res. 2014, 66, 219–232. [Google Scholar] [CrossRef]
- Oturan, N.; van Hullebusch, E.; Zhang, H.; Mazeas, L.; Budzinski, H.; Le Menach, K.; Oturan, M. Occurrence and Removal of Organic Micropollutants in Landfill Leachates Treated by Electrochemical Advanced Oxidation Processes. Environ. Sci. Technol. 2015, 49, 12187–12196. [Google Scholar] [CrossRef] [PubMed]
- Samet, Y.; Agengui, L.; Abdelhédi, R. Anodic oxidation of chlorpyrifos in aqueous solution at lead dioxide electrodes. J. Electroanal. Chem. 2010, 650, 152–158. [Google Scholar] [CrossRef]
- Jeon, B.Y.; Park, D.H. Improvement of ethanol production by electrochemical redox coupling of Zymomonas mobilis and Saccharomyces cerevisiae. J. Microbiol. Biotechnol. 2010, 20, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Tinkara, R.; Tina, E.e.; Maja, Z.i.J.; Andrej, M.; Domen, L.t.; Bojan, S. The effects of electrochemical oxidation on in-vivo fluorescence and toxin content in Microcystis aeruginosa culture. Chin. J. Oceanol. Limnol. 2018, 36, 1091–1102. [Google Scholar] [CrossRef]
- Oliveira, S.C.; Diculescu, V.C.; Palleschi, G.; Compagnone, D.; Oliveira-Brett, A.M. Electrochemical oxidation of ochratoxin A at a glassy carbon electrode and in situ evaluation of the interaction with deoxyribonucleic acid using an electrochemical deoxyribonucleic acid-biosensor. Anal. Chim. Acta 2007, 588, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Raju, G.B.; Karuppiah, M.T.; Latha, S.S.; Parvathy, S.; Prabhakar, S. Treatment of wastewater from synthetic textile industry by electrocoagulation–electrooxidation. Chem. Eng. J. 2008, 144, 51–58. [Google Scholar] [CrossRef]
- Gan, Y.P.; HUANG, H.; ZHANG, W.-k. Electrocatalytic oxidation of methanol on carbon-nanotubes/graphite electrode modified with platinum and molybdenum oxide nanoparticles. Trans. Nonferrous Met. Soc. China 2007, 17, 214–219. [Google Scholar] [CrossRef]
- Un, U.T.; Altay, U.; Koparal, A.S.; Ogutveren, U.B. Complete treatment of olive mill wastewaters by electrooxidation. Chem. Eng. J. 2008, 139, 445–452. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Keller, J.; Brillas, E.; Radjenovic, J. Removal of organic contaminants from secondary effluent by anodic oxidation with a boron-doped diamond anode as tertiary treatment. J. Hazard. Mater. 2015, 283, 551–557. [Google Scholar] [CrossRef]
- Kapałka, A.; Fóti, G.; Comninellis, C. Kinetic modelling of the electrochemical mineralization of organic pollutants for wastewater treatment. J. Appl. Electrochem. 2007, 38, 7–16. [Google Scholar] [CrossRef]
- Li, X.Y.; Cui, Y.H.; Feng, Y.J.; Xie, Z.M.; Gu, J.D. Reaction pathways and mechanisms of the electrochemical degradation of phenol on different electrodes. Water Res. 2005, 39, 1972–1981. [Google Scholar] [CrossRef]
- Vidal, A.; Sanchis, V.; Ramos, A.J.; Marín, S. Thermal stability and kinetics of degradation of deoxynivalenol, deoxynivalenol conjugates and ochratoxin A during baking of wheat bakery products. Food Chem. 2015, 178, 276–286. [Google Scholar] [CrossRef]
- Zou, J.; Peng, X.; Li, M.; Xiong, Y.; Wang, B.; Dong, F.; Wang, B. Electrochemical oxidation of COD from real textile wastewaters: Kinetic study and energy consumption. Chemosphere 2017, 171, 332–338. [Google Scholar] [CrossRef]
- Komtchou, S.; Dirany, A.; Drogui, P.; Bermond, A. Removal of carbamazepine from spiked municipal wastewater using electro-Fenton process. Environ. Sci. Pollut. Res. 2015, 22, 11513–11525. [Google Scholar] [CrossRef]
- Fajardo, A.S.; Seca, H.F.; Martins, R.C.; Corceiro, V.N.; Freitas, I.F.; Quinta-Ferreira, M.E.; Quinta-Ferreira, R.M. Electrochemical oxidation of phenolic wastewaters using a batch-stirred reactor with NaCl electrolyte and Ti/RuO 2 anodes. J. Electroanal. Chem. 2017, 785, 180–189. [Google Scholar] [CrossRef]
- Tang, W.Z.; Huang, C.P. Effect of chlorine content of chlorinated phenols on their oxidation kinetics by Fenton’s reagent. Chemosphere 1996, 33, 1621–1635. [Google Scholar] [CrossRef]
- Govindaraj, M.; Rathinam, R.; Sukumar, C.; Uthayasankar, M.; Pattabhi, S. Electrochemical oxidation of bisphenol-A from aqueous solution using graphite electrodes. Environ. Technol. 2013, 34, 503–511. [Google Scholar] [CrossRef]
- Boudenne, J.L.; Cerclier, O.; Galéa, J.; Van der Vlist, E. Electrochemical oxidation of aqueous phenol at a carbon black slurry electrode. Appl. Catal. A: Gen. 1996, 143, 185–202. [Google Scholar] [CrossRef]
- Frysz, C.A.; Chung, D.D.L. Electrochemical behavior of flexible graphite. Carbon 1997, 35, 858–860. [Google Scholar] [CrossRef]
- Pierson, H.O. 5 - Molded Graphite: Processing, Properties, and Applications. In Handbook of Carbon, Graphite, Diamonds and Fullerenes; Pierson, H.O., Ed.; William Andrew Publishing: Oxford, UK, 1993; pp. 87–121. [Google Scholar] [CrossRef]
- Nevskaia, D.M.; Castillejos-López, E.; Muñoz, V.; Ruiz, A. Adsorption of Aromatic Compounds from Water by Treated Carbon Materials. Environ. Sci. Technol. 2004, 38, 5786–5796. [Google Scholar] [CrossRef]
- Chen, J.-L.; Chiou, G.-C.; Wu, C.-C. Electrochemical oxidation of 4-chlorophenol with granular graphite electrodes. Desalination 2010, 264, 92–96. [Google Scholar] [CrossRef]
- Ghareeb, K.; Awad, W.; Böhm, J.; Zebeli, Q. Impacts of the feed contaminant deoxynivalenol on the intestine of monogastric animals: Poultry and swine. J. Appl. Toxicol. 2014, 35, 327–337. [Google Scholar] [CrossRef]
- Awad, W.; Aschenbach, J.R.; Zentek, J. Cytotoxicity and metabolic stress induced by deoxynivalenol in the porcine intestinal IPEC-J2 cell line. J. Anim. Physiol. Anim. Nutr. 2011, 96, 709–716. [Google Scholar] [CrossRef]
- Wan, L.Y.; Turner, P.C.; El-Nezami, H. Individual and combined cytotoxic effects of Fusarium toxins (deoxynivalenol, nivalenol, zearalenone and fumonisins B1) on swine jejunal epithelial cells. Food Chem. Toxicol. 2013, 57, 276–283. [Google Scholar] [CrossRef]
- Yunxia, Y.; Song, Y.; Na, L.; Haibin, X.; Yunyun, G.; Yongning, W.; Peilong, W.; Xiaoou, S.; Yucai, L.; Sarah, D.S. Transcription factor FOXO3a is a negative regulator of cytotoxicity of Fusarium mycotoxin in GES-1 cells. Toxicol. Sci. 2018, 166, 370–381. [Google Scholar] [CrossRef]
- Dai, Y.; Xie, H.; Xu, Y. Evaluation of deoxynivalenol-induced toxic effects on mouse endometrial stromal cells: Cell apoptosis and cell cycle. Biochem. Biophys. Res. Commun. 2017, 483, 572–577. [Google Scholar] [CrossRef]
- Chang, H.; Mi, M.; Ling, W.; Zhu, J.; Zhang, Q.; Wei, N.; Zhou, Y.; Tang, Y.; Yuan, J. Structurally related cytotoxic effects of flavonoids on human cancer cells in vitro. Arch. Pharmacal Res. 2008, 31, 1137–1144. [Google Scholar] [CrossRef]
- Kang, S.H.; Lee, H.J.; Jeong, S.J.; Kwon, H.Y.; Kim, J.H.; Yun, S.M.; Kim, J.H.; Lee, H.J.; Lee, E.O.; Ahn, K.S.; et al. Protective effect of Bojungbangdocktang on cisplatin-induced cytotoxicity and apoptosis in MCF-10A breast endothelial cells. Environ. Toxicol. Pharmacol. 2009, 28, 430–438. [Google Scholar] [CrossRef]
- Cromwell, G.L.; Herkelman, K.L.; Stahly, T.S. Physical, chemical, and nutritional characteristics of distillers dried grains with solubles for chicks and pigs. J. Anim. Sci. 1993, 71, 679–686. [Google Scholar] [CrossRef][Green Version]
- Wang, L.; Shao, H.; Luo, X.; Wang, R.; Li, Y.; Li, Y.; Luo, Y.; Chen, Z. Effect of Ozone Treatment on Deoxynivalenol and Wheat Quality. PLoS One 2016, 11, e0147613. [Google Scholar] [CrossRef]
- Fastinger, N.D.; Latshaw, J.D.; Mahan, D.C. Amino acid availability and true metabolizable energy content of corn distillers dried grains with solubles in adult cecectomized roosters. Poult. Sci. 2006, 85, 1212–1216. [Google Scholar] [CrossRef]
- Lyberg, K.; Borling, J.; Lindberg, J.E. Characterization and nutrient evaluation of wet and dried wheat distillers’ grain for growing pigs. Anim. Feed. Sci. Technol. 2013, 186, 45–52. [Google Scholar] [CrossRef]

| Treatment | L* | a* | b* |
|---|---|---|---|
| Before ECO | 62.72 ± 0.09 a | 8.97 ± 0.43 a | 35.92 ± 1.54 a |
| After ECO | 62.59 ± 0.66 a | 9.78 ± 0.94 a | 36.79 ± 0.88 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, S.; Li, X.; Zhao, C.; Gao, J.; Yuan, W.; Zhang, J. The Degradation of Deoxynivalenol by Using Electrochemical Oxidation with Graphite Electrodes and the Toxicity Assessment of Degradation Products. Toxins 2019, 11, 478. https://doi.org/10.3390/toxins11080478
Xiong S, Li X, Zhao C, Gao J, Yuan W, Zhang J. The Degradation of Deoxynivalenol by Using Electrochemical Oxidation with Graphite Electrodes and the Toxicity Assessment of Degradation Products. Toxins. 2019; 11(8):478. https://doi.org/10.3390/toxins11080478
Chicago/Turabian StyleXiong, Suli, Xiao Li, Changsong Zhao, Jingqi Gao, Wenjuan Yuan, and Jie Zhang. 2019. "The Degradation of Deoxynivalenol by Using Electrochemical Oxidation with Graphite Electrodes and the Toxicity Assessment of Degradation Products" Toxins 11, no. 8: 478. https://doi.org/10.3390/toxins11080478
APA StyleXiong, S., Li, X., Zhao, C., Gao, J., Yuan, W., & Zhang, J. (2019). The Degradation of Deoxynivalenol by Using Electrochemical Oxidation with Graphite Electrodes and the Toxicity Assessment of Degradation Products. Toxins, 11(8), 478. https://doi.org/10.3390/toxins11080478





