Pain and Lethality Induced by Insect Stings: An Exploratory and Correlational Study
Abstract
:1. Introduction
2. Results and Discussion
2.1. Pain Ratings of Insect Stings
2.2. Lethality of Stinging Insect Venoms
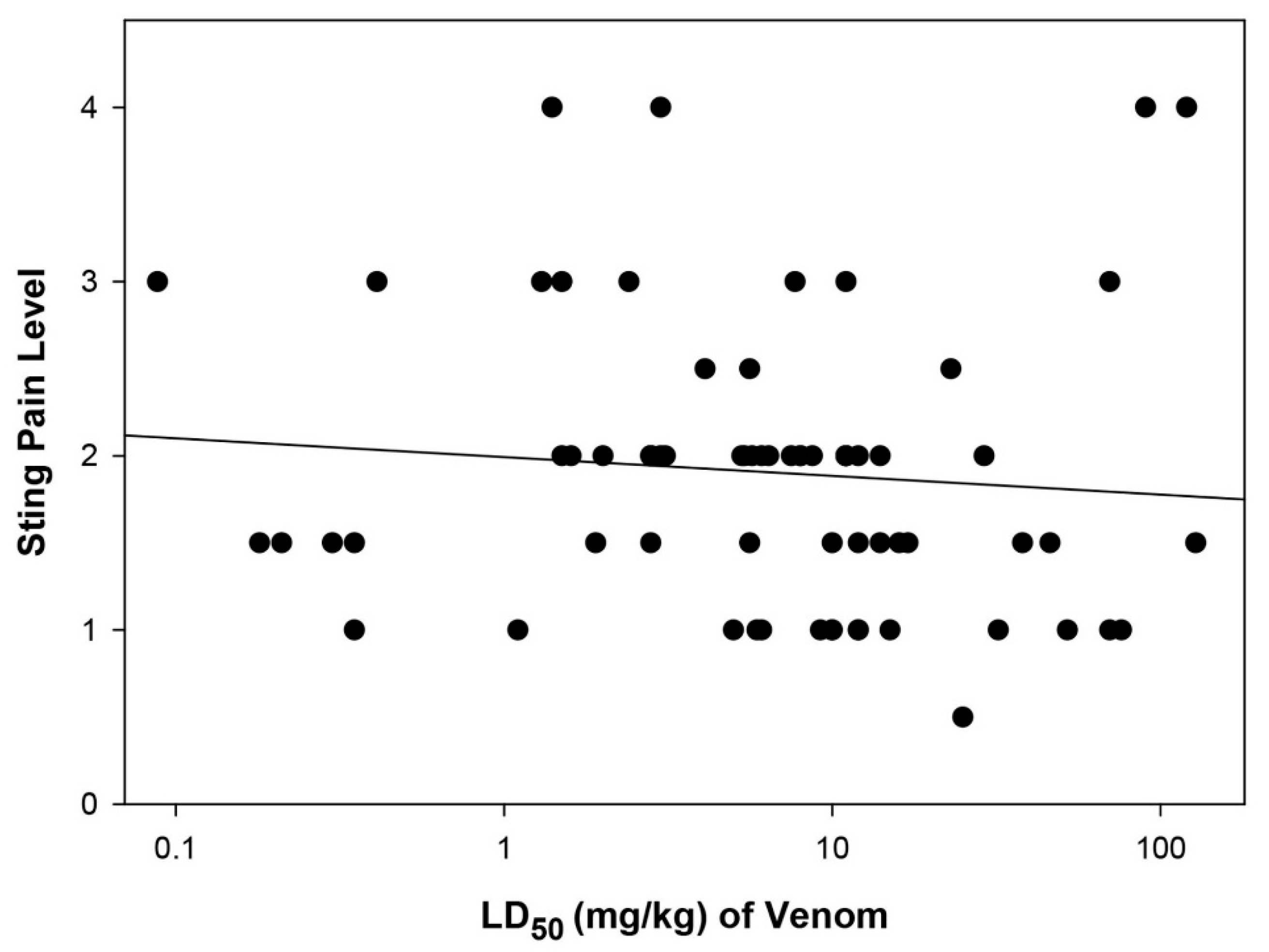
2.3. Relationship between Sting Pain Level and Lethality of Stinging Insect Venoms
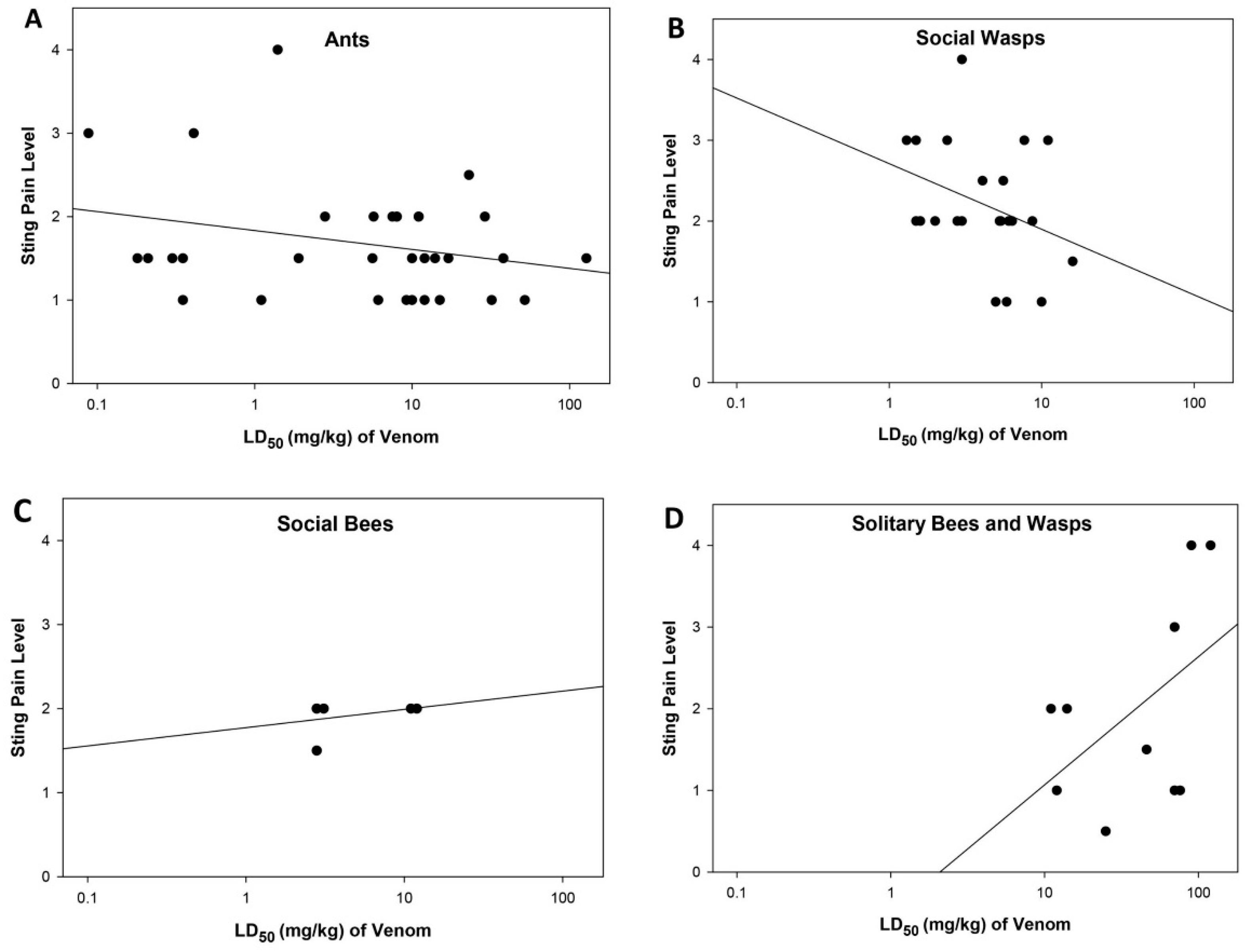
2.4. Relationship between Sting Pain Level, Lethality, and Sociality of Stinging Insects
2.5. Natural History of Stinging Insects and How It Can Help Guide Discovery of Interesting Venom Peptides, Proteins, and Other Natural Pharmaceuticals
2.6. Targeting Promising Species of Stinging Insects for Discovery of New Pharmaceuticals Based upon Sting Pain and Lethality
3. Materials and Methods
3.1. Insects
3.2. Pain Measurement
3.3. Venom
3.4. Physiological Measurements
Funding
Conflicts of Interest
References
- Freeman, T.; Tracy, J. (Eds.) Stinging Insect Allergy: A Clinician’s Guide; Springer: Heidelberg, Germany, 2017; p. 321. ISBN 978-3-319-46190-8. [Google Scholar]
- Bettini, S. (Ed.) Arthropod Venoms. In Handbook of Experimental Pharmacology; Springer: Berlin, Germany, 1978; Volume 48, p. 978. ISBN 3-540-082228-X. [Google Scholar]
- Maschwitz, U.; Hahn, M.; Schönegge, P. Paralysis of prey in ponerine ants. Naturwissenschaften 1979, 66, 213–214. [Google Scholar] [CrossRef]
- Piek, T. (Ed.) Venoms of the Hymenoptera: Biochemical, Pharmacological and Behavioral Aspects; Academic Press: London, UK, 1986; p. 570. ISBN 0-12-554770-6. [Google Scholar]
- Palma, M.S. Insect venom peptides. In Handbook of Biologically Active Peptides; Kastin, A.J., Ed.; Academic Press: Amsterdam, The Netherlands, 2006; pp. 409–416. ISBN 13: 978-0-12-369442-3. [Google Scholar]
- Schmidt, J.O. Biochemistry of insect venoms. Annu. Rev. Entomol. 1982, 27, 339–368. [Google Scholar] [CrossRef] [PubMed]
- Torres, A.F.C.; Quinet, Y.P.; Havt, A.; Rádis-Baptista, G.; Martins, A.M.C. Molecular pharmacology and toxinology of venom from ants. In Analytical Procedures to Biomedical Applications; Rádis Baptista, G., Ed.; IntechOpen: London, UK, 2013; pp. 207–222. [Google Scholar] [CrossRef]
- Touchard, A.; Samira, R.; Aili, S.R.; Gonçalves, E.; Fox, P.; Escoubas, P.; Orivel, J.; Nicholson, G.M.; Dejean, A. The Biochemical Toxin Arsenal from Ant Venoms. Toxins 2016, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.O. Hymenopteran venoms: Striving toward the ultimate defense against vertebrates. In Insect Defense: Adaptations and Strategies of Prey and Predators; Evans, D.L., Schmidt, J.O., Eds.; SUNY Press: Albany, NY, USA, 1990; pp. 387–419. ISBN 0-88706-896-0. [Google Scholar]
- Schmidt, J.O. Venom and the good life in tarantula hawks (Hymenoptera: Pompilidae): How to eat, not be eaten, and live long. J. Kansas Entomol. Soc. 2004, 77, 402–413. [Google Scholar] [CrossRef]
- Pessini, A.C.; Kanashiro, A.; Malvar, D.C.; Machado, R.R.; Soares, D.M.; Figueiredo, M.J.; Kalapothakis, E.; Souza, G.E.P. Inflammatory mediators involved in the nociceptive and oedematogenic responses induced by Tityus serrulatus scorpion venom injected into rat paws. Toxicon 2008, 52, 729–736. [Google Scholar] [CrossRef]
- Pucca, M.B.; Cerni, F.A.; Cordeiro, F.A.; Peigneur, S.; Cunha, T.M.; Tytgat, J.; Arantes, E.C. Ts8 scorpion toxin inhibits the Kv4.2 channel and produces nociception in vivo. Toxicon 2016, 119, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Julius, D. TRP channels and pain. Annu. Rev. Cell Dev. Biol. 2013, 29, 355–384. [Google Scholar] [CrossRef]
- Min, J.W.; Liu, W.H.; He, X.H.; Peng, B.W. Different types of toxins targeting TRPV1 in pain. Toxicon 2013, 71, 66–75. [Google Scholar] [CrossRef]
- Klint, J.K.; Smith, J.J.; Vetter, I.; Rupasinghe, D.B.; Er, S.Y.; Senff, S.; Herzig, V.; Mobli, M.; Lewis, R.J.; Bosmans, F.; et al. Seven novel modulators of the analgesic target NaV1.7 uncovered using a high-throughput venom-based discovery approach. Br. J. Pharmacol. 2015, 172, 2445–2458. [Google Scholar] [CrossRef]
- Chen, J.; Guan, S.M.; Sun, W.; Fu, H. Melittin, the major pain-producing substance of bee venom. Neurosci. Bull. 2016, 32, 265–272. [Google Scholar] [CrossRef]
- Smith, M.L. Honey bee sting pain index by body location. PeerJ 2014, 2, e338. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.O.; Blum, M.S.; Overal, W.L. Hemolytic activities of stinging insect venoms. Arch. Insect Biochem. Physiol. 1984, 1, 155–160. [Google Scholar] [CrossRef]
- Schmidt, J.O.; Blum, M.S.; Overal, W.L. Comparative enzymology of venoms from stinging Hymenoptera. Toxicon 1986, 24, 907–921. [Google Scholar] [CrossRef]
- Aili, S.R.; Touchard, A.; Koh, J.M.S.; Dejean, A.; Orivel, J.; Padula, M.P.; Escoubas, P.; Nicholson, G.M. Comparisons of protein and peptide complexity in poneroid and formicoid ant venoms. J. Proteome Res. 2016, 15, 3039–3054. [Google Scholar] [CrossRef] [PubMed]
- Aili, S.R.; Touchard, A.; Petitclerc, F.; Dejean, A.; Orivel, J.; Padula, M.P.; Escoubas, P.; Nicholson, G.M. Combined peptidomic and proteomic analysis of electrically stimulated and manually dissected venom from the South American bullet ant Paraponera clavata. J. Proteome Res. 2017, 16, 1339–1351. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Baek, J.H.; Yoon, K.A. Differential properties of venom peptides and proteins in solitary vs. social hunting wasps. Toxins 2016, 8, 32. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.R.; Rikli, H.G.; Schmidt, J.O.; Evans, M.S. A reexamination of poneratoxin from the bullet ant Paraponera clavata. Peptides 2017, 98, 51–62. [Google Scholar] [CrossRef]
- Robinson, S.D.; Mueller, A.; Clayton, D.; Starobova, H.; Hamilton, B.R.; Payne, R.J.; Vetter, I.; King, G.F.; Undheim, E.B. A comprehensive portrait of the venom of the giant red bull ant, Myrmecia gulosa, reveals a hyperdiverse hymenopteran toxin gene family. Sci. Adv. 2018, 4, eaau4640. [Google Scholar] [CrossRef]
- MacConnell, J.G.; Blum, M.S.; Fales, H.M. Alkaloid from fire ant venom: Identification and synthesis. Science 1970, 168, 840–841. [Google Scholar] [CrossRef]
- Bernheimer, A.W.; Avigad, L.S.; Schmidt, J.O. A hemolytic polypeptide from the venom of the red harvester ant, Pogonomyrmex barbatus. Toxicon 1980, 18, 271–278. [Google Scholar] [CrossRef]
- Argiolas, A.; Pisano, J.J. Bombolitins, a new class of mast cell degranulating peptides from the venom of the bumblebee Megabombus pennsylvanicus. J. Biol. Chem. 1985, 260, 1437–1444. [Google Scholar] [PubMed]
- Schmidt, J.O. Evolutionary responses of solitary and social Hymenoptera to predation by primates and overwhelmingly powerful vertebrate predators. J. Hum. Evol. 2014, 71, 12–19. [Google Scholar]
- Schmidt, J.O. The Sting of the Wild; Johns Hopkins Univ. Press: Baltimore, MD, USA, 2016; p. 280. ISBN 978-1-4214-1928-2. [Google Scholar]
- Mortari, J.M.R.; do Couto, L.L.; dos Anjos, L.C.; Mourão, C.B.F.; Camargos, T.S.; Vargas, J.A.G.; Oliveira, F.N.; Gati, C.C.; Schwartz, C.A.; Schwartz, E.F. Pharmacological characterization of Synoeca cyanea venom: An aggressive social wasp widely distributed in the Neotropical region. Toxicon 2012, 59, 163–170. [Google Scholar] [CrossRef] [PubMed]
- da Silva, J.R.; de Souza, A.Z.; Pirovani, C.P.; Costa, H.; Silva, A.; Dias, J.C.T.; Delabie, J.H.C.; Fontana, R. Assessing the proteomic activity of the venom of the ant Ectatomma tuberculatum (Hymenoptera: Formicidae: Ectatomminae). Psyche 2018, 2018, 7915464. [Google Scholar] [CrossRef]
- Schmidt, J.O.; Blum, M.S. A harvester ant venom: Chemistry and pharmacology. Science 1978, 200, 1064–1066. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.O.; Snelling, G.C. Pogonomyrmex anzensis Cole: Does an unusual harvester ant species have an unusual venom? J. Hymenopt. Res. 2009, 18, 322–325. [Google Scholar]
- Pessoa, W.F.B.; Silva, L.L.C.; Dias, L.O.; Delabie, J.H.C.; Costa, H.; Romano, C.C. Analysis of protein composition and bioactivity of Neoponera villosa venom (Hymenoptera: Formicidae). Int. J. Mol. Sci. 2016, 17, 513. [Google Scholar] [CrossRef]
- Starr, C.K. A simple pain scale for field comparison of hymenopteran stings. J. Entomol. Sci. 1985, 20, 225–232. [Google Scholar] [CrossRef]
- Schmidt, J.O. Chemistry, pharmacology and chemical ecology of ant venoms. In Venoms of the Hymenoptera; Piek, T., Ed.; Academic Press: London, UK, 1986; pp. 425–508. ISBN 0-12-554770-6. [Google Scholar]
- Schmidt, J.O. Allergy to venomous insects. In The Hive and the Honey Bee; Graham, J.M., Ed.; Dadant and Sons: Hamilton, IL, USA, 2015; pp. 907–952. ISBN 978-0-915698-16-5. [Google Scholar]
- Schmidt, J.O. Toxinology of venoms from the honeybee genus Apis. Toxicon 1995, 33, 917–927. [Google Scholar] [CrossRef]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
| Species (Common Name) | Sting Pain | LD50 (mg/kg) |
|---|---|---|
| Ants | ||
| Solenopsis invicta (red fire ant) | 1 | |
| S. xyloni (southern fire ant) | 1 | |
| S. geminata (tropical fire ant) | 1 | |
| Tetraponera sp. (Old World twig ant) | 1 | 0.35 |
| Daceton armigerum (trap-jawed ant) | 1 | 1.1 |
| Myrmica rubra (European fire ant) | 1 | 6.1 |
| Bothroponera strigulosa | 1 | 9.2 |
| Leptogenys kitteli | 1 | 10 |
| Pseudomyrmex gracilis (twig ant) | 1 | 12 |
| P. nigrocinctus (bullhorn acacia ant) | 1.5 | 1.9 |
| Ectatomma ruidum, | 1 | 15 |
| E. tuberculatum | 1.5 | 0.3 |
| E. quadridens | 1.5 | 17 |
| Ectatomma sp. | 17 | |
| Opthalmopone berthoudi (big-eye ant) | 1 | 32 |
| Harpegnathos venator | 1 | 52 |
| Brachyponera chinensis (needle ant) | 1 | |
| B. sennaarensis (Samsum ant) | 1.5 | 5.6 |
| Myrmecia gulosa (red bulldog ant) | 1.5 | 0.18 |
| M. browning (bulldog ant) | 0.18 | |
| M. tarsata (bulldog ant) | 0.18 | |
| M. simillima (bulldog ant) | 1.5 | 0.21 |
| M. rufinodis (bulldog ant) | 1.5 | 0.35 |
| M. pilosula (Jack jumper ant) | 2 | 5.7 |
| Eciton burchelli (army ant) | 1.5 | 10 |
| Anochetus inermis (a trap-jaw ant) | 1.5 | 12 |
| Dinoponera gigantea (giant ant) | 1.5 | 14 |
| Paltothyreus tarsatus (giant stink ant) | 1.5 | 38 |
| Megaponera analis (Matabele ant) | 1.5 | 128 |
| Pachycondyla crassinoda | 2 | 2.8 |
| Neoponera villosa | 2 | 7.5 |
| N. commutate (termite-hunting ant) | 2 | 11 |
| Streblognathus aethiopicus (African giant ant) | 2 | 8 |
| Diacamma rugosum | 2 | 8 |
| Platythyrea lamellose | 2 | 11 |
| P. cribrinodis | 42 | |
| Odontoponera transversa | 2 | 29 |
| Rhytidoponera metallica | 2 | |
| Odontomachus bauri (trap-jaw ant) | 2.5 | 23 |
| O. infandus (trap-jaw ant) | 33 | |
| O. chelifer (trap-jaw ant) | 37 | |
| Pogonomyrmex cunicularius (Argentine harvester ant) | 3 | 0.088 |
| Pogonomyrmex (North American harvester ants) (20 spp.) | 3 | 0.12–0.7 |
| Paraponera clavata (bullet ant) | 4 | 1.4 |
| Manica bradleyi | 6 | |
| Social Wasps | ||
| Polybia occidentalis (polybia wasp) | 1 | 5 |
| P. rejecta (polybia wasp) | 1.5 | 16 |
| P. simillima (polybia wasp) | 2.5 | 4.1 |
| P. sericea (polybia wasp) | 6.1 | |
| Ropalidia flavobrunnea | 1 | 5.9 |
| Ropalidia sp. | 1 | 10 |
| Ropalidia (Icarielia) sp. | 14 | |
| Belonogaster sp. (thin paper wasp) | 1.5 | |
| B. juncea colonialis (fire-tail wasp) | 2 | 3 |
| Brachygastra mellifica (honey wasp) | 2 | 1.5 |
| Vespula germanica (yellowjacket wasp) | 2 | 2.8 |
| V. vulgaris (yellowjacket wasp) | 2 | 5.4 |
| V. pensylvanica (yellowjacket wasp) | 2 | 6.4 |
| V. vidua (yellowjacket wasp) | 2.6 | |
| V. consobrina (yellowjacket wasp) | 2.8 | |
| Polistes instabilis (paper wasp) | 2 | 1.6 |
| P. arizonicus (paper wasp) | 2 | 2 |
| P. infuscatus (paper wasp) | 3 | 1.3 |
| P. erythrocephalus (paper wasp) | 3 | 1.5 |
| P. canadensis. (paper wasp) | 3 | 2.4 |
| P. tepidus (paper wasp) | 3 | 7.7 |
| P. annularis (paper wasp) | 3 | 11 |
| Parachartergus fraternus (artistic wasp) | 2 | 5.3 |
| Dolichovespula maculata (baldfaced hornet) | 2 | 6.1 |
| D. arenaria (aerial yellowjacket) | 2 | 8.7 |
| Mischocyttarus sp. (a paper wasp) | 2 | |
| Agelaia myrmecophila (fire wasp) | 2.5 | 5.6 |
| Provespa sp. (nocturnal hornet) | 2.5 | |
| Synoeca septentrionalis (warrior wasp) | 4 | 3 |
| Vespa luctuosa (hornet) | 1.6 | |
| V. tropica (hornet) | 2.8 | |
| V. simillima (hornet) | 3.1 | |
| V. mandarinia (giant hornet) | 4.1 | |
| Apoica pallens (night wasp) | 13.5 | |
| Social Bees | ||
| Apis florea (dwarf honey bee) | 1.5 | 2.8 |
| A. mellifera (honey bee) | 2 | 2.8 |
| A. dorsata (giant honey bee) | 2 | 2.8 |
| A. cerana (Eastern honey bee) | 2 | 3.1 |
| Bombus impatiens (bumble bee) | 2 | 11 |
| B. sonorus (bumble bee) | 2 | 12 |
| Solitary Bees | ||
| Dieunomia heteropoda (giant sweat bee) | 0.5 | 25 |
| Triepeolus sp. (cuckoo bee) | 0.5 | |
| Xenoglossa angustior (squash bee) | 1 | 12 |
| Habropoda pallida (white-faced bee) | 1 | 70 |
| Diadasia rinconis (cactus bee) | 1 | 76 |
| Emphoropsis pallida | 1 | |
| Lasioglossum spp. (sweat bee) | 1 | |
| Ericrocis lata (cuckoo bee) | 1 | |
| Euglossa dilemma (orchid bee) | 1.5 | |
| Xylocopa rufa (nocturnal carpenter bee) | 2 | 11 |
| X. californica (carpenter bee) | 2 | 14 |
| X. veripuncta (carpenter bee) | 33 | |
| Xylocopa sp. (giant Bornean bee) | 2.5 | |
| Centris pallida (palo verde bee) | 56 | |
| Solitary Wasps | ||
| Sapyga pumila (club-horned wasp) | 0.5 | |
| Eumeninae spp. (potter wasps) | 1 | |
| Sphecius convallis (cicada killer wasp) | 1 | |
| S. grandis (cicada killer wasp) | 1.5 | 46 |
| Sphex pensylvanicus (great black wasp) | 1 | |
| Chlorion cyaneum (cockroach-hunter wasp) | 1 | |
| Triscolia ardens (scarab-hunter wasp) | 1 | |
| Sceliphron caementarium (mud dauber wasp) | 1 | |
| Euodynerus crypticus (water walking wasp) | 1 | |
| Dasymutilla thetis (little velvet ant) | 1 | |
| D. gloriosa (velvet ant) | 2 | |
| D. klugii (cow killer velvet ant) | 3 | 70 |
| Pepsis grossa (tarantula hawk wasp) | 4 | 90 |
| P. thisbe (tarantula hawk wasp) | 4 | 120 |
| Mutillidae sp. (small nocturnal velvet ant) | 1.5 | |
| Crioscolia flammicoma (scoliid wasp) | 62 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, J.O. Pain and Lethality Induced by Insect Stings: An Exploratory and Correlational Study. Toxins 2019, 11, 427. https://doi.org/10.3390/toxins11070427
Schmidt JO. Pain and Lethality Induced by Insect Stings: An Exploratory and Correlational Study. Toxins. 2019; 11(7):427. https://doi.org/10.3390/toxins11070427
Chicago/Turabian StyleSchmidt, Justin O. 2019. "Pain and Lethality Induced by Insect Stings: An Exploratory and Correlational Study" Toxins 11, no. 7: 427. https://doi.org/10.3390/toxins11070427
APA StyleSchmidt, J. O. (2019). Pain and Lethality Induced by Insect Stings: An Exploratory and Correlational Study. Toxins, 11(7), 427. https://doi.org/10.3390/toxins11070427