The Mechanisms of Social Immunity Against Fungal Infections in Eusocial Insects
Abstract
1. Introduction
2. Multi-Defense Strategies of Social Immunity
2.1. Avoidance Strategy
2.2. Resistance Strategy
2.2.1. Sanitary Care of Contaminated Insects
2.2.2. Exclusion of Infected and Dead Insects
2.2.3. Food Protection
2.2.4. Antifungal Secretions
2.2.5. Social Immunization
2.3. Tolerance Strategy
2.3.1. Nutrition and Reproduction in Tolerance
2.3.2. Metabolic Tolerance
3. Mechanism of Social Immunity
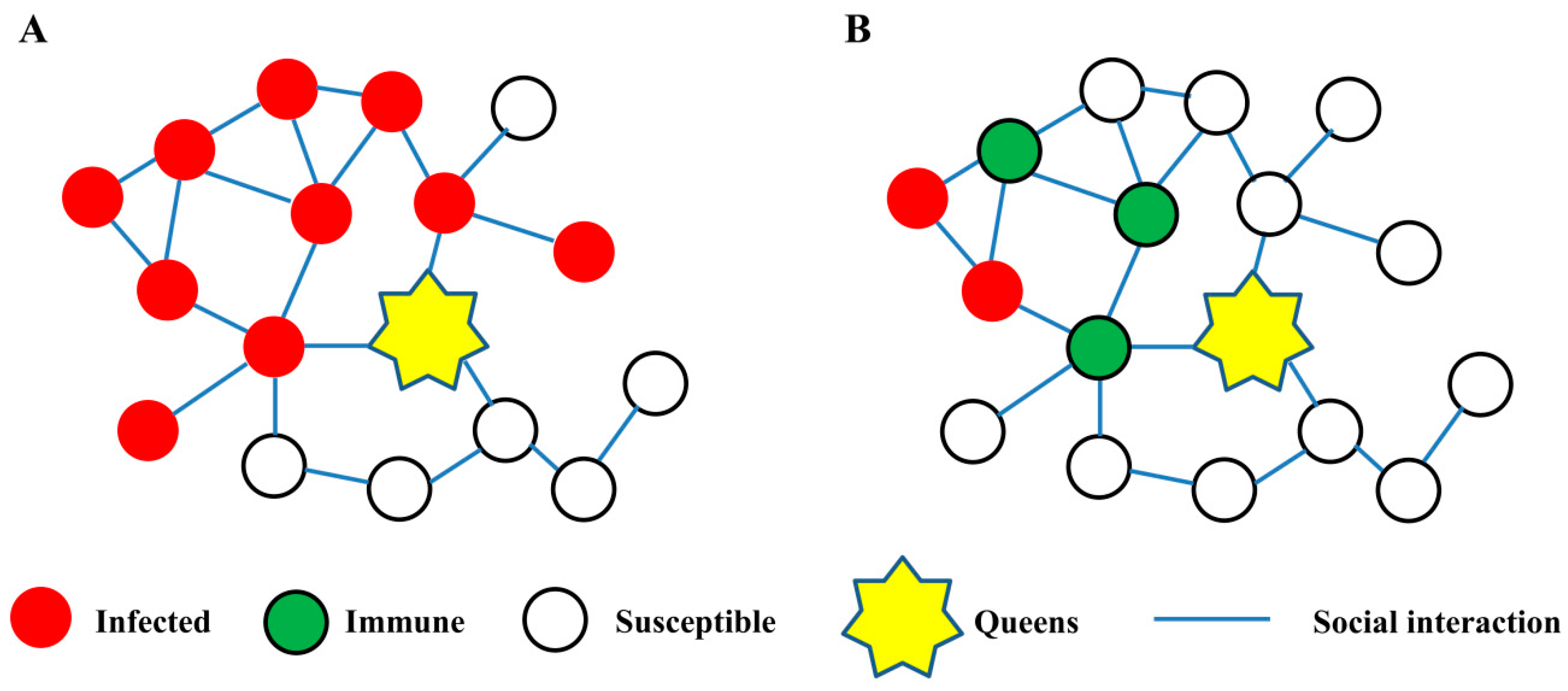
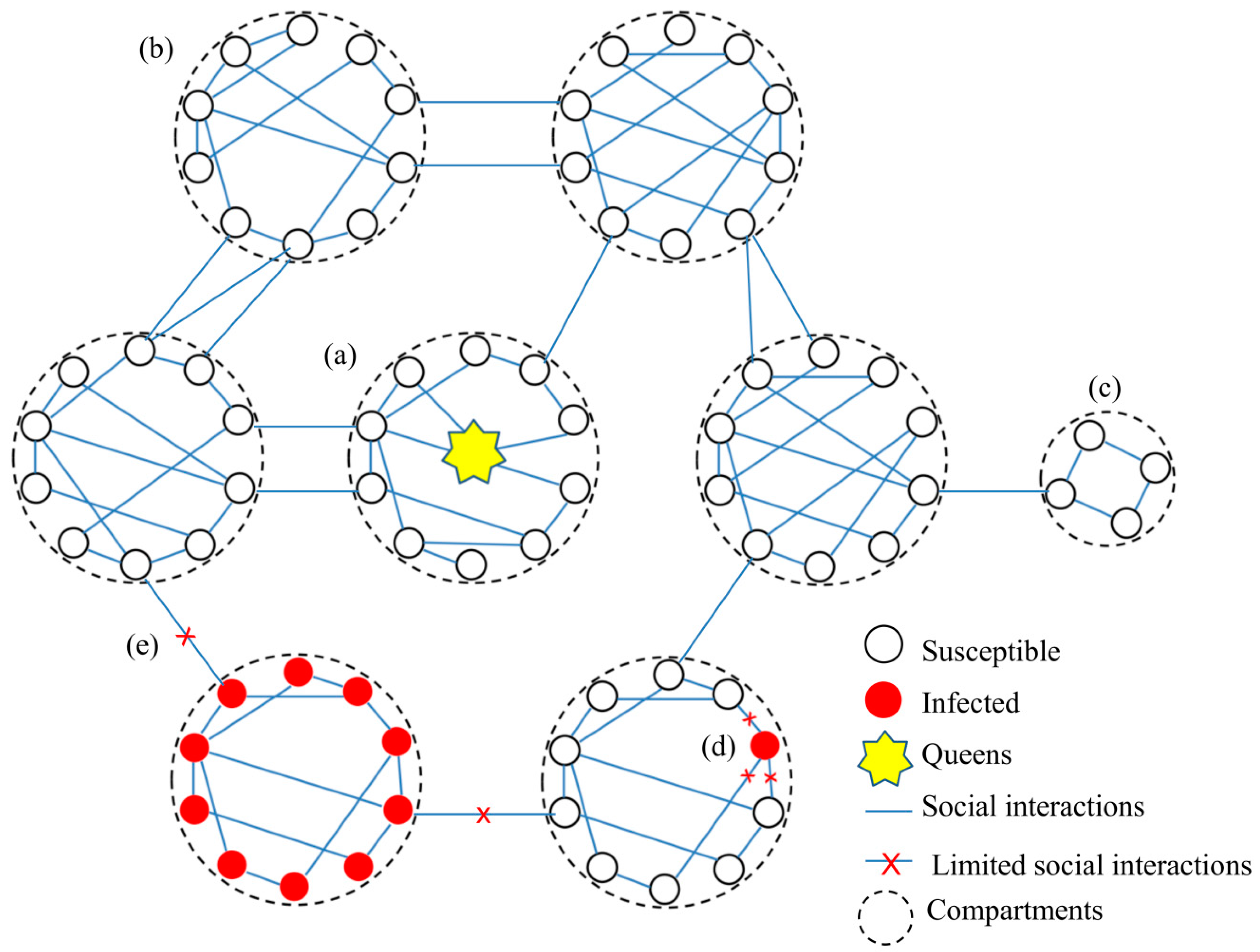
3.1. Social Interaction Network in Social Immunity
3.1.1. The Network Structure
3.1.2. The Network Plasticity
3.2. Molecular Basis of Social Immunity
3.2.1. Chemosensory Regulation of Social Immunity
3.2.2. Physiological Regulation of Social Immunity
4. The Role of Fungal Toxins in the Evolution of Social Immunity
5. Conclusions and Future Work
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cremer, S.; Armitage, S.A.; Schmid-Hempel, P. Social immunity. Curr. Biol. 2007, 17, R693–R702. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Traniello, J.F.A.; Bulmer, M.S. Ecology, behavior and evolution of disease resistance in termites. In Biology of Termites: A Modern Synthesis; Bignell, D.E., Roisin., Y., Lo, N., Eds.; Springer: New York, NY, USA, 2011; pp. 165–191. [Google Scholar]
- Cremer, S.; Pull, C.D.; Fürst, M.A. Social immunity: Emergence and evolution of colony-level disease protection. Annu. Rev. Entomol. 2018, 63, 105–123. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Hempel, P. Parasites in Social Insects; Princeton Univ. Press: Princeton, NJ, USA, 1998. [Google Scholar]
- Hamilton, W.D. Kinship, recognition, disease, and intelligence: Constraints of social evolution. In Animal Societies: Theories and Facts; Ito, Y., Brown, J., Kikkawa, J., Eds.; Sci. Soc. Press: Tokyo, Japan, 1987; pp. 81–102. [Google Scholar]
- Fefferman, N.H.; Traniello, J.F.A.; Rosengaus, R.B.; Calleri II, D.V. Disease prevention and resistance in social insects: Modeling the survival consequences of immunity, hygienic behavior, and colony organization. Behav. Ecol. Sociobiol. 2007, 61, 565–577. [Google Scholar] [CrossRef]
- Wilson-Rich, N.; Spivak, M.; Fefferman, N.H.; Starks, P.T. Genetic, individual, and group facilitation of disease resistance in Insect Societies. Annu. Rev. Entomol. 2009, 54, 405–423. [Google Scholar] [CrossRef]
- Stroeymeyt, N.; Grasse, A.V.; Crespi, A.; Mersch, D.P.; Cremer, S.; Keller, L. Social network plasticity decreases disease transmission in eusocial insect. Science 2018, 362, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Stroeymeyt, N.; Casillas-Pe´rez, B.; Cremer, S. Organisational immunity in social insects. Curr. Opin. Insect Sci. 2014, 5, 1–15. [Google Scholar] [CrossRef]
- Siebeneicher, S.R.; Bradleigh^Vinson, S.; Kenerley, C.M. Infection of the red imported fire ant by Beauveria bassiana through various routes of exposure. J. Invertebr. Pathol. 1992, 59, 280–285. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Maxmen, A.B.; Coates, L.E.; Traniello, J.F.A. Disease resistance: A benefit of sociality in the dampwood termite Zootermopsis angusticollis (Isoptera: Termopsidae). Behav. Ecol. Sociobiol. 1998, 44, 125–134. [Google Scholar] [CrossRef]
- Myles, T.G. Alarm, aggregation, and defense by Reticulitermes flavipes in response to a naturally occurring isolate of Metarhizium anisopliae. Sociobiology 2002, 40, 243–255. [Google Scholar]
- Yanagawa, A.; Yokohari, F.; Shimizu, S. Defense mechanism of the termite, Coptotermes formosanus Shiraki, to entomopathogenic fungi. J. Invertebr. Pathol. 2008, 97, 165–170. [Google Scholar] [CrossRef]
- Konrad, M.; Vyleta, M.L.; Theis, F.J.; Stock, M.; Tragust, S.; Klatt, M.; Drescher, V.; Marr, C.; Ugelvig, L.V.; Cremer, S. Social transfer of pathogenic fungus promotes active immunisation in ant colonies. PLoS Biol. 2012, 10, e1001300. [Google Scholar] [CrossRef]
- Qiu, H.L.; Lu, L.H.; Shi, Q.X.; He, Y.R. Fungus exposed Solenopsis invicta ants benefit from grooming. J. Insect Behav. 2014, 27, 678–691. [Google Scholar] [CrossRef]
- Konrad, M.; Pull, C.D.; Metzler, S.; Seif, K.; Naderlinger, E.; Grasse, A.V.; Cremer, S. Ants avoid superinfections by performing risk-adjusted sanitary care. Proc. Natl. Acad. Sci. USA 2018, 115, 2782–2787. [Google Scholar] [CrossRef]
- Hamilton, C.; Bulmer, M.S. Molecular antifungal defenses in subterranean termites: RNA interference reveals in vivo roles of termicins and GNBPs against a naturally encountered pathogen. Dev. Comp. Immunol. 2012, 36, 372–377. [Google Scholar] [CrossRef]
- Vestergaard, S.; Butt, T.M.; Bresciani, J.; Gillespie, A.T.; Eilenberg, J. Light and electron microscopy studies of the infection of the western flower thrips frankliniella occidentalis (Thysanoptera: thripidae) by the entomopathogenic fungus metarhizium anisopliae. J. Invertebr. Pathol. 1999, 73, 25–33. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.Y.; Robert, A. Inhibition of Metarhizium anisopliae in the alimentary tract of the eastern subterranean termite Reticulitermes flavipes. J. Invertebr. Pathol. 2009, 101, 130–136. [Google Scholar] [CrossRef]
- Chouvenc, T.; Su, N.Y.; Robert, A. Cellular encapsulation in the eastern subterranean termite, Reticulitermes flavipes (Isoptera), against infection by the entomopathogenic fungus Metarhizium anisopliae. J. Invertebr. Pathol. 2009, 101, 234–241. [Google Scholar] [CrossRef]
- Feng, P.; Shang, Y.; Cen, K.; Wang, C. Fungal biosynthesis of the bibenzoquinone oosporein to evade insect immunity. Proc. Natl. Acad. Sci. USA 2015, 112, 11365–11370. [Google Scholar] [CrossRef]
- Wei, G.; Lai, Y.; Wang, G.; Chen, H.; Li, F.; Wang, S. Insect pathogenic fungus interacts with the gut microbiota to accelerate mosquito mortality. Proc. Natl. Acad. Sci. USA 2017, 114, 5994–5999. [Google Scholar] [CrossRef]
- Cremer, S.; Sixt, M. Analogies in the evolution of individual and social immunity. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2009, 364, 129–142. [Google Scholar] [CrossRef]
- Rueppell, O.; Hayworth, M.K.; Ross, N.P. Altruistic self-removal of health-compromised honey bee workers from their hive. J. Evol. Biol. 2010, 23, 1538–1546. [Google Scholar] [CrossRef]
- de Roode, J.C.; Lefèvre, T. Behavioral immunity in insects. Insects 2012, 3, 789–820. [Google Scholar] [CrossRef] [PubMed]
- Tranter, C.; LeFevre, L.; Evison, S.E.F.; Hughes, W.O.H. Threat detection: Contextual recognition and response to parasites by ants. Behav. Ecol. 2014, 26, 396–405. [Google Scholar] [CrossRef]
- Mburu, D.M.; Ochola, L.; Maniania, N.K.; Njagi, P.G.; Gitonga, L.M.; Ndung’u, M.W.; Wanjoya, A.K.; Hassanali, A. Relationship between virulence and repellency of entomopathogenic isolates of Metarhizium anisopliae and Beauveria bassiana to the termite Macrotermes michaelseni. J. Insect Physiol. 2009, 55, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Yanagawa, A.; Shimizu, S. Resistance of the termite, Coptotermes formosanus Shiraki to Metarhizium anisopliae due to grooming. BioControl 2007, 52, 75–85. [Google Scholar] [CrossRef]
- Walker, T.N.; Hughes, W.O.H. Adaptive social immunity in leaf-cutting ants. Biol. Lett. 2009, 446–448. [Google Scholar] [CrossRef]
- Diehl-Fleig, E.; Lucchese, M.E. Reacoes comportamentais de operarias de Acromyrmex striatus (Hymenoptera, Formicidae) na presenca de fungos entomopatogenicos. Rev. Bras. Entomol. 1991, 35, 101–107. [Google Scholar]
- Rosengaus, R.B.; Jordan, C.; Lefebvre, M.L.; Traniello, J.F.A. Pathogen Alarm behavior in a termite: A new form of communication in social insects. Naturwissenschaften 1999, 86, 544–548. [Google Scholar] [CrossRef]
- Marikovsky, P.I. On some features of behavior of the ants Formica rufa L. infected with fungous disease. Insect. Soc. 1962, 9, 173–179. [Google Scholar] [CrossRef]
- Vieira-Neto, E.H.M.; Mundim, F.M.; Vasconcelos, H.L. Hitchhiking behaviour in leaf-cutter ants: An experimental evaluation of three hypotheses. Insect. Soc. 2006, 53, 326–332. [Google Scholar] [CrossRef]
- Christe, P.; Oppliger, A.; Bancala, F.; Castella, G.; Chapuisat, M. Evidence for collective medication in ants. Ecol. Lett. 2002, 6, 19–22. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Guldin, M.R.; Traniello, J.F.A. Inhibitory effect of termite fecal pellets on fungal spore germination. J. Chem. Ecol. 1998, 24, 1697–1706. [Google Scholar] [CrossRef]
- Bulmer, M.S.; Bachelet, I.; Raman, R.; Rosengaus, R.B.; Sasisekharan, R. Targeting an antimicrobial effector function in insect immunity as a pest control strategy. Proc. Natl. Acad. Sci. USA 2009, 106, 12652–12657. [Google Scholar] [CrossRef]
- Tranter, C.; Graystock, P.; Shaw, C.; Lopes, J.F.S.; Hughes, W.O.H. Sanitizing the fortress: Protection of ant brood and nest material by worker antibiotics. Behav. Ecol. Sociobiol. 2014, 68, 499–507. [Google Scholar] [CrossRef][Green Version]
- Baracchi, D.; Turillazzi, S. Differences in venom and cuticular peptides in individuals of Apis mellifera (Hymenoptera: Apidae) determined by MALDI-TOF MS. J. Insect Physiol. 2010, 56, 366–375. [Google Scholar] [CrossRef]
- Chouvenc, T.; Efstathion, C.A.; Elliott, M.L.; Su, N.Y. Extended disease resistance emerging from the faecal nest of a subterranean termite. Proc. R. Soc. Lond. B 2013, 280, 20131885. [Google Scholar] [CrossRef]
- Seipke, R.F.; Barke, J.; Brearley, C.; Hill, L.; Yu, D.W.; Goss, R.J.; Hutchings, M.I. A single Streptomyces symbiont makes multiple antifungals to support the fungus farming ant Acromyrmex octospinosus. PLoS ONE 2011, 6, e22028. [Google Scholar] [CrossRef]
- Gliński, Z.; Buczek, K. Response of the Apoidea to fungal infections. Apiacta 2003, 38, 183–189. [Google Scholar]
- Rosengaus, R.B.; Lefebvre, M.L.; Traniello, J.F.A. Inhibition of Fungal Spore Germination by Nasutitermes: Evidence for a possible antiseptic role of soldier defensive secretions. J. Chem. Ecol. 2000, 26, 21–39. [Google Scholar] [CrossRef]
- Mitaka, Y.; Mori, N.; Matsuura, K. Multi-functional roles of a soldier-specific volatile as a worker arrestant, primer pheromone and an antimicrobial agent in a termite. Proc. R. Soc. Lond. B 2017, 284, 20171134. [Google Scholar] [CrossRef]
- Tragust, S.; Mitteregger, B.; Barone, V.; Konrad, M.; Ugelvig, L.V.; Cremer, S. Ants disinfect fungus-exposed brood by oral uptake and spread of their poison. Curr. Biol. 2013, 23, 76–82. [Google Scholar] [CrossRef]
- Ugelvig, L.V.; Cremer, S. Social prophylaxis: Group interaction promotes collective immunity in ant colonies. Curr. Biol. 2007, 17, 1967–1971. [Google Scholar] [CrossRef]
- Heinze, J.; Walter, B. Moribund ants leave their nests to die in social isolation. Curr. Biol. 2010, 20, 249–252. [Google Scholar] [CrossRef]
- Leclerc, J.B.; Detrain, C. Loss of attraction for social cues leads to fungal-infected Myrmica rubra ants withdrawing from the nest. Anim. Behav. 2017, 129, 133–141. [Google Scholar] [CrossRef]
- Sun, Q.; Haynes, K.F.; Zhou, X.G. Dynamic changes in death cues modulate risks and rewards of corpse management in a social insect. Funct. Ecol. 2016, 31, 697–706. [Google Scholar] [CrossRef]
- Davis, H.E.; Meconcelli, S.; Radek, R.; Mcmahon, D.P. When to care and when to kill: Termites shape their collective response based on stage of infection. Sci. Rep.-UK 2018, 8, 14433. [Google Scholar] [CrossRef]
- Yanagawa, A.; Fujiwara-Tsujii, N.; Akino, T.; Yoshimura, T.; Yanagawa, T.; Shimizu, S. Musty odor of entomopathogens enhances disease-prevention behaviors in the termite Coptotermes formosanus. J. Invertebr. Pathol. 2011, 108, 1–6. [Google Scholar] [CrossRef]
- Renucci, M.; Tirard, A.; Provost, E. Complex undertaking behavior in Temnothorax lichtensteini ant colonies: From corpse-burying behavior to necrophoric behavior. Insect. Soc. 2011, 58, 9–16. [Google Scholar] [CrossRef]
- Diez, L.; Lejeune, P.; Detrain, C. Keep the nest clean: Survival advantages of corpse removal in ants. Biol. Lett. 2014, 10, 20140306. [Google Scholar] [CrossRef]
- Qiu, H.L.; Lu, L.H.; Shi, Q.X.; Tu, C.C.; Lin, T.; He, Y.R. Differential necrophoric behaviour of the ant Solenopsis invicta towards fungal-infected corpses of workers and pupae. Bull. Entomol. Res. 2015, 105, 607–614. [Google Scholar] [CrossRef]
- Swanson, J.A.; Torto, B.; Kells, S.A.; Mesce, K.A.; Tumlinson, J.H.; Spivak, M. Odorants that induce hygienic behavior in honeybees: Identification of volatile compounds in chalkbrood-infected honeybee larvae. J. Chem. Ecol. 2009, 35, 1108–1116. [Google Scholar] [CrossRef]
- Pull, C.D.; Ugelvig, L.V.; Wiesenhofer, F.; Grasse, A.V.; Tragust, S.; Schmitt, T.; Brown, M.J.F.; Cremer, S. Destructive disinfection of infected brood prevents systemic disease spread in ant colonies. eLife 2018, 7, e32073. [Google Scholar] [CrossRef]
- Bíliková, K.; Wub, G.; Simúth, J. Isolation of a peptide fraction from honeybee royal jelly as a potential antifoulbrood factor. Apidologie 2001, 32, 275–283. [Google Scholar] [CrossRef]
- Koç, A.N.; Silici, S.; Kasap, F.; Hörmet-Oz, H.T.; Mavus-Buldu, H.; Ercal, B.D. Antifungal activity of the honeybee products against Candida spp. and Trichosporon spp. J. Med. Food. 2011, 14, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Currie, C.R.; Stuart, A.E. Weeding and grooming of pathogens in agriculture by ants. Proc. R. Soc. Lond. B. 2001, 268, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Haeder, S.; Wirth, R.; Herz, H.; Spiteller, D. Candicidin-producing Streptomyces support leaf-cutting ants to protect their fungus garden against the pathogenic fungus Escovopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 4742–4746. [Google Scholar] [CrossRef] [PubMed]
- Cafaro, M.J.; Poulsen, M.; Little, A.E.; Price, S.L.; Gerardo, N.M.; Wong, B.; Stuart, A.E.; Larget, B.; Abbot, P.; Currie, C.R. Specificity in the symbiotic association between fungus-growing ants and protective Pseudonocardia bacteria. Proc. R. Soc. Lond. B 2011, 278, 1814–1822. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.G.; Ratnieks, F.L.W. Task partitioning, division of labour and nest compartmentalisation collectively isolate hazardous waste in the leafcutting ant Atta cephalotes. Behav. Ecol. Sociobiol. 2001, 49, 387–392. [Google Scholar] [CrossRef]
- Liu, L.; Li, G.H.; Sun, P.D.; Lei, C.L.; Huang, Q.Y. Experimental verification and molecular basis of active immunization against fungal pathogens in termites. Sci. Rep.-UK 2015, 5, 15106. [Google Scholar] [CrossRef] [PubMed]
- Bordoni, A.; Dapporto, L.; Tatini, I.; Celli, M.; Bercigli, M.; Ressurrección, B.S.; Perito, B.; Turillazzi, S. Trans-generational immunization in the acrobat ant Crematogaster scutellaris. Biol. Lett. 2018, 14, 20170761. [Google Scholar] [CrossRef]
- Kay, A.D.; Bruning, A.J.; van Alst, A.; Abrahamson, T.T.; Hughes, W.O.H.; Kaspari, M. A carbohydrate-rich diet increases social immunity in ants. Proc. R. Soc. Lond. B. 2014, 281, 20132374. [Google Scholar] [CrossRef]
- Qiu, H.L.; Lu, L.H.; Zalucki, M.P.; He, Y.R. Metarhizium anisopliae infection alters feeding and trophallactic behavior in the ant Solenopsis invicta. J. Invertebr. Pathol. 2016, 138, 24–29. [Google Scholar] [CrossRef]
- Alaux, C.; Ducloz, F.; Crauser, D.; Le Conte, Y. Diet effects on honeybee immunocompetence. Biol. Lett. 2010, 6, 562–565. [Google Scholar] [CrossRef]
- Giehr, J.; Grasse, A.V.; Cremer, S.; Heinze, J.; Schrempf, A. Ant queens increase their reproductive efforts after pathogen infection. R. Soc. Open Sci. 2017, 4, 170547. [Google Scholar] [CrossRef] [PubMed]
- Vanengelsdorp, D.; Evans, J.D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B.K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y.; et al. Colony collapse disorder: A descriptive study. PLoS ONE 2009, 4, e6481. [Google Scholar] [CrossRef] [PubMed]
- Dionne, M.S.; Pham, L.N.; Shirasu-Hiza, M.; Schneider, D.S. Akt and FOXO dysregulation contribute to infection-induced wasting in Drosophila. Curr. Biol. 2006, 16, 1977–1985. [Google Scholar] [CrossRef] [PubMed]
- Clem, R.J. The role of apoptosis in defense against baculovirus infection in insects. Curr. Top. Microbiol. Immunol. 2005, 289, 113–129. [Google Scholar]
- Freitak, D.; Ots, I.; Vanatoa, A.; Hörak, P. Immune response is energetically costly in white cabbage butterfly pupae. Proc. R. Soc. Lond. B. 2003, 270, S220–S222. [Google Scholar] [CrossRef]
- Baverstock, J.; Roy, H.E.; Pell, J.K. Entomopathogenic fungi and insect behaviour: From unsuspecting hosts to targeted vectors. BioControl 2010, 55, 89–102. [Google Scholar] [CrossRef]
- Delfinado-Baker, M.; Rath, W.; Boecking, O. Phoretic bee mites and honeybee grooming behavior. Int. J. Acarol. 1992, 18, 315–322. [Google Scholar] [CrossRef]
- Sumana, A.; Starks, P.T. Grooming patterns in the primitively eusocial wasp Polistes dominulus. Ethology 2004, 110, 825–833. [Google Scholar] [CrossRef]
- He, S.; Johnston, P.R.; Kuropka, B.; Lokatis, S.; Weise, C.; Plarre, R.; Kunte, H.J.; Mcmahon, D.P. Termite soldiers contribute to social immunity by synthesizing potent oral secretions. Insect Mol. Biol. 2018, 27, 564–576. [Google Scholar] [CrossRef]
- Yanagawa, A.; Fujiwara-Tsujii, N.; Akino, T.; Yoshimura, T.; Yanagawa, T.; Shimizu, S. Behavioral changes in the termite, Coptotermes formosanus (Isoptera), inoculated with six fungal isolates. J. Invertebr. Pathol. 2011, 107, 100–106. [Google Scholar] [CrossRef]
- Sun, Q.; Zhou, X.G. Corpse management in social insects. Int. J. Biol. Sci. 2013, 9, 313–321. [Google Scholar] [CrossRef]
- Lavine, M.D.; Strand, M.R. Insect hemocytes and their role in immunity. Insect Biochem. Mol. Biol. 2002, 32, 1295–1309. [Google Scholar] [CrossRef]
- Otti, O.; Tragust, S.; Feldhaar, H. Unifying external and internal immune defences. Trends Ecol. Evol. 2014, 29, 625–634. [Google Scholar] [CrossRef]
- Gao, Q.; Thompson, G.J. Social context affects immune gene expression in a subterranean termite. Insect. Soc. 2015, 62, 167–170. [Google Scholar] [CrossRef]
- Liu, L.; Wang, W.; Liu, Y.L.; Sun, P.D.; Lei, C.L.; Huang, Q.Y. The Influence of Allogrooming Behavior on Individual Innate Immunity in the Subterranean Termite Reticulitermes chinensis (Isoptera: Rhinotermitidae). J. Insect Sci. 2019, 19, 6. [Google Scholar] [CrossRef]
- Masri, L.; Cremer, S. Individual and social immunisation in insects. Trends Immunol. 2014, 35, 471–482. [Google Scholar] [CrossRef] [PubMed]
- Sadd, B.M.; Kleinlogel, Y.; Schmid-Hempel, R.; Schmid-Hempel, P. Trans-generational immune priming in a social insect. Biol. Lett. 2005, 1, 386–388. [Google Scholar] [CrossRef] [PubMed]
- Fine, P.; Eames, K.; Heymann, D.L. "Herd immunity": A rough guide. Clin. Infect. Dis. 2011, 52, 911–916. [Google Scholar] [CrossRef]
- Ardia, D.R.; Gantz, J.E.; Schneider, B.C.; Strebel, S. Costs of immunity in insects: An induced immune response increases metabolic rate and decreases antimicrobial activity. Funct. Ecol. 2012, 26, 732–739. [Google Scholar] [CrossRef]
- Dainat, B.; Evans, J.D.; Chen, Y.P.; Gauthier, L.; Neumann, P. Predictive markers of honey bee colony collapse. PLoS ONE 2012, 7, e32151. [Google Scholar] [CrossRef]
- Calleri, D.V.; Rosengaus, R.B.; Traniello, J.F.A. Immunity and reproduction during colony foundation in the dampwood termite, Zootermopsis angusticollis. Physiol. Entomol. 2007, 32, 136–142. [Google Scholar] [CrossRef]
- Dumas, C.; Matha, V.; Quiot, J.M.; Vey, A. Effects of destruxins, cyclic depsipeptide mycotoxins, on calcium balance and phosphorylation of intracellular proteins in lepidopteran cell lines. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1996, 114, 213–219. [Google Scholar] [CrossRef]
- Sree, K.S.; Padmaja, V. Destruxin from Metarhizium anisopliae induces oxidative stress effecting larval mortality of the polyphagous pest Spodoptera litura. J. Appl. Entomol. 2008, 132, 68–78. [Google Scholar] [CrossRef]
- Zhou, X.W.; Wang, X.F.; Li, Q.Z. Expression and characteristic of the Cu/Zn superoxide dismutase gene from the insect parasitizing fungus Cordyceps militaris. Mol. Biol. Rep. 2012, 39, 10303–10311. [Google Scholar] [CrossRef]
- Serebrov, V.V.; Gerber, O.N.; Malyarchuk, A.A.; Martemyanov, V.V.; Alekseev, A.A.; Glupov, V.V. Effect of entomopathogenic fungi on detoxification enzyme activity in greater wax moth Galleria mellonella L. (Lepidoptera, Pyralidae) and role of detoxification enzymes in development of insect resistance to entomopathogenic fungi. Biol. Bull. 2006, 33, 581. [Google Scholar] [CrossRef]
- Schmid-Hempel, P.; Schmid-Hempel, R. Transmission of a pathogen in Bombus terrestris, with a note on division of labour in social insects. Behav. Ecol. Sociobiol. 1993, 33, 319–327. [Google Scholar] [CrossRef]
- Naug, D.; Smith, B. Experimentally induced change in infectious period affects transmission dynamics in a social group. Proc. R. Soc. Lond. B. 2006, 274, 61–65. [Google Scholar] [CrossRef]
- Naug, D.; Camazine, S. The role of colony organization on pathogen transmission in social insects. J. Theor. Biol. 2002, 215, 427–439. [Google Scholar] [CrossRef]
- Trible, W.; Olivos-Cisneros, L.; McKenzie, S.K.; Saragosti, J.; Chang, N.C.; Matthews, B.J.; Oxley, P.R.; Kronauer, D.J.C. Orco mutagenesis causes loss of antennal lobe glomeruli and impaired social behavior in ants. Cell 2017, 170, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Parker, R.; Guarna, M.M.; Melathopoulos, A.P.; Moon, K.M.; White, R.; Huxter, E.; Pernal, S.F.; Foster, L.J. Correlation of proteome-wide changes with social immunity behaviors provides insight into resistance to the parasitic mite, Varroa destructor, in the honey bee (Apis mellifera). Genome Biol. 2012, 13, R81. [Google Scholar] [CrossRef]
- Naug, D. Structure of the social network and its influence on transmission dynamics in a honeybee colony. Behav. Ecol. Sociobiol. 2008, 62, 1719–1725. [Google Scholar] [CrossRef]
- Thompson, G.J.; Crozier, Y.C.; Crozier, R.H. Isolation and characterization of a termite transferrin gene up-regulated on infection. Insect Mol. Biol. 2003, 12, 1–7. [Google Scholar] [CrossRef]
- Aronstein, K.A.; Murray, K.D.; Saldivar, E. Transcriptional responses in honey bee larvae infected with chalkbrood fungus. BMC Genom. 2010, 11, 391. [Google Scholar] [CrossRef]
- Mersch, D.P.; Crespi, A.; Keller, L. Tracking individuals shows spatial fidelity is a key regulator of ant social organization. Science 2013, 340, 1090–1093. [Google Scholar] [CrossRef]
- Baracchi, D.; Cini, A. A socio-spatial combined approach confirms a highly compartmentalised structure in honeybees. Ethology 2014, 120, 1167–1176. [Google Scholar] [CrossRef]
- Schlüns, H.; Crozier, R.H. Molecular and chemical immune defenses in ants (Hymenoptera: Formicidae). Myrmecol. News 2009, 12, 237–249. [Google Scholar]
- Kavaliers, M.; Choleris, E.; Ågmo, A.; Pfaff, D.W. Olfactory-mediated parasite recognition and avoidance: Linking genes to behavior. Horm. Behav. 2004, 46, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Nei, M.; Niimura, Y.; Nozawa, M. The evolution of animal chemosensory receptor gene repertoires: Roles of chance and necessity. Nat. Rev. Genet. 2008, 9, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.M.; Carlson, J.R. Drosophila chemoreceptors: A molecular interface between the chemical world and the brain. Trends Genet. 2015, 31, 683–695. [Google Scholar] [CrossRef] [PubMed]
- Meunier, J. Social immunity and the evolution of group living in insects. Philos. Trans. R. Soc. Lond B Biol. Sci. 2015, 370, 20140102. [Google Scholar] [CrossRef]
- Yan, H.; Opachaloemphan, C.; Mancini, G.; Yang, H.; Gallitto, M.; Mlejnek, J.; Leibholz, A.; Haight, K.; Ghaninia, M.; Huo, L.; et al. An engineered orco mutation produces aberrant social behavior and defective neural development in Ants. Cell 2017, 170, 736–747. [Google Scholar] [CrossRef] [PubMed]
- Guarna, M.M.; Melathopoulos, A.P.; Huxter, E.; Iovinella, I.; Parker, R.; Stoynov, N.; Tam, A.; Moon, K.M.; Chan, Q.W.; Pelosi, P.; et al. A search for protein biomarkers links olfactory signal transduction to social immunity. BMC Genom. 2015, 16, 63. [Google Scholar] [CrossRef]
- Terrapon, N.; Li, C.; Robertson, H.M.; Ji, L.; Meng, X.; Booth, W.; Chen, Z.; Childers, C.P.; Glastad, K.M.; Gokhale, K.; et al. Molecular traces of alternative social organization in a termite genome. Nat. Commun. 2014, 5, 3636. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, B.; Nicolas, E.; Michaut, L.; Reichhart, J.M.; Hoffmann, J.A. The dorsoventral regulatory gene cassette spätzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell 1996, 86, 973–983. [Google Scholar] [CrossRef]
- Krutzik, S.R.; Sieling, P.A.; Modlin, R.L. The role of Toll-like receptors in host defense against microbial infection. Curr. Opin. Immunol. 2001, 13, 104–108. [Google Scholar] [CrossRef]
- Yek, S.H.; Boomsma, J.J.; Schiøtt, M. Differential gene expression in Acromyrmex leaf-cutting ants after challenges with two fungal pathogens. Mol. Ecol. 2013, 22, 2173–2187. [Google Scholar] [CrossRef]
- Evans, J.D.; Aronstein, K.; Chen, Y.P.; Hetru, C.; Imler, J.L.; Jiang, H.; Kanost, M.; Thompson, G.J.; Zou, Z.; Hultmark, D. Immune pathways and defence mechanisms in honey bees Apis mellifera. Insect Mol. Biol. 2006, 15, 645–656. [Google Scholar] [CrossRef]
- Barribeau, S.M.; Sadd, B.M.; du Plessis, L.; Brown, M.J.; Buechel, S.D.; Cappelle, K.; Carolan, J.C.; Christiaens, O.; Colgan, T.J.; Erler, S.; et al. A depauperate immune repertoire precedes evolution of sociality in bees. Genome Boil. 2015, 16, 83. [Google Scholar] [CrossRef]
- Laughton, A.M.; Boots, M.; Siva-Jothy, M.T. The ontogeny of immunity in the honey bee, Apis mellifera L. following an immune challenge. J. Insect Physiol. 2011, 57, 1023–1032. [Google Scholar] [CrossRef]
- Laissue, P.P.; Vosshall, L.B. The olfactory sensory map in Drosophila. Adv. Exp. Med. Biol. 2008, 628, 102–114. [Google Scholar]
- Zhou, X.; Slone, J.D.; Rokas, A.; Berger, S.L.; Liebig, J.; Ray, A.; Reinberg, D.; Zwiebel, L.J. Phylogenetic and transcriptomic analysis of chemosensory receptors in a pair of divergent ant species reveals sex-specific signatures of odor coding. PLoS Genet. 2012, 8, e1002930. [Google Scholar] [CrossRef]
- Robertson, H.M.; Wanner, K.W. The chemoreceptor superfamily in the honey bee, Apis mellifera: Expansion of the odorant, but not gustatory, receptor family. Genome Res. 2006, 16, 1395–1403. [Google Scholar] [CrossRef]
- Vosshall, L.B.; Hansson, B.S. A unified nomenclature system for the insect olfactory coreceptor. Chem. Senses 2011, 36, 497–498. [Google Scholar] [CrossRef]
- Yanagawa, A.; Yokohari, F.; Shimizu, S. The role of antennae in removing entomopathogenic fungi from cuticle of the termite, Coptotermes formosanus. J. Insect Sci. 2009, 9. [Google Scholar] [CrossRef]
- Richard, F.J.; Aubert, A.; Grozinger, C.M. Modulation of social interactions by immune stimulation in honey bee, Apis mellifera, workers. BMC Biol. 2008, 6, 50. [Google Scholar] [CrossRef]
- Richard, F.J.; Holt, H.L.; Grozinger, C.M. Effects of immunostimulation on social behavior, chemical communication and genome-wide gene expression in honey bee workers (Apis mellifera). BMC Genom. 2012, 13, 558. [Google Scholar] [CrossRef]
- López, J.H.; Riessberger-Gallé, U.; Crailsheim, K.; Schuehly, W. Cuticular hydrocarbon cues of immune-challenged workers elicit immune activation in honeybee queens. Mol. Ecol. 2017, 26, 3062–3073. [Google Scholar] [CrossRef]
- Zhang, J.; Huang, W.; Yuan, C.; Lu, Y.; Yang, B.; Wang, C.Y.; Zhang, P.; Dobens, L.; Zou, Z.; Wang, C.; et al. Prophenoloxidase-mediated ex vivo immunity to delay fungal infection after insect ecdysis. Front. Immunol. 2017, 8, 1445. [Google Scholar] [CrossRef]
- Kuhn-Nentwig, L. Antimicrobial and cytolytic peptides of venomous arthropods. Cell Mol. Life Sci. 2003, 60, 2651–2668. [Google Scholar] [CrossRef]
- Ideker, T.; Ozier, O.; Schwikowski, B.; Siegel, A.F. Discovering regulatory and signalling circuits in molecular interaction networks. Bioinformatics 2002, 18, S233–S240. [Google Scholar] [CrossRef]
- McGettrick, A.F.; O’Neill, L.A. How metabolism generates signals during innate immunity and inflammation. J. Biol. Chem. 2013, 288, 22893–22898. [Google Scholar] [CrossRef] [PubMed]
- Chandran, A.; Antony, C.; Jose, L.; Mundayoor, S.; Natarajan, K.; Kumar, R.A. Mycobacterium tuberculosis infection induces HDAC1-mediated suppression of IL-12B gene expression in macrophages. Front. Cell Infect. Microbiol. 2015, 5, 90. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Wang, Y.; Jia, Z.; Wang, L.; Wang, J.; Yang, D.; Song, J.; Wang, S.; Fan, Z. Demethylation of IGFBP5 by histone demethylase KDM6B promotes mesenchymal stem cell-mediated periodontal tissue regeneration by enhancing osteogenic differentiation and anti-inflammation potentials. Stem Cells 2015, 33, 2523–2536. [Google Scholar] [CrossRef]
- Arthur, J.S.; Ley, S.C. Mitogen-activated protein kinases in innate immunity. Nat. Rev. Immunol. 2013, 13, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Kashima, M. H1 histones contribute to candidacidal activities of human epidermal extract. J. Dermatol. 1991, 18, 695–706. [Google Scholar] [CrossRef]
- Patat, S.A.; Carnegie, R.B.; Kingsbury, C.; Gross, P.S.; Chapman, R.; Schey, K.L. Antimicrobial activity of histones from hemocytes of the Pacific white shrimp. Eur. J. Biochem. 2004, 271, 4825–4833. [Google Scholar] [CrossRef]
- Cooper, D.; Eleftherianos, I. Memory and specificity in the insect immune system: Current perspectives and future challenges. Front. Immunol. 2017, 8, 539. [Google Scholar] [CrossRef]
- Kurtz, J.; Franz, K. Evidence for memory in invertebrate immunity. Nature 2003, 425, 37–38. [Google Scholar] [CrossRef]
- Sadd, B.M.; Schmid-Hempel, P. Insect immunity shows specificity in protection upon secondary pathogen exposure. Curr. Biol. 2006, 16, 1206–1210. [Google Scholar] [CrossRef]
- Rosengaus, R.B.; Malak, T.; Mackintosh, C. Immune-priming in ant larvae: Social immunity does not undermine individual immunity. Biol Lett. 2013, 9, 20130563. [Google Scholar] [CrossRef]
- Traniello, J.F.A.; Rosengaus, R.B.; Savoie, K. The development of immunity in a social insect: Evidence for the group facilitation of disease resistance. Proc. Natl. Acad. Sci. USA 2002, 99, 6838–6842. [Google Scholar] [CrossRef]
- Reber, A.; Purcell, J.; Buechel, S.D.; Buri, P.; Chapuisat, M. The expression and impact of antifungal grooming in ants. J. Evol. Biol. 2011, 24, 954–964. [Google Scholar] [CrossRef] [PubMed]
- Reber, A.; Chapuisat, M. No evidence for immune priming in ants exposed to a fungal pathogen. PLoS ONE 2012, 7, e35372. [Google Scholar] [CrossRef] [PubMed]
- Nürnberger, T.; Brunner, F. Innate immunity in plants and animals: Emerging parallels between the recognition of general elicitors and pathogen-associated molecular patterns. Curr. Opin. Plant. Biol. 2002, 5, 318–324. [Google Scholar] [CrossRef]
- Meyling, N.V.; Pell, J.K. Detection and avoidance of an entomopathogenic fungus by a generalist insect predator. Ecol. Entomol. 2006, 31, 162–171. [Google Scholar] [CrossRef]
- Gottar, M.; Gobert, V.; Matskevich, A.A.; Reichhart, J.M.; Wang, C.; Butt, T.M.; Belvin, M.; Hoffmann, J.A.; Ferrandon, D. Dual detection of fungal infections in Drosophila via recognition of glucans and sensing of virulence factors. Cell 2006, 127, 1425–1437. [Google Scholar] [CrossRef]
- Clarke, T.E.; Clem, R.J. Insect defenses against virus infection: The role of apoptosis. Int. Rev. Immunol. 2003, 22, 401–424. [Google Scholar] [CrossRef]
- Gospocic, J.; Shields, E.J.; Glastad, K.M.; Lin, Y.; Penick, C.A.; Yan, H.; Mikheyev, A.S.; Linksvayer, T.A.; Garcia, B.A.; Berger, S.L.; et al. The neuropeptide corazonin controls social behavior and caste identity in ants. Cell 2017, 170, 748–759. [Google Scholar] [CrossRef] [PubMed]
| Strategy | Effect | Defense Mechanism | Host | Species and Reference |
|---|---|---|---|---|
| Avoiding infection | Protect insect colonies from becoming infected by preventing the entrance of pathogens into the colonies | Avoid fungus-infected areas | Termites | Macrotermes michaelseni [27] |
| Ants | Acromyrmex striatus [30] | |||
| Avoid fungus-infected individuals | Termites | Zootermopsis angusticollis [31] | ||
| Reticulitermes flavipes [12] | ||||
| Ants | Formica rufa [32] | |||
| Check before colony entrance | Ants | Atta sexdens [33] Atta laevigata [33] | ||
| Collect environmental compounds for nest materials | Ants | Formica paralugubris [34] | ||
| Use self-produced compounds for nest materials (antifungal secretions) | Termites | Zootermopsis angusticollis [35] | ||
| Nasutitermes corniger [36] | ||||
| Ants | Acromyrmex subterraneus [37] | |||
| Polyrhachis dives [37] | ||||
| Bees | Apis mellifera [38] | |||
| Use symbiotic microorganism for nest materials | Termites | Coptotermes formosanus [39] | ||
| Ants | Acromyrmex octospinosus [40] | |||
| Resisting infection | Eliminate pathogens quickly by clearing infectious sources and decreasing individual susceptibility in combination | Grooming | Termites | Zootermopsis angusticollis [11] |
| Coptotermes formosanus [28] | ||||
| Ants | Acromyrmex echinatior [29] | |||
| Solenopsis invicta [15] | ||||
| Bees | Unkown [41] | |||
| Chemical disinfection (antifungal secretions) | Termites | Reticulitermes flavipes [17] | ||
| Nasutitermes costalis [42] | ||||
| Nasutitermes nigriceps [42] | ||||
| Reticulitermes speratus [43] | ||||
| Ants | Acromyrmex subterraneus [37] | |||
| Polyrhachis dives [37] | ||||
| Lasius neglectus [44] | ||||
| Bees | Apis mellifera [38] | |||
| Active self-exclusions | Ants | Lasius neglectus [45] | ||
| Lasius niger [8] | ||||
| Temnothorax unifasciatus [46] | ||||
| Myrmica rubra [47] | ||||
| Bees | Apis mellifera [24] | |||
| Aggressive behavior | Termites | Reticulitermes flavipes [12] | ||
| Ants | Lasius neglectus [16] | |||
| Cannibalism/Burial | Termites | Reticulitermes flavipes [12,48] | ||
| Coptotermes formosanus [49,50] | ||||
| Ants | Formica rufa [32] | |||
| Temnothorax lichtensteini [51] | ||||
| Removal | Ants | Myrmica rubra [52] | ||
| Solenopsis invicta [53] | ||||
| Bees | Apis mellifera [54] | |||
| Destructive disinfection | Ants | Lasius neglectus [55] | ||
| ‘Graveyards’ | Ants | Solenopsis invicta [10] | ||
| Food protection (antifungal secretions) | Bees | Apis mellifera [56,57] | ||
| Ants | Atta colombica [58] | |||
| Acromyrmex species [59] | ||||
| Tribe Attini [60] | ||||
| ‘Garbage dump’ | Ants | Atta cephalotes [61] | ||
| Social immunization | Termites | Reticulitermes chinensis [62] | ||
| Ants | Lasius neglectus [14] | |||
| Crematogaster scutellaris [63] | ||||
| Tolerating infection | Cannot directly eliminate pathogens but play an important role in social immunity | Food/nutrition | Ants | Ectatomma ruidum [64] |
| Solenopsis invicta [65] | ||||
| Bees | Apis mellifera [66] | |||
| Reproduction | Ants | Cardiocondyla obscurior [67] | ||
| Detoxification/antioxidation | Termites | Reticulitermes chinensis [62] |
| Regulator | Function | Origin | Molecule | Species and Reference |
|---|---|---|---|---|
| Chemosensory regulation | Detecting pathogens, chemical communication and inducing behavioral and physiological defenses | Fungal pathogens | Odor substances | Termites: Macrotermes Michaelseni [27] Coptotermes formosanus [50] Reticulitermes flavipes [48] |
| Host | Chemical ‘sickness cues’ | Ants: Lasius neglectus [55] | ||
| Linoleic and oleic acids | Ants: Solenopsis invicta [53] | |||
| Phenethyl acetate | Bees: Apis mellifera [54] | |||
| OR and Orco genes | Ants: Ooceraea biroi [95] Harpegnathos saltator [107] Bees: Apis mellifera [108] | |||
| IR genes | Termites: Zootermopsis nevadensis [109] | |||
| Physiological regulation | External defense by sharing insect venoms with their nestmates and nest materials | Frontal gland | α-pinene and limonene | Termites: Nasutitermes costalis and N. nigriceps [42] |
| (-)-β-elemene | Termites: Reticulitermes speratus [43] | |||
| Oral secretions | Proteins and chemicals | Termites: Mastotermes darwiniensis [75] | ||
| Fecal material | Unknown | Termites: Zootermopsis angusticollis [35] | ||
| Salivary gland | Termicins and GNBPs | Termites: Reticulitermes flavipes [17] Nasutitermes corniger [36] | ||
| Venom gland | Formic acid | Ants: Lasius neglectus [44] | ||
| Melittin | Bees: Apis mellifera [38] | |||
| Metapleural gland | Unknown | Ants: Lasius neglectus [14] Acromyrmex subterraneus [37] | ||
| Hypopharyngeal gland | Royal jelly | Apis mellifera [56,57] | ||
| Internal defense by enhancing physiological resistance and tolerance to fungal infections | Immune signal and immune effector | Toll pathway | Model insect: Drosophila [110,111] | |
| Termites: transglutaminase and histone Ants: β-1,3-glucan-binding protein and defensin Bees: tyrosine kinase 3, MyD88 and abaecin | Termites: Reticulitermes flavipes [17] Reticulitermes chinensis [62] Ants: Lasius neglectus [14] Acromyrmex echinatior [112] Bees: Apis mellifera [38,99] | |||
| Detoxication | Glutathione S-transferase and cytochrome P450 | Termites: Reticulitermes chinensis [62] | ||
| Antioxidation | Termites: superoxide dismutase and catalase Bees: hexamerin 70b and vitellogenin | Termites: Reticulitermes chinensis [62] Bees: Apis mellifera [99] | ||
| Energy metabolism, biosynthesis, development and others | Others | Termites: Reticulitermes chinensis [62] Ants: Acromyrmex echinatior [112] Bees: Apis mellifera [99] | ||
| Other | Nest materials | Conifer (Picea abies) | Resin | Ants: Formica paralugubris [34] |
| Streptomyces | Unknown | Termites: Coptotermes formosanus [39] | ||
| Streptomyces | Candicidin and antimycins | Ants: Acromyrmex octospinosus [40] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Zhao, X.-Y.; Tang, Q.-B.; Lei, C.-L.; Huang, Q.-Y. The Mechanisms of Social Immunity Against Fungal Infections in Eusocial Insects. Toxins 2019, 11, 244. https://doi.org/10.3390/toxins11050244
Liu L, Zhao X-Y, Tang Q-B, Lei C-L, Huang Q-Y. The Mechanisms of Social Immunity Against Fungal Infections in Eusocial Insects. Toxins. 2019; 11(5):244. https://doi.org/10.3390/toxins11050244
Chicago/Turabian StyleLiu, Long, Xing-Ying Zhao, Qing-Bo Tang, Chao-Liang Lei, and Qiu-Ying Huang. 2019. "The Mechanisms of Social Immunity Against Fungal Infections in Eusocial Insects" Toxins 11, no. 5: 244. https://doi.org/10.3390/toxins11050244
APA StyleLiu, L., Zhao, X.-Y., Tang, Q.-B., Lei, C.-L., & Huang, Q.-Y. (2019). The Mechanisms of Social Immunity Against Fungal Infections in Eusocial Insects. Toxins, 11(5), 244. https://doi.org/10.3390/toxins11050244




